Abstract
Acute lymphoblastic leukemia (ALL) is the second most common acute leukemia in adults with a poor prognosis with relapsed or refractory (R/R) B-cell lineage ALL (B-ALL). Anti-CD19 chimeric antigen receptor (CAR) T-cell therapy has shown excellent response rates in RR B-ALL, but most patients relapse due to poor persistence of CAR T-cell therapy or other tumor-associated escape mechanisms. In addition, anti-CD19 CAR T-cell therapy causes several serious side effects such as cytokine release syndrome and neurotoxicity. In this review, we will discuss novel CAR targets, CAR constructs, and various strategies to boost CARs for the treatment of RR B-ALL. In addition, we discuss a few novel strategies developed to reduce the side effects of CAR.
Keywords
Introduction
Chimeric antigen receptor (CAR) T-cell therapy is transforming cancer care, especially in hematological malignancies. CAR T cells are genetically engineered T cells to recognize and efficiently kill tumor cells with the target antigen. CARs are synthetic receptors with an extracellular antigen recognition domain—single-chain variable fragment (scFv), intracellular co-stimulatory domains (CD28, 41BB, and OX40), and T-cell signaling domains. CD19 antigen is a specific B-cell surface protein that plays a crucial role in the differentiation of naive B cells into pre-B cells, and thus, CD19 can function as a B-lineage marker and a target for patients with acute lymphoblastic leukemia (ALL).
In a landmark phase II clinical trial, Tisagenlecleucel, an anti-CD19 CAR T-cell therapy, reported a remission rate of 81% in patients with relapsed or refractory (R/R) B-cell ALL. 1 Tisagenlecleucel is the only CAR T-cell therapy currently approved for R/R ALL in patients up to 25 years of age. 2 Multiple ongoing studies explore the response of other CAR T-cell therapies in R/R ALL with promising results (Table 1). Despite impressive early response rates, long-term follow-up reports relapse in almost one-third to one-half of the patients after anti-CD19 CAR T-cell therapy. Two distinct types of relapses have been identified, namely CD19-positive relapse and CD19-negative relapse. CD19-positive relapse, in which CD19 surface marker is still present in ALL cells, is associated with limited and poor persistence, decrease in potency, and exhaustion of CAR T cells (2). CD19-positive relapse is influenced by the type of CAR stimulatory domain (41BB vs CD28), source of the scFv (murine vs humanized), age-dependent quality of T cells (child/young adults vs adults), and T-cell phenotype (expression of LAG-3 and secretion of cytokines). In CD-19-negative relapse, there is a loss of CD19 antigen, resulting in tumors eluding CAR-mediated recognition and clearance despite CAR T-cell persistence. 3 Tumor heterogeneity, lineage switching, trogocytosis (T-cell-mediated killing), and cooperative killing are some mechanisms associated with CD19-negative relapse. 4
Results from clinical trials which did not use only CD19 as the therapeutic target.
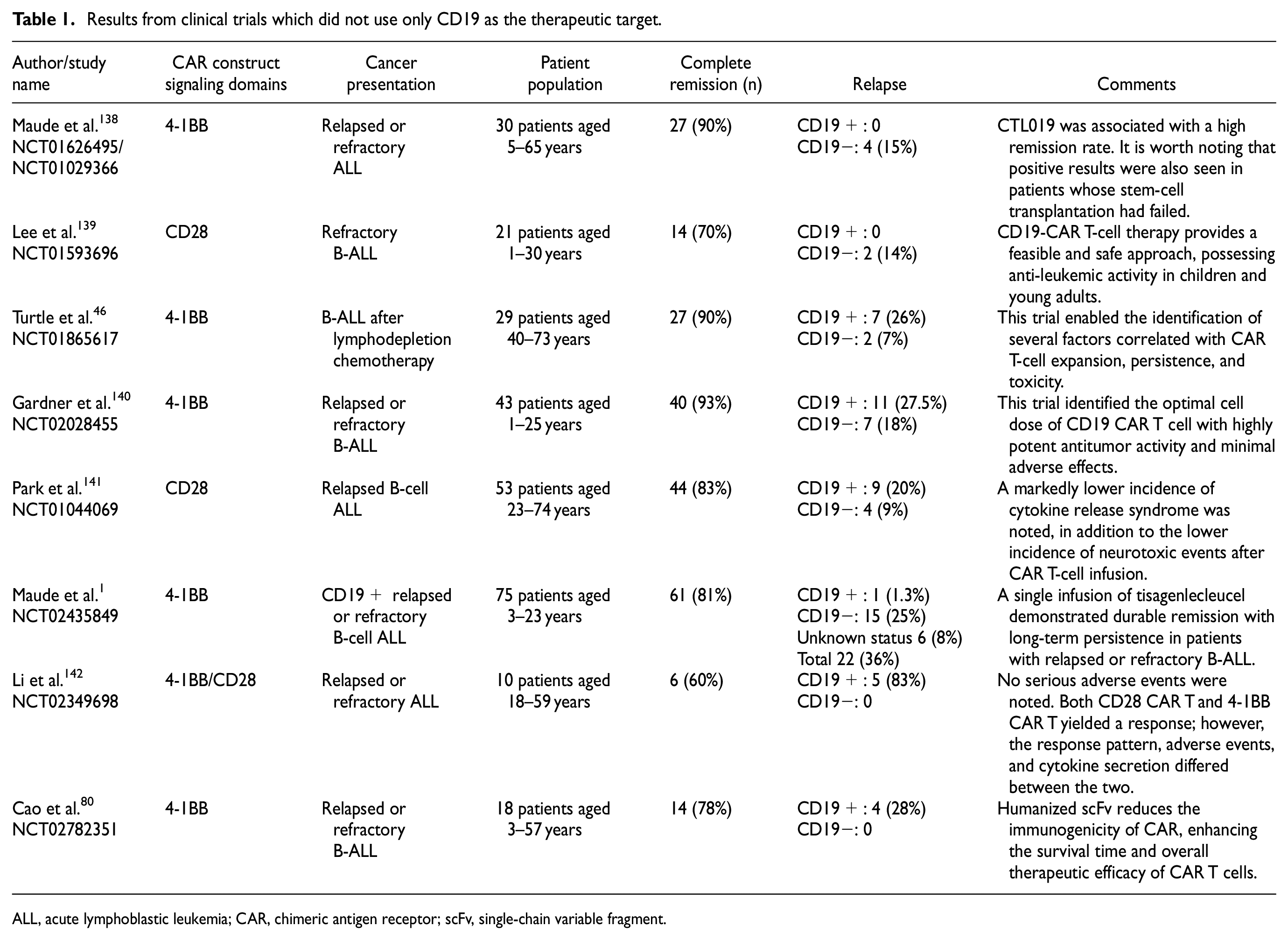
ALL, acute lymphoblastic leukemia; CAR, chimeric antigen receptor; scFv, single-chain variable fragment.
Besides CD19, substantial progress in genomics has resulted in the identification of several other B-cell surface markers such as CD123,5,6 CD22,7–9 BAFF-R,10–12 CD38, 13 CD133,14,15 CSPG4,16,17 CD20,13,18,19 and thymic stromal lymphopoietin receptor (TSLPR)20–23 as targets in the treatment of B-ALL. Table 2 shows selected ongoing clinical trials with CAR T-cell constructs other than CD19. In this review, we discuss the application of novel targets and multiple engineering and clinical strategies to decrease relapse and increase the efficacy of CAR T-cell therapy.
Selected ongoing clinical trials with CAR T-cell constructs other than CD19.
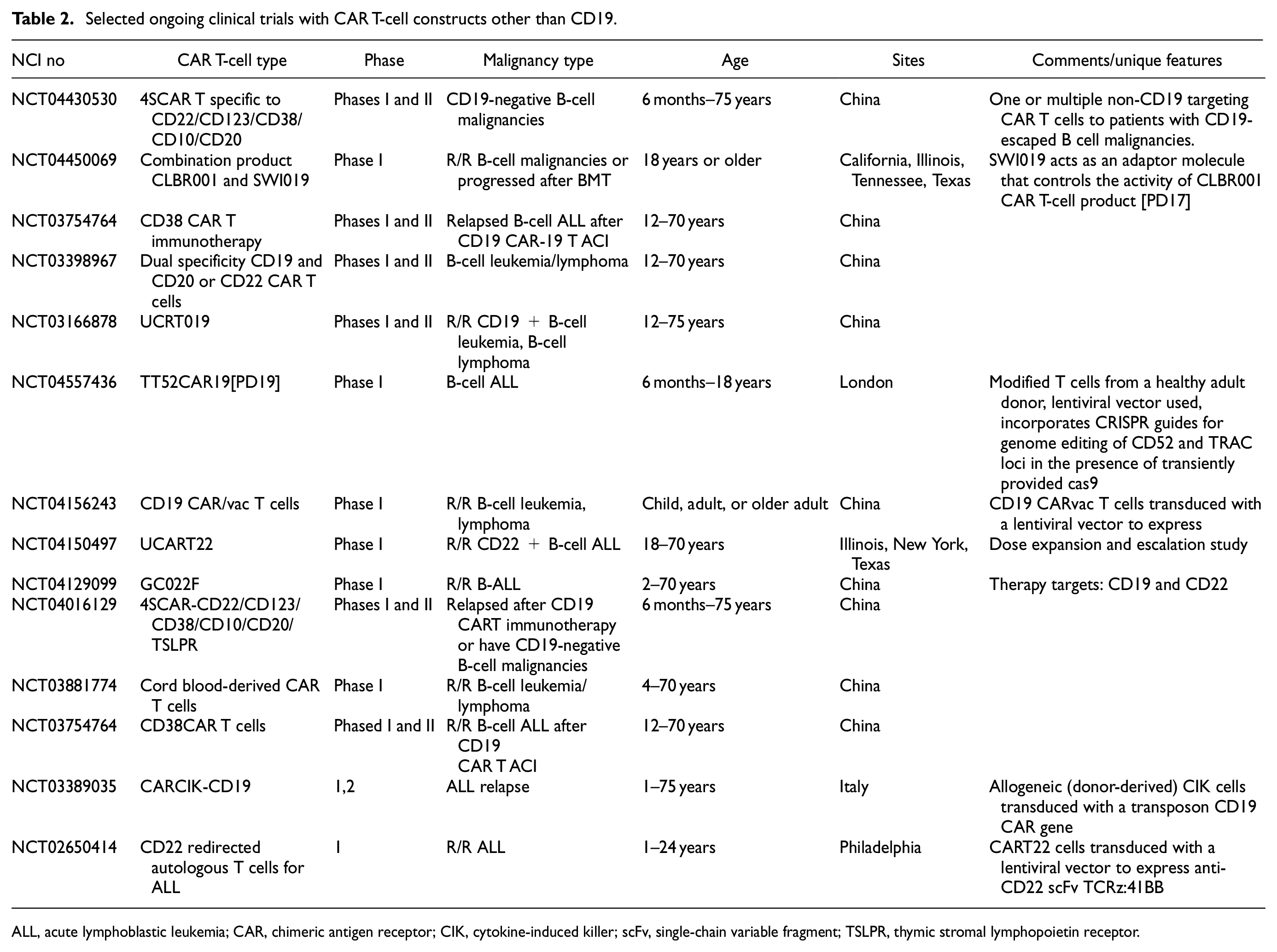
ALL, acute lymphoblastic leukemia; CAR, chimeric antigen receptor; CIK, cytokine-induced killer; scFv, single-chain variable fragment; TSLPR, thymic stromal lymphopoietin receptor.
Novel CAR T-cell antigen targets for B-cell ALL
CD123
IL-3 receptor α chain CD123 is highly expressed in leukemia-initiating cells (LICs) and most B cells; however, CD123 expression in normal hematopoietic stem cells is negligible. 24 Importantly, CD123 is retained in CD19-negative relapse in B-ALL after CAR19 therapy. Ruella et al. identified small subpopulations of CD19–CD123+CD45+(dim) cells in B-ALL patients with LIC-like phenotype (CD34+CD38–) with disease-associated genetic aberration, suggesting that targeting CD19 alone could lead to a subclonal relapse derived from preexisting CD19–CD123+cells. They found that combining anti-CD-123 CAR T cell and anti-CD19 CAR T cell prevented antigen-loss relapses and increased in vivo activity against tumors compared to single-expressing CAR T-cell constructs. In fact, dual-stimulated anti-CD19 and anti-CD123 CAR T-cells exhibited both short-term and long-term higher efficacy against the B-ALL cell lines than an equal number of single-stimulated anti-CD19 CAR T cells or single-stimulated anti-CD123 CAR T-cells or pooled anti-CD19 CAR T cells, and anti-CD123 CAR T cells. These findings provide the groundwork for further studies regarding multitargeted T cells, specifically anti-CD19 and anti-CD123 on leukemic blasts, as an effective strategy for preventing and treating antigen-negative relapses occurring after CD19-directed therapies. 6
CD22
CD22 is a lectin found on the surface of mature B cells and at lower levels on a few immature B cells, which functions as an inhibitor for B-cell receptor (BCR) signaling. CD22 expression is seen in 90% of B-cell ALL patients. Qin et al. investigated a combined anti-CD19 and anti-CD22 CAR construct; T cells expressing both CD19 and CD22 CAR eradicated ALL cell line xenografts and patient-derived xenografts (PDXs) with a CD19-negative relapse. 25 Pan et al. studied anti-CD22 CAR T-cell therapy in RR ALL patients who failed prior anti-CD19 CAR T-cell therapy with a CR rate of 71%. 8 A phase I trial using a bispecific CAR T cell that targets both CD19 and CD22 in RR B-ALL found it safe and achieved minimal residual disease (MRD) negative in all six enrolled patients. 26 Shah et al. tested 58 children and young adults with CD22+R/R B-ALL using anti-CD22 CAR T cells and found a CR rate of 70%, among the ones who achieved CR, the median relapse-free survival was 6.0 months. 27 A recent study sequentially combined CD19 and CD22 CAR T-cell therapy (1–6 months apart) in R/R B-ALL after allo-HSCT and achieved CR in 66% of cases. 28 A phase I clinical trial of UCART22 in RR CD22+ B-cell ALL is ongoing (NCT04150497).
BAFF-R
B-cell activating factor receptor (BAFF-R) is crucial for co-stimulation of T cells, survival of mature B-cell, 29 and growth of malignant B cells. Normal pre-B cells generally do not express BAFF-R but BAFF-R is universally expressed in pre-B-ALL cells, opening the possibility of a therapeutic target.10,30 In 2017, Fazio et al. reported that BAFF-R-positive blasts persist after early treatment and relapse in pediatric B-ALL. In 2018, their team headed by Turazzi et al. engineered CAR T cells (INVsh.BAFFR. CAR) toward BAFF-R, also known as TNFRSF13C. 11 Turazzi et al. proposed that BAFF-R targeting with anti-CD19 CAR T cell can avoid antigen escape and be used as a second-line treatment in CD19-negative relapse cases. 11 Qin et al. also found retention of BAFF-R in patient samples with CD19-negative relapse and reported a significant antitumor activity of anti-BAFF-R CAR T cell against CD19-negative PDX in vivo. 12 Recently, Dong et al. performed validation of the potency and efficacy of BAFF-R CAR T cells in xenograft mouse models. 31 A phase I clinical trial evaluating BAFFR-targeting CAR T cells in R/R ALL is ongoing (NCT04690595).
CD38
CD38 is a cyclic ADP ribose hydrolase, adhesive type II transmembrane glycoprotein 32 with functions of cell adhesion, signal transduction, and calcium signaling. 33 CD38 is expressed on B lymphocytes, T lymphocytes, monocytes, and natural killer (NK) cells. 34 Researchers have suggested CD38 as one of the leukemia-associated immunophenotype markers to detect MRD and a potential target in B-ALL. 35 CD38 CAR T cells are known to reduce the tumor burden in the bone marrow and blood after bispecific CD19/CD22 CAR T-cell failure; however, cytokine release syndrome has been reported. 36 Also, the CD38-based CAR T cells are at risk of apoptosis or T-cell-mediated killing which may be avoided by blocking the CD38 antigen with specific antibodies or proteins. 37 A phase I/II study is ongoing to evaluate the treatment of relapsed B-ALL after CD19 CAR T-cell therapy with anti-CD38 CAR T cells (NCT04150497).
CSPG4
Chondroitin sulfate proteoglycan 4 (CSPG4) is also known as neuron-glial antigen-2 (NG2), a heavily glycosylated transmembrane protein that is overexpressed in variable proportions by tumor cells in certain malignancies like malignant melanoma,38,39 triple-negative breast cancer, 40 and gliomas. 41 CSPG4 also expresses on the surface of MLL-rearranged leukemias, including ALL.42,43 MLL rearrangement is characterized by chromosome 11 translocation, confers resistance to standard chemotherapy regimens, and is associated with a poor prognosis. 44 MLL-rearranged ALL patients undergoing CD19 CAR T-cell therapy are at high risk of lineage switching to AML, which results in relapse.45,46 The expression of CSPG4 is not affected by lineage switching since it is detected in both MLL-rearranged ALL and myeloid lineage-switched ALL cells (AML). Harrer et al. studied anti-CSPG4-CAR T cells in CSPG4-positive MLL leukemias using MLL1-MLLT1-translocated KOPN8 B-ALL cells as target cells and found anti-CSPG4-CAR T cells to be effective in secreting cytokines and eradicating targeted cells. 47 Further studies are warranted to further validate the therapeutic potential of anti-CSPG4-CAR T cells in CSPG4-positive MLL leukemias.
TSLPR
TSLPR, a heterodimeric receptor complex, is composed of a TSLPR subunit and a CD127 subunit. Thymic stromal lymphopoietin (TSLP) cytokine binds to the TSLPR to activate the JAK/STAT signaling pathway. TSLPR is associated with cytokine signaling, including IL-7 receptor pathways, particularly CD127 (encoded by IL7RA). 48 CRLF2 translocations or deletions result in the utilization of alternative promoters leading to overexpression of TSLPR and occur in approximately 5%– 15% of children and adults with B-ALL.49–54 Activating the TSLPR signaling pathway is biologically necessary for ALL blasts.55,56 B-ALL with CRLF2 rearrangements is frequently associated with a gene expression profile similar to Philadelphia chromosome-positive ALL without the BCR-ABL1 fusion. 49 B-ALL-bearing CRLF2 rearrangements and TSLPR overexpression have poor relapse-free and overall survival rates in pediatric and adult ALL.57–63 TSLPR expression in normal tissues appears to be restricted. TSLPR CAR was capable of completely eradicating human CRLF2-rearranged (TSLPR overexpressing) ALL in multiple model systems, including PDXs). 21 TSLPR CAR constructs demonstrated activity in vitro when incubated with the B-ALL cell line (REH transduced to express TSLPR) and TSLPR overexpressing CRLF2-rearranged ALL line (MUTZ5). 21 More recently, Ross et al. demonstrated potency in vitro and in vivo functionality of novel bivalent CD19-TSLPR CAR T cell and CD22-TSLPR CAR T cell. 64 TSLPR alone or in combination with other antigens can be an effective immunotherapeutic strategy for CRLF2-rearranged B-ALL. Ongoing studies are evaluating bicistronic CD19 TSLPR CAR T-cell constructs. 64
Novel engineering strategies
Multitargeted CAR T cells
The role of multitargeted CAR T-cells is to provide combinatorial antigen targeting that simultaneously expresses two or more CAR molecules within the same T-cell population. Multiple studies have demonstrated the efficacy of this method in circumventing lineage switching, in addition to CD19 antigen loss resulting from trogocytosis and synergy.6,65
Dual-signaling CAR T cells
Dual-signaling CAR T cells incorporate two different scFvs in tandem on one T cell. Dual-signaling CAR T cells recognize and kill the CD19-negative malignant cells by aiming for alternative targets such as CD20, CD22, or CD123. Dual-targeted CAR constructs induce a higher amount of interferon gamma (IFN-γ) and interleukin (IL)-2 in vitro, paired with the eradication of PDXs produced in antigen-negative relapse following CD19-directed CAR T-cell therapy (ref). Gardner et al. designed a novel CAR construct with dual specificity, specifically targeting CD19 and CD22 simultaneously, and investigated the incidence of antigen escape and durability of remission following administration. Preclinical testing revealed a preference for signaling through CD22 over CD19; however, dual-transduced T cells signaled through both CD19 and CD22. Also, the lytic activity and cytokine production are similar to single transduced CAR T cells, demonstrating the same specificity. With these findings, a dual-signaling CAR T cell is currently being studied in phase I clinical trials in patients with CD19/CD22 relapsed or refractory B-ALL. 66 A phase I study evaluated the efficacy of AUTO3, a CD19/CD22 simultaneously targeted CAR T-cell therapy, in reducing the incidence of relapse as a result of antigen loss. The objective response rate was 75% at 1 month, suggesting that those treated at a higher dose of AUTO3 presented with superior outcomes, with an increased incidence of MRD-negative complete remission. Moreover, an enhanced CAR T-cell expansion occurred in those receiving a higher dose of AUTO3. 67 A phase I/II clinical trial investigates the feasibility of Universal dual specificity CD19 and CD20 or CD22 CAR T-cell immunotherapy in RR leukemia or lymphoma (NCT03398967).
Tandem CAR T cells
Tandem CAR T-cell (TanCAR) construct is a single CAR connected in tandem with two distinct antigen-binding domains using a single vector. 68 A preclinical Glioblastoma model created TanCAR by joining HER2-binding scFv and an IL13Rα2-binding IL-13 mutein to create a CAR exodomain to CD28.ζ endodomain. 68 TanCAR showed increased persistence and lowered exhaustion compared to Dual CAR T cells that co-expressed a HER2 CAR and an IL13Rα2 CAR, single antigen-specific CAR T cell, or pooled CAR T cells. 68 TanCAR overcame antigen escape and prolonged animal survival due to its enhanced antitumor efficacy. 68 Although not tested yet, TanCAR can be an effective strategy for RR B-ALL.
Trivalent CAR T cells
Trivalent CAR (TriCAR) constructs express three CARs individually, and a Side CAR T-cell construct expresses a mono-CAR and a Tandem CAR or vice versa. 19 Fousek et al. used both TriCAR and SideCAR (single CAR targeting CD19 and a second bispecific CAR targeting CD20 and CD22 via a tandem arrangement) to target primary B-ALL cells. The results showed that trivalent CAR killed ALL cells more robustly at low E:T ratios than CD19 CAR T cells alone. 19 ImageStream analysis of single-cell interactions between CAR T-cells and primary B-ALL cells showed increased actin polymerization of TriCAR T cells compared to CD19 CAR T cells, suggesting remodeling and increased cell activation. Trivalent CAR T cells also produced IFN-γ/TNF-α and killed CD19-negative primary ALL, thus mitigating CD19-negative relapse. This strategy can be used as an initial CAR therapy in RR ALL or a salvage therapy for patients with CD19-negative disease. 19 A phase 1 clinical trial named “Trivalent Autologous T-Lymphocytes Co-Expressing Three Chimeric Antigen Receptors Targeting CD19, CD20 and CD22 in Acute B-lineage Leukemia (TriCAR)” plans to start recruiting on October 1, 2021 (NCT05010564).
Universal CAR T cells
Universal CAR T-cell (UniCAR) therapy uses allogeneic T cells, while current CAR T-cell constructs employ autologous T cells. Case-by-case basis production of CAR T cells using autologous T cells has significantly limited the large-scale clinical applications of CAR T cells due to the lack of cost efficiency and the significant production process. Importantly, CAR T-cell production takes several days, and patients have to wait. UniCAR does not need patients’ T cells; thus, it can be used as an “off-the-shelf” product and produced on a large scale. Besides the wide-scale applications, UniCAR T cells can target multiple antigens and be delivered to several different recipients without re-editing the T cells. Unlike conventional CARs, the scFv in universal CARs recognizes a short non-immunogenic peptide (5B9 tag-a motif of 10 amino acids) derived from the human nuclear autoantigen La/SS-B and not a cell surface antigen. 69 The UniCAR system consists of a CAR for an inert manipulation of T cells and specific targeting modules (TMs) for redirecting UniCAR T cells in an individualized time and target-dependent manner. UniCAR T cells remain inactive after reinfusion, as the UniCAR target is not available on the surface of intact cells under physiological conditions. 70 TM is the component that provides ultimate antigen specificity since it is a binding domain directed against a tumor antigen (e.g., a tumor-specific scFv) fused to the 5B9 tag recognized by the UniCAR scFv. TMs are interchangeable, thus adding high flexibility to the system. 70 UniCAR T cells can target various tumor antigens simply by the replacement of the respective TM. Thus, we can target more than one antigen simultaneously or subsequently enhance efficacy and lower the risk of developing antigen-loss tumor variants under treatment. Allogeneic universal CAR T cells produced cytogenetic and molecular remissions in two infants with highly relapsed refractory CD19+B-ALL; they both later successfully received allogeneic stem cell transplantation. 71 In the AML bone marrow xenograft (NOD/SCID IL2Rγ−/−) mouse model, Cartellieri et al. provided proof of concept that retargeting UniCAR T cells to CD33 and/or CD123-specific TMs can eliminate AML cells. 72 Loff et al. demonstrated a preclinical proof of concept for the safety switch mechanism using a hematotoxicity mouse model wherein TM123-redirected UniCAR T showed reversible toxicity toward hematopoietic cells compared to CD123 CAR T cell. 73 In a phase Ia trial in RR AML, the first three patients who completed UniCAR CD123 showed that UniCAR CD123 was well tolerated and was efficacious even at the lowest dose levels. 74 An ongoing phase I/II clinical trial evaluates UCART019, a universal CAR construct, in patients with R/R CD19-positive leukemia andlymphoma (NCT03166878).
Armored CAR T cells
Armored CAR T cells are enhanced CAR T cells programmed to secrete various proteins such as cytokines with synergistic activity to augment CAR T-cell expansion, persistence, infiltrative capacity, and function in the immunosuppressive tumor microenvironment (TME). These armored CAR T cells have proven effective, especially in a hostile TME, utilizing different mechanisms. 75 The most common cytokines required for T-cell function and trafficking are interleukins, namely IL-7, IL-12, IL-15, IL-18, IL-21, and IL-23.
Interleukin armored CAR T cells: Gene-edited interleukin CAR T cells constructs can be engineered to co-express a single interleukin, two interleukins, an interleukin combined with other cytokines, interleukin receptors, interleukin subunits, and fusion inverted cytokine receptors. 76 Singh et al. reported a second extrinsic approach to numerically expand CAR T cells with the desired phenotype through alteration of the culture milieu by IL-21 showed an improvement in CAR-dependent T-cell effector functions. 77 The findings demonstrate a preferential numeric expansion of CD19-specific T cells in the presence of IL-21. 77 The collective findings of this study reveal that IL-21 can provide an extrinsic reprogramming signal to generate desired CAR T cells for the advancement of effective immunotherapies.77,78
CAR T with improved structure
CAR has continually evolved since its introduction as first-generation (1G) CARs which consisted only of the CD3ζ signaling domain. Subsequently, researchers developed 2G and 3G CARs with one or more co-stimulatory domains. Scientists recently introduced 4G and 5G CAR structures with more sophisticated regulatory capabilities and increased efficacy. 79
Single-chain variable fragment and transmembrane domain
Improving CAR structure regarding the scFv and transmembrane domain forms the first laboratory strategies for preventing and treating antigen-positive relapse. Cao et al. developed a novel humanized scFv for the murine FMC63 antibody. 80 This study evaluated the outcomes of humanized CD19-targeted T cells in 18 relapsed/refractory ALL patients who presented with or without prior murine CD19 CAR T therapy. A total of 14 patients had not received previous CAR T therapy, with the results revealing that 92.9% of this group achieved complete remission or complete remission with incomplete count recovery on day 30. On the other hand, one of the three patients who failed a second murine CAR T infusion reached complete remission after hCART19s infusion. 80 The primary outcomes measured here were the overall and leukemia-free survival rates, recorded at 65.8% and 71.4%, respectively, and the cumulative incidence of relapse, recorded at 22.6%. The most prevalent adverse outcome noted was grade 1-2 cytokine release syndrome, presenting in 13 participants. The findings of this study provide evidence that hCART19s, an improved CAR structure, has the potential to induce remission in patients with relapsed/refractory ALL, specifically in those who received a reinfusion of murine CAR T. 80
Intracellular costimulatory domain
The adoption of improved CAR structures in the prevention and treatment of CD19-positive relapse was reinforced by Guedan et al. who investigated the hypothesis that CD4+and CD8+T cells can exhibit distinct persistence and effector phenotypes depending on the identity of certain intracellular signaling domains used to generate the CAR construct. The findings of this study suggest that the persistence of CD8+T cells is predominantly dependent on a helper effect provided by the intracellular signaling domains used to redirect CD4+T cells. Moreover, combining the ICOS and 4-1BB intracellular signaling domains in a third-generation CAR construct yielded significantly superior antitumor effects and increased persistence in vivo. The evidence highlights that combining intracellular signaling domains in CAR constructs, as a method of improvement, may yield superior outcomes in solid tumor models compared to current 4-1BB-based CAR constructs and should be investigated further. 81 Gue et al. generated a new CD19 CAR T (HI19α-4-1BB-ζ CAR T, or CNCT19), including an scFv that interacts with an epitope of the human CD19 antigen different from that recognized by the current FMC63 clone. The results suggested that CNCT19 cells have potent anti-leukemic activities in patients with R/R B-ALL. 82
Genome editing
Vector-based semi-random DNA integration is currently used to manufacture CAR T cells on a large scale
83
; this method is expensive, production is cumbersome, and prone to introducing errors limiting its clinical application. Therefore, alternatives such as genome editing platforms are thus increasingly studied to solve this issue. Genome editing using platforms such as ZFN, transcription activator-like effector nuclease, and CRISPR/Cas9 can disrupt TCR expression and/or MHC on allogeneic third-party T cells to reduce the risk of GVHD. Advanced genome-editing technologies may reduce the chances of introducing abnormal genotypes or off-target effects. Split, universal, and programmable (SUPRA) CARs can provide flexibility and controllability of UniCAR T cells. Rupp et al. found that Cas9-edited CAR T cells to disrupt programmed death-ligand 1 (PD-L1) increased the efficacy of CAR T-cell-mediated killing of tumor cells in vitro. Moreover, in vivo investigations, these constructs demonstrated augmentation of PD-L1+tumor xenograft clearance. These findings highlight the potential of CRISPR/Cas9 genome editing to enhance cell therapies.
84
Eyquem et al. evaluated similar objectives and demonstrated that directing a CD19-specific CAR to the T-cell receptor alpha constant (TRAC) locus has several beneficial outcomes, including uniform CAR expression in human peripheral blood T cells and enhancement of T-cell potency.
85
The findings of this study validate that targeting the CAR to the TRAC locus prevents tonic CAR signaling, establishing an effective re-expression of the CAR construct after repeated exposure to antigen delaying the effector T-cell differentiation and exhaustion.
85
These findings provided supportive evidence for the potential of CRISPR/Cas9 genome editing to advance adoptive cell therapies.
85
Clinical trials of CRISPR/Cas9 gene-edited universal allogeneic CAR T cells (CD19-targeted CAR T cells) have been initiated (
Measures to reduce side effects of CAR
Inhibitory CARs
Inhibitory immune receptors such as programmed death 1 receptor (PD-1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) attenuate or terminate the T-cell function. Fedorov et al. showed that PD-1- and CTLA-4 based inhibitory CARs (iCARs) could reversibly restrict critical TCR or activate CAR functions to an antigen and prevent off-target T-cell toxicity. 86 More recently, in vitro and Burkitt’s lymphoma xenograft models, Tao et al. demonstrated that the KIR/PD-1-based iCAR could be a promising strategy for preventing B-cell aplasia induced by CD19-CAR T-cell therapy. 87
Synthetic Notch AND gate T cells
Synthetic Notch (SynNotch) AND gate T-cell system is an endogenous safety switch; the activation of SynNotch by tumor antigen leads to initiation of transcription of a CAR structure that recognizes a second tumor antigen.88,89 Srivatsava et al. found that CAR T-cell targeting ROR1 recognized ROR1+stromal cells in the bone marrow causing lethal bone marrow failure. Srivatsava et al. generated T cells with synthetic Notch (synNotch) receptors specific to EpCAM or B7-H3, restricted to ROR1+tumor cells but not to ROR1 stromal cells. The results showed that synNotch T cells exhibited tumor-restricted accumulation, CAR expression, and antitumor activity sparing ROR1+stromal cells in the bone marrow and spleen. 90 Moghimi et al. found that the murine GD2 CAR in neuroblastoma (NBL) caused fatal neurotoxicity, so they created constructed synNotch AND gate T-cell by linking signal peptide derived from the human CD8 to an anti-GD2 or anti-B7H3 scFv. In the model, GD2 was the gate, and B7H3 was the target, and it showed a remarkable degree of therapeutic discrimination in vitro and in vivo, sparing single antigen “bystander” cells while eradicating GD2+B7H3+NBL cells. He also found that GD2-B7H3 CAR T cells maintain high metabolic fitness comparable to resting T cells, thus more resistant to exhaustion, and have better in vivo efficacy post-exhaustion than conventional B7H3 CAR T cells. 91
Switch on and off CAR T cells
CAR T cells can lead to life-threatening off-tumor, on-target side effects if CAR T cells cross-react with healthy tissues. Scientists proposed that switching on and off CAR T cells can reduce the risk of on-target side effects through rapid and reversible control of the CAR T-cell reactivity. Lipocalin-based on-switch was proposed based on the findings of the human retinol-binding protein 4 (hRBP4, a member of the lipocalin family) successfully regulating the activity of CAR T cells by interacting with engineered hRBP4 binders in the presence of a small molecule named A1120. 92 Giordano-Attianese et al. developed an off switch named STOP-CAR T by incorporating a chemically disruptable heterodimer (CDH) into a synthetic heterodimeric CAR T-cell. 93 CDH has a high-affinity protein interface that can be disrupted by small-molecule drugs, allowing CAR T’s dynamic inactivation under timed administration [87]. Fms-related tyrosine kinase 3 (FLT3)-specific CAR T engineered with a rituximab-responsive off-switch effectively recovered bone marrow after achieving AML remission. 94
Suicide genes and mRNA mediated modified biodegradation
To minimize the adverse events associated with CAR T-cell therapy, researchers introduced the inducible caspase 9 suicide gene as a safety feature in human T-cell therapies to remove CAR T cells when necessary or shorten their life span.95–99 Wang et al. showed transgene-encoded cell surface polypeptide huEGFRt could be used for selection, in vivo tracking, and ablation of CAR-engineered T cells. 100 Casucci et al. explored the effect of co-expressing a suicide gene enabling fast and efficient pharmacologic ablation of CD44v6-targeted T cells and complete rescue from hyperacute xenogeneic graft-versus-host disease (GvHD) modeling early and generalized toxicity. 101 Mutated Herpes simplex virus type 1 thymidine kinase -HSV1-tk gene (sr39tk) incorporated B7H3-specific sr39tk CAR T cell got completely ablated after the intraperitoneal administration of prodrug ganciclovir in osteosarcoma models. 102 Kenderian et al. studied mRNA-modified CAR33 in the AML xenograft model and found high-level expression of CAR that gradually diminished over 7 days. He then proposed that CAR can be mRNA modified to be biodegradable as a safety mechanism to avoid prolonged toxicity. 103
TMs in UniCAR
A novel modular universal CAR platform technology termed UniCAR was found to reduce the risk of on-target side effects by a rapid and reversible control of CAR T-cell reactivity. UniCAR T cells are inactive in the absence of a TM. UniCAR T cells become active after cross-linkage with tumor cells via a TM. Therefore, UniCAR T cells can be turned “on” just by infusion of the TM and turned “off” by stopping the infusion followed by elimination of the TM. UniCAR uses TM with properties to rapidly reach the target, rapidly dissociate (high off rate) from the UniCAR complex, and rapid elimination from the bloodstream. scFvs or nanobodies with elimination half-lives between 15 and 45 min are ideal TM to be used in UniCAR. Cartellieri et al. provided evidence of retargeting of UniCAR T-cells to CD33-negative and CD123-positive AML blasts in vitro and in vivo. 72
Chemically regulated and SH2-delivered inhibitory tail
Researchers recently developed chemically regulated and SH2-delivered inhibitory tail (CRASH-IT), a titratable and reversible safety switch platform to reduce the side effects. 104 CRASH-IT comprises Zap70 SH2 domains, the PD-1 tail, and the SMASh tag or FKBP12F36V domain. These three functional elements induce proximity to the antigen receptor, provide the inhibitory signal, and allow the possibility to regulate the strength of this inhibitory signal. 104 Furthermore, CRASH-IT can be retrofitted with CARs so the existing CARs do not need a structural redesign so that CRASH-IT can be used along with CAR designs already being used in clinical settings. Ongoing efforts are evaluating the ability of CRASH-IT to achieve optimal control of NK cells and other cells and inhibit other activating immune receptors. Researchers are also exploring alternative inhibitory domains from receptors containing immuno-receptor tyrosine-based inhibitory motifs or immunoreceptor tyrosine-based switch motifs to vary the level of T-cell suppression or to direct such suppression to specific T-cell output signals. 104 The optimization of functional modules, including the improvement of small-molecule ligands, will increase the clinical utility of the CRASH-IT system. 104
Strategies to complement CAR
CAR T combined with immunotherapy (specific and nonspecific immunotherapies)
Specific immunotherapies
Tumor cells can evade immune surveillance by upregulating immune checkpoint inhibitors such as PD-1, CTLA-4, and TIM-3, thereby increasing the risk of relapse or recurrence.105–107 Disruption of the PD-L1/PD-1 immune inhibitory axis using antibodies to block the PD-1 on T cells can regress tumor growth.106,108–111 Li et al. evaluated the use of PD-1/PD-L1 blockade (pembrolizumab, n = 13; nivolumab n = 1) following the failure of CD19 CAR T cell in ALL. Studies have shown favorable responses in patients with early B-cell recovery and substantial extramedullary involvement. In B-ALL patients with poor initial marrow response to CAR T-cell therapy alone, PD-1 inhibition had a partial response suggesting that checkpoint inhibitors can be safe and effective when combined with CAR T-cell therapy in relapsed B-ALL and supports enhancing the CAR T-cell antitumor effect and its persistence.112,113
Nonspecific immunotherapies
A phase II, single-center clinical trial, NCT04534634, is currently recruiting patients to assess the efficacy and safety of interferon-α in combination with CAR T-cell therapy for those with either relapsed or refractory ALL. Enrolled patients will receive IFN-α combined with an infusion of CAR T cells, with participation spanning 5 years, including treatment and follow-up periods. The primary outcome measure is the overall response rate in a 2-year time frame, including complete remission, complete remission with incomplete hematologic recovery, morphologic leukemia-free state, and partial remission. The secondary outcomes measured are overall survival, leukemia-free survival, the cumulative incidence of relapse, the duration of CAR T cells in patients, and the number of adverse events, all assessed over 2 years. The anticipated study completion date is July 2025.
Artificial antigen-presenting cells
Antigen-presenting cells (APCs) are custodians of the immune system. aAPCs are synthetic versions of these sentinel cells and are made by attaching the specific T-cell stimulating signals to various macro- and micro-biocompatible surfaces. aAPCs remove the need to harvest patient-specific APCs such as dendritic cells (DCs) and the process of activating the DCs in the stimulation of antigen-specific T cells. aAPCs can be cell based, 114 or microparticles or nanoparticles115,116 and do not have any variability unlike APC. Also aAPC can be engineered to deliver specific co-stimulatory signals and cytokines for the expansion of specific cell lineages (IL-15 for memory NK and memory T-cell expansion and IL-21 for priming of naive cells and expansion of memory T cells).
Turtle et al. suggested that the use of aAPCs platform as an “off-the-shelf” reagent improves the manufacturing process by avoiding the procurement and use of allogeneic peripheral blood mononuclear cells (PBMCs) as irradiated “feeders.” 117 Kebriaei et al. first used aAPC to propagate CAR T cells in 2012. 118 Hasan et al. discussed artificial APCs (aAPCs) to allow optimal control over T-cell signals and substantiated that aAPCs enhance the efficacy and persistence of infused T cells to combat CD19-positive relapse. 119 The role of aAPC in UniCAR is yet to be studied; if UniCAR becomes one of the mainstream CAR strategies, it would be interesting to evaluate if aAPC would improve its efficacy.
Role of epigenetics in boosting CAR
Epigenetic modifications may play a role in tumor growth or promotion by (1) methylation or hydroxymethylation of cytosine; (2) modification of histone (acetylation, methylation, or phosphorylation), (3) non-coding RNA (ncRNA) association modifications that alter microRNA-associated gene expression, and (4) higher-order associated modification.120,121 Epigenetically modulating CAR T cells or cells in their surrounding microenvironment might also help prevent antigen heterogeneity or loss, impaired trafficking, T-cell intrinsic regulation, and make CAR T-cell therapy more effective in the context of a harsh TME. 122 In addition, epigenetic reprogramming of CAR T cells to promote central memory and retain stem cells, the differentiated progeny, can improve the efficacy of CAR T-cell therapy. 122
The use of low-dose decitabine priming of CAR T cell has emerged as one of the most studied epigenetic strategies to improve CAR T effectiveness.123–125 Inhibition of de novo DNA methylation enhances T-cell rejuvenation in vivo and prevents T-cell exhaustion. Wang et al. studied decitabine’s effect on CAR T cells, a DNA methyltransferase inhibitor, to check its efficacy in modifying exhaustion-associated DNA methylation. CAR T cells pretreated with a low-dose decitabine (5-aza-2′-deoxycytidine) showed a more potent antitumor effect, increased proliferation of CAR T cells, and enhanced CAR T-cell cytokine release capacities. In both in vivo and in vitro settings, CAR T cells treated with decitabine (dCAR T-cells) maintained a higher memory-associated and relatively lower exhaustion-associated gene expression under tumor cell stimulation. In addition, the use of methylation inhibitors demonstrated improvement in CAR T-cells’ exhaustion and promoted the maintenance of memory phenotype and effector function. 124
CAR NK cells
CAR NK cells have several advantages over CAR T cells, such as limited undesirable “off-target” toxicities due to a relatively shorter life span, low risk of GvHD,126–130 and their ability to kill tumor cells with CAR-independent mechanisms. CAR-independent mechanisms include natural cytotoxicity against tumor cells, 131 activated using NCRs, NKG2D, co-stimulatory receptor DNAM-1 aka CD226, activating KIRs such as KIR2DS1, KIR2DS4, KIR2DL4,132,133 and CD16-mediated ADCC. 134 The risk of GVHD remains low even with allogeneic cells. 135 Therefore, CAR NKs can be generated from various sources such as NK92 cell lines, PBMCs, umbilical cord blood, and induced pluripotent stem cells. 136 HLA-mismatched anti-CD19 CAR NK cells induced complete remission in most of the patients with NHL or chronic lymphocytic leukemia without the development of significant adverse effects. 137 In this study, the inducible expression of caspase 9 as a safety switch further enhanced the safety of anti-CD19 CAR NK cells. 137 Several promising CAR-NK cell candidates are currently in clinical testing, and novel genetic engineering avenues are testing the next generation of CAR-NK cell immunotherapies. Compared to CAR T cells, CAR NKs have a lower risk of GVHD due to a lack of TCRs126–129 and less side effects due to their shorter life span. The low risk of GVHD associated with CAR NK can enable allogeneic CAR NK cell transplantation, further reducing costs. However, the short life span may increase the costs since it may need multiple administrations periodically. There are no studies to evaluate the combined use of CAR NK and CAR T cells.
Conclusion and future directions
Several ongoing studies are exploring alternative antigen targets alone or in combination with anti-CD19 CAR to solve the shortcomings of a single anti-CD19 CAR. Engineering strategies such as multitargeting constructs and universal CAR T cells improve CAR’s efficacy and prevent negative antigen relapse. Armored CAR T cells—Gene-Edited, Interleukin CAR T cells—improve the efficacy and survival in harsh TME. CAR will continually evolve with periodic updates to scFv and transmembrane domain. The use of advanced gene editing platforms will further reduce the creation of abnormal genotypes or off-target effects. Similarly, the introduction of CAR NKs, iCARs, synNotch AND gate T-cells, suicide gene switches, mRNA-modified CARs, TMs in UniCARs, and CRASH-IT are promising approaches to reduce adverse events. With all this novel research, the future of CAR T-cell therapy looks exciting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Prior presentation
None.
