Abstract
This paper aims to determine the surface patterns of corneal diseases under the effects of swelling, such as in keratoconus (KC). Swelling is a mechanism that responds to changes in pressure within a medium. The cornea allows aqueous humor to flow from the stroma into the anterior chamber, following the laws of mechanics to reach mechanical equilibrium. The geometry of the cornea is altered to change its curvature, and it is reformed through the process of inflating and shearing. The theory of shells is divided into two theories: membrane theory and bending theory. These theories are used to determine stress and its corresponding shell deformation, providing solutions for bending, changes in shell thickness, and axially symmetrical load deformation, respectively. The change in corneal shape due to swelling is calculated and demonstrated under constant intraocular pressure (IOP) and the corneal load. This estimates the potential deformations of the corneal structure in response to the pressure and load imposed by the equilibrium equations. Moreover, the model can estimate the tensile properties of corneal tissue, such as its elastic modulus and stiffness. This model helps understand the mechanical stress–strain relations, permanent structural changes, and surface patterns of corneal deformations.
1. Introduction
Sight can be assumed to be a simple physical optical phenomenon, but the eye involves complex, relatively perpetual biomechanical processes. The anatomy of the anterior part of the human eye and of the different corneal layers, which, altogether, constitute the corneal tissue, are schematically seen in Figure 1. Biologically, the sclera and cornea are two critical tissues of the eye. Nevertheless, studies of eye diseases can generally be performed on the cornea rather than on the sclera because of its opacity. It is essential to investigate the biomechanical processes in the cornea to understand its physical properties. The cornea is thin (approximate thickness of 540 μm to 560 μm) and has soft tissue. It has an inertial dynamic that mainly balances atmospheric pressure and intraocular pressure (IOP). The pressure sources on the cornea are not limited to these two; a more complex mechanism is involved. Therefore, structural deformations occur during dynamical processes, and the cornea’s geometry changes due to its biomechanics [1].
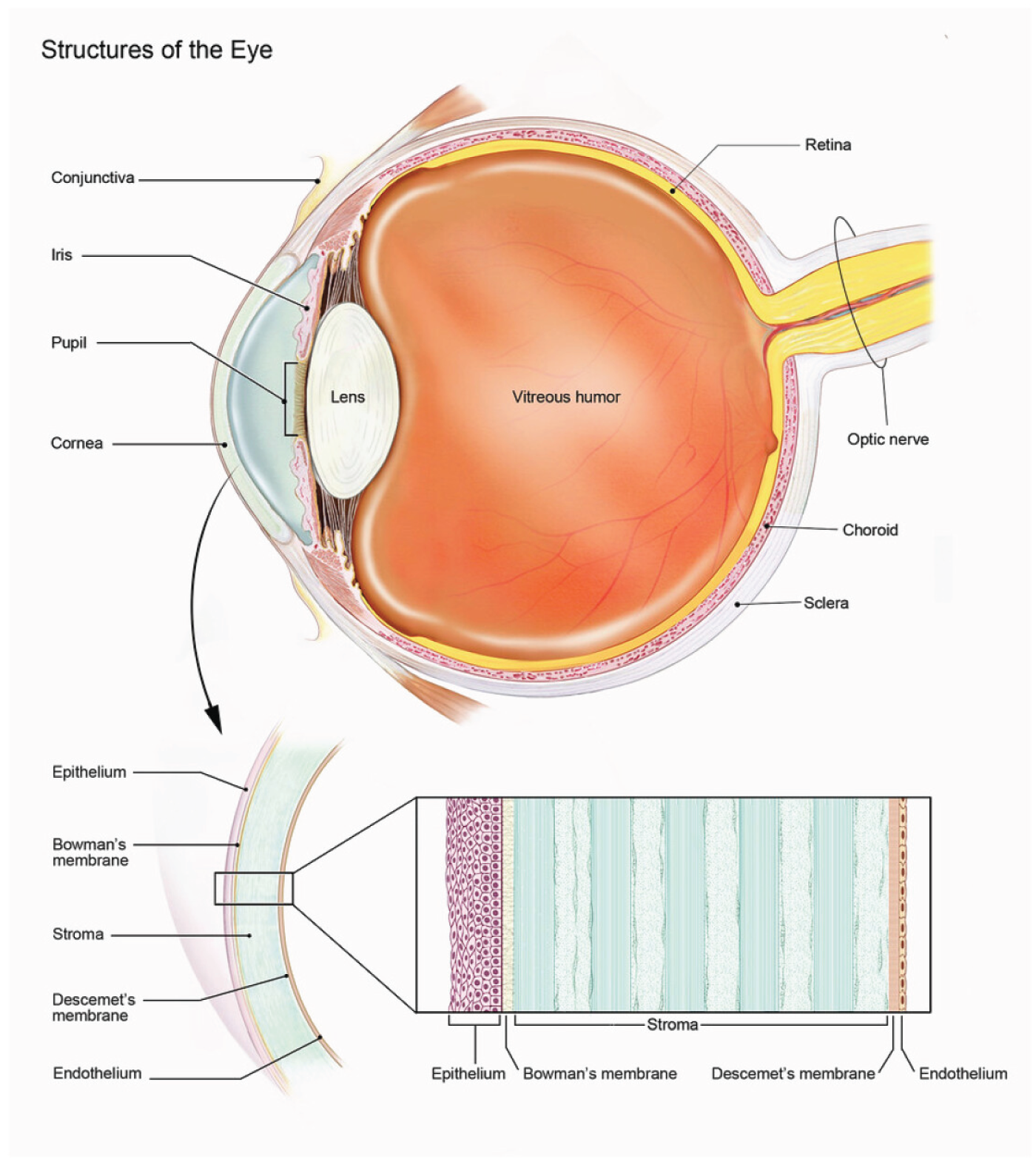
A schematic representation of the human eye.
The geometric structure of the cornea is an oblate spheroid (or approximately hemisphere-like) shaped patch stitched on the sclera via limbus, and its central role is to ensure that the light that comes from outside passes directly into the center of focus of the eye via a right angle. The cornea is a tissue composed of layers, and the thickness of the cornea varies from its center to the limbus, and the anterior and posterior radii are also different. Approximately 90 percent of the cornea consists of stroma, which is made up of lamellar layers. Each lamella contains collagen fibrils that are mostly regularly distributed within the stroma. The elastic performance of the cornea under meridional stress was discussed in [2].
Corneal studies encompass a wide range of subjects, including corneal diseases and the mechanical properties of the cornea. The model analyzed and prescribed the cornea’s mechanical attitude, assuming the cornea was a thin, rigid shell, and the experimental tonometry results were reported in [3]. Another model of corneal biomechanics based on membrane structure was developed for tonometric studies in [4]. A constitutive formulation was constructed in the context of continuum mechanics regarding the anisotropy of collagen fibrils, and the numerical solution was given in [5]. The etiology of keratoconus (KC) was discussed based on the observed collagen anisotropies under the effect of IOP. Anisotropy of collagen fibrils in the cornea was identified by the Inverse Finite Element Method based on the fibril-reinforced continuum material [6]. Molecular origins of corneal biomechanics were reviewed in [7]. Collagen fibril arrangements were identified in three dimensions by scanning electron microscopy in [8]. The response of the bovine cornea to inflation was studied, and according to [9], the deformation primarily occurred in the limbus. [10] analyzed correlations between IOP and biomechanical parameters of the cornea. [11] demonstrated that the strain distribution over the cornea was not uniform.
Another issue is the sources and mechanisms of corneal pressure. Here, we also observe multiple sophisticated pressure sources, swelling, IOP, osmotic, Donnan, and imbibition pressure. However, analyzing the mechanical effects of these pressures across the entire cornea poses a significant mathematical challenge; we can identify the pressures and their corresponding effects. Swelling is defined as a volumetric growth of a body part not caused by cells [12]. In the cornea, the swelling pressure is used to balance fluctuations in IOP, allowing lamellae to expand through coupling to the fluid distribution via electromagnetic interactions between fibrils. The cornea exhibits distinct hydrodynamic features governed by dynamic and poromechanical processes during swelling. Thus, holistic and reversible changes in corneal volume are observed, and the corneal surface can develop into many different patterns. The response of the corneal system can be evaluated mechanically and statistically. The statistical response is an equilibrium property of the cornea, and the mechanical response is a geometric property of the cornea that shapes the mechanics of swelling. Mathematical modeling of soft-tissue biomechanics during swelling has been studied within the framework of continuum mechanics [13,14]. Swelling in the cornea was discussed in [15,16].
On the contrary, swelling is the cornea’s response mechanism to balance IOP. As mentioned, the cornea grows along a specific path determined by its geometry, infrastructure, swelling mechanism, and minor effects [1]. The biomechanics of the cornea should be evaluated with respect to both swelling and shear. Eventually, the cornea’s shape, lamellar structure, and elastic properties undergo permanent changes [17–19]. Geometric distortion is the primary origin of the vision problem. These are progressive and irreversible, and their origins are mostly unknown. Regular corneal deformations can lead to corneal dystrophies, including KC [20]. Experimental and theoretical research suggests that surface analysis linked to the internal parameters of the cornea may provide an efficient method for identifying the origins of corneal impairment.
Biomechanical properties of the cornea are primarily based on the stroma, so the lamellar structure defines its viscoelasticity. Stroma is a chemoelastic tissue that governs the corneal response to variation in IOP. The system embedded in the eye evolves the geometry by pumping chemicals into the cornea, causing the cornea to swell. Swelling is a volumetric growth that results in a surface pattern differing from the previous one. As a result of swelling, the cornea deforms due to its viscoelastic characteristics. The geometry of the cornea evolves from an oblate spheroid to a non-axially deformed ellipsoid, then recovers its original shape in hourly cycles. In that context, the evolution of corneal curvature was analyzed in [21,22] without accounting for the biomechanical properties of the corneal infrastructure.
KC is an asymmetric corneal ectasia that exhibits abnormal geometric and biomechanical progression, including reduced corneal thickness, high local curvature, and topographic irregularity [23,24]. The curvature of the outer corneal surface is displaced around a point below, and the thickness is relatively symmetrical around the central point of the cornea, as shown in Figure 2, the corneal topography of KC. The structural weakening, conical protrusion, and inferior-superior asymmetry are commonly detected in geometrical deformations associated with KC [25]. KC is also relevant to the shifted corneal vertical apex and thinnest point location but not with horizontal location [26,27]. The typical geometrical evolution of KC changes the cornea from a dome-shaped to an asymmetrically cone-shaped shape, as shown in Figure 3. The general deformation pattern can be seen as abnormal inferior steepening on retinoscopy, keratometry, keratoscopy, or corneal topography [28]. Thus, the goal is to determine the general deformation pattern of ectatic corneal disorder (KC) using suitable geometric arguments and instruments. For this reason, shell theory offers the most potent approaches and geometric tools for addressing corneal disorders, as we have seen. Our motivation is to derive the theoretical background of the geometrical deformation that monitors the classical resultant geometry of KC.
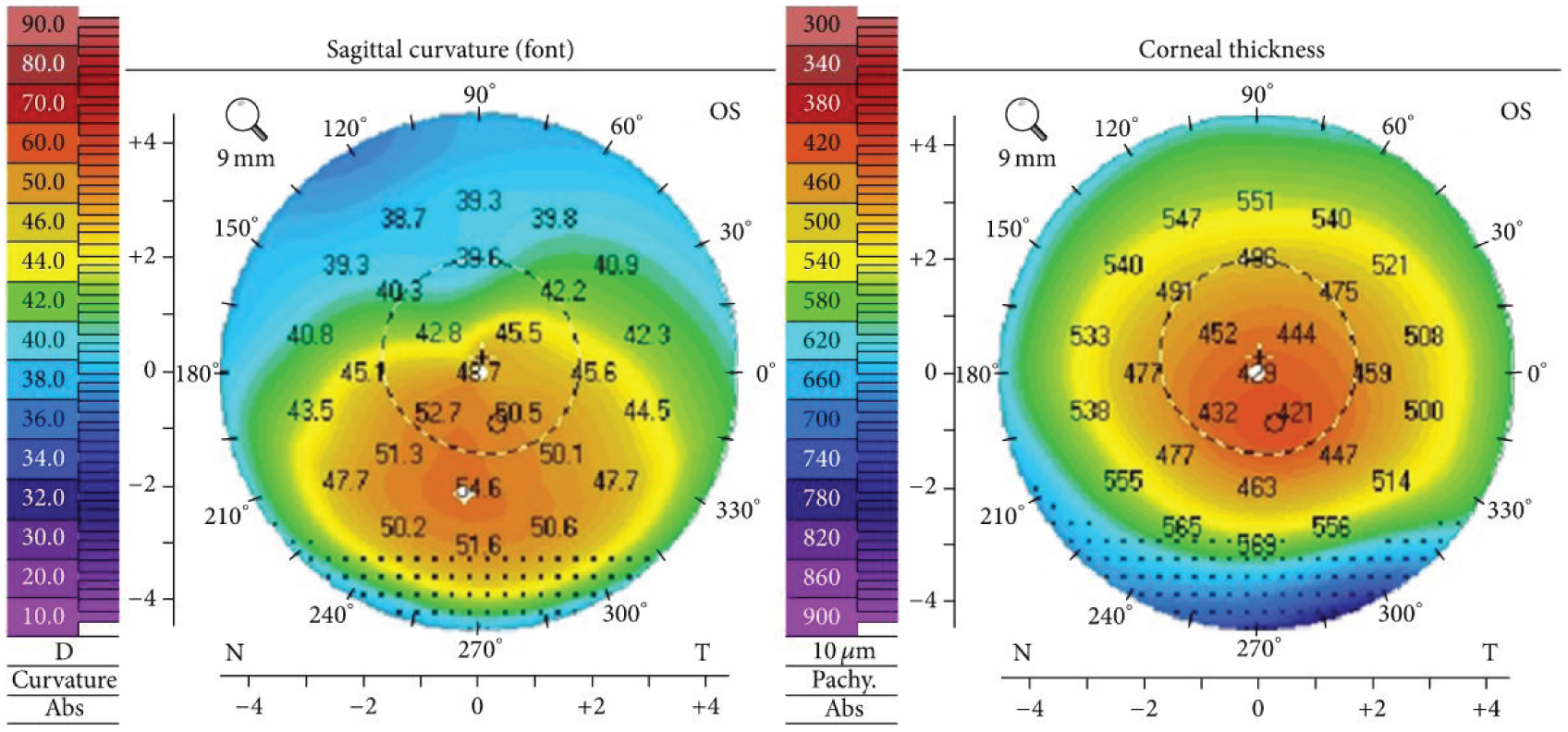
Corneal topography in typical keratoconus.
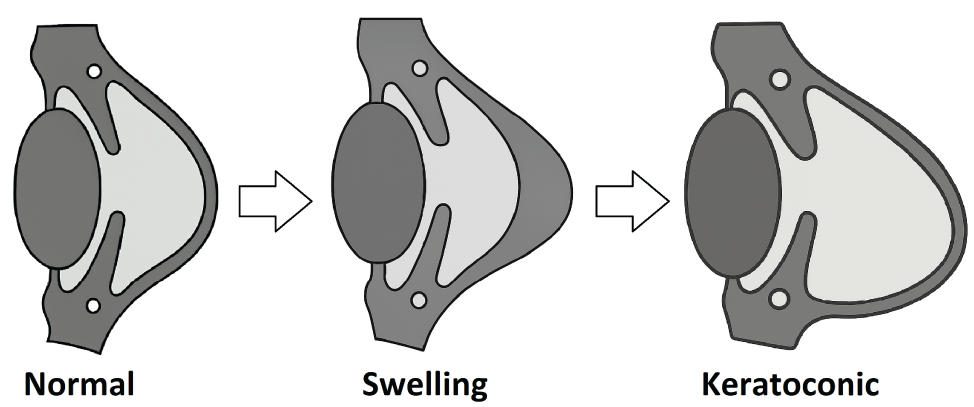
Normal, swelling, and keratoconic corneal states.
KC is a corneal disorder that permanently alters its geometry, viscoelasticity, stiffness, and biomechanical properties [28,29]. In addition, corneal surface patterns change during swelling, and some patterns can result in permanent geometric deformations of the cornea. Thus, swelling can be regarded as one of the responsible mechanisms of ectatic corneal deformations, such as in KC, due to irreversible, inelastic changes that can occur during the process. The geometrical analysis of general deformation patterns associated with KC reveals common distortions, including corneal aberrations, asphericity, and conical protrusion [30]. No theory has been proposed to explain corneal reshaping related to KC. Thus, a realistic model of KC is required to determine the cone-shaped, vertically asymmetric geometry of the cornea.
This paper provides a brief description of corneal biomechanics and offers a concise review of swelling in the following section. We evaluate the swelling–shear relation and discuss its strong correlation. We present shell theory, derive the shear- and angular-displacement fields via bending theory, and obtain the resultant geometry via membrane theory. We load an external factor (the corneal load) to induce asymmetric growth. We find the resultant general geometrical deformation of KC that any other mathematical approach has not yet obtained. Finally, we present our results, illustrations, and prospects for corneal biomechanics.
2. Biomechanics of cornea
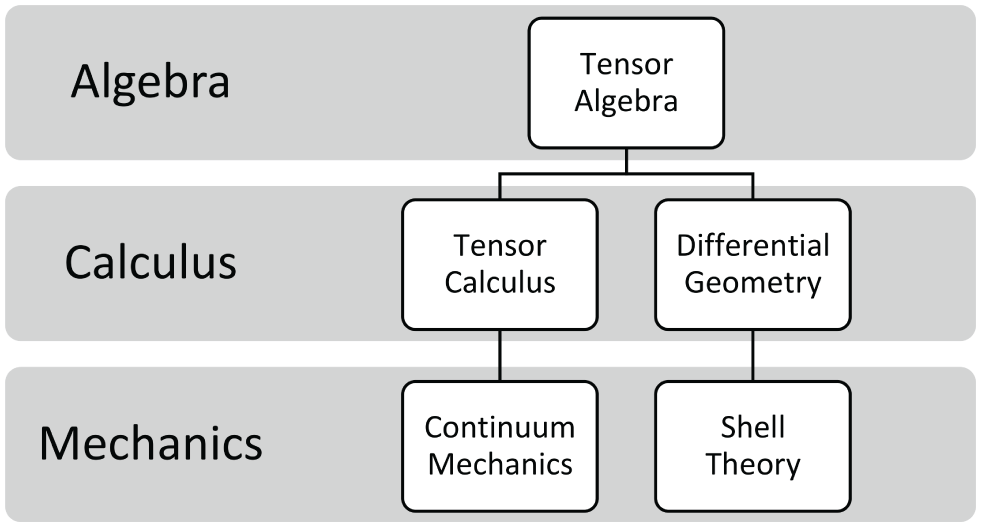
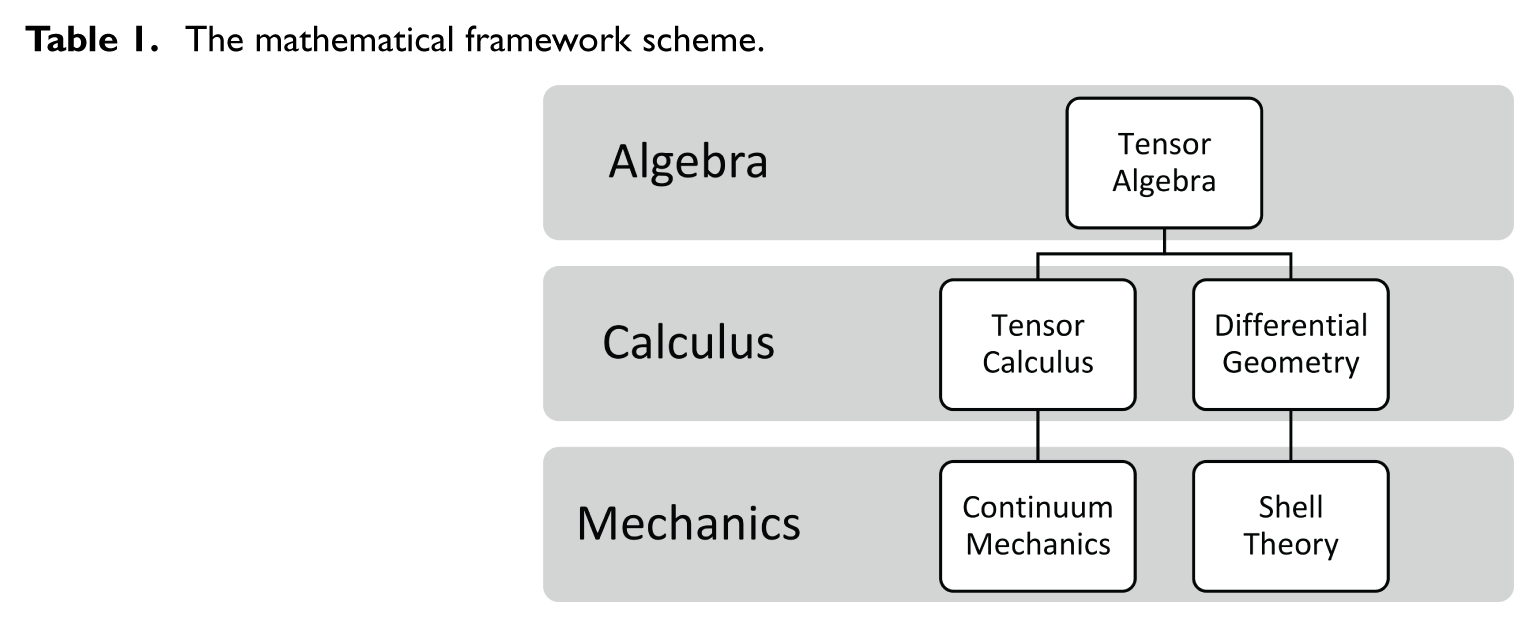
The mathematical modeling of corneal biomechanics has developed into two theoretical traditions: one is continuum-mechanical models based on tensor algebra and tensor calculus, and the other is shell-theoretical models based on tensor algebra and differential geometry, as shown in Table 1. Understanding this branching in modeling studies is essential for placing the present work within the mathematical framework of the biomechanical literature.
The mathematical framework scheme.
In general, many research models the cornea as an anisotropic, fiber-reinforced structure governed by continuum mechanics. These formulations apply tensor calculus to describe deformation, stress redistribution, collagen orientation, and stromal remodeling. Works such as [5,6,31] employ hyperelastic and anisotropic constitutive models to determine the mechanical response of corneal stroma. More recent papers derive surface mechanics from volumetric elasticity using enriched kinematics that include thickness variation, transverse shear, and higher-order gradients. Notably, Dell’Isola et al. [32] introduces a generalized plate model with kinematically independent thickness to model geometrical patterns of corneal pathologies, and Dell’Isola, et al. [33] formulates an elastic surface theory for fibers of fabrics accounting for higher-order strain-gradient effects. Continuum-mechanical approaches provide detailed representations of internal mechanical behavior but generally require numerical analysis and do not give closed-form geometric descriptions.
Another research tradition treats the cornea as an elastic shell governed by shell theory, using differential geometry to describe changes in surface patterns. Membrane and bending theories, formulated by [34–36], have been adapted to corneal biomechanics to study deformation under IOP, tonometry, and inflation testing. These models operate on a curved surface representing the corneal mid-surface and provide analytical relations among load, swelling, and the deformation patterns. Shell-theoretical approaches are efficient for exploring geometric patterns of deformation, including anterior bulging, curvature steepening, and asymmetric deformation patterns that characterize disorders such as KC.
Although the cornea has a complex structure, some simplifications can be made to model it mathematically. Initially, the cornea is a soft tissue exposed to atmospheric pressure on the outside and to IOP on the inside. It can be assumed to be homogeneous, isotropic, continuous, and independent of any particular coordinate system. Under these conditions, the cornea is modeled as a continuum in continuum mechanics to study deformations and associated stresses [37]. Woo et al. [38] investigated the nonlinear behavior of the cornea-sclera structure by using the finite element method. In this mathematical model, they used applanation tonometry to compute the nonlinear relationship between IOP and volume, as well as the relationship between force and volume.
In [31], the cornea is considered an anisotropic structure, so they use the experimental data of the air-puff technique to simulate the behavior of the cornea. The swelling procedure during osmotic flow between the epithelial and endothelial membrane layers is modeled and tested in [15], which is based on experimental data. Vito and Carnell [39] used different models to simulate deformations. Even so, the limbus has been treated as a boundary condition on the cornea; some models, such as membrane and bending theories, are derived from the non-limbus type, which is assumed to be isotropic, linear elastic, and semi-incompressible. Bending and membrane theories are crucial mathematical tools because pressure and load distributions are not uniform on the corneal surface.
We have summarized the correlations between biomechanics and the cornea’s internal structure above. In this manner, we can consider the cornea as a viscoelastic/hydrogel material that allows deformation while protecting against deformation through swelling. The geometry of an oblate spheroid initially evolves to the conical surface through a swelling procedure. Collagen fibril/lamellae orientations are getting nonuniform from the center to the limbus. Corneal elasticity comprises various types of collagens, each with distinct functions. Mow et al. developed a corneal tonometry model by treating the cornea as a spherical membrane with constant thickness [3,4]. The stress–strain relation of the cornea can be constructed for reinforced material by fibril orientations via continuum mechanics [5,40]. However, defining fibril orientations across the entire cornea is challenging. In the literature, alternative methods have been derived to model the swelling effect on fibril-reinforced materials, focusing on calculating the shear of the corneal structure using beam theory [37,41]. We argue that surface analysis can be used to combine all information collected from surveys. Membrane and bending theories provide limited information and only partial solutions regarding the deformation of the cornea. Ultimately, the next step is to develop an algorithm that simulates the evolution of the cornea surface.
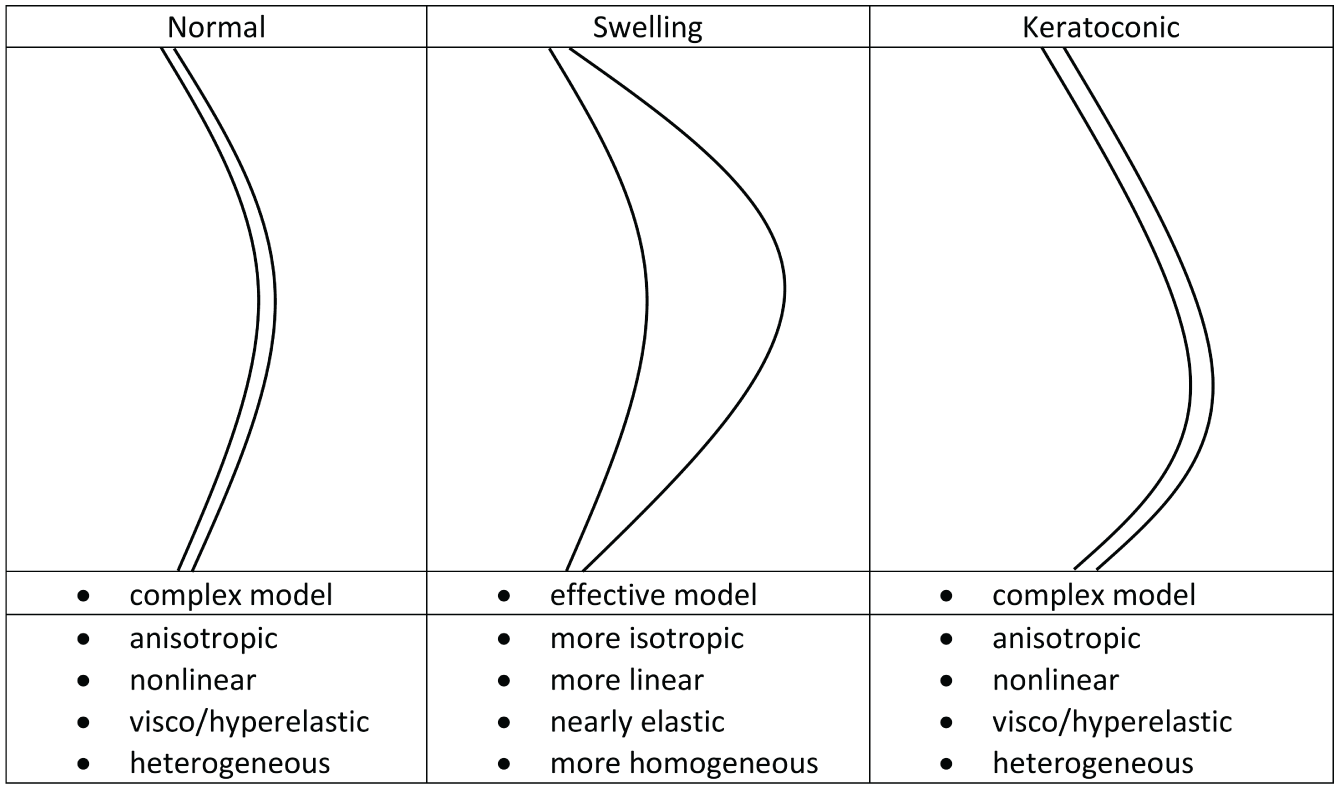
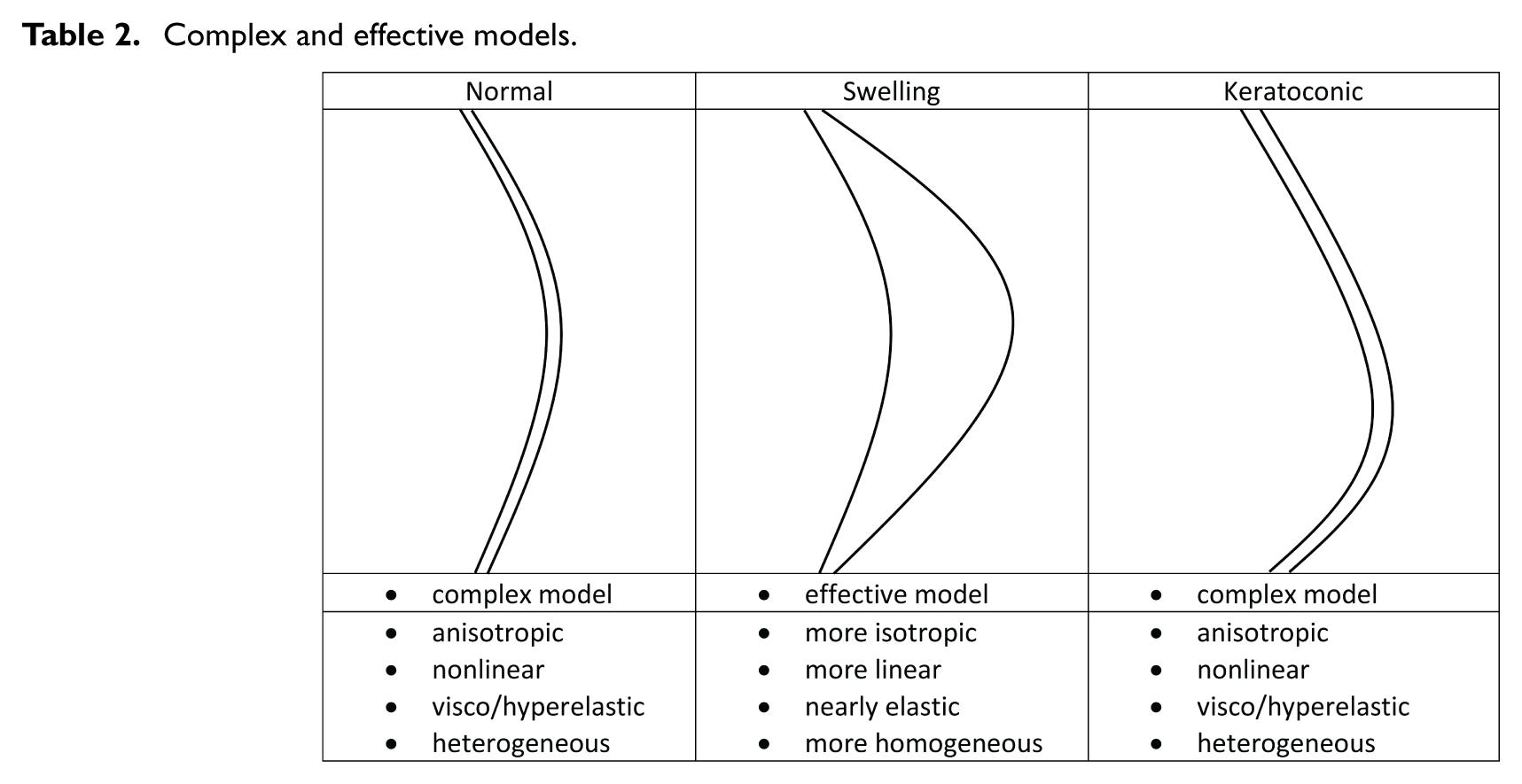
At this point, we can reconsider the modeling strategies for corneal deformation, which are determined by the normal, swelling, and keratoconic corneal states, and group them into two: complex and effective models, as shown in Table 2. The complex models exhibit mechanical behaviors arising from their organized collagen fibril orientations or lamellar architecture, which produce biomechanical properties such as anisotropy, nonlinearity, heterogeneity, and stiffness, as in [42]. Hydration-dependent structural studies have shown that swelling increases interfibrillar spacing and lamellar separation and reduces collagen fibril alignment and weakens directional stiffness, indicating that the stromal microstructure becomes less directionally ordered under hydration [43,44]. Swelling also tends to diminish depth-dependent variations in stromal organization, contributing to a more uniform distribution of collagen fibrils across the corneal tissue thickness [45]. Mechanical testing further demonstrates that corneal swelling reduces overall stiffness and weakens the characteristic nonlinear stiffening associated with fiber recruitment, yielding a smoother and more matrix-dominated macroscopic response [46,47]. While these hydration-induced changes do not eliminate anisotropy or nonlinearity, they collectively indicate a trend toward more isotropic, more homogeneous, and nearly elastic effective behavior at the macroscopic scale. In light of these studies, we can model the cornea in the swelling state using the effective model, represented by an isotropic, linearly elastic spherical shell with uniform material properties, which captures the dominant mechanical effects of swelling while retaining analytical tractability for axisymmetric loading analysis.
Complex and effective models.
3. Shell theory on cornea
The theory examines the effects of swelling and shear on corneal deformation, providing a more comprehensive understanding of the cornea’s biomechanical behavior by addressing factors often overlooked in existing models. The model also introduces a nonlinear representation of radial growth due to swelling, thereby providing more accurate predictions of the cornea’s response to changes in IOP and external loads, particularly in conditions such as KC. Applying shell theory, specifically membrane and bending theories, to the cornea, which is modeled as a spherical shell, enhances understanding of non-axial deformation and surface patterns, which are crucial for understanding mechanical stress–strain responses.
The theory of shells is divided into two theories: the membrane theory and the bending theory. The membrane theory can determine the stress and its corresponding shell deformation. In some cases, membrane theory cannot provide solutions that are compatible with the actual deformation conditions. Bending theory provides solutions for bending and shear stiffness, shell-thickness changes, and deformation under axially symmetric loads. Therefore, an analytical approach to a shell of revolution problem combines the two theories. For details in computations, please see [34–36,48,49].
For a general shell of revolution, as shown in Figure 4, let us consider an infinitesimal spherical patch ABCD bounded by two meridians and two parallels with principal radii
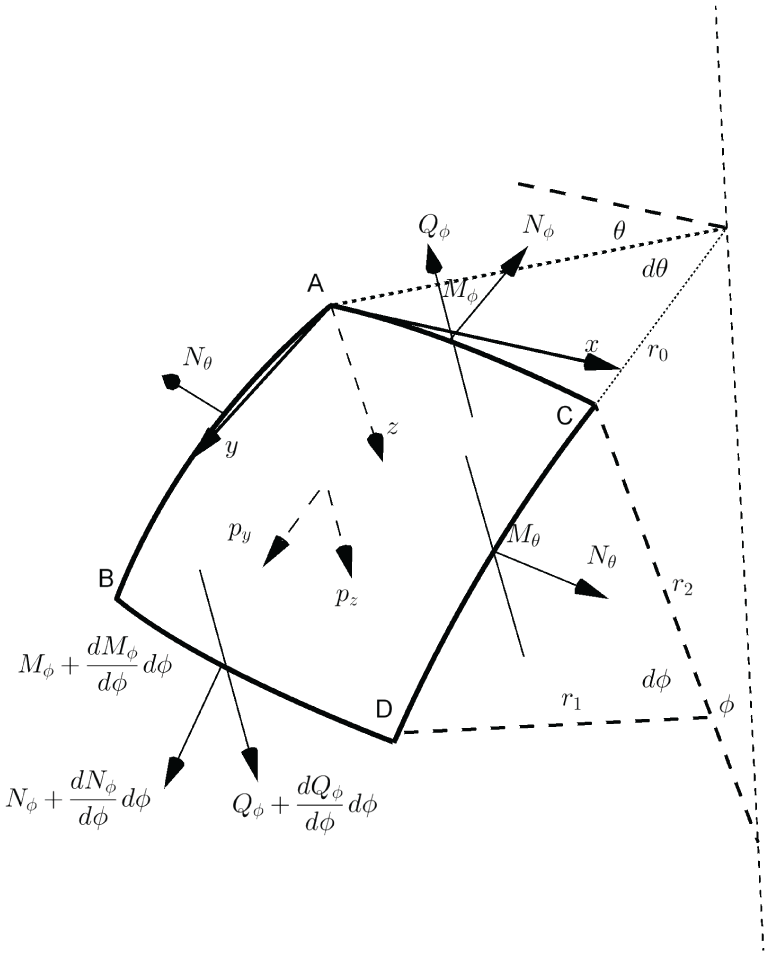
The displacement of a spherical patch.
Terminology of the shell theory.
The equilibrium equations for forces, moments, and shear in bending theory are depicted as follows:

The equilibrium equations for the forces and shear in membrane theory are depicted as follows:

When the cornea swells, it retains the extra water, depositing it and causing a corneal hydro-load. We refer to the corneal self-load and hydro-load collectively as the corneal load, as shown in Figure 5. Both pressure and corneal load must be evaluated simultaneously; however, no existing method simultaneously evaluates them. We approach the problem by considering the corneal load in conjunction with IOP. Therefore, we can consider the IOP p and the symmetric corneal load
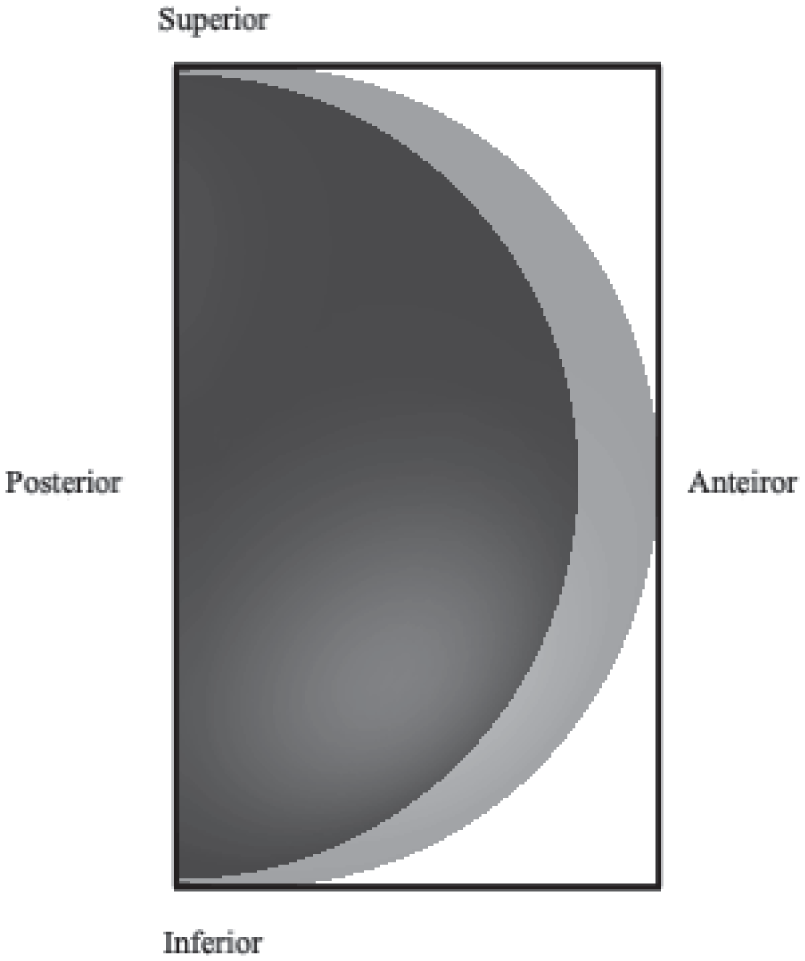
Symmetric corneal load (shaded) on the sclera (black-colored) via limbus.
The components of the loading for the sphere are expressed, as shown in Figure 6:

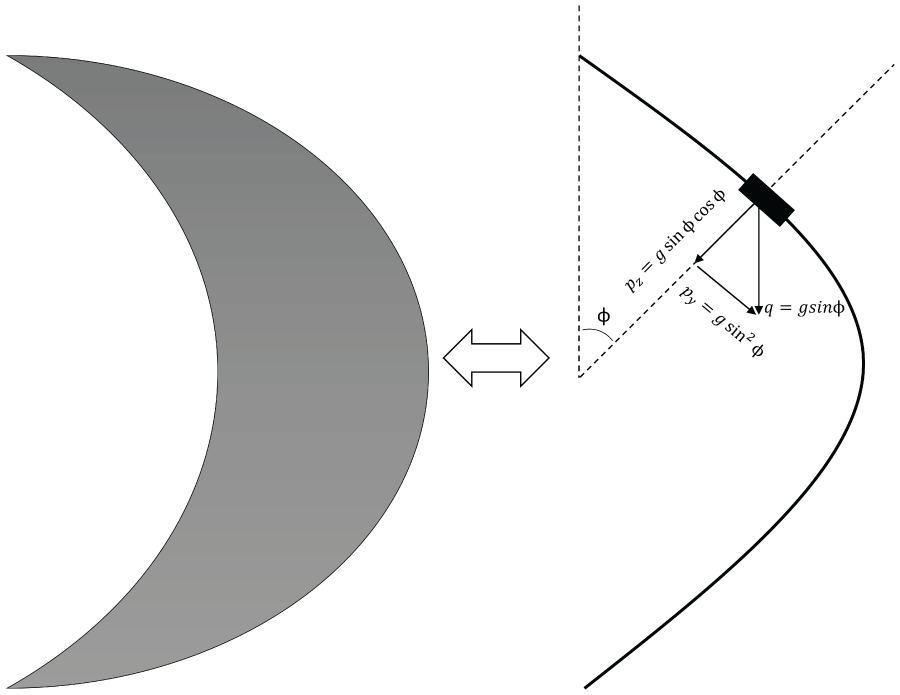
Corneal load of swelling state.
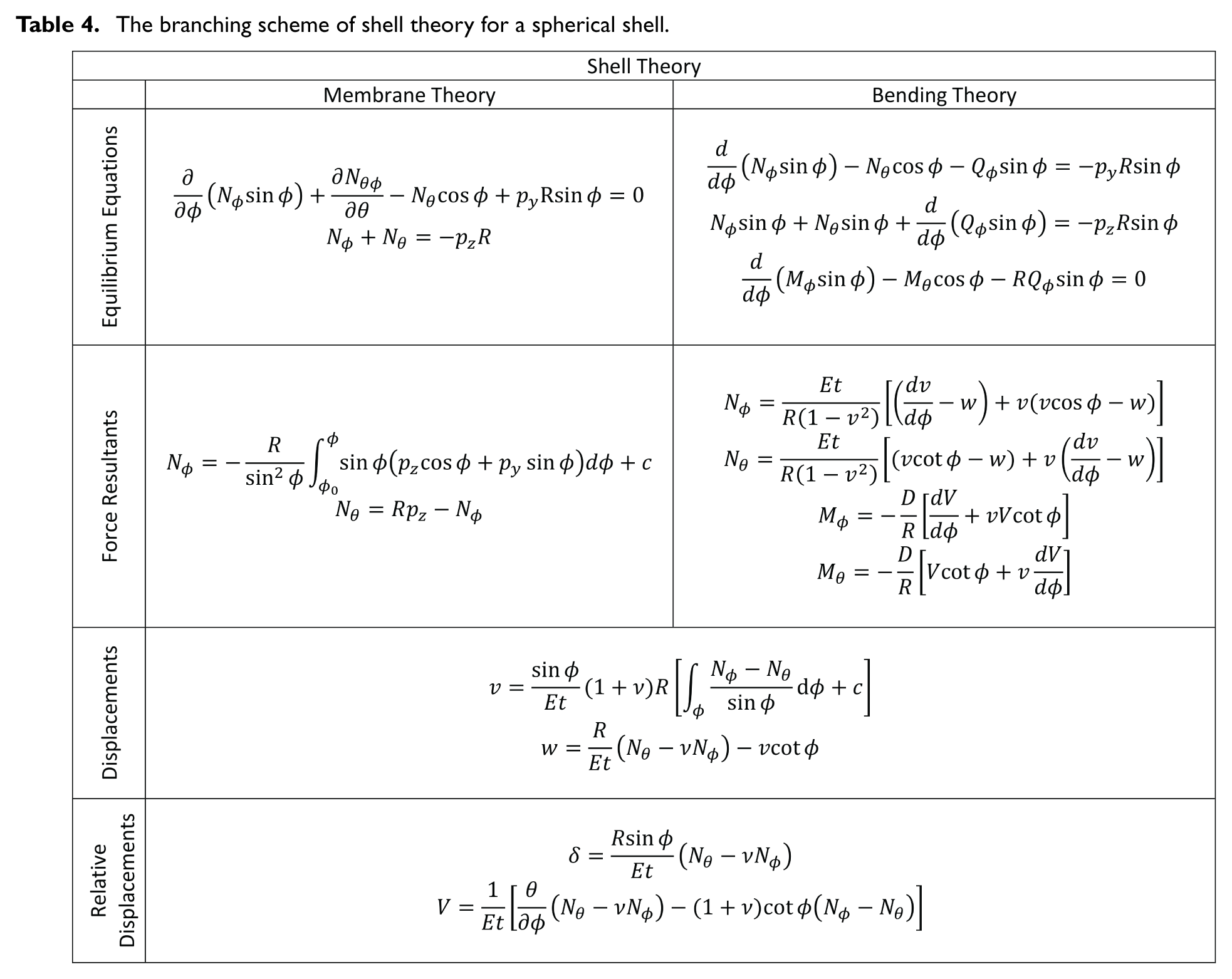
The equilibrium equations, force resultants, and the displacements for the spherical shell in membrane and bending theories are shown in Table 4.
The branching scheme of shell theory for a spherical shell.
The equilibrium equations are reduced to two differential equations for the shear force


Therefore, the problem of bending the corneal shell under uniform IOP and the corneal load is reduced to two differential equations. However, a complete solution to these two differential equations in terms of a hypergeometric series is relatively complex. To get rid of complex terms in the solution, we truncate the first-order terms and the incompatible term


In this case, we obtain a simplified solution for an axisymmetric load. From the preceding, by eliminating

The general solution of the foregoing equation is:

where
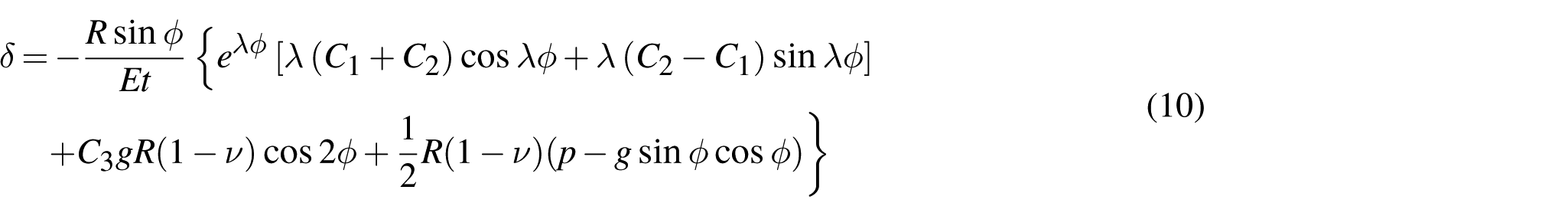
Calculating displacements v and w from the resulting coupled equilibrium equations, using bending theory, is computationally more demanding, so we derive relative displacement relations. Instead, applying membrane theory to the same corneal load on the spherical shell readily yields displacements, providing a simple and accurate description of deformation patterns. The components of the loading in the equation for the sphere can be given again as:

The components of the stress tensor are found as:


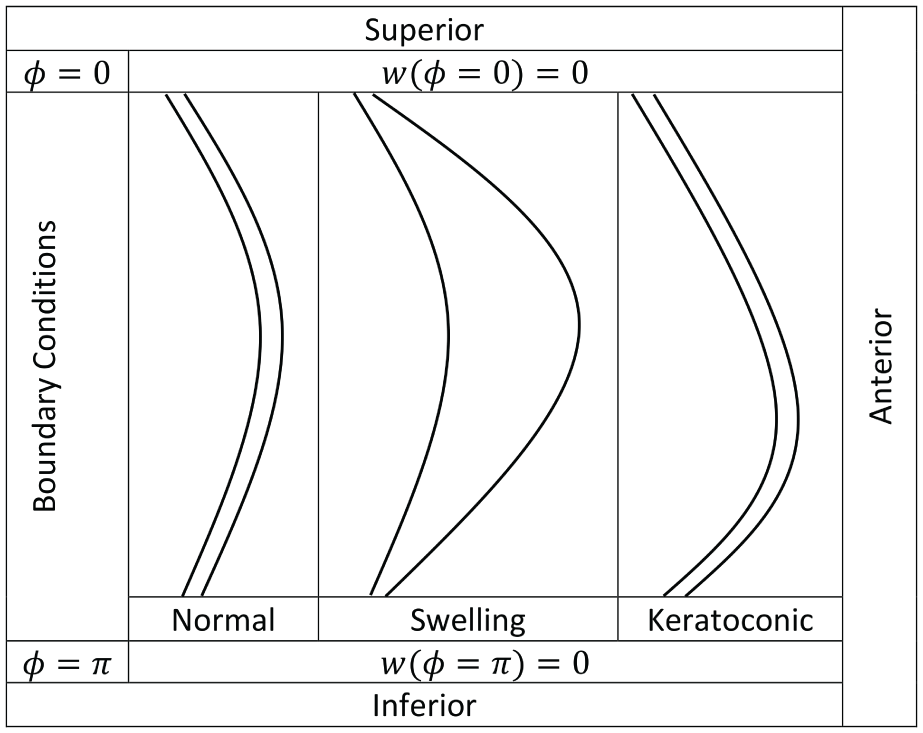
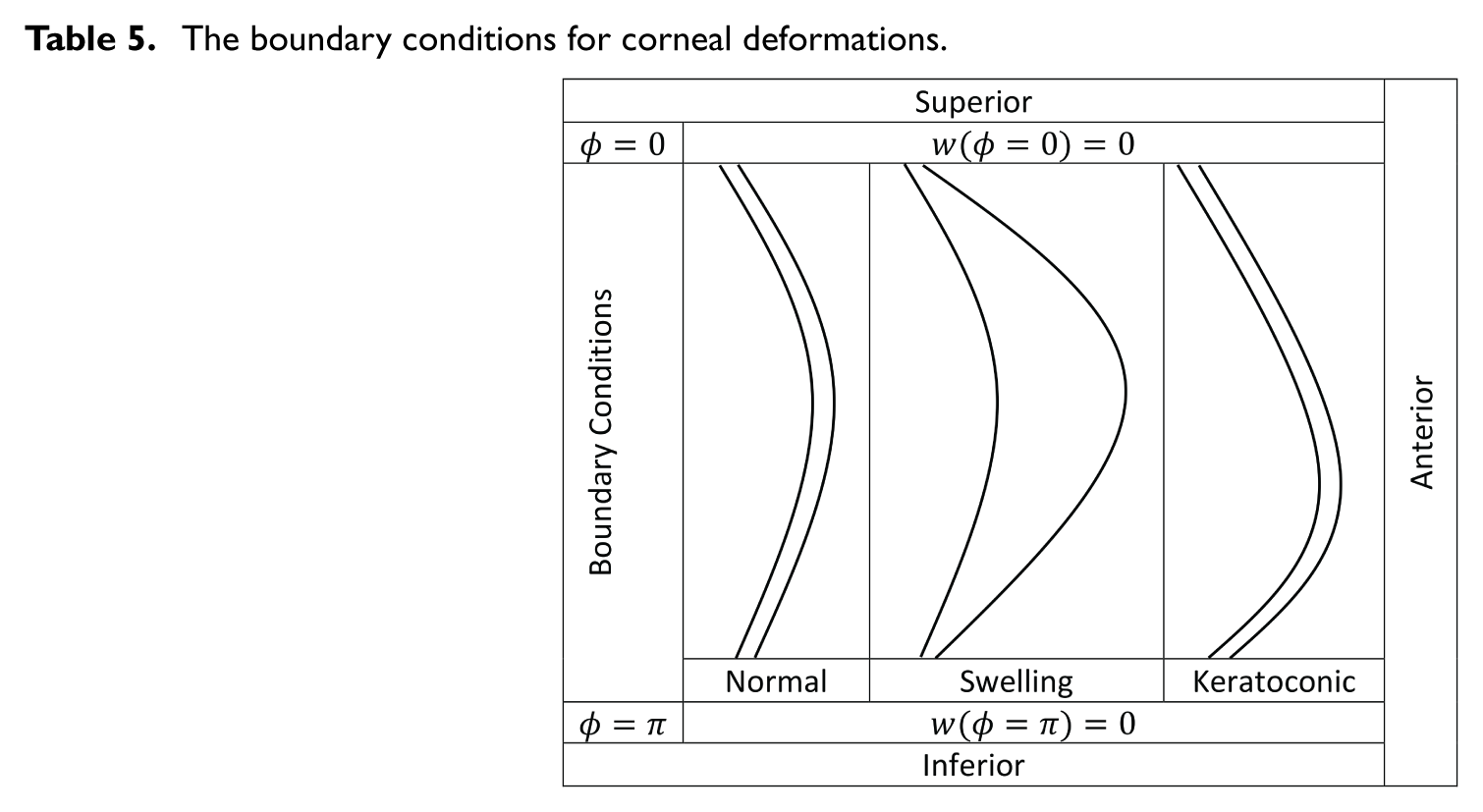
The deformation of a shell of revolution in the direction of the tangent to the meridian and in the direction normal to the surface υ and w, with the presented boundary conditions given in Table 5, obtained as:


The boundary conditions for corneal deformations.
We obtained a couple of displacement relations for the corneal shell sphere under corneal loading and a uniform IOP. We provided representative graphs of the radial displacement and the change in radius, as shown in Figures 7 and 8. The results obtained support the construction of stress–strain and shear–angular displacement relations. We get the general surface pattern of anomalous corneal deformation, which describes an asymmetrically swelling shell. Nevertheless, the information about the elastic properties of the cornea is not the complete story of corneal biomechanics. Since many parameters affect deformation, we focus on its geometric aspects rather than discussing the elastic properties of the cornea. It entails defining the displacements on all spheroid corneal geometries from the initial to the final one.
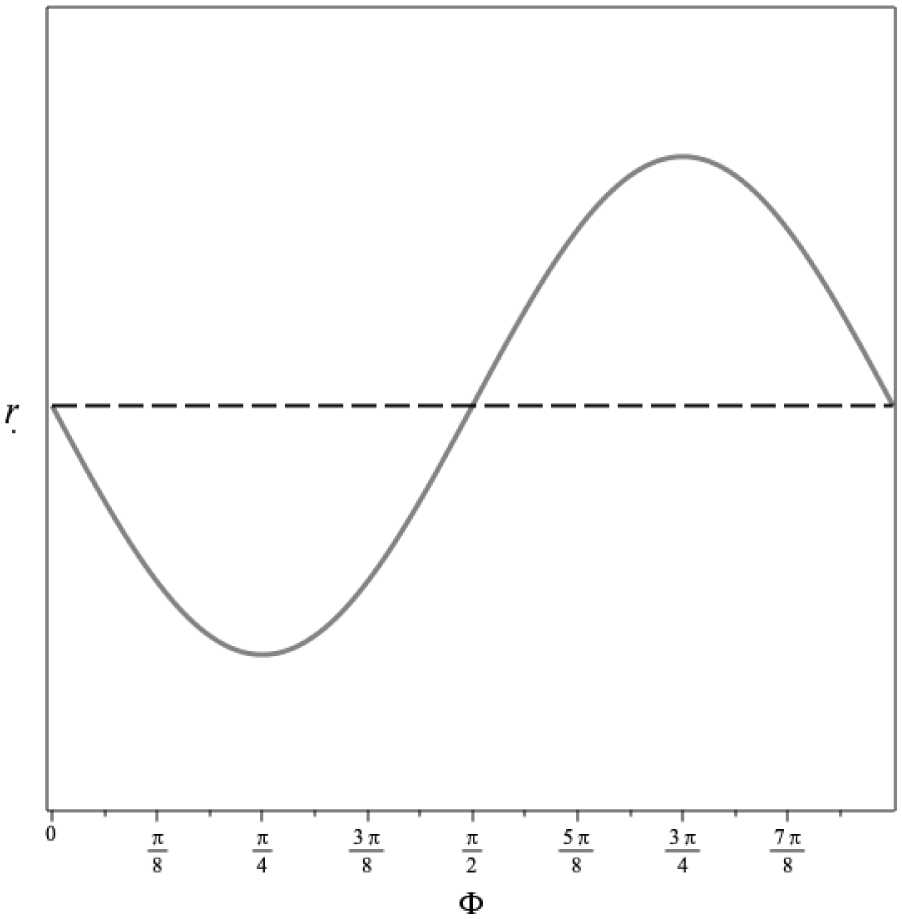
The representative graph of the change in radius.
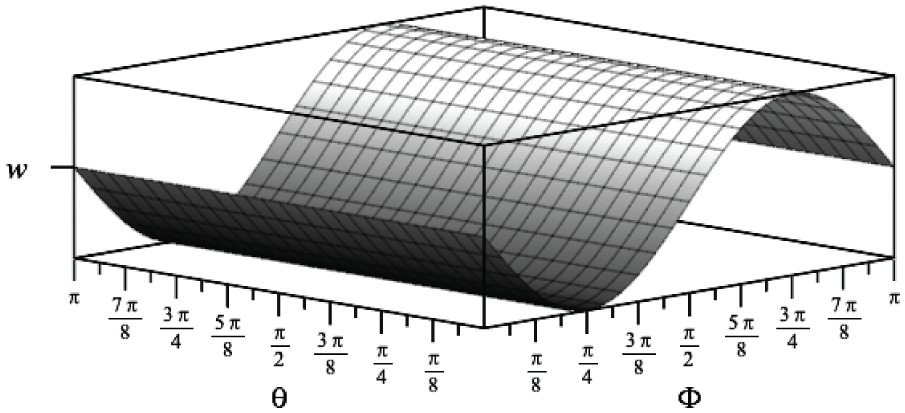
The representative graph of radial displacement.
A judicious choice of displacement direction, informed by effective pressures, can shape the deformed surface. Here, we monitor the deformation to understand its geometry using the method mentioned above. This led us to understand which forces act on the cornea’s surface, how they act, and in which direction they exert their effect. Thus, the displacement equation was consistent with the assumed geometry, and the results confirmed that the correct deformation was obtained. The asymmetric shapes, as shown in Figures 9 and 10 are the best proof of this.
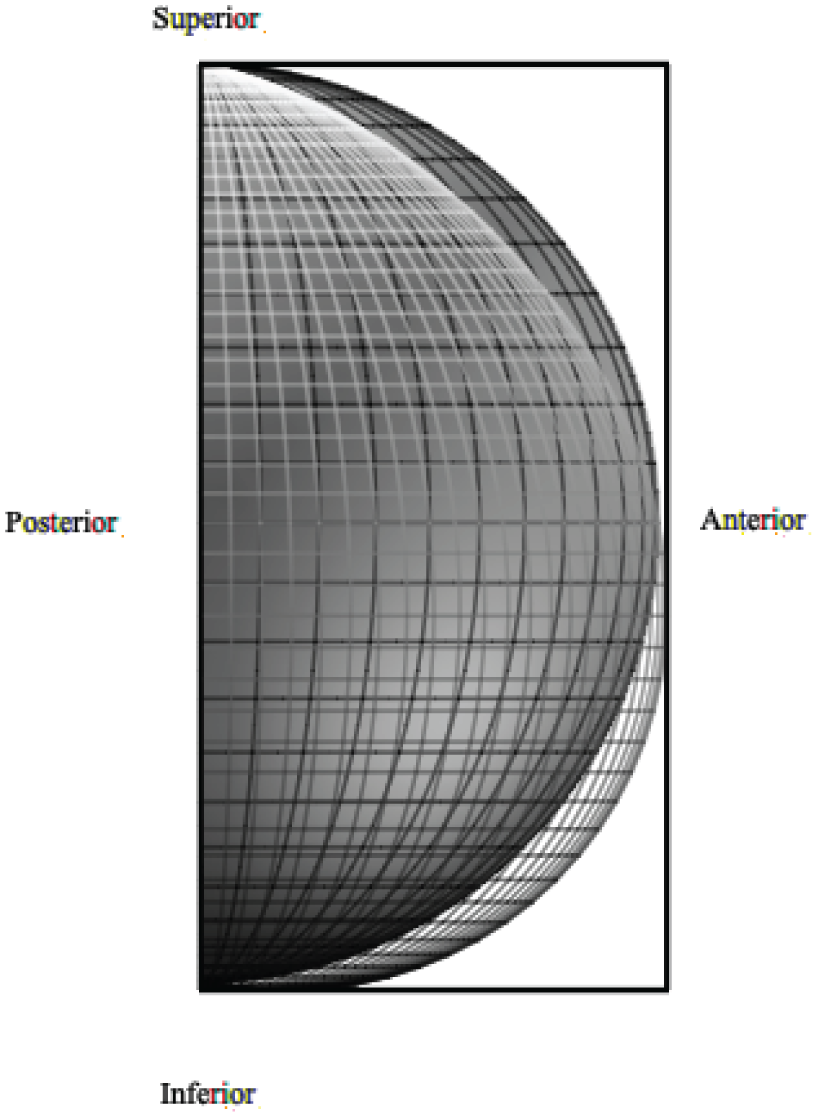
(Patch) symmetric corneal geometry and (line) the resulting corneal geometry after the asymmetric displacement.
Pre(line)-post(point) cross-sectional view of corneal deformation.
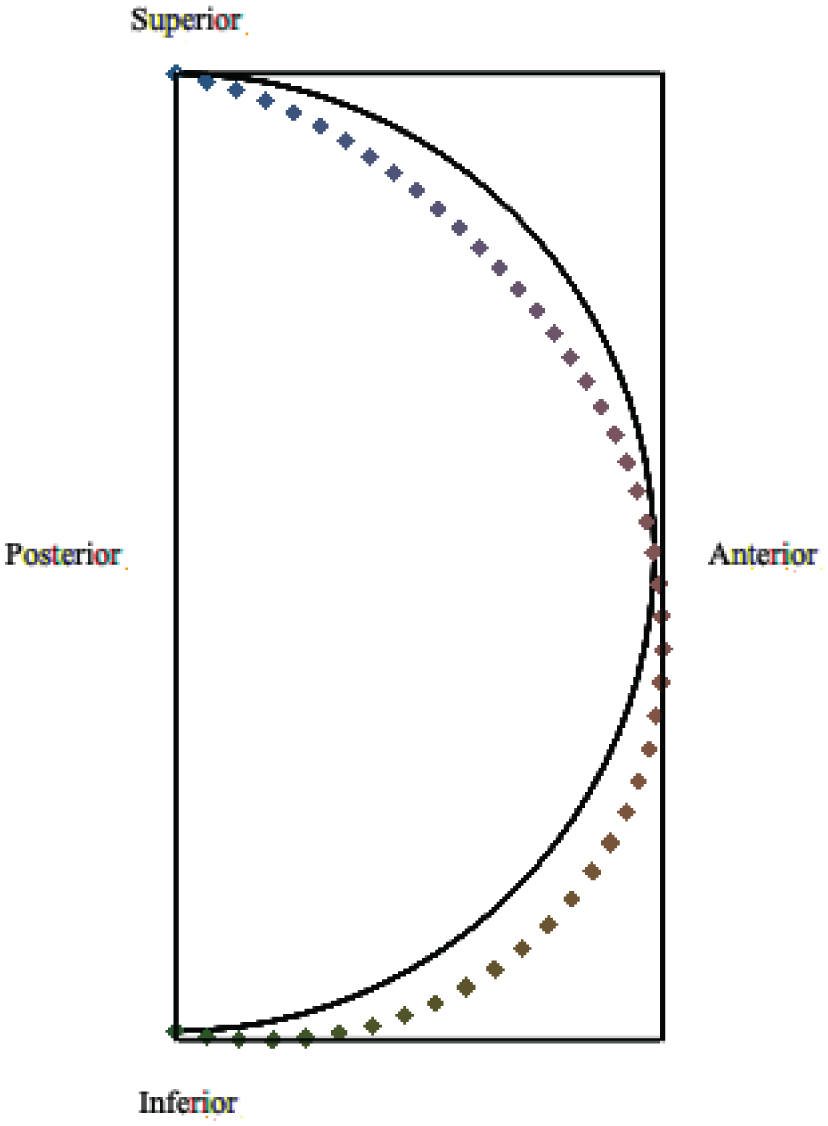
The deformation direction can be indicated through arrows, as shown in Figure 11, and is consequently deduced to be mainly in the anterior–inferior direction, which is consistent with the test data [9,37,41,51].
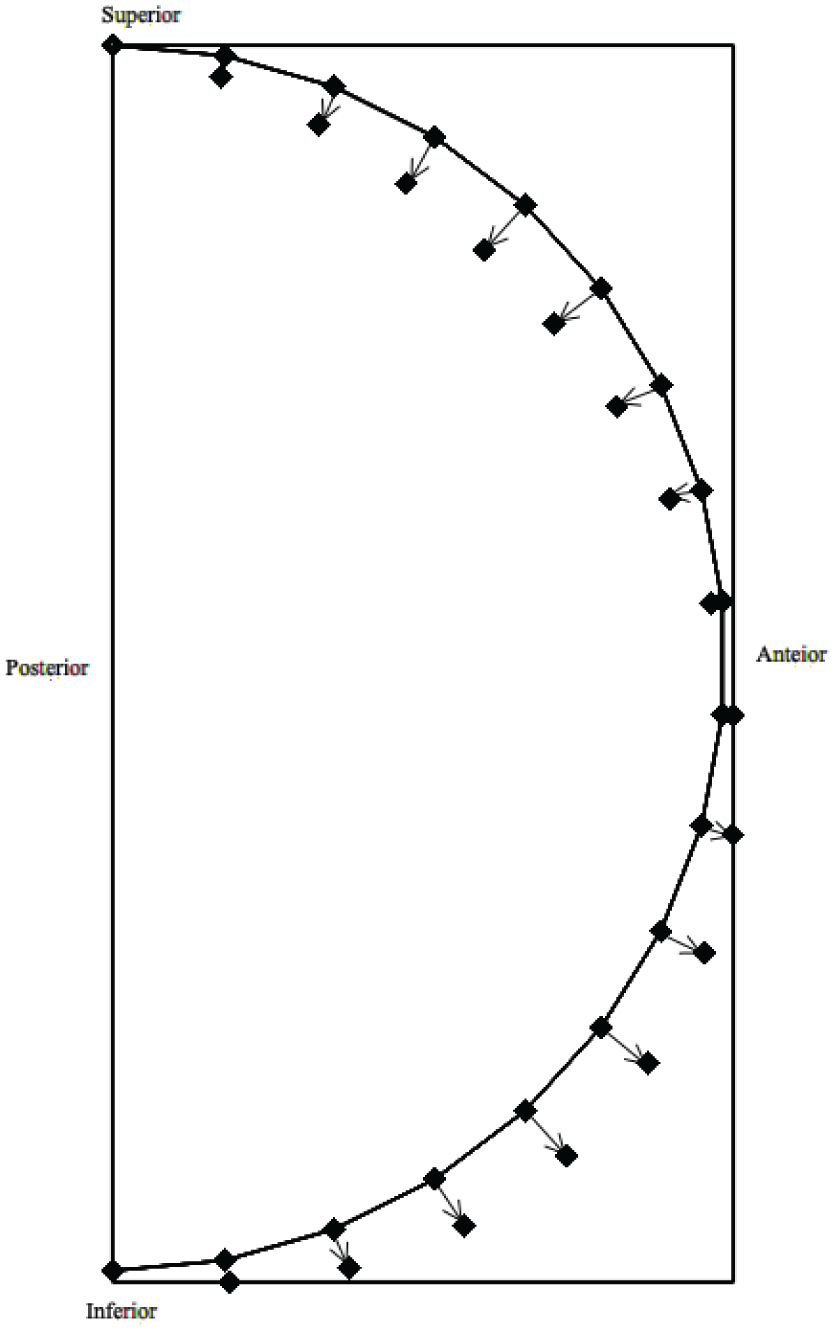
Pre(line)-post(point) cross-sectional point to point evolution of corneal deformation.
4. Discussion and conclusion
This study aims to develop a comprehensive mathematical model that accurately represents corneal deformation under swelling and shear forces, thereby capturing the patterns of corneal curvature. It describes the cornea as a spherical shell, applies shell theories, such as membrane and bending theories, and elucidates the patterns of non-axial deformations, which are crucial for understanding mechanical stress–strain responses. It also seeks to investigate the relationship between swelling and shear, particularly their impact on corneal deformation and disease progression, such as in KC. In addition, it aims to analyze the biomechanical properties of the cornea and its response to changes in IOP and to external corneal load. We strive to determine how IOP and corneal load, which lead to permanent structural changes, contribute to the patterns of corneal deformations.
We mentioned that swelling and shear forces must be evaluated together in corneal research. Membrane theory is primarily used to analyze swelling-induced increases in corneal load without a shear effect, as reported in the literature. On the contrary, the shear effect was discussed using bending theory, which is independent of its origin. In this paper, we ask that the mathematical model be investigated using two approaches within the same method. Swelling is attributed to the nonlinear radial growth of the cornea under additional load. The shear effect is introduced via bending theory on a spherical shell. In the long term, we observe the collective impact of both. Naturally, solutions yield volumetric growth, with the expected angular distortion. This model can mimic the eventual deformation of the cornea, or the deformed cornea, as in KC. Our geometric model generates surface patterns of the cornea, applied in conjunction with membrane theory, on a spherical shell subjected to external forces. Eventually, we encounter non-axial deformation in the geometry, indicating that it is not axially symmetric.
In this paper, we complement the analytical results with representative KC topography and schematic illustrations to relate the predicted deformation pattern to reported KC features in the literature. This comparison is qualitative and relies on experimental data (e.g., the direction of apex displacement, curvature changes, and surface-pattern evolution), with the interpretation intended to demonstrate consistency with established observations. Although the present model is validated qualitatively, an inverse FE analysis employing an effective model can be used in future work to estimate biomechanical parameters. This model predicts changes in displacement and curvature, which can be compared directly with measured shape evolution. However, in this paper, we limit our intention to provide qualitative consistency between the model and the deformation patterns of KC.
Swelling is a suspected process causing corneal diseases, such as KC. The swelling–shear relation must be analytically exposed to the correlations between mechanical processes and corneal deformation. Corneal ectatic diseases are closely linked to corneal biomechanics, particularly swelling, which can lead to persistent angular distortion. The amount of angular distortion can damage lamellae inside the cornea, altering the material’s elasticity and stiffness. We plan to develop more sophisticated models, including those that incorporate inner processes. Furthermore, the sclera’s role in reshaping the corneal surface is vague. Our analysis assumes that the limbus, as a stationary boundary, causes resonance modes on the corneal surface, which tear down the cornea. This observation suggests that the sclera’s surface also adjusts to damp possible oscillations on the cornea’s surface.
Footnotes
Ethical approval and consent to participate
Not applicable.
Author contributions
U.S. contributed to the investigation, conceptualization, methodology, modeling, and writing—original draft preparation. F.T. conducted the literature review and wrote the introduction, with review and editing and supervision. E.U.S. conducted the literature review and wrote the introduction, with review, editing, and supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for publication
Not applicable.
Informed consent
All the authors have approved the manuscript and the submission.
Data availability statement
No data sets were generated or analyzed during the current study.