Abstract
Affecting an estimated 88 million Americans, prediabetes increases the risk for developing type 2 diabetes mellitus (T2DM), and independently, cardiovascular disease, retinopathy, nephropathy, and neuropathy. Nevertheless, little is known about the use of metformin for diabetes prevention among patients in the Veterans Health Administration, the largest integrated healthcare system in the U.S. This is a retrospective observational cohort study of the proportion of Veterans with incident prediabetes who were prescribed metformin at the Veterans Health Administration from October 2010 to September 2019. Among 1,059,605 Veterans with incident prediabetes, 12,009 (1.1%) were prescribed metformin during an average 3.4 years of observation after diagnosis. Metformin prescribing was marginally higher (1.6%) among those with body mass index (BMI) ≥35 kg/m2, age <60 years, HbA1c≥6.0%, or those with a history of gestational diabetes, all subgroups at a higher risk for progression to T2DM. In a multivariable model, metformin was more likely to be prescribed for those with BMI ≥35 kg/m2 incidence rate ratio [IRR] 2.6 [95% confidence intervals (CI): 2.1–3.3], female sex IRR, 2.4 [95% CI: 1.8–3.3], HbA1c≥6% IRR, 1.93 [95% CI: 1.5–2.4], age <60 years IRR, 1.7 [95% CI: 1.3–2.3], hypertriglyceridemia IRR, 1.5 [95% CI: 1.2–1.9], hypertension IRR, 1.5 [95% CI: 1.1–2.1], Major Depressive Disorder IRR, 1.5 [95% CI: 1.1–2.0], or schizophrenia IRR, 2.1 [95% CI: 1.2–3.8]. Over 20% of Veterans with prediabetes attended a comprehensive structured lifestyle modification clinic or program. Among Veterans with prediabetes, metformin was prescribed to 1.1% overall, a proportion that marginally increased to 1.6% in the subset of individuals at highest risk for progression to T2DM.
Keywords
Little is known about the use of metformin for diabetes prevention among patients in the Veterans Health Administration, the largest integrated healthcare system in the U.S.
An observational cohort study of 1,059,605 Veterans with incident prediabetes revealed that metformin prescription patterns within the Veterans Health Administration are low (1.1%) but comparable to prescription patterns within the general population.
Prescription interventions are not well utilized for patients with prediabetes, suggesting that system-level approaches are necessary to identify patients who would benefit most from pharmaco-prevention.
Introduction
Diabetes mellitus continues to increase in prevalence, affecting an estimated 34.2 million Americans in 2018. 1 The associated total direct and indirect estimated costs of diabetes care were $412.9 billion in 2019. 2 Primary prevention with safe and cost-effective interventions is needed to reduce the incidence of diabetes.
Prediabetes, described as an intermediate state between normoglycemia and type 2 diabetes mellitus (T2DM), is a complex, multifactorial disorder associated with an increased risk for progression to T2DM, and independently, other conditions such as cardiovascular disease, retinopathy, nephropathy, and neuropathy.3–6
As of 2018, the Centers for Disease Control and Prevention (CDC) estimated that 88 million Americans (one in three) have prediabetes, of whom 84% had not been told of their condition by a health professional, despite an increased risk of progression to T2DM and availability of effective interventions. 1
Given the high prevalence of prediabetes, its increased risk for diabetes, and the deleterious health consequences of both conditions, much research on treating prediabetes with lifestyle changes and/or pharmacological agents, including metformin, has been conducted in multiple countries. Several clinical trials produced compelling evidence that both intensive lifestyle intervention (ILI) and metformin reduce risk of progression to diabetes.7–9 The largest diabetes prevention trial worldwide, the Diabetes Prevention Program (DPP), showed metformin to be safe, well-tolerated, and effective, while also reducing progression to diabetes by 31% over approximately 3 years and demonstrating durable clinical benefit over 15 years of follow-up.9–11 Despite such benefit, studies over the past decade revealed that pharmaco-prevention with metformin has been low, generally ranging from <1% to 3.7% of those with prediabetes.12–15 Moreover, no diabetes-prevention treatment has been approved by the U.S. Food and Drug Administration (FDA).
During the past dozen years, diagnostic criteria and treatment guidelines for prediabetes have evolved. Notably, the American Diabetes Association (ADA) added HbA1c in the 5.7%–6.4% range to the criteria for prediabetes, making identification of individuals at higher risk for progression to diabetes more straightforward. 16 The ADA clarified its recommendations to consider metformin for individuals with prediabetes at the highest risk for progression to diabetes, including those with risk factors: body mass index (BMI) ≥35 kg/m2, age <60 years, women with a history of gestational diabetes, or a rising HbA1c despite lifestyle intervention.17,18
Few studies have examined the temporal impact of these diagnostic and treatment guidelines on metformin prescribing for diabetes prevention in real-world clinical settings, especially in U.S. Veterans. Moreover, little is known about the prevalence of prediabetes in Veterans or the metformin prescribing pattern for Veterans with prediabetes. Limited studies in Veterans Affairs (VA) populations reveal that over 55% of Veterans at one VA facility without a history of diabetes had HbA1c levels in the prediabetes range, 19 and that prediabetes is common among Veterans with schizophrenia (42%), and those who are overweight or obese (28%).20,21
To answer these questions, we examined metformin prescribing patterns by the Veterans Health Administration from 2010-2019, using a national database of U.S. Veterans with incident prediabetes and associated clinical and demographic predictors of metformin prescription.
Materials and methods
Study population
The source population included Veterans (n = 2,042,097) who received care at a VA healthcare facility between October 1, 2010 and September 30, 2019, and had confirmed euglycemia in the 2 years prior to the prediabetes diagnosis (index) date. Veterans were included in the prediabetes cohort for this analysis if they had at least one outpatient or inpatient International Classification of Diseases (ICD-9/10) code consistent with prediabetes (790.21, 790.22, R73.01, R73.02, and R73.03), or an HbA1c of 5.7% to 6.4%. Fasting plasma glucose was not used to define prediabetes because it was not possible to reliably determine that the patient had fasted. Furthermore, evidence of normoglycemia was required throughout the two years prior to the index date defined as either an HbA1c <5.7% or a random glucose <100 mg/dL.
Veterans were excluded if they were diagnosed with prediabetes or diabetes based on a prediabetes ICD code or HbA1c laboratory measurement of 5.7% or greater or were prescribed any glucose-lowering medication within the 2 years prior to the start of the study period, or with type 1 diabetes. 22 Individuals were also excluded if their estimated glomerular filtration rate was <45 ml/min/1.73 m2. Figure 1 illustrates the derivation of the study cohort.
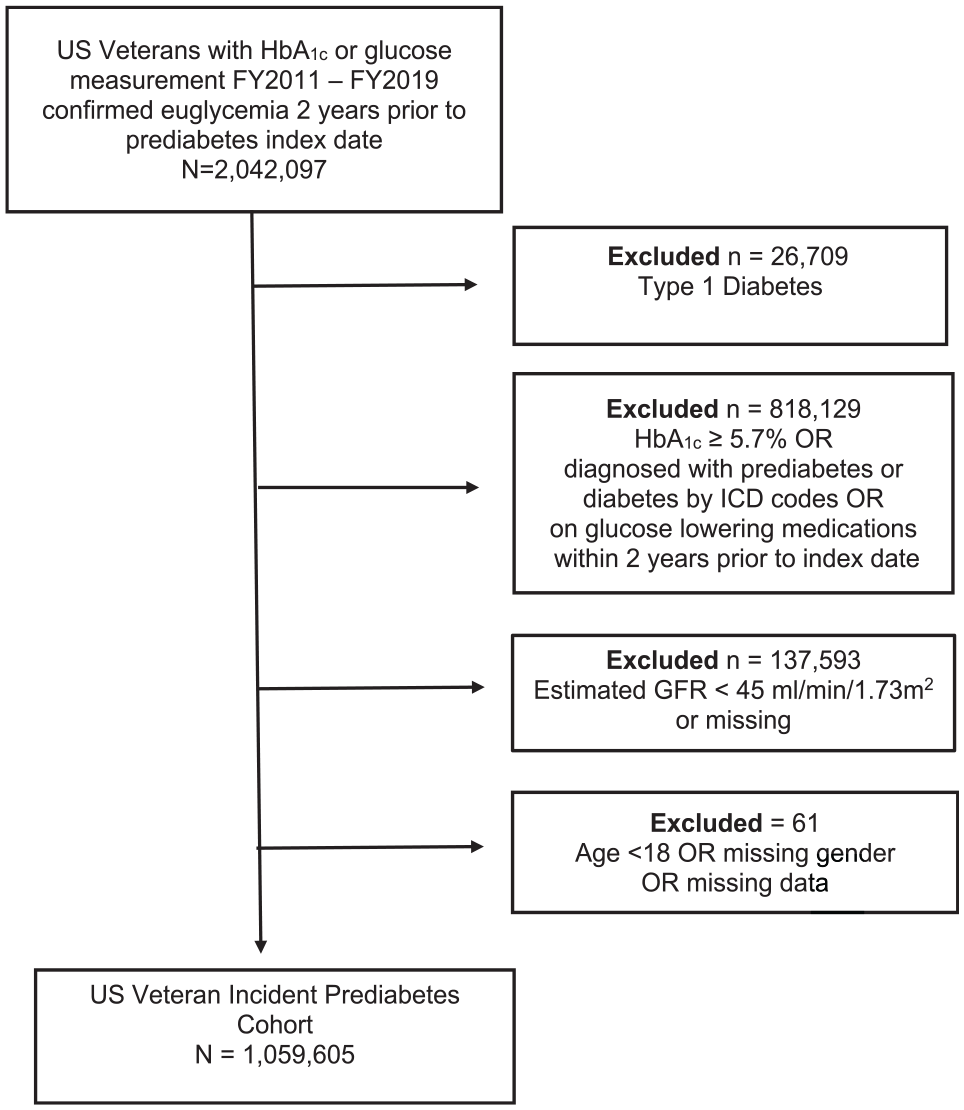
Flowchart of incident prediabetes cohort.
Veterans with prediabetes were included up until October 1, 2018, to allow for at least one full year of observation for metformin prescription. Demographic and clinical data were extracted from the electronic clinical and administrative data sources through the Veterans Health Administration Corporate Data Warehouse. Data outliers were excluded if outside predefined thresholds. This study was approved by the VA Central Institutional Review Board.
Veterans were followed from their prediabetes index date for up to 5 years until any of the following occurred: prescription of metformin, diagnosis of T2DM (based on either ICD code or HbA1c≥6.5%), death (date determined from the VA Beneficiary Identification Records Locator Subsystem), or September 30, 2019, whichever came first.
Measures
Covariates included sociodemographic variables (age, sex, race and/or ethnicity, and marital status), comorbid diagnoses (hypertension, coronary heart disease, stroke, and peripheral vascular disease), body mass index (BMI), blood pressure, and cardiometabolic laboratory variables (HDL-cholesterol, triglycerides, and HbA1c). Diagnosis of serious mental health diagnoses (schizophrenia and major depressive disorder [MDD]), which have a relatively high prevalence among Veterans and may be associated with metformin prescribing were also included. Charlson Comorbidity Index (CCI) conditions and other comorbid diagnoses were required to have ≥ 1 or 2 outpatient ICD-9/10 codes (respectively) or ≥ 1 inpatient ICD-9/10 code in the year prior to and up to 6 months after the prediabetes index date. Similar timeframes were used for all laboratory, blood pressure, and BMI variables. Lifestyle modification included attendance at comprehensive structured programs such as the Nutrition or Dietetics Clinic, participation in the MOVE! Program, a national VA program focused on healthy eating behaviors and increased physical activity, or Diabetes Self-Management Education class (DSME).23,24
Outcome
Our study outcome was receipt of a metformin prescription from a VA facility. Metformin prescription was identified based on the earliest date that metformin was released from the VA Pharmacy for delivery to the Veteran.
Statistical analyses
Descriptive statistics for covariates and metformin prescription were computed using t-tests for continuous variables and chi-square, linear trend, or Fisher exact tests for categorical variables. We used Poisson regression with a log link and robust variance estimates to calculate incidence rate ratios (IRR), and 95% confidence intervals (CI). The model contained an offset equal to the natural logarithm of person-time, calculated as the duration from time of prediabetes diagnosis until the event of interest (receipt of metformin), or conversion to T2DM, death, or the end of the study period for those who did not receive metformin. To control for potential confounding, all demographic and clinical variables (except for polycystic ovary syndrome which occurs only in women) entered into the multivariate model as covariates.
Missing values were addressed by multiple imputations using the R MICE package 25 and the SAS PROC MIANALYSE procedure. 26 Analyses were conducted using SAS 9.4 (SAS, Cary, NC, USA). Study protocol is available upon request.
Results
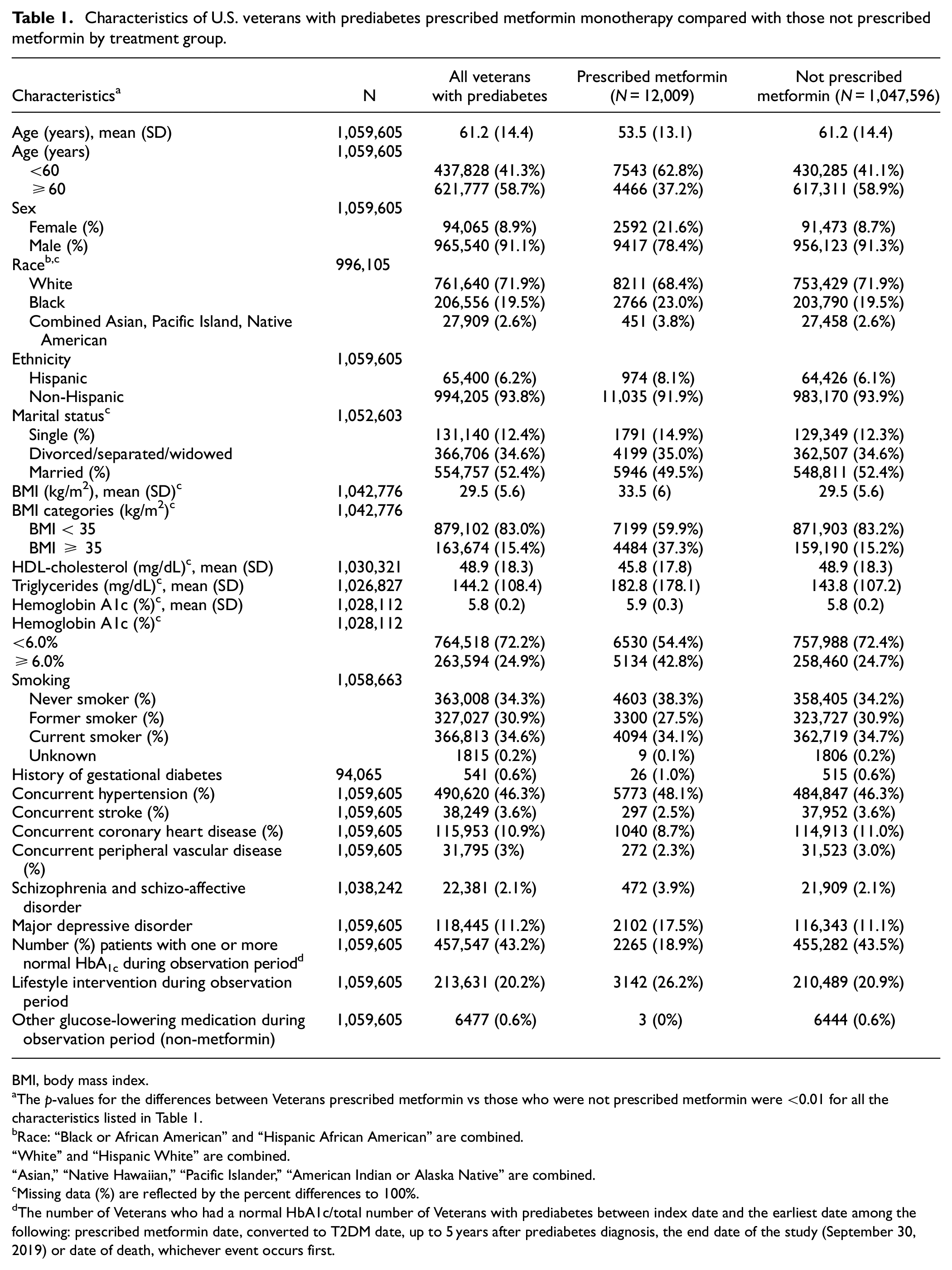
Data were analyzed for 1,059,605 U.S. Veterans diagnosed with incident prediabetes from 2010 to 2018. Virtually all Veterans in this cohort (99.9%) had at least 2 outpatient visits in the baseline period. This study population was 91.1% male, 71.9% White, 19.5% Black, and 6.2% Hispanic (Table 1). Mean age was 61.2 years, mean BMI 29.5 kg/m2, and mean HbA1c 5.8%. During a mean observation period of 3.4 years after diagnosis, metformin was prescribed for 1.1% (12,009) of these Veterans (Table 2).
Characteristics of U.S. veterans with prediabetes prescribed metformin monotherapy compared with those not prescribed metformin by treatment group.
BMI, body mass index.
The p-values for the differences between Veterans prescribed metformin vs those who were not prescribed metformin were <0.01 for all the characteristics listed in Table 1.
Race: “Black or African American” and “Hispanic African American” are combined.
“White” and “Hispanic White” are combined.
“Asian,” “Native Hawaiian,” “Pacific Islander,” “American Indian or Alaska Native” are combined.
Missing data (%) are reflected by the percent differences to 100%.
The number of Veterans who had a normal HbA1c/total number of Veterans with prediabetes between index date and the earliest date among the following: prescribed metformin date, converted to T2DM date, up to 5 years after prediabetes diagnosis, the end date of the study (September 30, 2019) or date of death, whichever event occurs first.
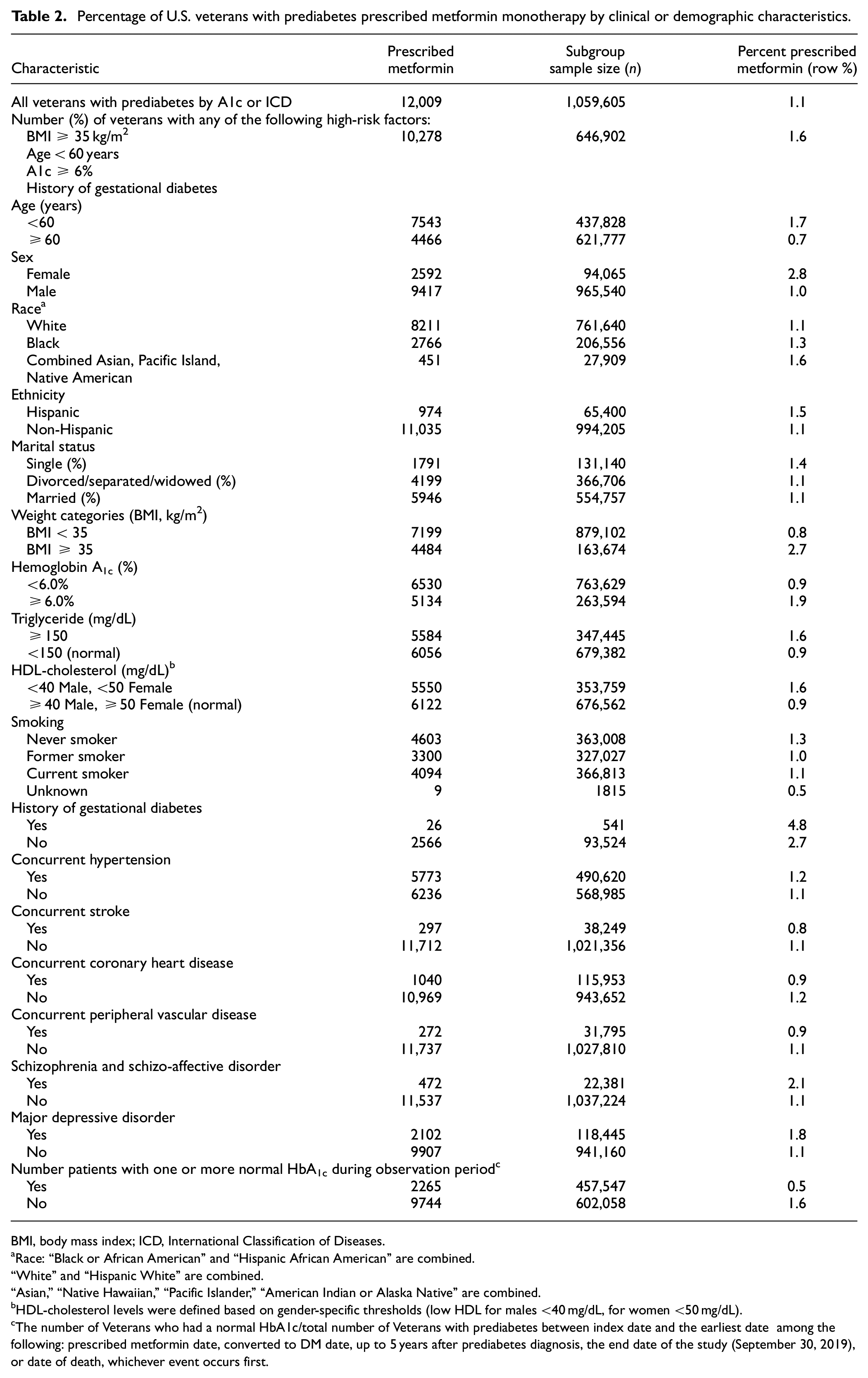
Percentage of U.S. veterans with prediabetes prescribed metformin monotherapy by clinical or demographic characteristics.
BMI, body mass index; ICD, International Classification of Diseases.
Race: “Black or African American” and “Hispanic African American” are combined.
“White” and “Hispanic White” are combined.
“Asian,” “Native Hawaiian,” “Pacific Islander,” “American Indian or Alaska Native” are combined.
HDL-cholesterol levels were defined based on gender-specific thresholds (low HDL for males <40 mg/dL, for women <50 mg/dL).
The number of Veterans who had a normal HbA1c/total number of Veterans with prediabetes between index date and the earliest date among the following: prescribed metformin date, converted to DM date, up to 5 years after prediabetes diagnosis, the end date of the study (September 30, 2019), or date of death, whichever event occurs first.
Overall, 61.1% had at least one additional risk factor for progression to T2DM for which metformin may be considered: BMI ≥35 kg/m2 (15.4%), age <60 years (41.3%), HbA1c 6.0–6.4% (25.9%), and females with a history of gestational diabetes (0.6%) (Tables 1 and 2). However, metformin was initiated in 1.6% of this Veteran subpopulation (Table 2).
Veterans with prediabetes who were prescribed metformin compared to those who were not were younger (mean age 53.5 vs 61.2 years) and had higher BMI (33.5 vs 29.5 kg/m2), as well as other clinical features of metabolic syndrome, including elevated triglycerides and lower HDL-cholesterol; all p-values <0.001 (Table 1).
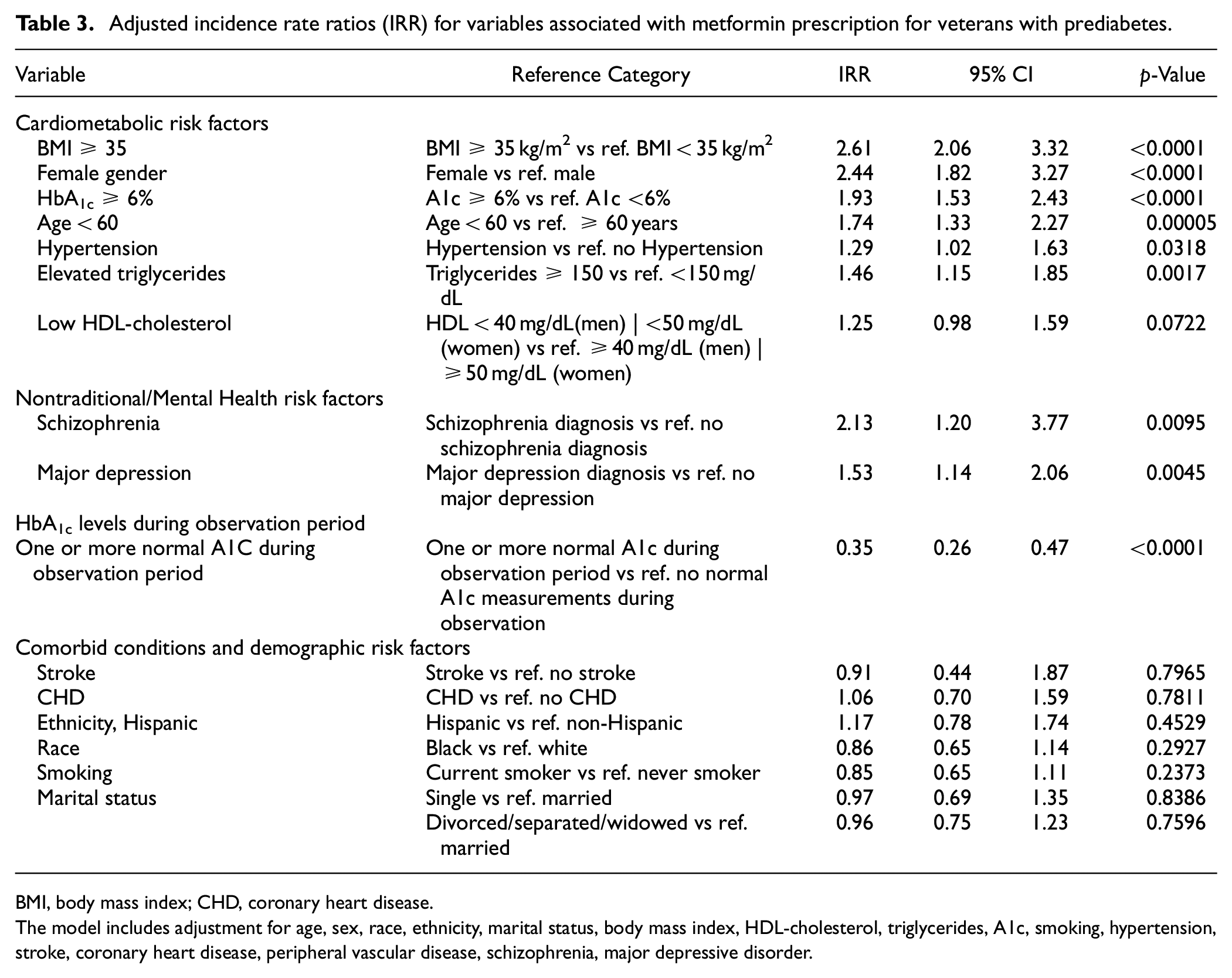
In the multivariate model, Veterans with the following characteristics were significantly more likely than those without to be prescribed metformin: BMI ≥35 kg/m2 IRR, 2.6 [95% CI: 2.1–3.3], female sex IRR, 2.4 [95% CI: 1.8–3.3], HbA1c≥6% IRR, 1.93 [95% CI: 1.5–2.4], age <60 years IRR, 1.7 [95% CI: 1.3–2.3], hypertriglyceridemia IRR, 1.5 [95% CI: 1.2–1.9], hypertension IRR, 1.3 [95% CI: 1.0–1.6], as well as serious mental health illnesses, including schizophrenia IRR, 2.1 [95% CI: 1.2–3.8], and major depressive disorder (MDD) IRR, 1.5 [95% CI: 1.1–2.0] (Table 3).
Adjusted incidence rate ratios (IRR) for variables associated with metformin prescription for veterans with prediabetes.
BMI, body mass index; CHD, coronary heart disease.
The model includes adjustment for age, sex, race, ethnicity, marital status, body mass index, HDL-cholesterol, triglycerides, A1c, smoking, hypertension, stroke, coronary heart disease, peripheral vascular disease, schizophrenia, major depressive disorder.
No associations were found for metformin prescribing with concurrent macrovascular or microvascular disease, low HDL-cholesterol, race/ethnicity (Table 3), or any of the individual CCI components (data not shown).
After the prediabetes index date, HbA1c was measured on average 3.3 times during the mean 3.4-year observation period. Almost all Veterans with prediabetes (>98%) had at least one follow-up HbA1c level measured during the observation period; the majority had between 1 and 5 HbA1c measurements. The large majority of A1c measurements, nearly 80%, were ordered by primary care providers. There were no clinically relevant differences in metformin prescription by geographic region across the U.S. (data not shown). During the follow-up period, a large majority of of HbA1c levels either remained in the prediabetes range or fluctuated between normal and prediabetes, (Supplemental Tables 1 and 2). Veterans with HbA1c levels persistently in the prediabetes range throughout the observation period were 3-fold more likely to be prescribed metformin IRR, 3.02 [95% CI: 2.17–4.2] than those with fluctuating HbA1c levels or no follow-up HbA1c values (data not shown). Based on HbA1c levels and/or ICD codes, 13.4% of Veterans with prediabetes transitioned to T2DM during the observation period (data not shown).
For those prescribed metformin, the median time to dispense a prescription was 476 days (IQR 65-1055) after the index date. A small proportion of Veterans (0.6%) who were not prescribed metformin were treated with an alternative glucose-lowering medication (Table 1).
Over 20% of Veterans with prediabetes attended a comprehensive lifestyle modification clinic or program (e.g., Nutrition or Dietetics clinic, the MOVE! Program, DSME Program) (Table 1).
Discussion
During 2010-2019, among a national sample of 1,059,605 U.S. Veterans with incident prediabetes eligible for diabetes prevention with metformin, 12,009 (1.1%) were prescribed metformin over a mean observation period of 3.4 years after diagnosis. The prescribing of metformin was slightly higher (1.6%) among the subset of Veterans with prediabetes who are most likely to benefit from treatment, namely those at higher risk for T2DM, including BMI ≥35 kg/m2, age <60 years, HbA1c≥6%, or a history of gestational diabetes. Veterans with components of the metabolic syndrome (specifically, hypertension and elevated triglycerides) were also more likely to be prescribed metformin, as were those with MDD or schizophrenia.
Despite strong clinical trial evidence accumulated over the years supporting metformin use to prevent or delay diabetes, metformin prescribing is low in the U.S. Veteran population. Nevertheless, the prescribing frequency is comparable to that in the general U.S. population with prediabetes.12,13,27,28
Evidence from numerous randomized controlled trials worldwide, including the Diabetes Prevention Program (DPP), suggests that intensive lifestyle interventions (ILIs) are more effective than pharmacological intervention, although the latter is not without benefit.9,11,29 However, large-scale ILIs are resource-intensive, expensive, and challenging to implement. For those with prediabetes who are either unable to adhere to or fail to benefit from ILI, pharmacologic treatment can be an effective option. Among the different pharmaceuticals studied for diabetes prevention, metformin has the strongest evidence: it has been shown to be safe, cost-effective, and more affordable than many other classes of antihyperglycemics.30–32 Notably, subgroup analyses demonstrated that ILI and metformin were equally effective for diabetes prevention among those with BMI ≥35 kg/m2 and for women with a history of gestational diabetes.9,33,34
The ADA has recommended that all individuals with prediabetes be referred to a structured lifestyle modification program and metformin therapy be considered for very high risk individuals such as those with a BMI ≥35 kg/m2, age <60 years, those with more severe or progressive hyperglycemia, or history of gestational diabetes. Nevertheless, it is clear from real-world clinical practice data that pharmaco-prevention of diabetes with metformin is low, mostly ranging from <1.0% to 3.7% of those with prediabetes12–14,28,35, and up to 7.4%–8.1% in higher risk populations.28,36 More recent data from the U.S. National Ambulatory Medical Survey found that lifestyle management for prediabetes was addressed in fewer than 25% of clinic visits and metformin was prescribed 2.5% of the time, 15 comparable to the results of our analysis.
Over the years, since most of these studies were published, several factors have facilitated the diagnosis and treatment of prediabetes. In 2010, ADA included the use of HbA1c to identify individuals with prediabetes, making diagnosis more straightforward. 16 In 2016, the FDA relaxed restrictions on metformin prescribing in those with reduced renal function, thus expanding the population eligible for metformin. 37 Our study, which encompasses the timeframe of implementation of these revisions, suggests that the changes in diagnostic criteria for prediabetes and relaxation of renal restriction guidelines have not increased prescription of metformin for diabetes prevention over the past decade.
The reasons for the low rate of metformin use are likely multifactorial. 32 Safety concerns include gastrointestinal intolerance, risk for vitamin B12 deficiency, 38 and lactic acidosis, although a Cochrane review in 2010 noted no evidence that metformin is associated with a higher risk for lactic acidosis than other antihyperglycemic agents. 39
A recent review of diabetes prevention identified other barriers to metformin use including a lack of FDA labeling for metformin to treat prediabetes, which can adversely impact prescribing patterns, reluctancy to assign a label of prediabetes and thus “medicalize” prediabetes, and limited awareness of the screening and laboratory criteria for prediabetes and recommended management guidelines.32,40–42 On one hand, although surveys show that most primary care providers believe that prediabetes is an important healthcare issue for screening and management, only a fraction of eligible patients had laboratory testing or treatment, and many providers cited multiple barriers to care (e.g., time constraints during clinical encounters to address prediabetes screening and management, medication compliance).12,42,43 On the other hand, 25% of survey responders were skeptical of the need for prediabetes screening and the benefit of metformin. 44 Differing opinions about the utility of prediabetes as a clinical construct and the value of treatment may influence provider opinions.41,45,46 Although the DPP showed significant reductions in progression to diabetes with metformin, long-term follow-up has not shown significant improvement in cardiovascular outcomes. 47
Inconsistent recommendations across practice guidelines may also contribute to screening and management uncertainty among primary care providers during the timeline of our study. Although the ADA has recommended screening and treatment for prediabetes since 2007, the U.S. Preventive Services Task Force (USPSTF) did not recommend screening for abnormal glucose until 2015. 48 For those with abnormal glucose levels, USPSTF recommended referral for intensive behavioral counseling for healthful diet and physical activity and cited insufficient evidence for pharmacologic treatment. National professional societies for primary care providers, such as the American College of Physicians, do not have practice guidelines for prediabetes. The present study underscores the dissonance surrounding metformin prescription for prediabetes management, resulting in low utilization.
In recent years, obesity pharmaco-therapeutics such as semaglutide, a glucagon like peptide-1 receptor agonist (GLP-1ra), have shown significant improvement in HbA1c levels among patients with prediabetes and resulting in normoglycemia in a substantial fraction of patients 49 . The expanding use of GLP-1ra agents and recent availability of newer neurometabolic agents such as tirzepatide for obesity management have the potential to further diminish the utilization of metformin for prediabetes management.
To better understand the reasons for low metformin utilization in the Veteran cohort, we examined the distribution of clinical risk factors. In our study population, potential reasons for low metformin utilization may include an early stage of prediabetes (mean HbA1c 5.8%) and a generally older patient population. HbA1c was less than 6% in approximately 72% of the prediabetes cohort at their index date, which may have influenced the decision to defer pharmacotherapy given that metformin is generally considered for individuals with higher HbA1c levels above 6%. Sixty percent of the Veterans were over age 60, a subpopulation that generally benefits more from lifestyle modification than metformin. Altogether these factors may have contributed to the low metformin prescribing for Veterans with prediabetes.
As recommended by clinical guidelines, referral to a structured lifestyle modification program is the initial intervention for prediabetes management. Overall, 20% of Veterans with prediabetes attended a clinic or program for lifestyle modification. However, this proportion may significantly underrepresent the Veteran population that were referred to lifestyle modification but elected not to participate as well as those who received lifestyle modification guidance through informal programs and one-on-one counseling during routine clinical encounters.
Based on the low utilization of prediabetes interventions in real-world practice settings, system-level approaches to diabetes prevention have been advocated, including expanded utilization of nonphysician providers, information technology, and team-based interventions.50,51 Given the magnitude of prediabetes prevalence, effective diabetes prevention requires healthcare system approaches to support providers in their efforts to screen and manage prediabetes, as earlier VA studies suggest.21,52,53 The Prediabetes Informed Decision and Education (PRIDE) Trial, a structured shared decision-making (SDM) intervention facilitated by clinical pharmacists within primary care practices showed improvement in prediabetes awareness and demonstrated significantly increased utilization of lifestyle intervention and metformin. 54
The strengths of our study include a large and nationally representative population of Veterans with incident prediabetes reflecting contemporary metformin prescribing patterns through 2019. To date, this is one of the largest national studies evaluating metformin use for prevention of T2DM in a real-world clinical practice setting and involves a broad range of geographic locations and varied practice settings.
Limitations
Limitations of our study include lack of data on metformin prescriptions filled at non-VA pharmacies and the lack of data regarding the number of referrals made by providers to structured lifestyle programs that Veterans did not attend, to nonstructured prevention programs, or one-on-one lifestyle modification counseling provided during a clinical encounter. Because the population examined in this study are Veterans who are primarily white and male, the study is not generalizable to other populations, including female Veterans, Veterans of color and those who receive their care outside the VA.
Conclusions
Despite almost two decades of strong clinical trial evidence supporting the safety, efficacy, tolerability, and cost-effectiveness of metformin for diabetes prevention, metformin prescription for Veterans with prediabetes is low and comparable to that reported in the private sector. Potential causes for low utilization of metformin include multiple barriers at the provider level, inconsistencies in clinical practice guidelines and expert opinion, a lack of evidence for long-term benefit in cardiovascular outcomes, and clinical factors of our Veteran population with prediabetes including mean HbA1c in the lower end of the prediabetes range and older age for which metformin is less beneficial than lifestyle modification. For higher risk individuals, system-based approaches may expand the identification and treatment of those who have been shown to benefit from pharmaco-prevention.
Footnotes
Acknowledgements
We thank Natalie K. Fullenkamp MS and Patricia Crutchfield for scientific editing and administrative support.
Author’s note
John Ko is now affiliated to The Medicare and Medicaid Analysis Center, Braintree, MA, USA, a field office of the VHA Chief Strategy Office.
Author contributions
Study concept and design: Mihaela Aslan, John Concato, Joseph Goulet, Barbara Gulanski, Kent Heberer, Jennifer Lee, Yuli Li, Julie Lynch, Krishnan Radhakrishnan, Mei-Chiung Shih
Acquisition of data: Jennifer Lee, Julie Lynch, Pradeep Mutalik, Nallakandi Rajeevan
Analysis and interpretation of data: Mihaela Aslan, Joseph Goulet, Barbara Gulanski, Jennifer Lee, Yuli Li, Julie Lynch, Nallakandi Rajeevan, Mei-Chiung Shih
Drafting of the manuscript: Mihaela Aslan, Joseph Goulet, Barbara Gulanski, Krishnan Radhakrishnan
Critical revision of the manuscript for important intellectual content: Mihaela Aslan, John Concato, Joseph Goulet, Barbara Gulanski, Kent Heberer, Jennifer Lee, Julie Lynch, Pradeep Mutalik, Krishnan Radhakrishnan, Mei-Chiung Shih, Elani Streja
Statistical analysis: Mei-Chiung Shih
Administrative, technical, or material support and supervision: Mihaela Aslan, Kei-Hoi Cheung, John Concato, Kent Heberer, Julie Lynch, Jennifer Lee, Pradeep Mutalik
Final approval of the version to be published: Mihaela Aslan, Kei-Hoi Cheung, John Concato, Joseph Goulet, Barbara Gulanski, Jennifer Lee, Julie Lynch, Pradeep Mutalik, Mei-Chiung Shih
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Veterans Affairs (VA) Cooperative Study #2012 from the U.S. Department of Veterans Affairs, Office of Research and Development, Cooperative Studies Program.
