Abstract
Acute lung injury (ALI)/acute respiratory distress syndrome (ARDS), induced by sepsis, is predominantly caused by inflammation injury. However, there is no clear consensus on how to regulate the inflammatory response. The TNF pathway is one of the primary inflammatory pathways activated in sepsis. cIAP1/2, an essential E3 ubiquitin ligase in the TNF pathway, plays a pivotal role in positively regulating the activation of nuclear factor κB (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathways to promote inflammation while inhibiting apoptosis. We found that Birc2 is the only differential expression gene in TNF pathway, and both cIAP1/2 upregulated in lung lysate with worsen lung injury. However, upon inhibiting cIAP1/2 using AZD5582, lung cell apoptosis was reactivated, and a significant improvement in lung injury was observed. Our study shows that cIAP1/2 expression increased in the lung tissue of a CLP rat ALI model. Inhibiting cIAP1/2 with AZD5582, a second mitochondria-derived activator of caspases (SMAC) mimetic, induced increased apoptosis and reduced lung injury. Therefore, inhibiting cIAP1/2 can alleviate sepsis-induced ALI, providing a new target for regulating organ damage induced by sepsis-induced inflammatory responses.
TNF pathway is an important inflammatory pathway in sepsis and can induce apoptosis.
Previous studies have found that apoptosis of peripheral blood monocyte mediates sepsis immunosuppression.
cIAP1/2 is a protein in the TNF pathway that inhibits cell apoptosis and promotes the expression of inflammatory factors.
The expression of cIAP1/2 increases in sepsis lung tissue and inhibits apoptosis.
By inhibiting cIAP1/2, lung tissue apoptosis increases, which can alleviate sepsis lung injury.
Indicating that apoptosis of non-immune cells may be beneficial for reducing organ damage in sepsis.
This study provides a new direction that inhibiting cIAP1/2 can alleviate sepsis-induced ALI.
Introduction
Acute respiratory distress syndrome (ARDS)/acute lung injury (ALI) resulting from sepsis is characterized by severe hypoxia and acute respiratory failure, causing pulmonary vascular damage, interstitial edema, and inflammation.1,2 The primary pathophysiological feature of ARDS/ALI is microvascular endothelial barrier dysfunction, which results in capillary leakage and edema, thereby exacerbating inflammatory injury, and ultimately leading to high morbidity and mortality. 2 Tumor necrosis factor (TNF) is a potent cytokine that plays diverse roles in regulating immunity, inflammation, cellular proliferation, differentiation, and apoptosis.3,4 Although the binding of TNF and TNFR1 is believed to cause cytokine storms in sepsis, the mechanism of how this signal become uncontrolled remains unclear.
Cell apoptosis, which is closely associated with the activation of the TNF pathway, has been observed to inhibit the inflammatory response in sepsis. Hotchkiss et al. 5 demonstrated that in mice subjected to cecal ligation and puncture (CLP) and transplanted with apoptotic splenic cells, the expression of inflammatory factors was downregulated. Although the induction of immune cell apoptosis suppressed the immune response, it also alleviated the inflammatory response, which may benefit the organ damage caused by sepsis-induced cytokine storm. Subsequently, Wang et al. 6 found that inhibiting neutrophil apoptosis was beneficial in promoting lung injury in the CLP model. However, no relevant research is currently investigating whether promoting cell apoptosis helps reduce sepsis-induced lung injury.
Inhibitor of apoptosis proteins (IAPs), an E3 ubiquitin ligase in the TNF pathway, can promote the inflammation signal from TNFR1 and limit the formation of apoptosis-inducing complexes. 7 Cells missing both cIAP1 and cIAP2 were sensitized to TNF-α-associated apoptosis and resistant to NF-κB activation. 8 In addition to directly interfering with cell death pathways by inhibiting apoptosis-inducing proteases (i.e. caspases), cIAP1/2 promotes inflammation through the activation of nuclear factor κB (NF-κB) and mitogen-activated protein kinase (MAPK) signaling pathways.8–10 Second mitochondria-derived activator of caspases (SMAC) mimetics (SMs), small molecule antagonists of cIAP1/2, have been widely studied in chronic inflammatory diseases such as inflammatory bowel disease (IBD), rheumatoid arthritis, and psoriasis.11–14 In IBD and psoriasis, the increase in cIAP1/2 expression is derived from the increase in the number of infiltrating leukocytes producing cIAP1/2 and the increased expression of cIAP2 in intestinal epithelial cells.15,16 This phenomenon is similar to the large number of inflammatory cell infiltrations observed in sepsis. Therefore, SMs are potential small molecules that can be employed to block inflammation.
After searching the Gene Expression Omnibus (GEO) database to identify datasets related to sepsis-induced lung injury, we found that the BIRC3 gene (which encodes the cIAP2 protein) was differentially expressed within the TNF pathway. This finding suggests that cIAP1/2 may represent an important intervention target in the TNF pathway of sepsis, with the potential to regulate sepsis-induced lung injury. Based on previous research, we believe that inhibiting cIAP1/2 may be a promising approach for alleviating lung inflammation and reducing ALI induced by sepsis.
Materials and methods
Identification of differentially expressed TNF-related genes
To obtain gene expression datasets for this study, we downloaded three assays, which contain four matrices in total, from the GEO database (http://www.ncbi.nlm.nih.gov/geo/): GSE165226, GSE165410, and GSE169532. GSE165226 determined mRNA expression profiles in the CLP mouse model of septic lung cells. GSE165410 determined mRNA by human lung microvascular endothelial cells treated with siControl+PBS, siControl+ TNF-α. GSE169532 induced sepsis in male C57BL/6 mice by CLP, then sorted capillaries (capEC) and post-capillary venules by FACS, both of cell types were involved in this study. Despite the large amount of gene expression data in the assays, we only analyzed genes related to the TNF pathway from Kyoto Encyclopedia of Genes and Genomes (KEGG) database.
Despite assays has been processed, The Limma package version 3.4.3 in R software version 4.0.3 was used to identify differentially expressed genes (DEGs) by comparing the mRNA expression of TNF pathway-associated genes. Those genes with p < 0.05 and |log2 FC| >1.0 were selected for further analysis.
Western blotting
Total proteins were extracted from cells using radio-immunoprecipitation assay buffer (Cat# PC101, EpiZyme Biotechnology, China) supplemented with protease inhibitor (1:100, Cat# P1265, Applygen, China) and phosphatase inhibitor (1:100, Cat# P1260, Applygen, China). Protein concentration was measured by BCA Protein Assay Kit (Cat# 23225, Thermo Fisher Scientific). Equal amounts of protein were separated by electrophoresis on 7.5% polyacrylamide gels (Cat# PG211, EpiZyme Biotechnology, China), transblotted onto polyvinylidene difluoride membranes, and incubated with antibodies against cIAP1 (1:1000, Cat# A19688, ABclonal, China), cIAP2 (1:1000, Cat# 3130, Cell Signaling Technology), beta-actin (1:10000, Cat# 4970, Cell Signaling Technology), and Caspase-3(1:1000, Cat# 9662, Cell Signaling Technology) at 4°C overnight. Afterward, the membranes were incubated with HRP-conjugated Goat-anti-rabbit IgG (1:10000, Cat# ZB-2301, ZSGB-BIO, China) secondary antibodies for 1 h at room temperature. Immunoreactive bands were visualized using Bio-Rad ChemiDoc XRS+ imaging systems. In some experiments, the previous primary antibody and secondary antibody were stripped from the PVDF membrane with stripping buffer (Cat# P1650, Applygen, China) for 15 min. After being re-blocked, the membrane was re-incubated with another primary antibody at 4°C overnight. The following steps were the same as described above.
Total RNA extraction and absolute RNA quantification by qPCR
Total RNAs were extracted from Fast RNA Extraction kit(Cat# RK30120, ABclonal, China) according to the instructions. RNA was reversely transcribed to cDNA templates using ABScript III RT Master Mix for qPCR with gDNA Remover (Cat# 20429, ABclonal, China) and amplified. Semiquantitative polymerase chain reaction (PCR) was performed using BrightCycle Universal SYBR Green qPCR mix with UDG (Cat# 21219, ABclonal, China) and QuantStudio 5 qPCR Platform (Applied Biosystems) with following the cycle conditions: 37°C for 2 min, 95°C for 3 min; 40 cycles of denaturation at 95°C for 5 s; annealing and amplification at 65°C for 30 s. qPCR primers were as follows: IL-10 F: 5′ CAAGGCGCATGTGAACTCC 3′ R: 5′ GGCCTTGCTCTTGTTTTCACAG 3′; IL-6 F: 5′ GGCAGAAAACAACCTGAACCT 3′ R: 5′ ATTTTCACCAGGCAAGTCTCC 3′; TNF-α F: 5′ ATCCTGGGGGACCCAATGTA 3′ R: 5′ AAAAGAAGGCACAGAGGCCA 3′; IL-1β F: 5′ ACCTCCAGGGACAGGATATGG 3′ R: 5′ ACACGCAGGACAGGTACAG 3′.
Animal treatment
The animal study was approved by the Ethics Committee of China-Japan Friendship Hospital (No. ZRDWLL230030) and was reported in compliance with the ARRIVE guidelines 17 and raised in the Animal Center of Clinical Medical Research Institute of China–Japan Friendship Hospital. Male Sprague Dawley (SD) rats, weighing 200 ± 10 g, were purchased from the Vital River Laboratories (Beijing, China, Certificate No. SCXK 2021-0011) and randomly assigned to three experimental groups using Completely Random Design, including sham, CLP and CLP+AZD5582 group. The CLP group is divided into 12 h and 24 h groups, and each group including 10 SD rats. The complete CLP model was carried out by following operation, while sham-operated rats were carried out with the same surgery without cecum ligation or puncture. Rats were anesthetized by inhaling isoflurane (2.5%). AZD5582 (0.5 mg/kg) was injected into the tail vein, whereas sham-operated mice were carried out with the same surgery without cecum ligation or puncture. Rats were sacrificed at 12 h and 24 h after CLP surgery or CLP+AZD5582 injection. The lung tissues were immediately frozen and stored in liquid nitrogen for subsequent experiments.
Cecal ligation and puncture
The rats were anesthetized with 5% isoflurane vaporized with air; 2.5% isoflurane was used for maintenance. After the mice had no response to external stimuli, their limbs were fixed in the test position. Abdominal hair removal and skin preparation: iodophor disinfection, sterile gauze coverage, a 2.5 cm incision in the center of the abdomen, and separated into the abdominal cavity according to the anatomical level. The cecum in the abdominal cavity was explored with smooth forceps. Rats model with moderate sepsis: 1/2 distal cecum was ligated with nylon suture (avoid ligating ileum and cecal mesenteric vessels). At the midpoint between the ligation and the end of the cecum, a 18-G sterile syringe needle was used to make a hole from the mesenteric edge to the mesenteric edge. After manually extruding part of the feces, the cecum was placed into the abdominal cavity and the abdominal tissues were sutured with 3-0 suture. A total of 2 mL sterile normal saline was injected intraperitoneally (i.p) at 37°C. Cecal perforation and ligation were not performed in the Sham group. 18
Histological analysis
Briefly, lung tissues were fixed in 4% paraformaldehyde at room temperature for 24 h. After processing for paraffin embedding, lung tissues were sectioned at 5 μm. Hematoxylin and eosin (HE) staining was performed by following the method routinely used in our laboratory. 19 To detect apoptosis of the lung tissue, a TUNNEL kit (Servicebio G1501, Wuhan, Hubei, China) was used to stain sections following the manufacturer’s instructions. The images were acquired using a Pannoramic MIDI system (3DHistech, Hungary), and the lung injury score was assessed by five easily identifiable pathological processes according to the official American Thoracic Society (ATS) workshop report,20,21 ImageJ software (with IHC Profiler plugins) was used for TUNNEL scoring.
Statistics
The results were expressed as mean ± SE, and R software version 4.1.1 was employed for statistical analysis. Statistical analysis for two groups employed by Student’s t-test; all experiments were performed with randomization of group assignment via allocation concealment, blinding of operators, blinding of measurements, and blinding of analyses. p < 0.05 was considered significant. The number of replicates was 3 per group for each dataset.
Results
BIRC3 is the only upregulated regulatory gene of the TNF pathway in lung cells of septic mice
We randomly selected three datasets from the GEO database using the methods previously described. The DEGs in the TNF pathway were identified from the four matrices in the three datasets, as depicted in Figure 1(a)–(d). The common DEGs in the four matrices were BIRC3, TNFAIP3, CXCL1, TNF, and IL-6, as shown in Figure 1E. We mapped these genes onto the TNF pathway diagram in KEGG (Figure 1(g)) and found that BIRC3 (also known as cIAP2) was the only upstream gene that was differentially expressed in all four groups (Figure 1(e)). The common DEGs in the four matrixes were BIRC3, TNFAIP3, CXCL1, TNF, and IL-6 (Figure 1(e)). There were no commonly downregulated genes, which can be observed in Figure 1F. Both cIAP1/2 are E3 ubiquitin ligases that ubiquitinate RIPK1 in the TNF pathway. They activate downstream inflammatory pathways by prolonging the K63 ubiquitin chain of RIPK1 and inhibit cell apoptosis, thus producing pro-survival inflammatory responses. 22 Interestingly, both BIRC2 and BIRC3 were upregulated in GSE165410 (Figure 1(b)), which is in line with previous studies showing that cIAP1/2 have similar functions and can substitute for each other.8,22 Despite their extensive study in chronic inflammation and tumors, their role in sepsis is still not well understood. Therefore, we investigated the expression of cIAP1/2 in the CLP model in SD rats.
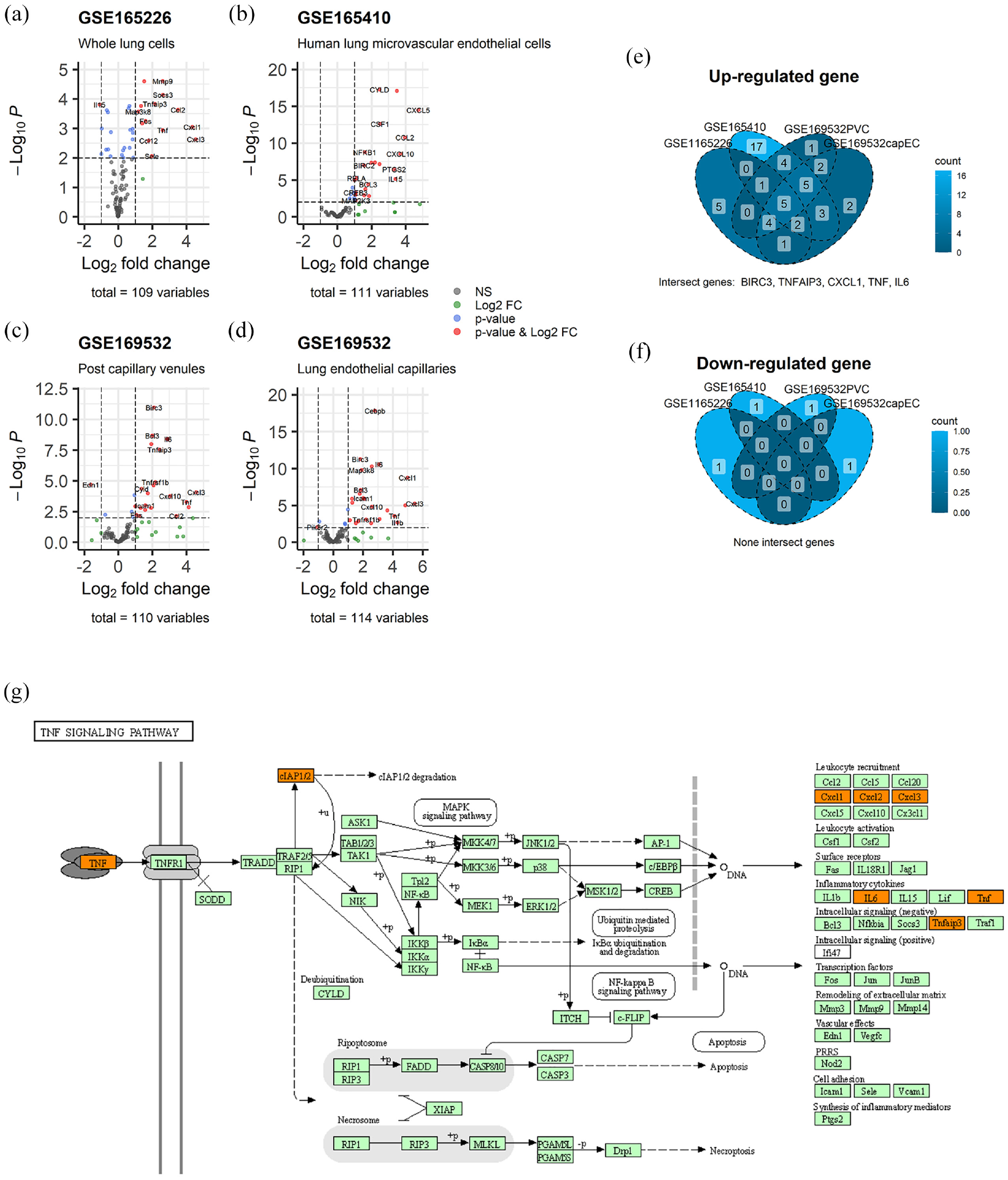
Volcano map and common differential gene Venn map of TNF pathway differential gene analysis (a)–(d): TNF pathway gene expression quantity and difference, the horizontal axis is log2fold, the vertical axis is p-log10, gray indicates no difference, green indicates difference in expression, blue indicates difference in p-value, red indicates difference in p-value and expression. (e) and (f) Common differential expression Venn diagram, each closed-loop region is an intersecting data set, and the number in each region indicates the number of genes in the region. (g) Location of common differentially expressed genes in the TNF pathway: Proteins with an orange background represent genes that are differentially expressed in the above datasets.
Increased expression of cIAP1/2 in lung tissue of septic rats
We induced sepsis in SD rats through CLP surgery and collected lung tissues for histological observation at 12 and 24 h after the procedure. All the rats in the 12 h group survived, whereas six rats died in the 24 h group. The surviving rats were used to continue the subsequent experiment. HE staining of lung tissue sections (Figure 2(a)) indicated an increase in inflammatory cells in the lung tissue of SD rats compared to the sham-operated group at both time points. Additionally, thickening of the alveolar wall and interstitium was observed. We observed more severe congestion with red blood cell accumulation in the lung tissue of the 24-h group, indicating that lung injury in CLP rats worsens over time and leads to sepsis-induced ALI. The lung injury score based on HE staining also revealed more severe lung injury in the 24-h group (Figure 2(b)).
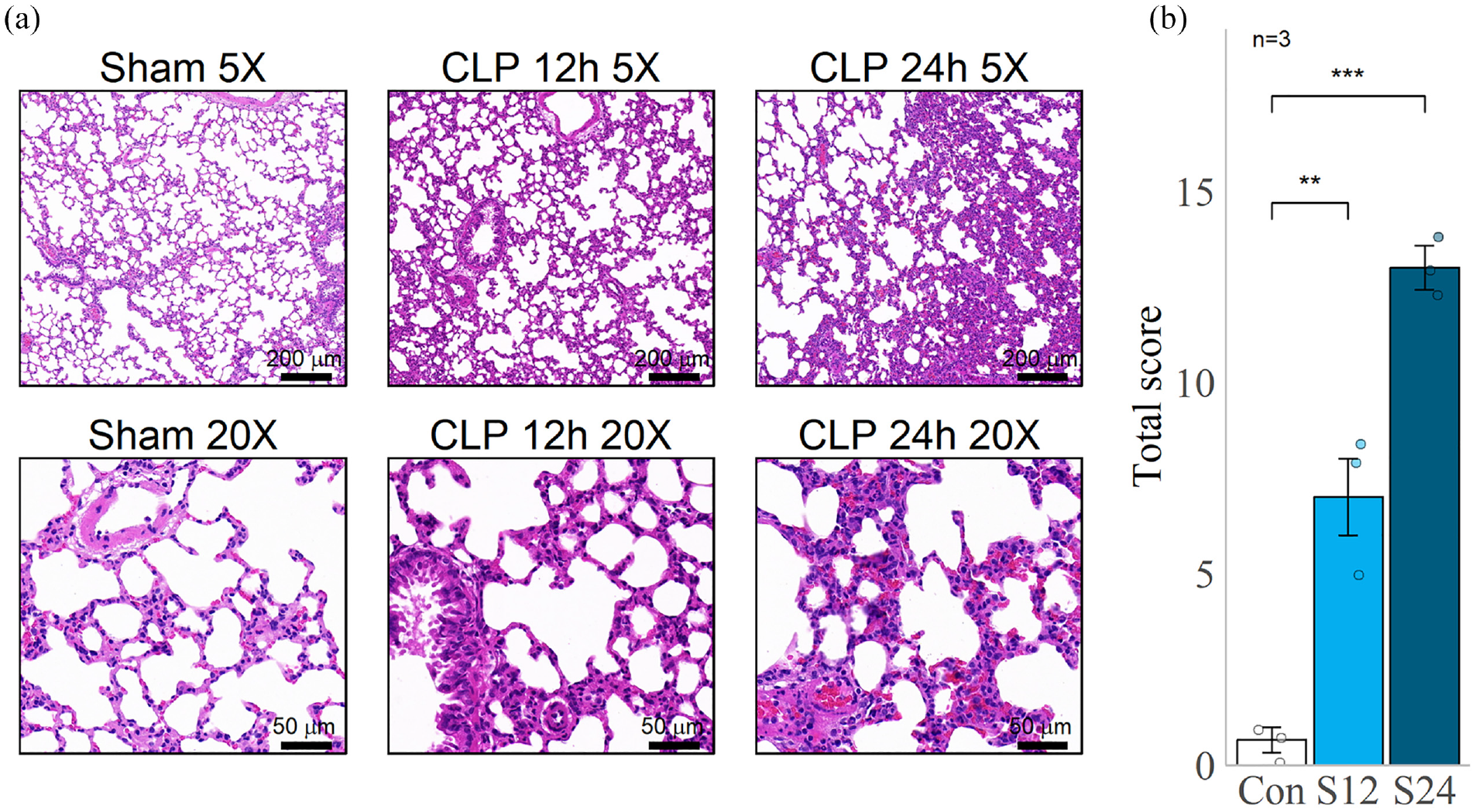
HE staining section of rat lung tissue. (a) HE staining sections of lung tissue of SD rats with different magnifications, the lower left corner is the ruler. (b) Lung histopathological score under 20× field of view, the horizontal axis is the group, and the vertical axis is the score. Each group selects three visual fields and selects the average of the three scores as the final score of the group. Sham and Con: Sham operation group.
We also quantified the expression of cIAP1/2 in lung tissue lysates from SD rats. As depicted in Figure 3(a), similarly to the differential gene expression results, the expression of cIAP1/2 was upregulated, with cIAP2 being significantly more upregulated. Importantly, despite the lack of BIRC2 gene differential expression in the previous results, we observed a significant increase in cIAP1 expression at 24 h, which could be due to differences in intervention time or severity among the randomly selected datasets. Therefore, our experimental data supported the RNA-seq result that cIAP1/2 were upregulated in the lung tissues of SD rats with sepsis.
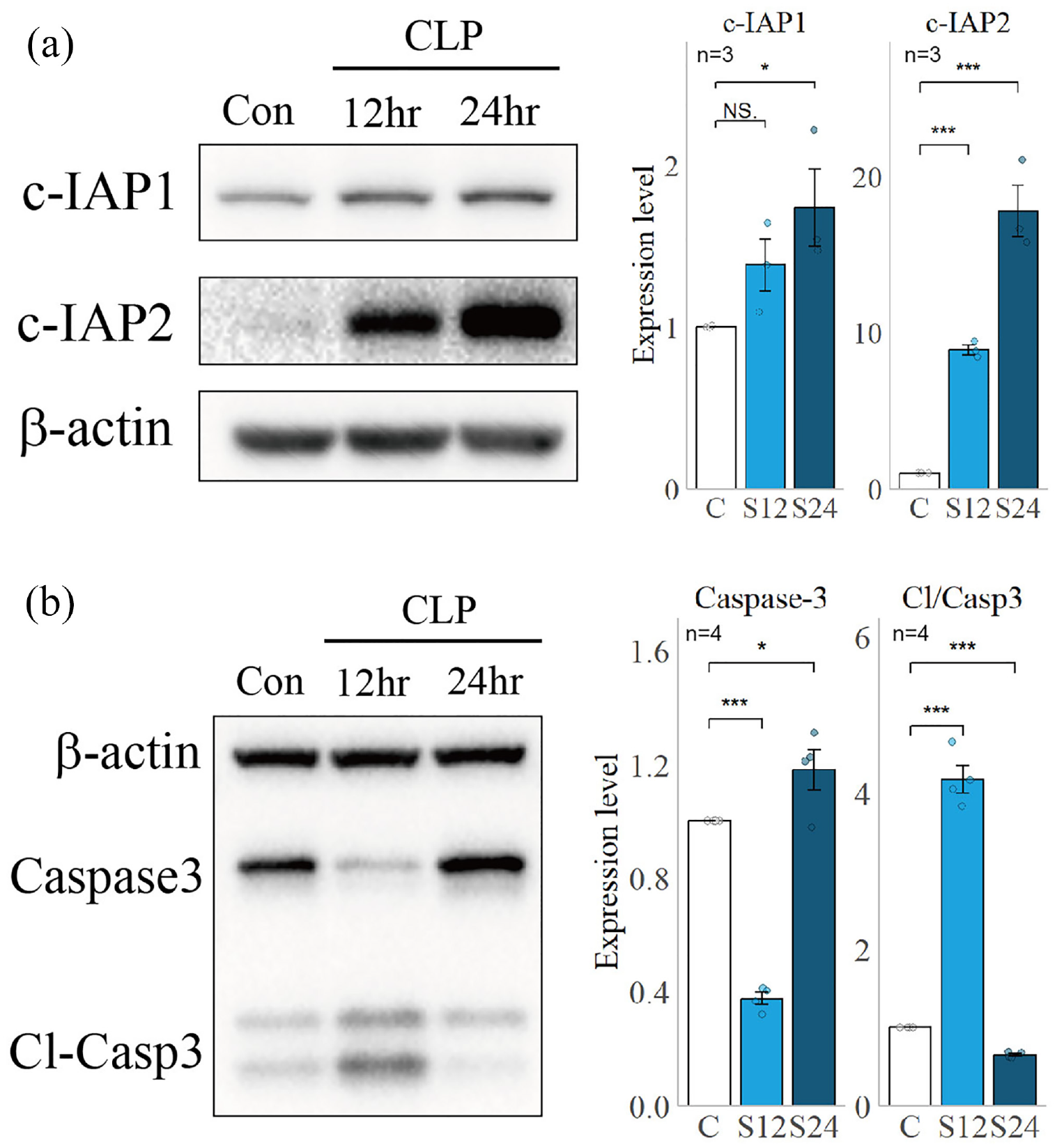
Expression of cIAP1/2, Caspase-3, and Cl-Caspase3/Caspase3 in rat lung lysate. The left side shows the expression levels of cIAP1/2, Caspase-3 and Cl-Caspase3/Caspase3 in the control, CLP 12h and 24h groups detected by Western blots, and the right side shows the relative grayscale map. (a) Expression of cIAP1/2 in rat lung lysate and relative grayscale analysis. (b) Expression of Caspase3 and Cleaved-caspase3 in rat lung lysate and relative grayscale analysis.
Next, we examined the impact of cIAP1/2 on TNF-mediated cell apoptosis by measuring Caspase3 and Cleaved-Caspase3 expression in the lung tissues of SD rats (Figure 3(b)). Our results showed an initial upregulation followed by a downregulation in the Cleaved-Caspase3/Caspase3 ratio in the lung tissues of SD rats with sepsis, indicating a transient increase in apoptosis. By analyzing the changes in cIAP1/2 expression, we concluded that its upregulation at 24 h inhibited the level of apoptosis in lung tissue. Previous studies have demonstrated that during apoptosis, the cell membrane can maintain its integrity, and cellular components do not leak out, leading to the clearance of severely damaged cells without causing severe inflammation. 23 Based on our findings, we speculate that the upregulation of cIAP1/2 in SD rats with lung injury caused by sepsis inhibits lung tissue apoptosis, thereby exacerbating ALI.
Inhibiting cIAP1/2 can enhance lung cells apoptosis and inhibit inflammation in septic rats
SMAC can bind to cIAP1/2 protein, which leads to its degradation and produces an inhibitory effect on cIAP1/2. Currently, numerous synthetic SMAC mimetics are used to suppress cIAP1/2. In this study, we intervened in SD rats by administering intravenous injection of AZD5582 24 30 min before CLP surgery and euthanized them at 24 h to collect lung tissue for analysis. Finally, 5 of 10 rats survived for subsequent experiments. As shown in Figure 4A, both cIAP1/2 underwent significant downregulation, indicating that AZD5582 successfully inhibited the expression of cIAP1/2 in the lung tissue of SD rats.
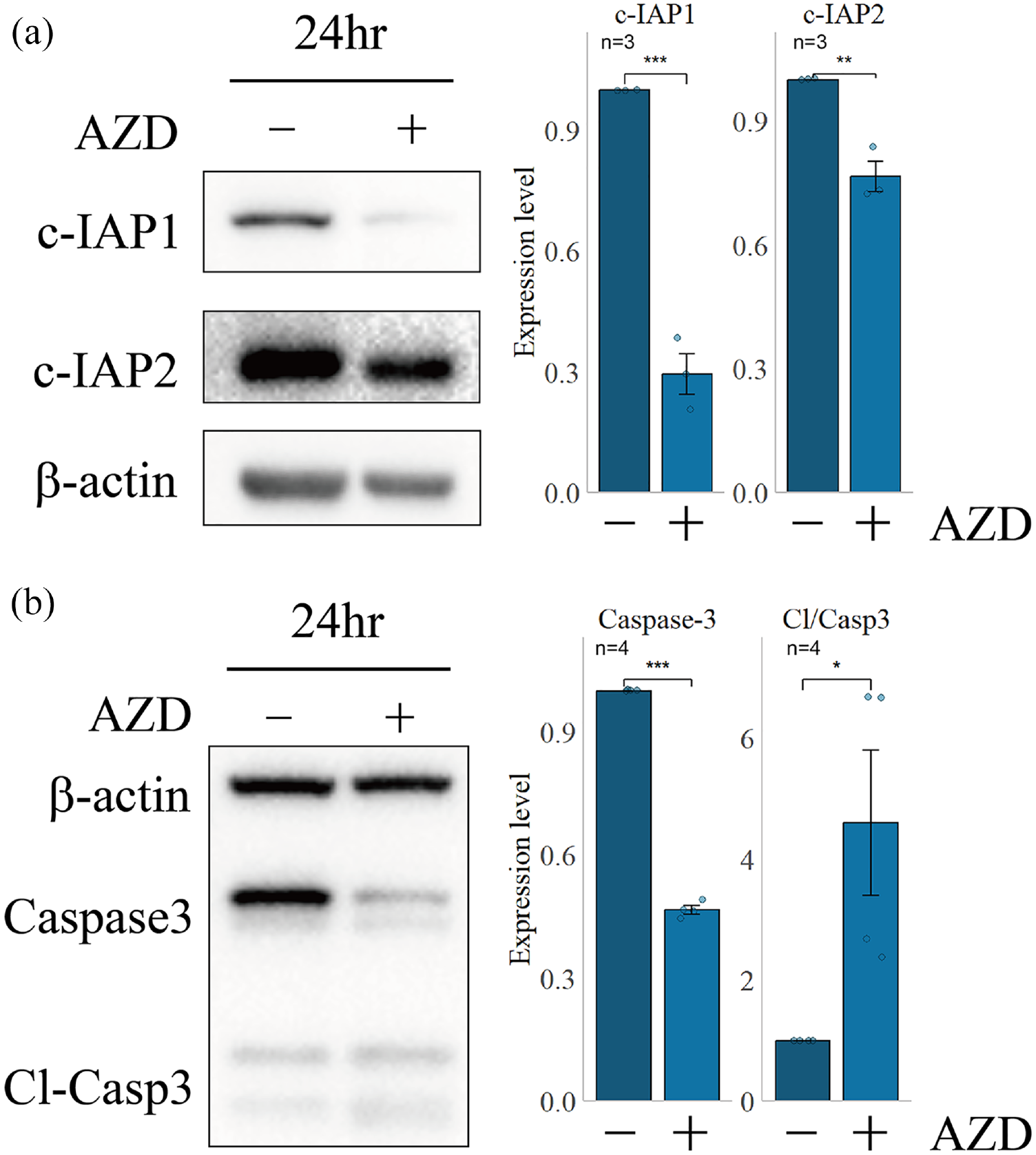
cIAP1/2, Caspase-3, and Cl-Caspase-3/Caspase3 expression level in lung tissue after AZD5582 intervention. The left side shows the expression level of cIAP1/2, Caspase-3 and Cl-Caspase-3/Caspase3 using AZD5582 intervene or not by Western blots to detect lung lysate after 24 h, whereas the right side shows the relative grayscale statistical map. Cl-Casp3: Cleaved-Caspase3, Cl/Casp3: ratio of Cl-Casp3/Caspase3. (a) Expression of cIAP1/2 in rat lung lysate after using AZD5582 and relative grayscale analysis. (b) Expression of Caspase3 and Cleaved-caspase3 in rat lung lysate after using AZD5582 and relative grayscale analysis.
In addition, we analyzed the ratio of Cleaved-Caspase3 to Caspase3 and observed a significant upregulation of this ratio after using AZD5582 to inhibit cIAP1/2 (Figure 4B). This finding suggests a significant increase in apoptotic levels in lung tissue. Thus, our results demonstrate that AZD5582 is capable of efficiently suppressing the expression of cIAP1/2 and promoting lung tissue apoptosis in septic SD rats.
Furthermore, we investigated the pathological changes of CLP rat lung slices after 24 h of HE staining following AZD5582 intervention (Figure 5). We observed a reduction in the number of inflammatory cells in the lung tissue, pulmonary interstitial edema, and alveolar wall thickness, as well as pulmonary congestion, compared to those without AZD5582 intervention. Moreover, the overall degree of lung injury and the ALI score was decreased. In TUNNEL staining, compared with the control group, a small number of TUNNEL staining positive cells appeared at 12 and 24 h. After the intervention of AZD5582, the positive cells increased significantly (Figure 6), which was in line with the change trend of Cl-Caspase3 (Figure 3(b) and Figure 4(b)), which further confirmed that AZD5582 could induce apoptosis of lung cells in CLP rats. Therefore, our findings indicate that from a pathological perspective, AZD5582 can alleviate ALI induced by sepsis.
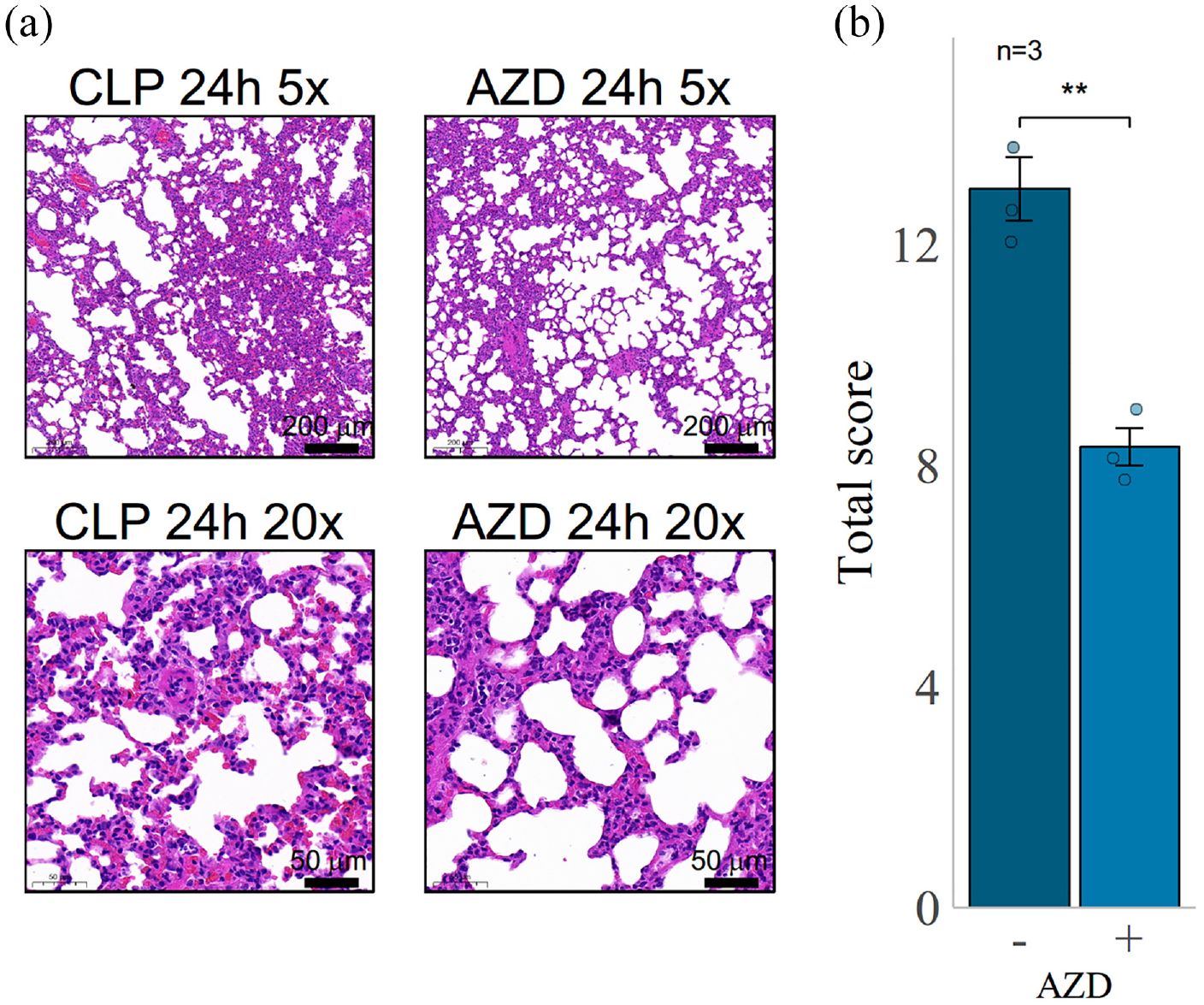
Rat lung HE staining section of CLP versus AZD5582 group at 24 h. (a) HE staining sections of lung tissue of SD rats with different magnifications, the lower left corner is the ruler. (b) Lung histopathological score under 20× field of view, the horizontal axis is the group, and the vertical axis is the score. Each group selects three visual fields and calculates the average of the three scores as the final pathological score of the group.

Rat lung TUNNEL staining section of CLP and AZD5582 group. (a) TUNNEL staining sections of lung tissue of SD rats with different magnifications, the lower left corner is the ruler. (b) Percent of TUNNEL positive cells were analyzed under 20× field of view, the horizontal axis is the group, and the vertical axis is the percentage. Each group selects three visual fields and calculates the average of the three percentages as the final percentage. Con: Sham group. S12, S24: CLP Group of 12 h and 24 h, respectively.
In addition, we analyzed the gene expression of IL-1b, IL-6, TNF-α, and IL-10 in the lung tissue of SD rats (Figure 7). Our results showed that IL-1b, IL-6, and TNF-α levels increased significantly over time, whereas IL-10 expression was not sensitive to time changes. After AZD5582 intervention, the levels of pro-inflammatory markers such as IL-1b, IL-6, and TNF-α were significantly decreased compared to the CLP surgery group, whereas the expression of the anti-inflammatory marker IL-10 was significantly upregulated. These findings suggest that in CLP surgery-intervened SD rats, ALI worsens over time, mainly manifested as an increase in inflammatory cells, thickening of the alveolar wall and pulmonary interstitium, and pulmonary congestion. Apoptosis of lung tissue cells increases initially and then decreases, while inflammatory factors increase. After the AZD5582 intervention, the level of cell apoptosis significantly increased compared with the non-intervention group, inflammatory factors were down-regulated, anti-inflammatory factors were upregulated, and lung injury was relieved. Thus, our results indicate that inhibiting cIAP1/2 can increase cell apoptosis in lung tissue, reduce inflammatory reactions, and ultimately alleviate sepsis-induced lung injury.
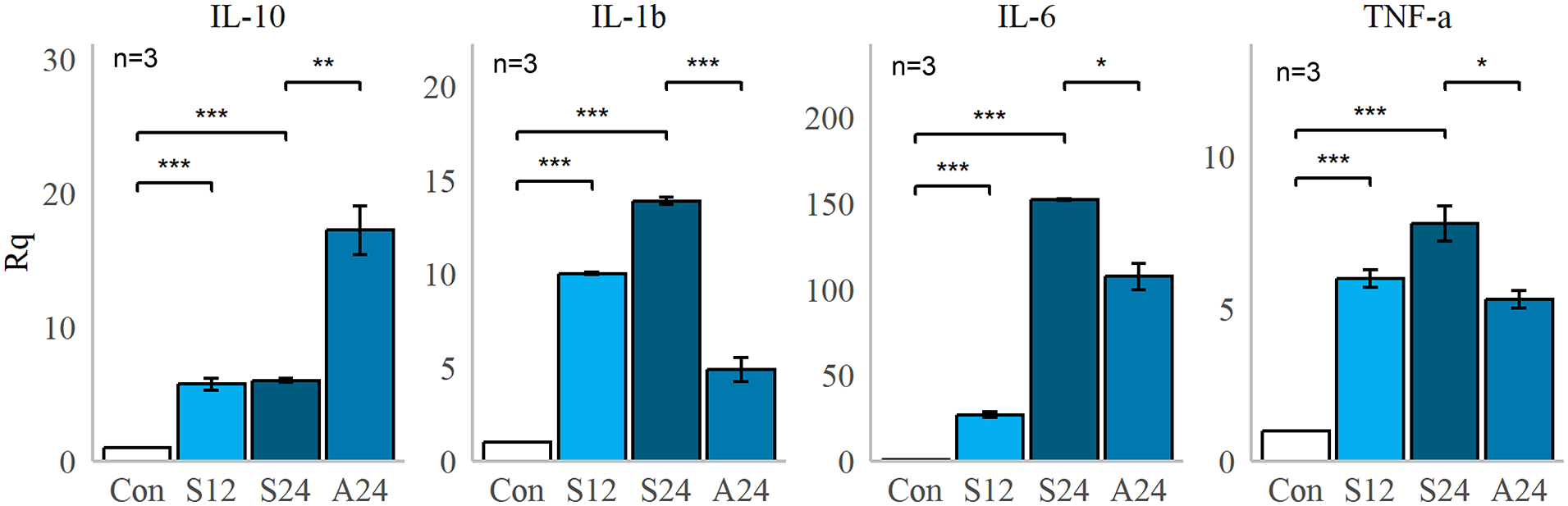
Expression of inflammatory markers by qPCR. The horizontal axis is the group, and the vertical axis is the relative quantification. Con: Sham group. S12, S24: CLP Group of 12 h and 24 h, respectively. A24: CLP 24h with intravenous AZD5582 group.
Discussion
Sepsis is a serious condition that is associated with high mortality and morbidity due to multi-organ dysfunction, including ARDS/ALI that causes severe systemic inflammatory response syndrome. The development of ARDS/ALI in sepsis involves the rapid activation of the systemic inflammatory response, which triggers a quick and effective defense mechanism aimed at reducing microvascular endothelial barrier permeability, further promoting the production and migration of immune cells such as monocytes and macrophages to resist the invasion of pathogenic microorganisms and endotoxins.25,26 However, sepsis leads to excessive cytokine release, causing an imbalance in microvascular endothelial barrier permeability, worsening microvascular leakage, and ultimately leading to circulatory failure and multi-organ dysfunction.27,28 The TNF receptor (TNFR)-mediated activation of the NF-κB and MAPK pathways induces an inflammatory response that exacerbates the sepsis inflammatory response,29–32 However, TNFR also mediates cell apoptosis, which helps to mitigate the inflammatory response.5,6 Thus, the regulation of the two opposing factions in the TNF pathway is an important topic worth further exploration.
In our study, we have found an exacerbation of lung injury due to cIAP1/2-mediated apoptosis inhibition. cIAP1/2 functions as an E3 ubiquitin ligase in the TNF pathway. Our prior RNA sequencing analysis revealed BIRC3 as the singular, common differentially expressed regulator found in the TNF pathway. BIRC3 is known to promote inflammatory signals from TNFR1 and limit the formation of apoptotic complexe. 3 However, research on the regulation of lung inflammation and injury in sepsis by cIAP1/2 remains scarce. Our experiments have shown that the expression of cIAP1/2 is upregulated in a time-dependent manner in the CLP-induced ALI model, accompanied by a decrease in the proportion of Cleaved-Caspase3 and an increase in the expression of IL-6, TNF-α, and IL-1β, as well as the lung injury score. These results indicate that the upregulation of cIAP1/2 in lung tissue during sepsis inhibits cell apoptosis, thereby exacerbating lung injury. Further confirmation of our hypothesis has been achieved through the use of AZD5582, which inhibits cIAP1/2 and relieves the inhibition of apoptosis in lung tissue, leading to a reduction in inflammation and subsequent alleviation of lung injury. However, in terms of survival rate, AZD5582 intervention did not show a significant improvement (6 of 10 died in the 24 h group and 5 of 10 died in the AZD5582 group). This may be because sepsis is often accompanied by multiple organ dysfunction syndrome, and reducing inflammation alone may not prevent organ dysfunction. Therefore, further experiments are required to investigate the effect of AZD5582 on the overall sepsis condition.
Prior research has suggested that lymphocyte apoptosis plays a role in sepsis-induced immunosuppression, which results in insufficient immune responses to combat infection. 33 However, immune responses during the progression of sepsis often shift from overactivation to suppression. Initially, overactive immune responses lead to excessive systemic inflammation, which results in septic shock and cytokine-induced systemic organ damage. Subsequently, suppressed immune responses lead to secondary infections. Therefore, early apoptosis may not necessarily indicate a poor prognosis. Our study indicates that promoting cell apoptosis during the early stages of sepsis can alleviate sepsis-induced lung injury. However, the current study has some limitations that need to be acknowledged. First, the study failed to differentiate between endothelial and lymphocyte apoptosis. Further investigations are needed to distinguish the impact of lymphocyte or endothelial cell apoptosis on ALI, which may help to understand the mechanism of injury resulting from inflammatory cytokines produced by specific cells. Second, the study only demonstrated the benefits of promoting apoptosis in mitigating inflammation-induced lung injury from a lung injury perspective. However, the study did not discuss the potential effects of this approach on the prognosis or the later-stage immune suppression. Therefore, additional experimental investigations are warranted to address these questions and provide a better understanding of the therapeutic potential of apoptosis in treating ALI.
Conclusions
While it is understood that cIAP1/2 inhibitors, such as SMs, can improve chronic inflammatory responses, there have been no reports on their effects on sepsis. Our study presents, for the first time, evidence that the inhibition of cIAP1/2 can alleviate sepsis-induced lung injury by promoting cell apoptosis in SD rats with sepsis. This leads to the suppression of inflammatory responses and the protection of lung tissue from the impacts of the cytokine storm. These findings identify a potential therapeutic target and a novel direction for sepsis-induced lung protection.
Footnotes
Acknowledgements
Thanks for the technical support of the Clinical Medical Research Center/Institute of Clinical Medicine of China–Japan Friendship Hospital.
Author contributions
Xiaoyu Liu designed this study and drafted the manuscript. Jie Chen and Xiaoyu Liu performed the animal model. Zhonghao Li conducted the data analysis. Nan Gao performed the WB and qPCR. Guoqiang Zhang revised this manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China [No. 82272196].
Ethics approval and consent to participate
Mouse experiments were approved by the Animal Ethics Committee of China-Japan friendship hospital (No. ZRDWLL230030). All experiments were performed following relevant guidelines and regulations for animals.
