Abstract
Metabolic dysfunction-associated fatty liver disease (MAFLD) is strongly associated with disturbances in the intestinal microbiota. Herein, the biological effects and mechanism of Bifidobacterium bifidum BGN4 fractions in regulating hepatocyte ferroptosis during MAFLD progression were investigated. To establish an in vitro model of MAFLD, LO2 cells were subjected to palmitic acid (PA). The mRNA and protein expressions were assessed using quantitative real-time polymerase chain reaction (qRT-PCR) and Western blot, respectively. LO2 cell proliferation was examined using 5-diphenyltetrazolium bromide (MTT) and ethynyl-2′-deoxyuridine (EdU) assays, whereas its apoptosis was evaluated by flow cytometry. Furthermore, level of reactive oxygen species (ROS) was measured using 2', 7,-Dichlorodihydrofluorescein diacetate (DCFH-DA) staining. Additionally, the levels of Fe2+, malondialdehyde (MDA), and glutathione (GSH), as well as the activities of superoxide dismutase (SOD) and glutathione peroxidase (GPX) were detected using corresponding kits. Chromatin immunoprecipitation and dual-luciferase reporter gene assays were performed to analyze the interaction between sterol-regulatory element binding protein 1 (SREBP1) and cytochrome P450-2E1 (CYP2E1) promoter. Our results revealed that Bifidobacterium bifidum BGN4 fractions effectively ameliorated PA-induced hepatocyte injury, oxidative stress, and ferroptosis. However, these beneficial effects of BGN4 fractions on PA-induced hepatocyte were dramatically reversed by SREBP1 overexpression, suggesting that BGN4 attenuated MAFLD by acting on SREBP1. Moreover, we observed that BGN4 fractions inhibited CYP2E1 transcription by suppressing SREBP1 nuclear translocation. In addition, CYP2E1 overexpression eliminated the inhibitory effect of BGN4 fractions on PA-induced hepatocyte oxidative stress and ferroptosis. These findings collectively indicated that BGN4 fractions reduced CYP2E1 expression by inhibiting SREBP1 nuclear translocation, thereby suppressing hepatocyte oxidative stress and ferroptosis during the development of MAFLD.
Keywords
Significance of this study
Hepatocyte ferroptosis plays a significant role in the pathogenesis and progression of MAFLD.
Bifidobacterium bifidum BGN4, as a probiotic, has demonstrated therapeutic effects in the treatment of MAFLD.
Beneficial effects of BGN4 fractions: BGN4 fractions were found to remarkably ameliorate palmitic acid (PA)-induced hepatocyte injury, oxidative stress, and ferroptosis.
Mechanism of action: The study revealed that BGN4 fractions exerted their effects by suppressing the transcription of CYP2E1, a gene involved in MAFLD, through the inhibition of SREBP1 nuclear translocation in hepatocytes.
This study not only offers a potential treatment option for MAFLD but also contributes to the advancement of scientific knowledge, facilitating the development of innovative approaches to tackle this disease.
Highlights
The fractions of BGN4 demonstrated a beneficial effect by improving hepatocyte injury, reducing oxidative stress, and inhibiting ferroptosis induced by palmitic acid (PA).
BGN4 fractions exerted their inhibitory effect on CYP2E1 expression through the inhibition of SREBP1 nuclear translocation.
The protective effects of BGN4 fractions on PA-induced hepatocyte damage were greatly reversed by the SREBP1-CYP2E1 signaling pathway.
Introduction
Nonalcoholic fatty liver disease (NAFLD) is a prevalent metabolic disorder characterized by excessive fat accumulation in hepatocytes, with the potential to progress to cirrhosis and liver cancer. 1 Recently, the term metabolic dysfunction-associated fatty liver disease (MAFLD) has been proposed to more accurately represent the underlying pathogenesis and metabolic disorders associated with the disease. The global prevalence of NAFLD is approximately 25% and up to 75% of individuals with obesity.2,3 Unfortunately, due to the unknown pathogenesis of MAFLD, there is no currently satisfactory treatment strategy. Urgently, new treatment options are needed to improve the clinical prognosis of MAFLD. The pathogenesis of MAFLD is very complex and involves various factors such as inflammation, oxidative stress, and lipid metabolism disorder. 4 Ferroptosis, an iron-dependent form of cell death characterized by the depletion of glutathione (GSH), accumulation of lipid peroxidation products, and generation of lethal ROS, 5 has been identified as a key player in the pathological process of MAFLD.6,7 Inhibition of ferroptosis has been suggested as a potential therapeutic strategy for NAFLD. 8 Consequently, pharmacological agents with multiple functions, including anti-ferroptosis properties, hold promise as potential therapeutic strategies for MAFLD.
The intestinal microbiota refers to the diverse the diverse population of bacteria residing in the gastrointestinal tract, consisting of thousands of different species. 9 The composition and distribution of the intestinal microbiome are dynamic and can be influenced by various factors such as nutrition, immune response, and the environment. Imbalances in the intestinal flora can have a significant impact on the host’s metabolism, inflammation, immune homeostasis, and other physiological processes and may contribute to the development of various diseases, including MAFLD. 10 As widely illustrated, the intestinal microbiota is a newly recognized environmental factor associated with obesity and NAFLD. 10 Certain metabolites produced by the intestinal flora can promote disease development by increasing liver steatosis and altering intestinal mucosal permeability. Targeting the intestinal microbiota has emerged as a novel therapeutic approach for managing NAFLD. 11
Live probiotics, heat-killed probiotics, and their metabolites have been shown to provide significant benefits in disease prevention and treatment. 12 As revealed by Lee et al., 13 both the live Bifidobacterium bifidum BGN4, heat-killed Bifidobacterium bifidum BGN4 and lysozyme-treated Bifidobacterium bifidum BGN4 improved inflammation-associated dysbiosis during the development of inflammatory bowel disease. Bifidobacterium bifidum is a probiotic strain known for its diverse biological activities and its potential to alleviate MAFLD. 11 As proof, Lee et al. 14 demonstrated that Bifidobacterium bifidum could markedly reduce H2O2-induced oxidative stress in hepatocytes, as well as decreased levels of hepatic aspartate transaminase, alanine transaminase, and lipid peroxidation in a mouse model of tert-Butyl hydroperoxide (tBHP)-induced liver injury. In addition, Wang et al. 15 demonstrated that Bifidobacterium bifidum alleviated hepatic steatosis in high-fat-induced NAFLD mice by modulating the gut microbiota and improving insulin resistance. Notably, previous reports have indicated bifidobacterial strains can inhibit inflammation induced by lipopolysaccharide (LPS) and iron through the regulation of hepcidin- and iron-signaling pathways. 16 Nevertheless, the specific role and regulatory mechanism of Bifidobacterium bifidum in modulating hepatocyte ferroptosis during MAFLD progression remain unclear, and further research is warranted to investigate these aspects.
Sterol-regulatory element binding protein 1 (SREBP1) is a transmembrane protein located in the endoplasmic reticulum that serves as a key regulator of lipid homeostasis. It acts as a signaling switch, receiving metabolic cues and activating the transcription of crucial enzyme genes involved in lipid metabolism pathways. 17 Dysregulation of SREBP1 is associated with impaired liver lipid metabolism, causing lipid accumulation, thereby contributing to the progression of MAFLD. 18 Interestingly, studies have shown that oral administration of Bifidobacterium could alleviate MAFLD symptoms by reducing the expression of SREBP1. 19 However, whether Bifidobacterium bifidum exerts its protective effects against hepatocyte ferroptosis through modulation of SREBP1 remains unclear, and further investigation is warranted to elucidate this relationship.
Based on the findings from the aforementioned studies, we hypothesized that Bifidobacterium bifidum has the potential to suppress hepatocyte ferroptosis during the progression of MAFLD by inhibiting the SREBP1-CYP2E1 pathway. This mechanism could contribute to the attenuation of MAFLD. The results obtained from our study provide valuable evidence for the treatment of MAFLD and shed light on the underlying therapeutic mechanism.
Materials and methods
Preparation of Bifidobacterium bifidum BGN4 fractions
Bifidobacterium bifidum BGN4 fractions, purchased from American Type Culture Collection (ATCC, VA, USA), were cultured in de Man, Rogosa and Sharpe (MRS) medium (Oxoid, Basingstoke, UK) supplemented with 0.01% L-cysteine hydrochloride monohydrate at 37°C for 24 h under anerobic condition. After incubation, the bacterial cells were collected by centrifugation. The resulting cell pellet was resuspended in phosphate-buffered saline (PBS). Subsequently, the cells were treated with lysozyme at 37°C for 2 h to facilitate cell lysis. The cell fractions were further disrupted using sonication. Finally, the BGN4 fractions obtained from this process were stored at −80°C until further use.
Cell culture and treatment
LO2 cells, purchased from ATCC, were cultured in RPMI 1640 medium (Gibco, MD, USA) supplemented with 10% fetal bovine serum (FBS, Gibco) at 37°C with 5% CO2. Once the cells reached 80% confluence, LO2 cells were treated with 0.35 mM palmitic acid (PA, MedChemExpress, NJ, USA) for 24 h. Following the PA treatment, the LO2 cells were exposed to bacterial fractions of BGN4 (1 × 108 CFU/mL) for various time points including 0, 12, 24, 48, and 72 h.
Cell transfection
The overexpression plasmid of SREBP1 (pcDNA3.1-SREBP1), CYP2E1 (pcDNA3.1-CYP2E1), and the negative control (pcDNA3.1) were obtained from GenePharma (Shanghai, China). The transfection was performed using Lipofectamine™ 3000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. After 48 h of transfection, the efficiency of transfection was assessed by quantitative real-time polymerase chain reaction (qRT-PCR).
3-(4, 5-Dimethylthiazolyl2)-2, 5-diphenyltetrazolium bromide assay
LO2 cells were seeded in a 96-well plate at a density of 5 × 103 cells/well and incubated for 12 h. Subsequently, the cells were treated with 5 mg/mL 5-diphenyltetrazolium bromide (MTT; Beyotime, Shanghai, China) and incubated for 4 h at room temperature. After the incubation period, the formazan crystals were dissolved by adding dimethyl sulfoxide (DMSO) (Sigma-Aldrich, MO, USA). The absorbance of the solution was measured at 490 nm using a microplate reader (Bioteke, Beijing, China).
Ethynyl-2′-deoxyuridine assay
LO2 cells were plated in 48-well plates at a density of 1 × 105 cells/well and incubated with ethynyl-2′-deoxyuridine (EdU; Beyotime) for 2 h at 37°C. Cells were subsequently fixed, permeabilized, and incubated with the staining solution for 30 min. Hoechst 33,342 was employed to stain the DNA. Cells were then observed under a fluorescence microscope (Olympus, Tokyo, Japan).
Cell apoptosis assay
After collection, LO2 cells were resuspended in 500 μL of 1x Annexin-binding buffer (Beyotime). Next, the cells were incubated with 10 μL Annexin V-FITC and 5 μL PI stain (Beyotime) for 10 min. Subsequently, the samples were immediately analyzed using flow cytometry (BD, NJ, USA).
Quantitative real-time polymerase chain reaction
Total RNA was extracted from LO2 cells with TRIzol regent (ThermoFisher Scientific, MA, USA). The cDNA was synthesized using the Reverse Transcription Kit (Toyobo, Tokyo, Japan) and subjected to qRT-PCR assay utilizing PowerUp™ SYBR™ Green Master Mix (ThermoFisher Scientific, MA, USA). The mRNA expression levels were normalized to the reference gene GAPDH using the 2−ΔΔCT method. The primers sequences used were as follows (5’-3’):
SREBP1 (F): ACAGTGACTTCCCTGGCCTAT
SREBP1 (R): GCATGGACGGGTACATCTTCAA
CYP2E1 (F): ATGTCTGCCCTCGGAGTCA
CYP2E1 (R): CGATGATGGGAAGCGGGAAA
β-actin (F): CCACGAAACTACCTTCAACTCC
β-actin (R): GTGATCTCCTTCTGCATCCTGT
Western blot
Total proteins were extracted using RIPA lysis buffer (Beyotime), and their concentrations were determined using a BCA kit (Beyotime). The total proteins were denatured by boiling and separated by SDS-PAGE gel. Subsequently, the proteins were transferred onto PVDF membranes (Millipore, MA, USA). The membranes were incubated overnight with the following primary antibodies: B-cell lymphoma-2 (Bcl-2, Abcam, Cambridge, UK, 1:1000, ab32124), Bax (Abcam, 1:1000, ab32503), glutathione peroxidase 4 (GPX4, Abcam, 1:1000, ab125066), TRF1 (Abcam, 1:1000, ab10579), SREBP1 (Abcam, 1:1000, ab28481), cytochrome P450-2E1 (CYP2E1, Abcam, 1:1000, ab28146), Lamin B1 (Abcam, 1:5000, ab16048), and β-actin (Abcam, 1:5000, ab8226). After washing with PBS-T, the membranes were then incubated with a secondary antibody (Abcam, 1:10000, ab7090) for 60 min. The protein bands were visualized using a GEL imaging system (Bio-Rad, CA, USA) and quantified by Image J software (National Institutes of Health, MD, USA).
Measurement of Fe2+
Following the specified treatment, cells were collected and homogenized using PBS. The resulting supernatant was used to measure the level of Fe2+ using an iron assay kit (Abcam, ab83366). Briefly, the samples were added to wells and incubated with assay buffer at 37°C for 30 min. Then the iron probe was added and incubated at 37°C for 60 min. The absorbance was measured using a microplate reader (Bioteke, Beijing, China).
Measurement of superoxide dismutase (SOD) activity, glutathione peroxidase (GPX) activity, glutathione (GSH) level, and malondialdehyde (MDA) level.
The activities of SOD and GPX, as well as the concentrations of GSH and MDA, in the supernatants of treated LO2 cells were determined using the SOD activity assay kit (S0101S, Beyotime), GPX activity assay kit (S0058, Beyotime), GSH assay kit (S0052, Beyotime), and MDA assay kit (S0131S, Beyotime), respectively, following the respective assay protocols.
Measurement of ROS
To detect intracellular ROS, the treated cells (1 × 106) were incubated with 10 μM 2′,7′-dichlorofluorescein diacetate (DCFH-DA, Beyotime) for 25 min in the dark. Following the incubation, the cells were washed twice with PBS to remove any excess DCFH-DA. The cells were then imaged using a fluorescence microscope (Olympus) to visualize the fluorescence signal indicative of intracellular ROS levels.
Chromatin immunoprecipitation assay
The cells were treated with 1% formaldehyde solution for 10 min to cross-link proteins and DNA. To stop the cross-linking, 125 mM glycine was added for 5 min. The cells were then fragmented into 200–500 bp length fragments using sonication. The cell lysate containing the fragmented chromatin was incubated overnight at 4°C with an anti-SREBP1 antibody (Abcam, 1:100, ab191857) or an anti-IgG antibody (Abcam, 1:100, ab172730). Following the incubation, the cross-links of the enriched and input DNA samples were reversed, and the DNA was purified. The purified DNA fragments were then analyzed using qRT-PCR to measure the enrichment of specific DNA regions.
Dual-luciferase reporter gene assay
The fragment of CYP2E1 promoter was amplified by PCR and subsequently inserted into the pGL3 vector (Promega, WI, USA). The resulting recombinant pGL3 vectors were co-transfected into LO2 cells along with either pcDNA3.1 or pcDNA3.1-SREBP1 using Lipofectamine™ 3000 (Invitrogen). After 48 h, the luciferase activity was assessed by a dual-luciferase reporter assay system (Promega).
Statistical analysis
All data were obtained from three independent experiments and analyzed using SPSS 19.0 (IBM, NY, USA). The results were expressed as means ± standard deviation (SD). The statistical significance of differences between two groups was assessed using Student’s t-test, whereas one-way analysis of variance followed by Tukey’s post hoc test was employed for multiple group comparisons. A p-value of less than 0.05 was considered statistically significant.
Results
Bifidobacterium bifidum BGN4 fractions prevented PA-induced hepatocyte damage
In this study, the effect of Bifidobacterium bifidum BGN4 fractions on LO2 cells was investigated. Initially, the viability of LO2 cells was assessed using the MTT assay after treatment with BGN4 fractions for various time points (12, 24, 48, and 72 h). The result showed no significant effect on LO2 cell viability, indicating that BGN4 fractions did not exhibit any toxic effects on LO2 cells (Figure 1(a)). Subsequently, LO2 cells were subjected to PA to establish an in vitro model of MAFLD. Obviously, PA treatment significantly inhibited LO2 cell viability and proliferation based on the results of MTT and EdU assays. However, the adverse effects of PA were reversed when cells were co-treated with BGN4 fractions (Figure 1(b) and (c)). In addition, it was observed that PA treatment significantly facilitated LO2 cell apoptosis, whereas this effect of PA was abolished by BGN4 (Figure 1(d)). Moreover, Western blot analysis revealed that PA treatment led to an elevation in Bax protein level and a reduction in Bcl-2 protein level. However, BGN4 fractions treatment counteracted these changes induced by PA, restoring the levels of Bax and Bcl-2 proteins (Figure 1(e)). Taken together, these findings demonstrated that Bifidobacterium bifidum BGN4 fractions treatment has a protective effect in PA-induced hepatocyte injury.
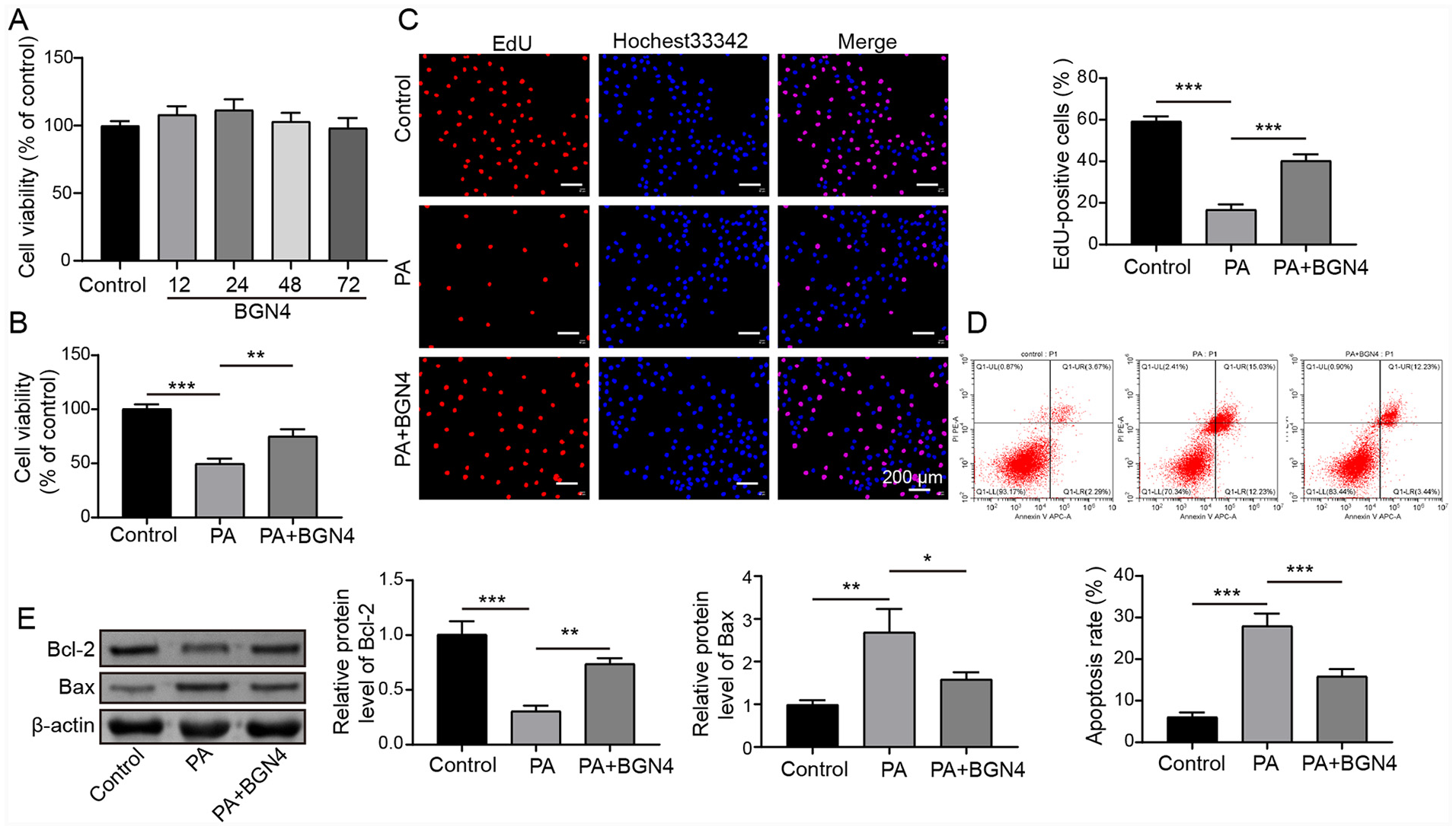
Bifidobacterium bifidum BGN4 fractions protected LO2 cells from PA-induced cell injury. (a) Viability of LO2 cells treated with bacteria fractions of BGN4 (1 × 108 CFU/mL) for different time points (0, 12, 24, 48, and 72 h) was assessed using MTT assay. LO2 cells were treated with 0.35 mM PA for 24 h and then incubated with bacteria fractions of BGN4 for an additional 24 h. (b) MTT assay was performed to evaluate the viability of LO2 cells. (c) EdU assay was conducted to measure cell proliferation. (d) Flow cytometry was performed to access LO2 cell apoptosis. (e) Protein levels of Bax and Bcl-2 in LO2 cells were examined using Western blot analysis. The measurement data were presented as mean ± SD. All data was obtained from at least three replicate experiments.
Bifidobacterium bifidum BGN4 suppressed PA-induced hepatocyte ferroptosis
In order to investigate the role of Bifidobacterium bifidum BGN4 fractions in regulating PA-induced hepatocyte ferroptosis, several key markers associated with ferroptosis were examined. It was observed that PA treatment led to an increase of MDA level, and a decrease of GSH level and SOD and GPX activities, further indicating impaired antioxidant defense mechanisms (Figure 2(a)–(d)). However, co-treatment with BGN4 fractions attenuated these changes induced by PA, restoring MDA levels, GSH levels, and the activities of SOD and GPX (Figure 2(a)–(d)). Furthermore, the intracellular ROS and Fe2+ levels were assessed. PA stimulation led to a significant increase in ROS production and Fe2+ level, whereas co-treatment with BGN4 fractions effectively reduced the ROS levels induced by PA (Figure 2(e) and (f)). Consistently, the decreased GPX4 level and increased TRF1 level were observed in PA-treated LO2 cells, whereas these changes caused by PA were eliminated by BGN4 fractions (Figure 2(g)). All these results suggested that PA-induced hepatocyte ferroptosis was ameliorated by Bifidobacterium bifidum BGN4 fractions treatment.
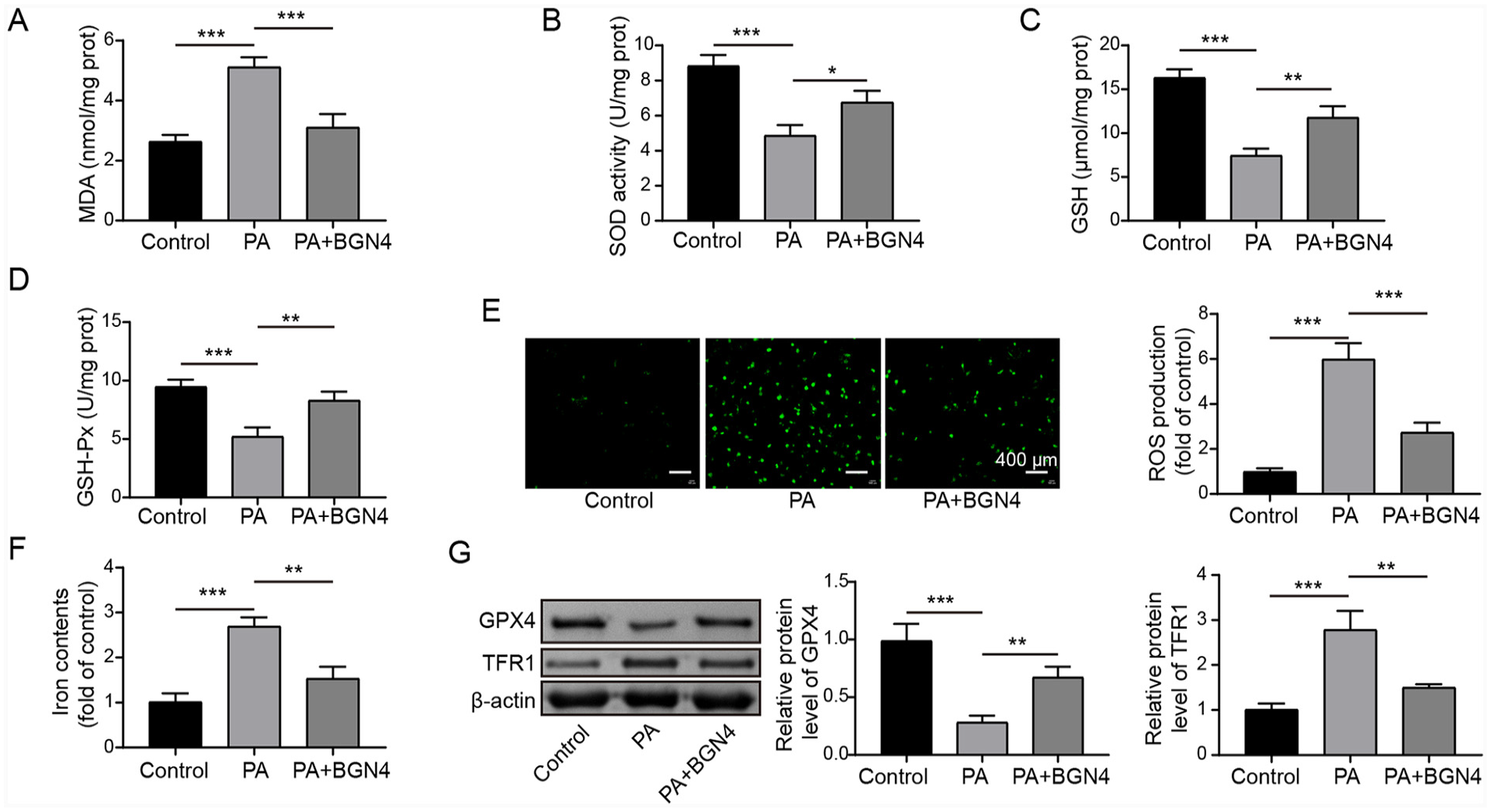
Bifidobacterium bifidum BGN4 fractions suppressed PA-induced hepatocyte ferroptosis. LO2 cells were treated with 0.35 mM PA for 24 h, followed by treated with bacteria fractions of BGN4 for another 24 h. (a–d) MDA and GSH levels, as well as SOD and GPX activities were detected by corresponding assay kits. (e) ROS level was assessed using DCFH-DA staining. (f) Fe2+ level was determined using an iron assay kit. (g) Western blot analysis was performed to evaluate TRF1 and GPX4 levels. The measurement data were presented as mean ± SD. All data were obtained from at least three replicate experiments.
SREBP1 overexpression reversed the protective effect of Bifidobacterium bifidum BGN4 fractions on PA-induced hepatocyte ferroptosis
The transcription factor SREBP1 plays a crucial role in promoting liver lipid accumulation and driving the progression of MAFLD. 18 In this study, it was observed that both the mRNA and protein levels of SREBP1 were significantly increased in LO2 cells upon PA stimulation, indicating an upregulation of SREBP1 expression in response to PA. Notably, treatment with BGN4 fractions resulted in a marked downregulation of SREBP1 expression in PA-induced LO2 cells (Figure 3(a) and (b)). To investigate the involvement of SREBP1 in the regulatory effects of BGN4 fractions in PA-induced LO2 cells, SREBP1 overexpressing LO2 cells were generated by transfecting pcDNA3.1-SREBP1 vectors. As confirmed in Figure 3(c), the expression of SREBP1 was strikingly upregulated when pcDNA3.1-SREBP1 vectors were transfected. Next, MTT assay demonstrated that the protective role of BGN4 fractions on cell proliferation in PA-induced LO2 cells was abolished by SREBP1 overexpression (Figure 3(d)). Additionally, SREBP1 overexpression abrogated the role of BGN4 treatment on the inhibitory roles on MDA level and the promoting roles on GSH level and SOD and GPX activities in PA-induced LO2 cells (Figure 3(e)–(h)). Moreover, SREBP1 overexpression counteracted the inhibitory effects of BGN4 fractions on ROS and Fe2+ levels in PA-induced LO2 cells (Figure 3(i) and (j)). Moreover, SREBP1 overexpression also elevated the protein level of TRF1 but reduced GPX4 protein levels, which greatly abrogated the biological role of BGN4 (Figure 3(k)). Taken together, these findings suggest that the protective role of BGN4 fractions in ameliorating the increase in hepatocyte ferroptosis induced by PA was abrogated by SREBP1 overexpression.
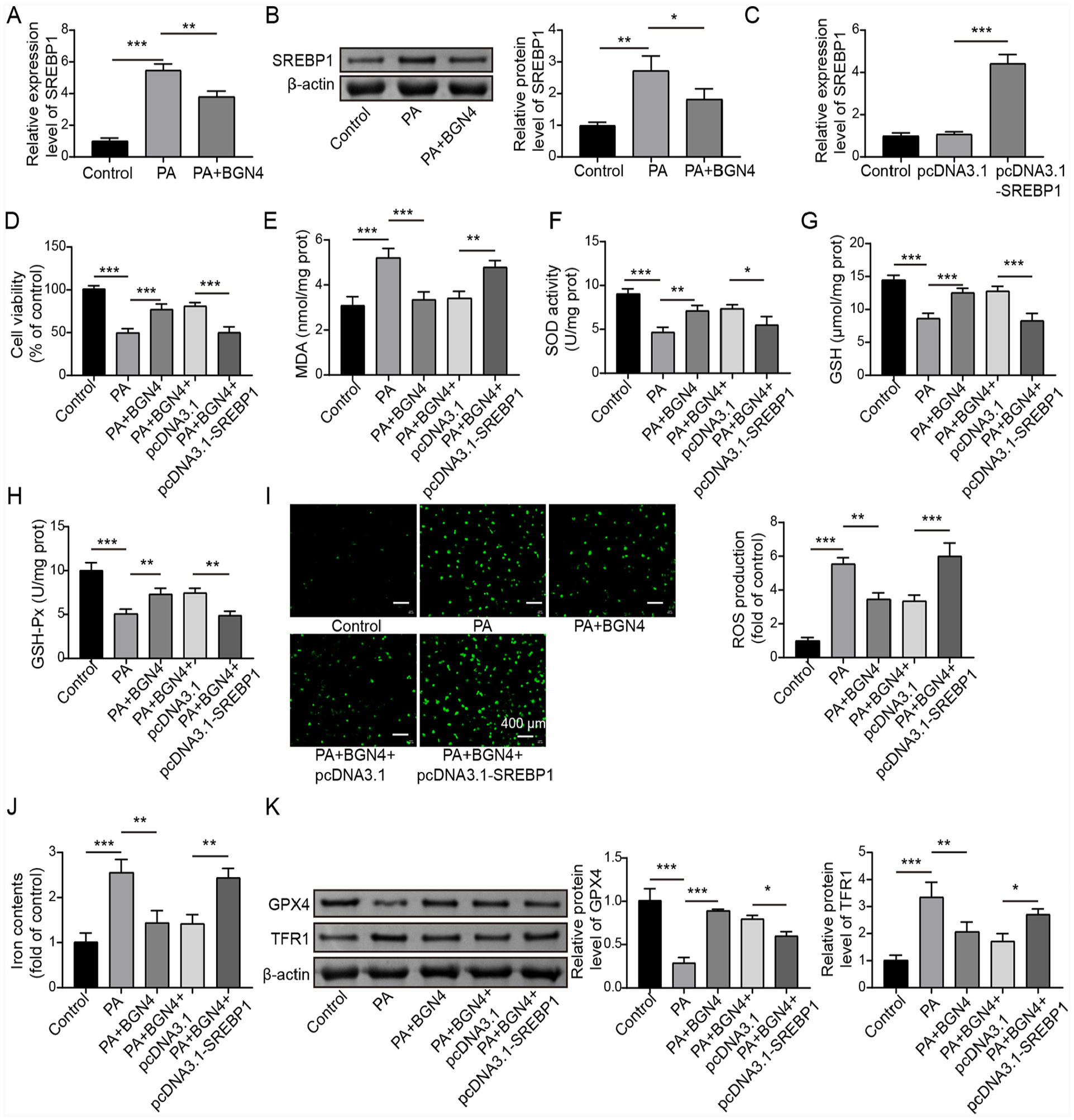
SREBP1 overexpression reversed the inhibitory effect of Bifidobacterium bifidum BGN4 fractions on PA-induced hepatocyte ferroptosis. (a and b) LO2 cells were treated with PA alone or co-treated with PA and BGN4 fractions, and the SREBP1 expression was examined using qRT-PCR and Western blot. (c) SREBP1 expression in LO2 cells transfected with pcDNA3.1 or pcDNA3.1-SREBP1 was determined by qRT-PCR. SREBP1 was overexpressed in PA-treated LO2 cells, followed by co-treatment of PA and BGN4 fractions. (d) LO2 cell viability was evaluated by the MTT assay. (e–h) MDA and GSH levels, as well as SOD and GPX activities, were detected by corresponding kits. (i) ROS level in cells was determined by DCFH-DA staining. (j) Cellular Fe2+ level was assessed by the iron assay kit. (k) TRF1 and GPX4 protein levels were examined by Western blot. The measurement data were presented as mean ± SD. All data were obtained from at least three replicate experiments.
BGN4 fractions inhibited CYP2E1 transcription by inhibiting SREBP1 nuclear accumulation
Previous studies have shown that once SREBP1 is activated, its transcriptional active fragment will be transported from the Golgi body to the nucleus to initiate the expression of downstream genes. 20 Herein, we further investigated the effects of BGN4 fractions on the subcellular distribution of SREBP1. After nuclear and cytoplasm separation, it was observed that PA treatment led to an increase in SREBP1 expression in both the nucleus and cytoplasm, with a more significant increase observed in the nucleus; however, treatment with BGN4 fractions markedly reversed this effect (Figure 4(a)), suggesting that BGN4 inhibited PA-induced SREBP1 nuclear accumulation. CYP2E1, as an important inducer of oxidative free radicals in cells, functions in promoting alcoholic or nonalcoholic mitochondrial dysfunction and liver injury, 21 and it was also involved in ethanol-induced hepatocyte ferroptosis. 22 Using the JASPAR database, we identified four potential binding sites of SREBP1 on the CYP2E1 promoter (designated as site1, site2, site3, and site4) (Figure 4(b)). The region of −1850∼1500 bp upstream of the transcriptional start site (TSS) was used as P1 fragment (including site1, site2, and site3), and the region of −400 to −200 bp upstream of the TSS site was used as P2 fragment (including site4). The result of chromatin immunoprecipitation (ChIP) assay subsequently demonstrated that P1 fragment rather than P2 fragment was significantly enriched in SREBP1 antibody pull-down complex compared with the IgG group (Figure 4(c)). Meanwhile, a dual-luciferase reporter gene assay revealed that SREBP1 overexpression significantly enhanced the luciferase activity of a recombinant luciferase vector containing the CYP2E1 promoter (Figure 4(d)), suggesting that SREBP1 transcriptionally activated CYP2E1. Moreover, SREBP1 overexpression markedly increased CYP2E1 expression in LO2 cells (Figure 4(e)). In addition, it was observed that PA significantly promoted CYP2E1 expression in LO2 cells, whereas BGN4 co-culture significantly reduced CYP2E1 expression, and the inhibitory effect of BGN4 on CYP2E1 expression was reversed by SREBP1 overexpression (Figure 4(f)). These findings indicated that BGN4 fractions inhibited CYP2E1 expression by suppressing SREBP1 nuclear translocation.
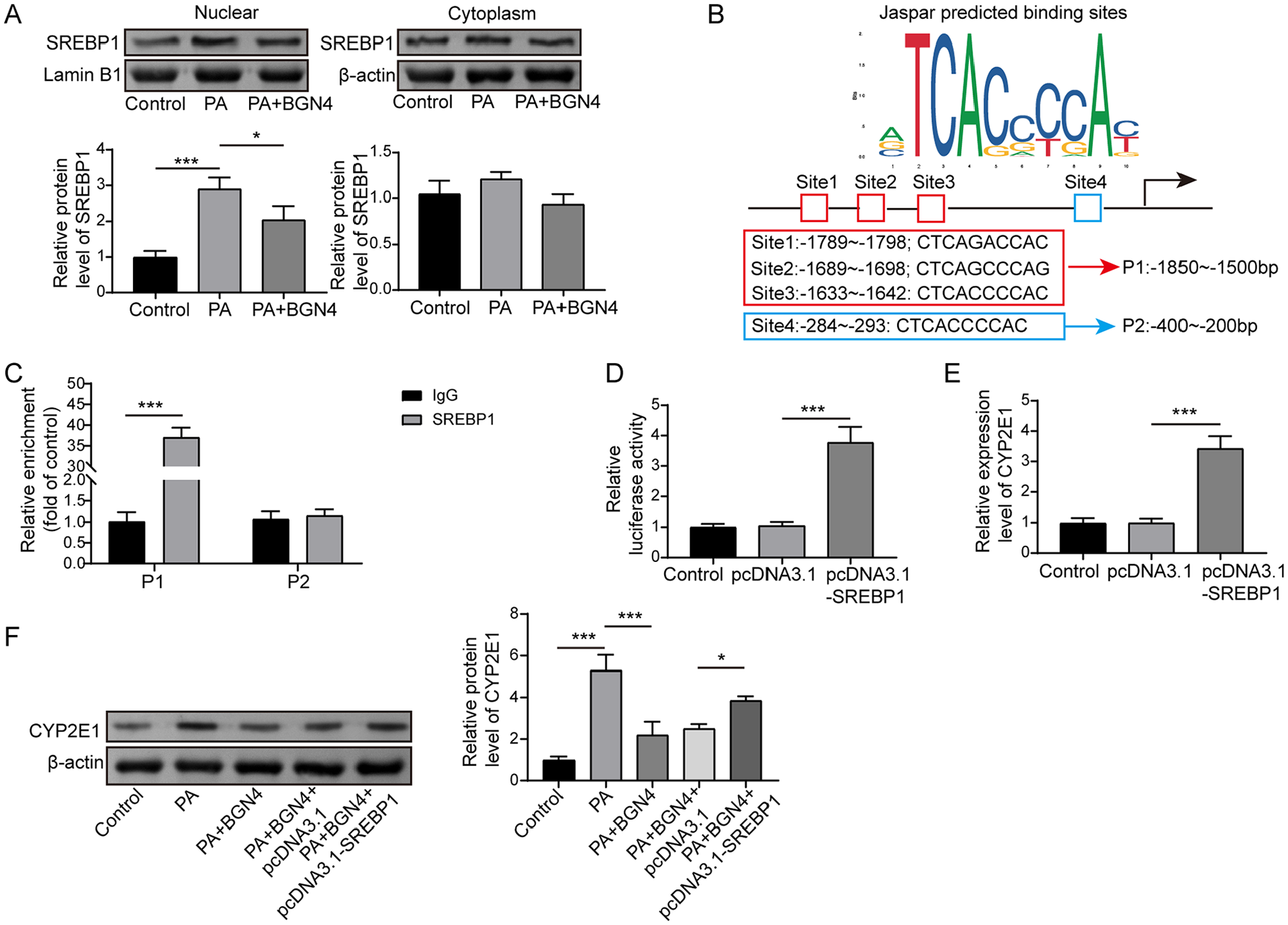
BGN4 fractions inhibited CYP2E1 transcription by inhibiting SREBP1 nuclear translocation. (a) LO2 cells were treated with PA alone or co-treated with PA and BGN4 fractions, and the SREBP1 level in the cytoplasm and nucleus was measured by Western blot. (b) Diagram showing the transcription recognition site of SREBP1 in the promoter region of CYE2P1. (c and d) ChIP and dual-luciferase reporter gene assays were performed to analyze the interaction between SREBP1 and CYP2E1 promoter. (e) CYP2E1 expression in LO2 cells after pcDNA3.1 or pcDNA3.1-SREBP1 transfection was examined by qRT-PCR. (f) SREBP1 was overexpressed in PA-treated LO2 cells and followed by co-treatment of PA and BGN4 fractions, and the protein level of CYP2E1 was measured using Western blot. The measurement data were presented as mean ± SD. All data were obtained from at least three replicate experiments.
CYP2E1 overexpression reversed the inhibitory effect of BGN4 fractions on PA-triggered hepatocyte ferroptosis
We next investigated the role of CYP2E1 in mediating the biological effects of BGN4 fractions in PA-treated hepatocytes. First, pcDNA3.1-CYP2E1 was transfected into LO2 cells and markedly enhanced CYP2E1 expression (Figure 5(a)). The result from MTT assay subsequently revealed that CYP2E1 overexpression abrogated the promoting roles of BGN4 treatment on the proliferation in PA-treated LO2 cells (Figure 5(b)). Similarly, the beneficial effects of BGN4 fractions on SOD and GPX activities, GSH levels, and the inhibitory role on MDA levels in PA-treated LO2 cells were reversed by CYP2E1 overexpression (Figure 5(c)–(f)). Moreover, the inhibitory effects of BGN4 fractions on ROS and Fe2+ levels in PA-treated LO2 cells were diminished by CYP2E1 overexpression (Figure 5(g) and (h)). Western blot analysis revealed that BGN4 fractions significantly reduced TFR1 levels and increased GPX4 levels in PA-treated LO2 cells, but these effects were reversed by CYP2E1 overexpression (Figure 5(i)). In summary, the inhibitory effect of BGN4 fractions on PA-triggered hepatocyte ferroptosis was abolished by CYP2E1 overexpression. These findings indicated that CYP2E1 plays a crucial role in mediating the protective effects of BGN4 fractions in PA-treated hepatocytes.
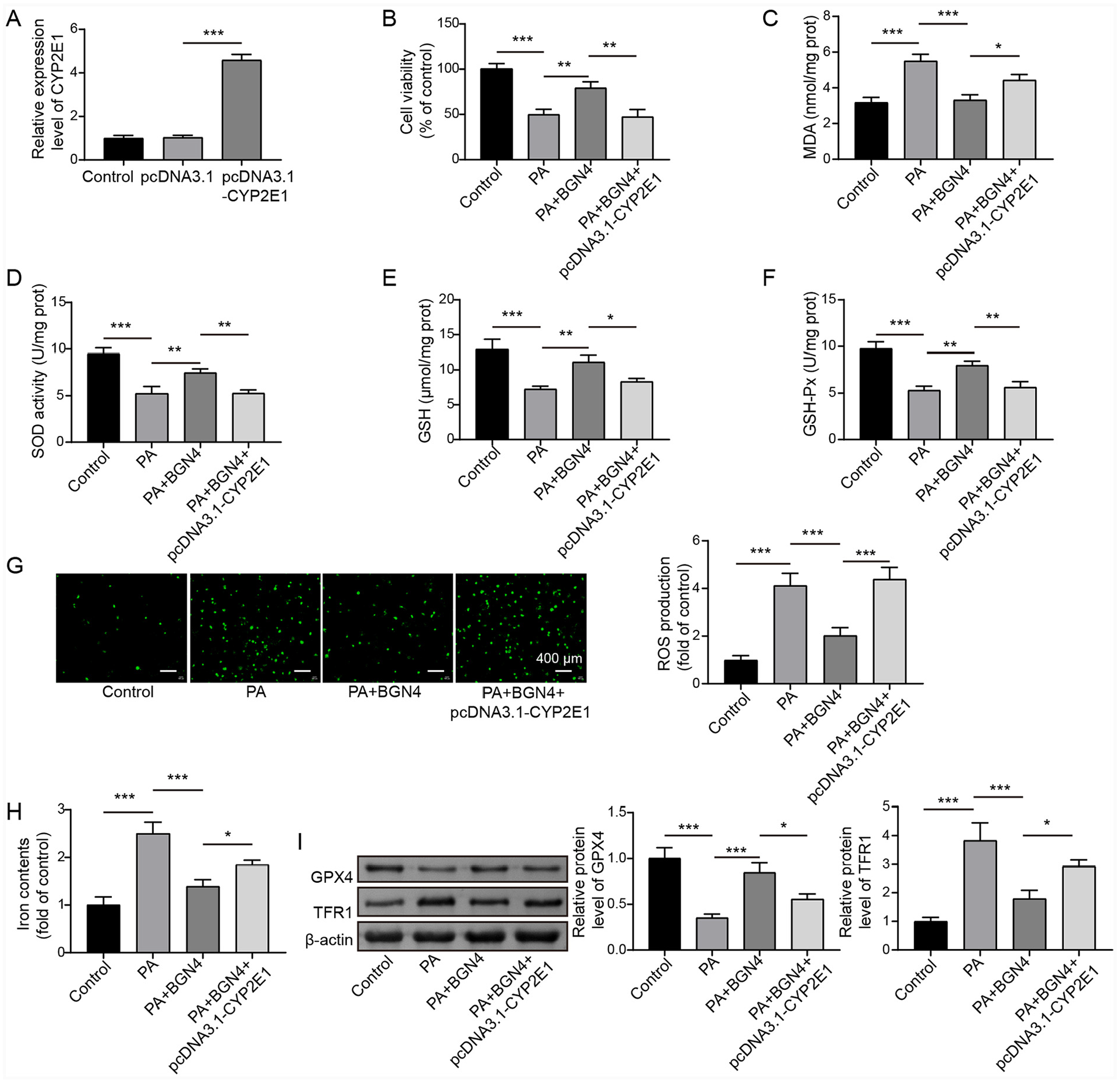
CYP2E1 overexpression reversed the inhibitory effect of BGN4 fractions on PA-triggered hepatocyte ferroptosis. (a) The expression of CYP2E1 in LO2 cells after pcDNA3.1 or pcDNA3.1- CYP2E1 transfection was examined by qRT-PCR. CYP2E1 overexpression was induced in PA-treated LO2 cells combined with BGN4 fractions treatment. (b) LO2 cell viability was analyzed by the MTT assay. (c–f) MDA and GSH levels, as well as SOD and GPX activities, were detected by the corresponding kits. (g) ROS level was assessed using DCFH-DA staining. (h) Cellular Fe2+ level was examined by the iron assay kit. (i) Western blot was conducted to detect protein levels of TRF1 and GPX4. The measurement data were presented as mean ± SD. All data were obtained from at least three replicate experiments.
Discussion
The composition of intestinal microbiota is complex and dynamic. There is at least 1000–1150 kinds of bacteria in human intestinal tract, including probiotics and harmful bacteria, and their composition and distribution will change under different nutritional, immune, and environmental conditions and are associated with the progression of many diseases, including liver diseases.23,24 Much evidence has revealed that NAFLD pathogenesis is closely related to intestinal microflora imbalance.25,26 It has been widely reported that appropriate increase probiotics content and reduce pathogenic bacteria content can change the host of the gut microbe composition and activity, to play a positive role in disease prevention and control, also including MAFLD.27,28 It was observed gut dysbiosis is related to intestinal hyperpermeability and endotoxemia in alcoholic patients, and alcoholics further exhibit reduced numbers of the beneficial Lactobacillus and Bifidobacterium. 29 Moreover, the abundance of various probiotics including Bifidobacterium bifidum is reduced in MAFLD patients. 30 Bifidobacterium bifidum is one of the common microbiotas colonized in human intestinal tract, which has shown a mitigating effect on liver fat accumulation and inflammatory responses during MAFLD progression. 15 In addition, Bifidobacterium bifidum also presents anti-oxidative stress effect in metabolic diseases, such as diabetes. 31 Notably, live probiotics, dead probiotics and their metabolites also convey significant benefits in prevention and treatment of diseases. 12 As Lee et al.’s 13 highlighted, both the live Bifidobacterium bifidum BGN4, heat-killed Bifidobacterium bifidum BGN4 and lysozyme-treated Bifidobacterium bifidum BGN4 have anti-inflammatory properties. 13 In this study, we used lysozyme-treated bacterial fractions of BGN4 to obtain the BGN4 fraction and further investigated its role and mechanism in vitro. The results revealed that Bifidobacterium bifidum BGN4 fractions prevented PA-induced decrease in LO2 cell proliferation and ameliorated PA-induced increase in the apoptosis. It has been widely described that ROS accumulation, GSH depletion, lipid peroxidation, and redox-active iron overload are critical events in the ferroptosis process. 32 Herein, it was observed that BGN4 fractions ameliorated PA-induced increase in MDA, ROS, Fe2+, and TRF1 levels in LO2 cells and prevented PA-induced decrease in GSH and GPX4 levels as well as SOD and GPX activities. All our results suggested that BGN4 fractions could inhibit PA-induced hepatocyte injury, oxidative stress, and ferroptosis.
The mechanism of BGN4 fractions in ameliorating hepatocyte ferroptosis during MAFLD progression was further investigated. SREBP1 is a key transcription factor for genes related to the de novo adipogenesis pathway of the liver and plays an important role in the pathogenesis of MAFLD. 33 As proof, Ziamajidi et al. 34 revealed that SREBP1 expression in HepG2 cells was markedly increased by oleic acid treatment, and its inhibition ameliorated oleic acid-induced steatosis and lipid accumulation in hepatocytes. Notably, it was observed that Bifidobacterium improved MAFLD symptoms by downregulating SREBP1. 19 Herein, our results revealed that SREBP1 level in LO2 cells was markedly increased by PA stimulation, whereas this effect was reversed by BGN4 fractions treatment, suggesting that BGN4 fractions may achieve its biological effects in MAFLD by acting on SREBP1. A previous study displayed that SREBP1 upregulation further exacerbated ovarian granulosa cell oxidative stress in polycystic ovary syndrome, whereas SREBP1 knockdown showed the opposite effect. 35 Nevertheless, the function of SREBP1 in regulating ferroptosis has never been reported before. In the present study, SREBP1 overexpression was induced in PA-treated LO2 cells combined with BGN4 fractions treatment, and it was found that SREBP1 overexpression reversed the inhibitory effect of Bifidobacterium bifidum BGN4 fractions on PA-induced hepatocyte oxidative stress and ferroptosis, which was never reported before. SREBP1 activation is closely related to its intracellular localization; the transcriptional active fragment of SREBP enters the nucleus from the cytoplasm to activate gene expression. 36 In the current research, it was observed that BGN4 inhibited PA-induced SREBP1 nuclear translocation in LO2 cells. All these results suggested that BGN4 fractions ameliorated PA-induced hepatocyte oxidative stress and ferroptosis by inhibiting SREBP1 nuclear translocation.
CYP2E1 is a phase I toxin metabolizing enzyme widely expressed in mammalian liver and extrahepatic tissues, which functions in metabolizing some exogenous and endogenous compounds. 37 CYP2E1 is well-established to be upregulated in pathological conditions related to the alteration of metabolic homeostasis, especially lipid dyshomeostasis, such as MAFLD. 38 Interestingly, compared with other members of the family, CYP2E1 has a very high NADPH oxidase activity, leading to an increase in ROS production 39 and can produce peroxynitrite through interaction with nitric oxide, or under toxic conditions, increase the levels of lipid peroxidation. 40 It was also reported that CYP2E1 was involved in alcohol-driven hepatocyte ferroptosis. 22 However, the role of CYP2E1 in regulating hepatocyte ferroptosis during MAFLD development remains unclear. Herein, our results revealed that SREBP1 transcriptionally activated CYP2E1 by directly binding to the CYP2E1 promoter. In addition, PA significantly promoted CYP2E1 expression in LO2 cells, while BGN4 fractions co-culture significantly reduced CYP2E1 expression, and the inhibitory effect of BGN4 fractions on CYP2E1 expression was reversed by SREBP1 overexpression, suggesting that BGN4 fractions inhibited CYP2E1 expression by repressing SREBP1 nuclear translocation in PA-treated LO2 cell. Moreover, as expected, CYP2E1 overexpression abrogated the inhibitory effect of BGN4 fractions on PA-induced hepatocyte oxidative stress and ferroptosis. The above findings suggested that CYP2E1 was a functional target of BGN4 fractions in protecting LO2 cells from PA-induced oxidative stress and ferroptosis.
Taken together, our research provides evidence for the beneficial effects of Bifidobacterium bifidum BGN4 fractions in alleviating hepatocyte oxidative stress and ferroptosis in PA-induced LO2 cells by repressing SREBP1/CYP2E1 signaling axis. Moreover, our findings contribute to the understanding of potential therapeutic strategies for MAFLD using Bifidobacterium bifidum. Future studies will focus on investigating the effects of live Bifidobacterium bifidum BGN4 and BGN4 metabolites on PA-induced oxidative stress and ferroptosis in hepatocytes, comparing the effects with the outcomes observed in this study. Additionally, we validated our findings in animal models to further consolidate the results obtained thus far. Moreover, we aimed to the impact of Bifidobacterium bifidum BGN4 fraction supplementation on the composition of intestinal microflora in MAFLD animal model.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science Research Project of Education Department of Hunan Province (No. 21C0026).
