Abstract
Purpose:
To compare the performance of two preservative-free artificial tears with sodium hyaluronate (SH) in patients with moderate-to-severe dry eye: a 0.24% SH eye drop with carbomer (CB) and triglycerides (TGs) as lipids (SH-CB-TG), and a comparator with 0.18% SH (C-SH).
Methods:
Relief of Eye Surface by Triple Action (RESTA), a multicenter, investigator-masked, noninferiority study (NCT03368404) assessed patients with moderate-to-severe dry eye (N = 79). Patients were randomized 1:1 to receive drops containing SH, CB, and medium-chain TGs [0.24% SH, 0.0625% CB, and 0.2% TGs; SH-CB-TG (Artelac® Complete); n = 45] versus an SH-only drop (0.18%; C-SH; n = 34) instilled 3–6 times daily for 90 days. The primary endpoint was change from baseline to Day 28 in total ocular surface fluorescein staining (OSFS) with noninferiority defined as a between-group 95% confidence interval (CI) upper bound <2 grades. Secondary endpoints included global OSFS at Day 90, individual OSFS component scores, dry eye symptoms, tear film break-up time, Schirmer’s test, quality of life (QoL) measures, and instillation frequency.
Results:
At Day 28, mean OSFS score in the SH-CB-TG group decreased by 2.07 ± 1.67 versus 1.50 ± 1.64 for C-SH; the 95% CI upper limit was 0.13, confirming noninferiority. Dry eye signs, symptoms, and QoL measures improved continuously in both groups, with the SH-CB-TG group showing significantly improved QoL globally at Day 90 (P = 0.0306) and across several individual QoL parameters, plus an acceptable safety profile.
Conclusions:
Including medium-chain TGs in dry eye drops provides noninferior improvements of dry eye signs and symptoms versus viscosity agent-only drops and may enhance patient QoL.
Keywords
Introduction
Dry eye symptoms affect an estimated 344 million people globally, 1 with its incidence exacerbated by modern lifestyles, including contact lens and digital screen use. 2 Dry eye is a multifactorial condition: while patients with dry eye share ocular discomfort and visual disturbance, 3 the complexity of the tear film coupled with a gamut of environmental stressors allow dry eye to arise from a number of possible underlying causes. 4 This complexity makes restoring proper tear film function a complex clinical challenge, often requiring personalized management beyond aqueous tear replacement.5–7
While aqueous artificial tears are the mainstay of initial dry eye treatment, they often have short ocular surface residence times 8 and may provide limited symptom relief. Adding viscosity modifiers such as sodium hyaluronate (SH) and carbomer (CB) to the formulation can increase artificial tear viscosity.9–11 These mucoadhesive polymers provide hydration while increasing the residence time of larger molecules important for ocular surface health, such as growth factors and triglycerides (TGs).9,11 Additionally, many patients with dry eye also have at least some tear film hyperevaporation due to insufficiency of the tear film’s overlying, anti-evaporative lipid layer, which can further promote tear film instability.4,12,13 To this end, several marketed tear substitutes have emerged containing not only viscosity modifiers but also medium-chain TGs to supplement the tear film’s lipid layer and inhibit evaporation, therefore potentially providing relief of dry eye signs and symptoms. 14
The multi-component tear substitute SH-CB-TG (marketed as Artelac® Complete MDO/Artelac® Triple Action/Nereya by Bausch + Lomb) was designed to provide long-lasting moisturization via the viscosity agents SH and CB and inhibit tear film evaporation via TG. Previously, short- and long-term effects of SH-CB-TG were assessed in 32 patients with mild-to-moderate evaporative dry eye at 90 min following instillation and after 21 days of use. At both the short- and longer-term time points, eyes receiving SH-CB-TG had significantly increased tear film stability as measured by noninvasive tear film break-up time (TBUT), and reduced tear evaporation. 15 Additionally, these patients reported significantly improved symptoms on the Ocular Surface Disease Index (OSDI) questionnaire.
To further understand the utility of SH-CB-TG in improving the signs and symptoms of dry eye, the Relief of Eye Surface by Triple Action (RESTA) study (NCT03368404), a 3-month, investigator-masked, randomized study, was conducted to demonstrate the noninferiority of SH-CB-TG to an SH-only comparator (C-SH; 0.18% SH from fermentation). C-SH was selected as a comparator because, like SH-CB-TG, it is a preservative-free—as recommended by the Tear Film & Ocular Surface Society Dry Eye Workshop II (TFOS DEWS II) 5 —commercially available (marketed as VISMED® MULTI Eye Drops, TRB Chemedica International, Carouge, Switzerland), and supported by a substantial number of studies demonstrating its utility in treating the signs and symptoms of dry eye.16–20
Methods
Study design
The RESTA study (NCT03368404) was a 3-month, investigator-masked, randomized study conducted over 106 ± 10 days at 16 sites across France. Similar to a study previously performed in France and Belgium, 21 the RESTA study assessed the noninferiority of two preservative-free drops: the lipid-containing study eye drop SH-CB-TG and the comparator C-SH.
The RESTA study was performed in compliance with the Declaration of Helsinki, Good Clinical Practices, International Organization for Standardization 14155, and local regulations. The study was approved by an independent ethics committee (Comité de Protection des Personnes du Sud-Ouest et Outre Mer IV), and all patients gave written informed consent.
Study population
Included patients were adults with moderate-to-severe dry eye defined as a score of 4–9 on the Oxford scheme of ocular surface fluorescein staining (OSFS), 22 a TBUT ≤10 s in at least one eye, and a symptoms score ≥1 for at least 2 out of 7 symptoms assessed on a 0-to-4 scale (sensation of dryness, foreign body, burning, stinging, itching, blurred vision, and sensitivity to light).
In addition, patients needed to have used a tear substitute for at least 2.5 months prior to screening, have a best corrected distance visual acuity of 0.1 or better, and approved contraception and negative urine pregnancy test for those of childbearing potential. Patients were excluded if they had severe blepharitis or other eye disease, used contact lenses, had a history of eye surgery or trauma in the previous 90 days, or suffered from ocular allergic disease in the past year. Patients were also excluded if they used ocular medications or therapies other than tear substitutes.
Study procedures
Patients were screened 14–16 days prior to randomization. In each patient, the study eye was the eye that fit the inclusion criteria and had the highest total ocular surface staining score at baseline. If both eyes were eligible and had the same ocular surface staining score, the right eye was used as the study eye.
Included patients underwent a 2-week run-in period where they received preservative-free povidone 2% (Aqualarm® UP, Dr. Mann Pharma, Berlin, Germany and Bausch + Lomb UK Ltd, Kingston, UK) up to 6 drops daily. On study Day 0, patients were randomized 1:1 to receive SH-CB-TG or C-SH, both instilled 3–6 times per day. Patients received a follow-up safety call after 1 week and were assessed at follow-up visits on Days 28 ± 3 and 90 ± 10.
Study materials
SH-CB-TG, C-SH, and povidone 2% are all Conformité Européenne-marked class IIb medical devices. They are all preservative-free and used in a multidose manner. To maintain patient masking, the product bottles had identical over-labels and boxes. On site, they could be identified only by the randomization number. The site dispensed and retrieved sealed boxes to maintain product masking for the investigator.
Endpoints and assessments
The primary effectiveness endpoint was the mean change from baseline (CFB) at 1 month (Day 28 ± 3 days) in study eye total OSFS score, which combines corneal, nasal, and temporal bulbar conjunctival fluorescein staining, each graded 0–5 using the Oxford scheme. 22
Secondary effectiveness endpoints included raw global OSFS at Day 28, and at Days 28 and 90, CFB in each of the OSFS components, patient-reported dry eye symptoms (sensation of dryness, foreign body sensation, burning, stinging, itching, blurred vision, and sensitivity to light, each on a 0-to-4 scale), and TBUT. An unanesthetized Schirmer’s test was performed on Day 28, and patients reported their frequency of daily instillations at Day 90. Additionally, patients also completed the Ocular Surface Disease-Quality of Life (OSD-QoL®) validated questionnaire 23 at Day 90.
Safety was evaluated as the incidence of adverse events (AEs), and tolerability upon instillation was also monitored.
Statistical analysis
Sample size was calculated using a validated macro program developed under Statistical Analysis Software® (SAS, Cary, NC) version 9.4. Assumptions included a unilateral α risk of 2.5%, a β risk of 10% (90% power), and a standard deviation (SD) of 2.5 on the primary endpoint; from this, it was determined that each treatment group would require 33 patients to demonstrate non-inferiority. With two treatment groups, this yielded a total group size of 66 patients. Based on the analysis of covariance (ANCOVA) model, a 95% two-sided confidence interval (CI) was calculated for the effect difference. Additionally, based on previous work, the noninferiority limit was set to 2 grades on the Oxford scheme.18–20
The primary analysis was performed on the per-protocol (PP) population, as is typically considered best practice in noninferiority studies, which included all patients who received treatments, had the primary endpoint assessment at Day 28, and had no major protocol deviations (ie, no impact on primary endpoint). Supporting analyses were performed on the intent-to-treat (ITT) population, which included all patients who received treatment and had at least one post-baseline assessment. Safety endpoints analyses were performed on the safety population (ie, all patients who received treatment and had follow-up information).
The primary endpoint was analyzed using an ANCOVA model with main effects of treatment and baseline OSFS score as covariates. A 95% two-sided CI based on the ANCOVA model was computed for the effect difference. Between-group differences were considered noninferior if the upper bound of the 95% CI was <2 grades in the PP analysis.
The same ANCOVA model was used to analyze secondary continuous endpoints. Categorical endpoints were analyzed using Fisher’s exact test or chi-square test where appropriate. All the statistical tests were performed at a significance level of α = 0.05.
Results
Study population
From the 87 randomized patients, 82 completed the study, and 5 patients withdrew prematurely: 3 had AEs requiring early withdrawal, and 2 withdrew consent (Fig. 1).
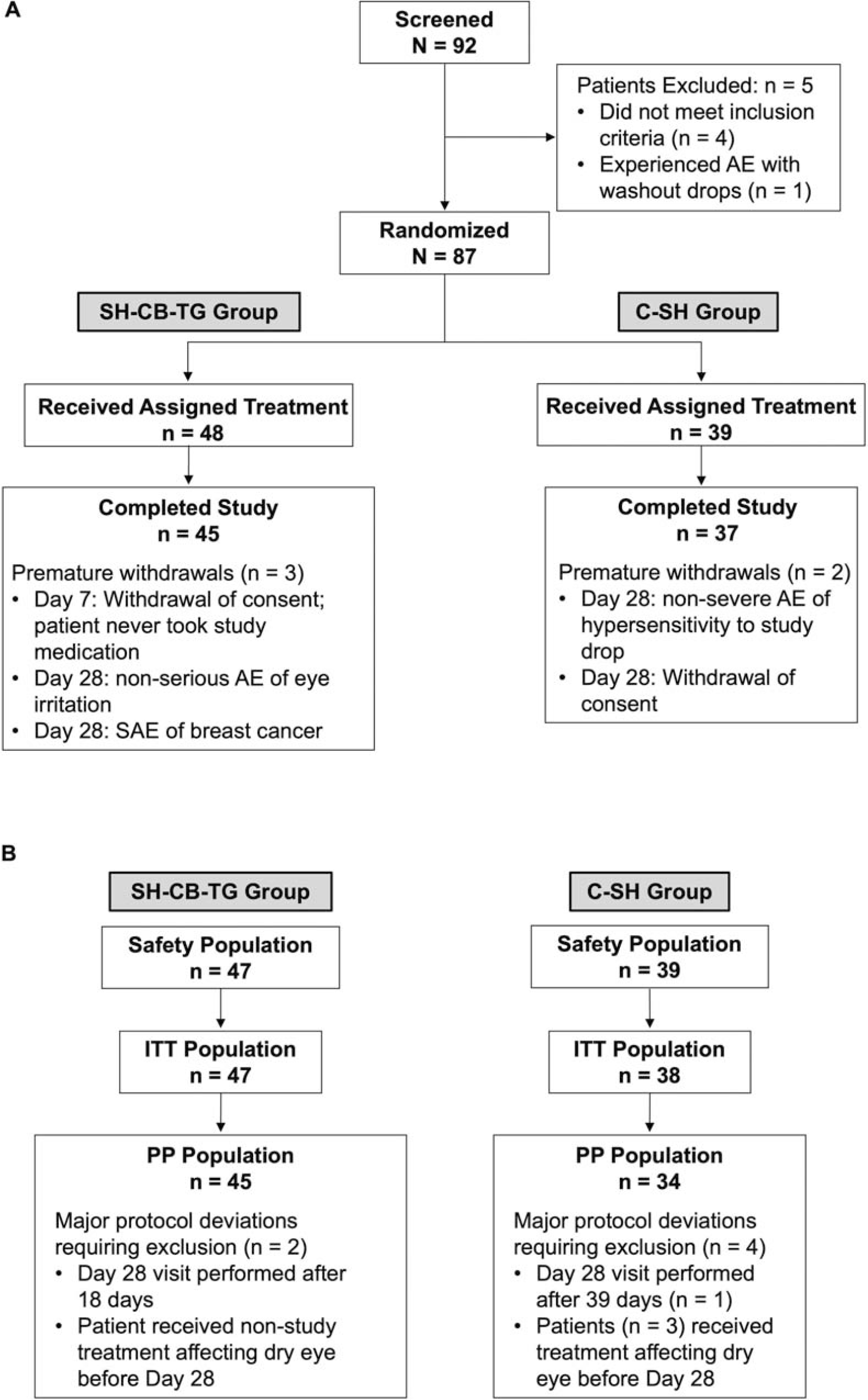
Study flow and populations. (
Overall, demographic and baseline characteristics were similar between treatment groups, though patients in the SH-CB-TG group had a slightly higher global dry eye symptom score (sensation of dryness, foreign body, burning, stinging, itching, blurred vision, and sensitivity to light, each graded from 0 to 4) at baseline (Table 1). The trend toward an older age (mean 64 years) range and more female patients (69%) is in accordance with the previous dry eye epidemiology studies in which most patients with dry eye were women aged over 50. 24 Demographic and baseline characteristics of the PP population are consistent with those of the ITT population (Supplementary Table S1).
Demographic and Baseline Characteristics (ITT Population)
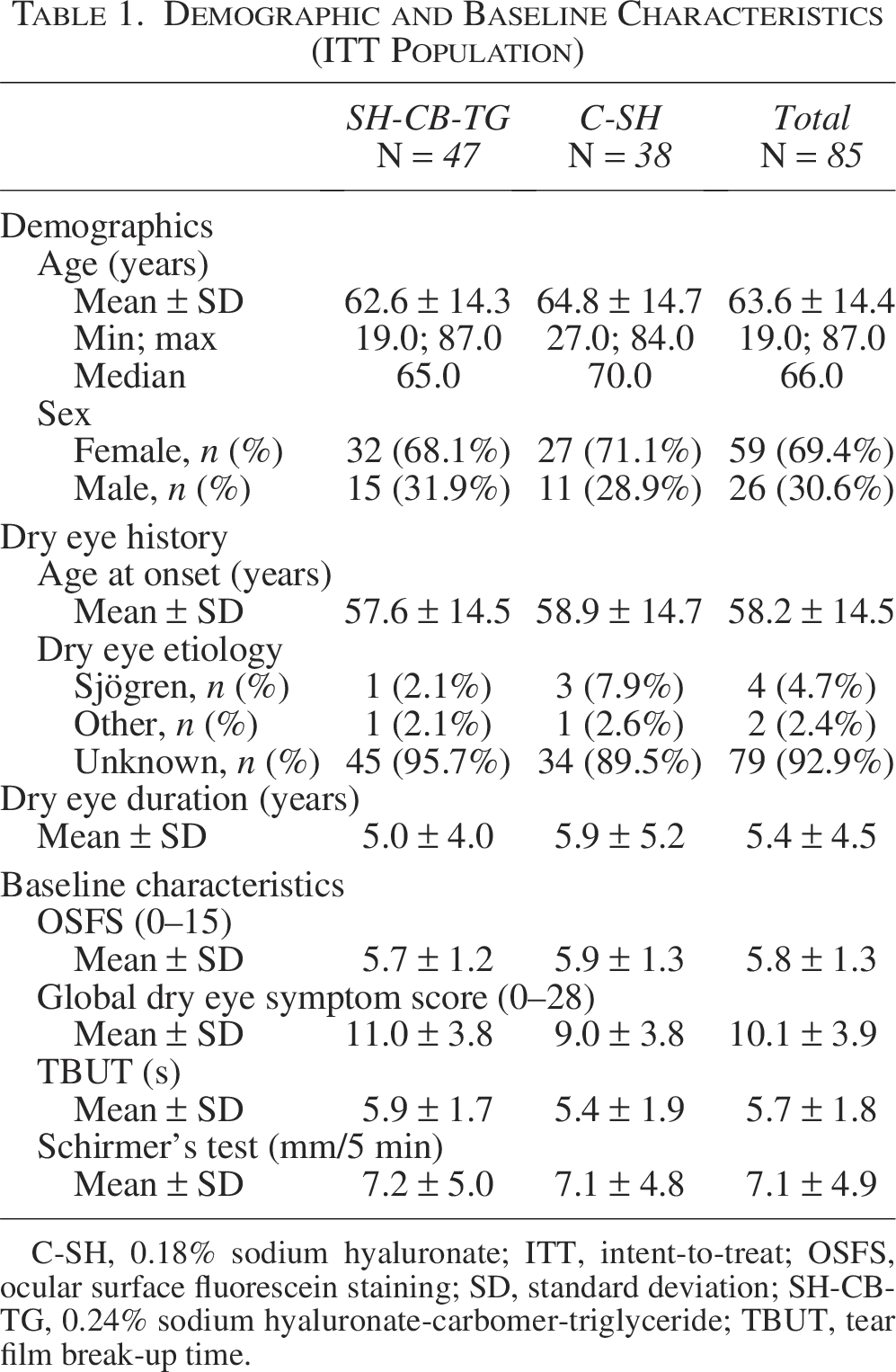
C-SH, 0.18% sodium hyaluronate; ITT, intent-to-treat; OSFS, ocular surface fluorescein staining; SD, standard deviation; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride; TBUT, tear film break-up time.
Primary and secondary effectiveness outcomes
At Day 28, the mean global OSFS score ± standard error (SE) decreased by 2.07 ± 1.67 in the SH-CB-TG group and 1.50 ± 1.64 in the C-SH group (Fig. 2). The upper bound of the 95% CI of the adjusted difference between the CFB values of both groups (−0.60 ± 0.37) was 0.13, less than the predefined noninferiority upper limit of 2 Oxford scheme grades, within the noninferiority range.
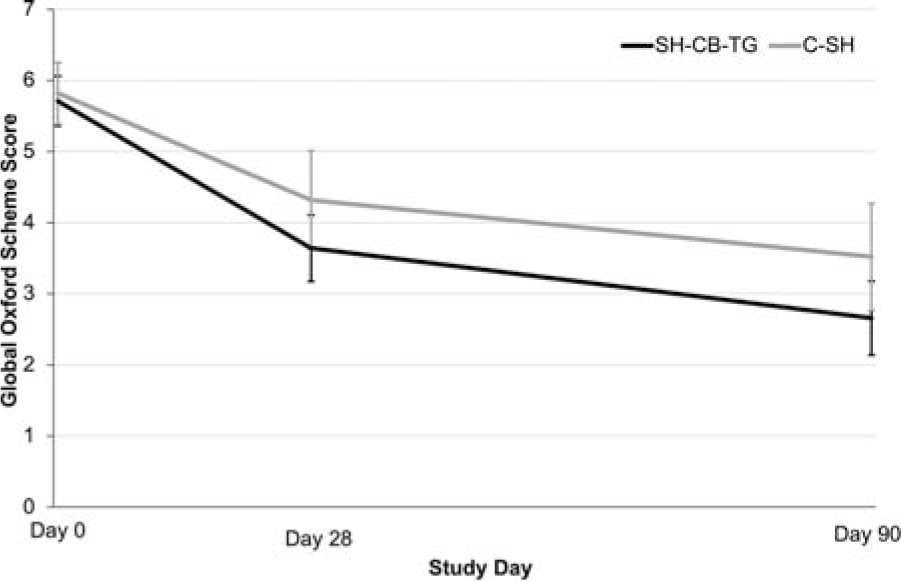
Ocular surface fluorescein staining score (mean ±95% CI; PP population). C-SH, 0.18% sodium hyaluronate; CI, confidence interval; PP, per-protocol; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride.
All individual OSFS components improved from baseline in both the SH-CB-TG and C-SH groups, with numerically better scores for the SH-CB-TG group, none of which reached statistical significance (Table 2).
CFB in Individual OSFS Component Scores at Days 28 and 90 (PP Population)
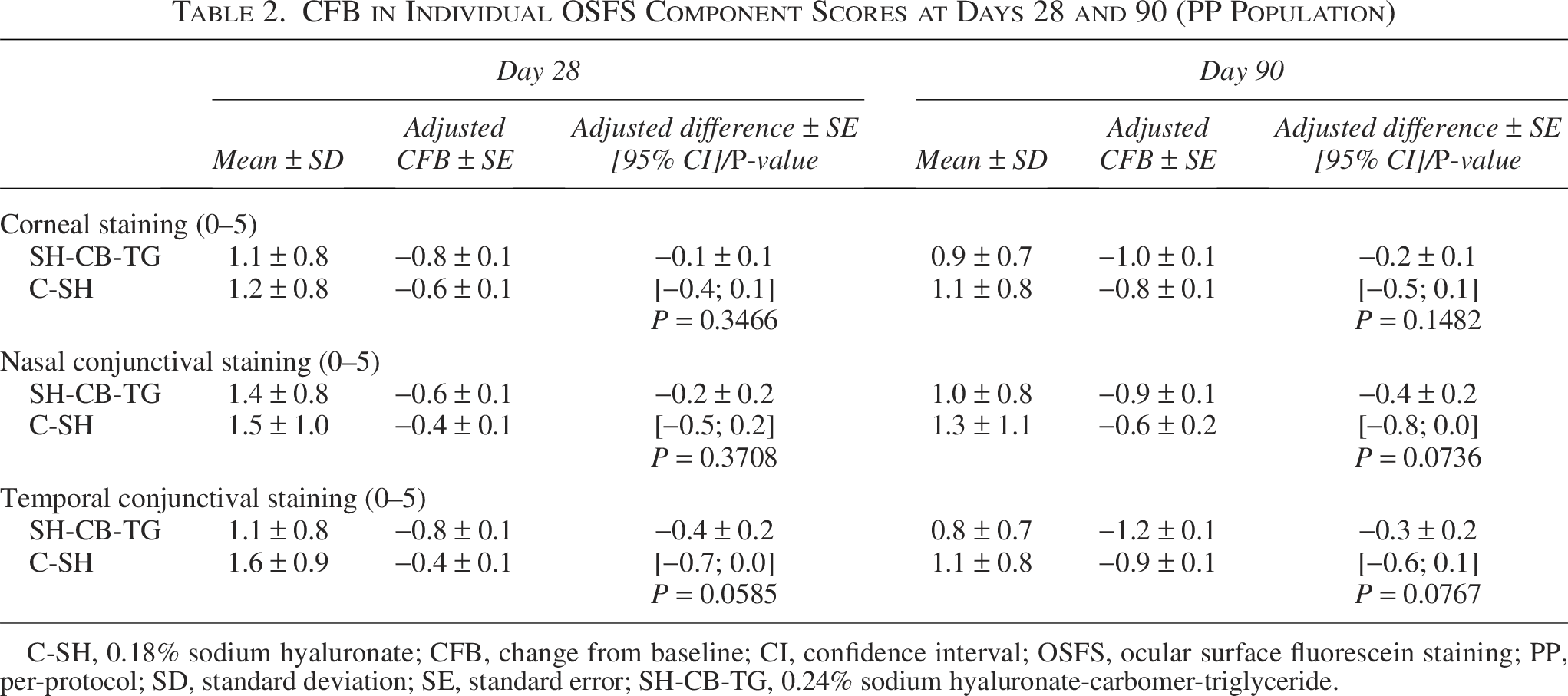
C-SH, 0.18% sodium hyaluronate; CFB, change from baseline; CI, confidence interval; OSFS, ocular surface fluorescein staining; PP, per-protocol; SD, standard deviation; SE, standard error; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride.
There were also improvements in TBUT and Schirmer’s test scores in both groups, though without significant difference (Table 3). The improvement of the global sum score of dry eye symptoms was significantly better in the SH-CB-TG group at both Day 28 (P = 0.0176) and Day 90 (P = 0.001) than in the C-SH group (with an adjusted difference of –1.75 ± 0.72 and –2.42 ± 0.71, respectively; Fig. 3).
CFB in TBUT And Schirmer’s Test Scores following SH-CB-TG or C-SH (PP Population)
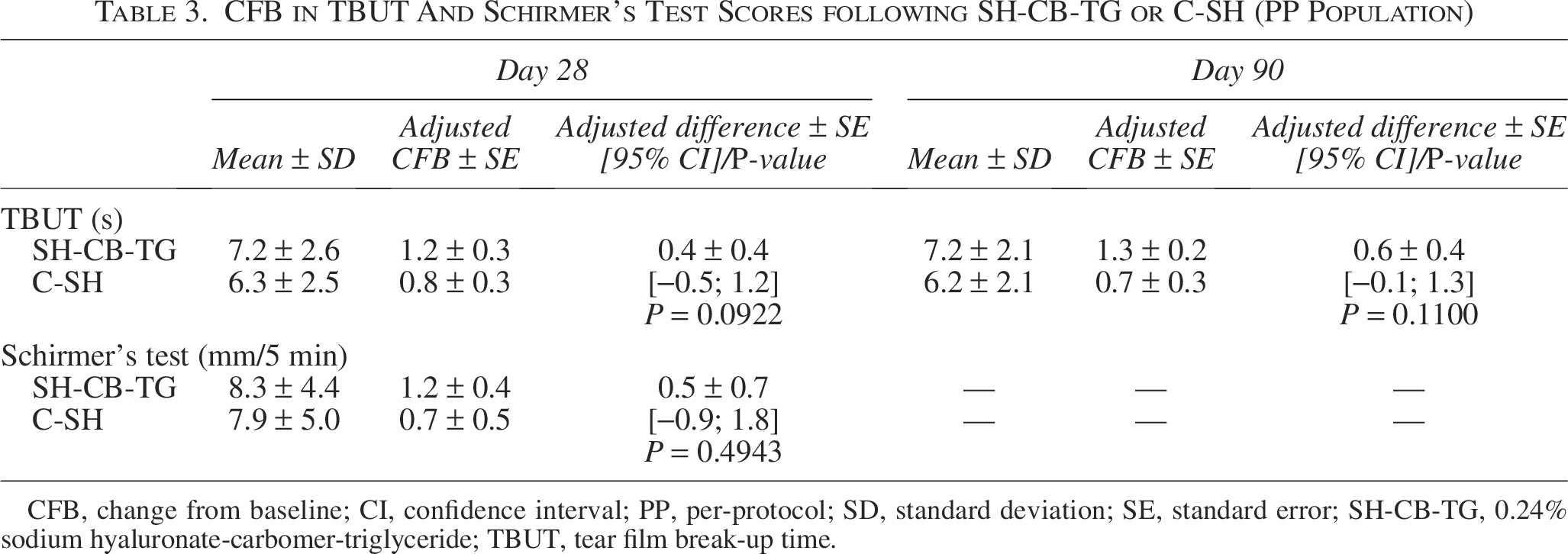
CFB, change from baseline; CI, confidence interval; PP, per-protocol; SD, standard deviation; SE, standard error; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride; TBUT, tear film break-up time.
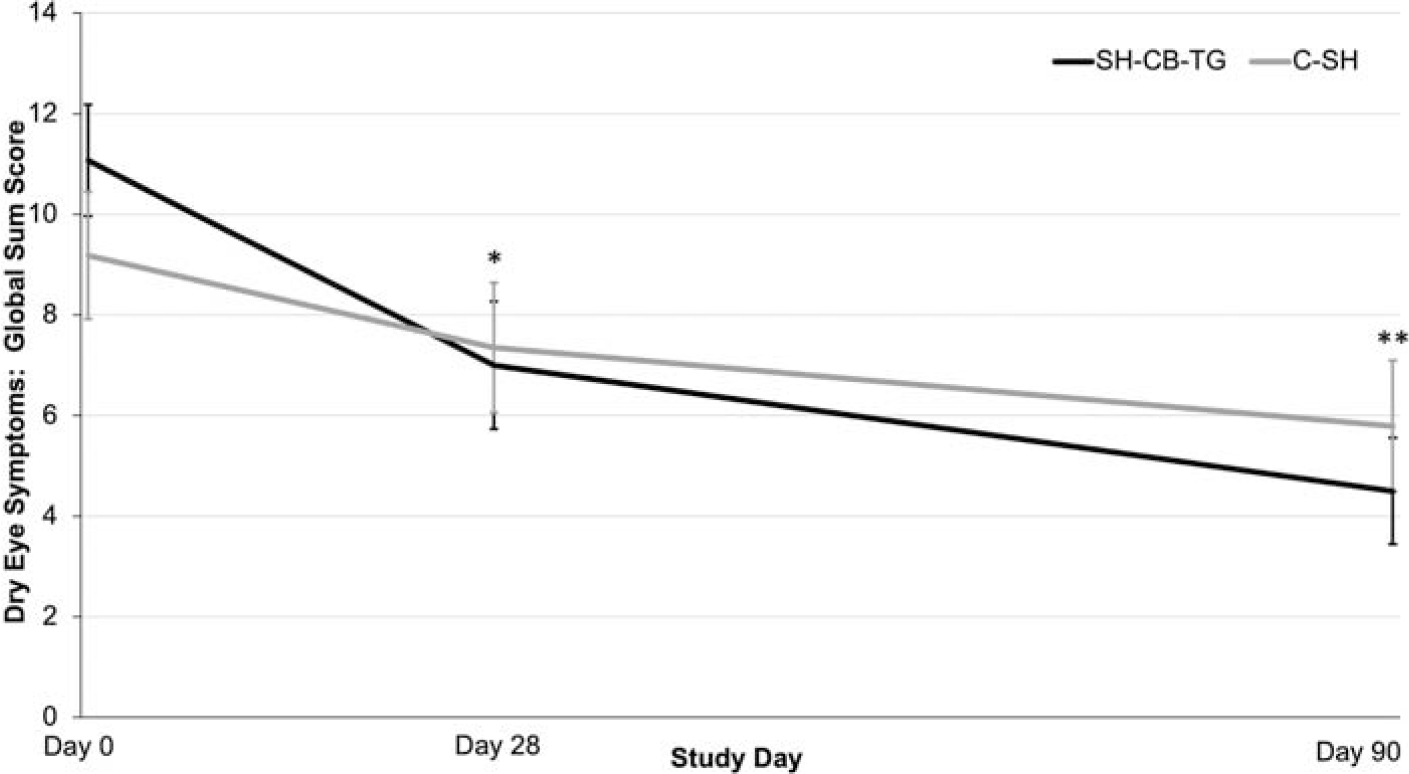
The global sum score of dry eye symptoms (mean ±95% CI) (PP population). C-SH, 0.18% sodium hyaluronate; CI, confidence interval; PP, per-protocol; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride. *P = 0.0176; **P = 0.001.
In alignment with symptom improvement, OSD-QoL scores were numerically improved at Day 90 in 6 dimensions in the SH-CB-TG group and 5 dimensions in the C-SH group (Fig. 4). In 3 dimensions, the SH-CB-TG group was significantly improved (P < 0.05) versus the C-SH group: daily activities, handicap and work difficulties, and acknowledgement of the disease. Furthermore, there was a significant difference in favor of SH-CB-TG versus C-SH for the OSD-QoL global question, which was assessed separately (P = 0.0306).
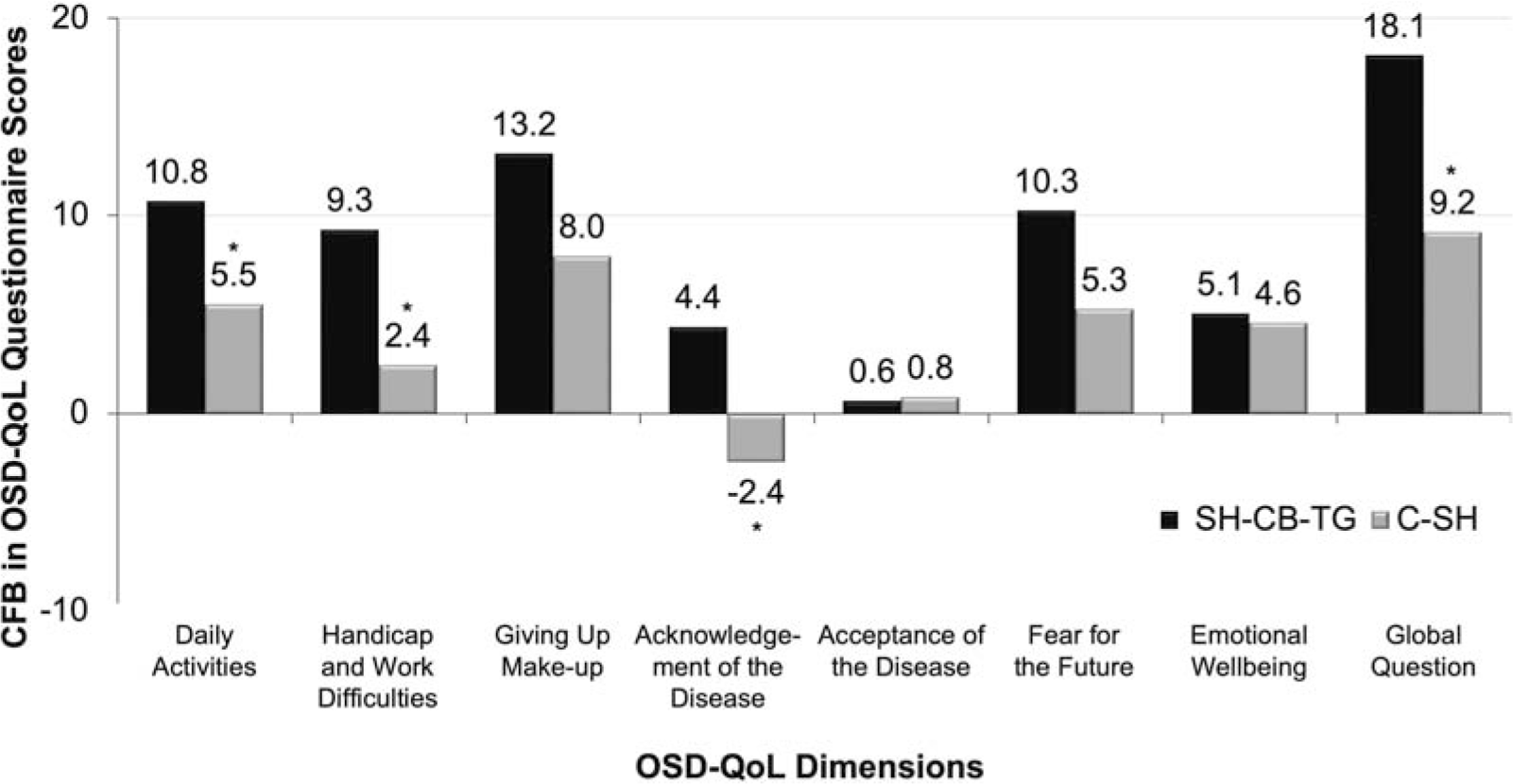
CFB to Day 90 in QoL assessments using the OSD-QoL questionnaire (PP population). C-SH, 0.18% sodium hyaluronate; CFB, change from baseline; OSD-QoL, Ocular Surface Disease Quality of Life; PP, per-protocol; QoL, quality of life; SH-CB-TG, 0.24% sodium hyaluronate-carbomer-triglyceride. *P < 0.05.
Concerning daily frequency of instillations by study end, patients in the SH-CB-TG group instilled the SH-CB-TG drop significantly less frequently compared with those in the C-SH group (means ± SD of 3.4 ± 0.6 vs. 4.0 ± 1.0, respectively; P = 0.0017).
Safety
The safety of the SH-CB-TG and C-SH drops, as measured by the incidence of AEs, was comparable during the 3-month administration period, and both drops were well-tolerated. Three patients withdrew prematurely due to AEs. In the SH-CB-TG group, one patient experienced a serious AE of breast cancer, which was assessed as unrelated to the study drop, and another patient had non-serious moderate eye irritation possibly related to the product. The third patient, who withdrew from the C-SH group, developed a severe eye allergy assessed by the investigator as certainly related to the product. Altogether, ocular AEs were reported by six patients in each group, where three ocular events per group were assessed as related to the assigned product: eyelid edema, blurred vision, and eye irritation in the SH-CB-TG group, and eye allergy, eye pain, and eye discharge in the C-SH group. No device deficiency was reported. Visual acuity and slit-lamp examinations showed no safety concerns and were comparable between the two groups. Conjunctival hyperemia was the most frequent sign at baseline but improved with the treatments. In both groups, at least 80% of patients agreed that the eye drops were comfortable upon instillation. At all study points (Days 7, 28, and 90), there was no statistically significant difference between the SH-CB-TG and C-SH groups in comfort during drop instillation.
Discussion
RESTA was a 3-month, investigator-masked, randomized study designed to establish the noninferiority of two preservative-free drops, one containing medium-chain TGs (SH-CB-TG) and an SH-only comparator (C-SH). SH is well-known as a safe and effective treatment for dry eye,16–20 but because of the high prevalence of dry eye with an evaporative component 1 , particularly due to Meibomian Gland Dysfunction 4 , this study evaluated the effects of adding medium-chain TGs toward alleviating dry eye signs and symptoms. The data presented here demonstrate that the SH-CB-TG eye drop is noninferior to the C-SH drop in relieving dry eye signs and symptoms from baseline to Day 28 in patients with moderate-to-severe dry eye, as measured by global OSFS score. In addition to meeting its primary endpoint, SH-CB-TG was not significantly different than C-SH across a number of effectiveness measures, including individual OSFS components and TBUT at Day 28 or Day 90, and Schirmer’s test at Day 28. Despite noninferiority in objective measures, the SH-CB-TG group reported significantly greater improvements in QoL versus the C-SH group.
The effectiveness of SH-CB-TG in reducing dry eye signs and symptoms in the present study is consistent with previous work in dry eye using similar molecules.14,25 A study comparing a cationic emulsion (CE) with mineral oil nanodroplets to C-SH found that, similar to the present study, CE + mineral oil drops was noninferior to C-SH drops in improving ocular surface staining versus baseline at 28 days or 3 months. 26 Another study comparing medium-chain-TG drops versus viscosity-agent-only drops [hydroxypropyl (HP)-guar gel] found the TG-containing drop noninferior to the HP-guar gel drop at 2 and 4 weeks; though both provided dry eye relief 25 Schirmer’s test values showed significantly greater improvements from baseline in tear film production at both 2 and 4 weeks in the TG group over the HP-guar gel group (P < 0.05). 25
In addition to objective dry eye measures, this study assessed the patient experience of dry eye using patient-reported outcomes. The SH-CB-TG group reported significantly better improvements in global dry eye symptoms versus at both Day 28 and Day 90 (P = 0.0176 and P = 0.001, respectively) compared with the C-SH group. Previous work has also examined patient-reported outcomes regarding dry eye symptoms with positive results.25,26 Here, patient QoL was assessed using the OSD-QoL questionnaire, 23 where patients in the SH-CB-TG group experienced significant improvements versus the C-SH group in multiple individual QoL measures—daily activities, handicap and work difficulties, acknowledgement of the disease—and in global QoL score, which is perhaps a point of further exploration.
Lastly, the SH-CB-TG drop demonstrated similar safety data to the C-SH drop, with low rates of ocular AEs and the only serious AE (breast cancer) assessed as unrelated to the study drop. Overall, both drops were well-tolerated.
As with all clinical trials, some limitations exist in the current study. Due to recruitment difficulties across study sites, the overall testing population was smaller than initially expected. Because of this, randomization was somewhat suboptimal, leading to more patients receiving SH-CB-TG versus C-SH. The SD of the primary endpoint in both groups, however, was below those used for sample size calculation, so the small study size likely had little effect on the validity of the reported data. Apart from the difference in initial dry eye symptom score, baseline characteristics were relatively comparable between the two groups. Additionally, because dry eye symptoms were more severe at baseline in the SH-TG-CB group than in the C-SH group, it is possible that the greater QoL improvements seen in the SH-CB-TG group were due to there being more room for improvement than in the C-SH group.
Unlike the majority of aqueous drops, the components of the SH-CB-TG drop were thoughtfully selected to help support both the mucoaqueous and lipid tear film layers. This study demonstrated the noninferiority of SH-CB-TG to C-SH in the study population. Its effectiveness combined with its satisfactory safety profile indicates that SH-CB-TG could be a helpful alternative in the management of moderate-to-severe dry eye.
Authors’ Contributions
M.L.: Methodology, supervision, validation, writing—original draft, and writing—review and editing. J.M.B.C.: Writing—review and editing. O.U.G.: Writing—review and editing. K.B.: Writing—review and editing. S.C.J.: Writing—review and editing. The RESTA Study group (P.B., N.B., A.C., A.C.M., F.D.S.E., A.D., C.F., P.G., F.J., A.K., M.L., V.M., J.F.P., P.P., N.R., A.S.): Investigation and resources. Authors had full control of the content and made the final decision on all aspects of this publication.
Footnotes
Acknowledgment
The authors thank the patients for their participation in the study.
Catherine Allaire and Raphaële Siou-Mermet contributed to the design, supervision, analyses, and reporting of the study on behalf of Laboratoire Chauvin (a Bausch + Lomb company) as employees of Laboratoire Chauvin at that time. Monitoring, study management, and statistical analyses were provided by Vivactis C.R.O., France. Medical writing assistance was provided under the direction of the authors by Hayley Clay and Lee-Jay Bannister (Illuminate Medical) and funded by Bausch + Lomb.
Author Disclosure Statement
Marc Labetoulle has acted as an occasional consultant (in the last 5 years) for: Abbvie, Alcon, Allergan, Bausch + Lomb, DMG, Dompé, GSK, Horus, MSD, Nordic, Novartis, PHE, Quantel, Santen, Shire, SIFI, Sun, Théa, and Topivert.
José M Benítez del Castillo has consulted for or received research grants from Alcon, Angelini, Brill, and Bausch + Lomb.
Omür Uçakhan-Gündüz has consulted for or received research grants from Alcon, Allergan, Bausch + Lomb, Santen, and Théa.
Kostas Boboridis has consulted for or received research grants from Allergan, Bausch + Lomb, Santen, and Théa.
Stephanie Joachim has acted as an occasional consultant in the last 5 years for: Bausch + Lomb and ebiga-VISION.
Funding Information
Funding for the study, publication fee, and technical, editorial, and medical writing assistance were provided by Bausch + Lomb U.K. Ltd.
Data Access Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
