Abstract
Purpose:
Refractive surgery may exacerbate the existing dry eye symptoms in patients with dry eye syndrome. Therefore, we explore the therapeutic and protective effects of using optimal intense pulsed light (IPL) therapy combined with meibomian gland expression (MGX) before and after surgery on post-laser corneal refractive surgery dry eyes.
Methods:
This was a prospective, self-controlled, single-center clinical trial. Using optimal IPL therapy and MGX, a total of 68 patients with mild-to-moderate evaporative dry eye or meibomian gland dysfunction who underwent refractive surgery received 2 sessions of treatment. The treatment was administered once a week preoperatively and once a week postoperatively. Baseline measurements were taken before optimal IPL therapy and MGX, and the parameters were assessed at 1 week and 1 month after surgery.
Results:
Out of 68 patients, 54 patients received small-incision lenticule extraction (SMILE), while 14 patients received femtosecond laser-assisted in situ keratomileusis (FS-LASIK) (results primarily reflect SMILE outcomes due to sample imbalance). In the SMILE group, significant improvement was already observed in uncorrected/corrected distance visual acuity (P = 1.42 × 10−3), meibomian gland expressibility score (MGYSS) (P = 0.01), and meibum yielding secretion score (MGS) (P = 5.39 × 10−4) between 1 week before and after the surgery. First noninvasive breakup time (NIBUTf) (P = 8.98 × 10−3), average noninvasive breakup time (NIBUTav) (P = 2.30 × 10−4), and meibomian gland dropout score (bottom) (P = 9.05 × 10−3) were observed to have significant improvement till 1 month after the surgery. Patients in the FS-LASIK group exhibited a comparable trend of change.
Conclusion:
IPL treatment combined with MGX may improve ocular discomfort and dry eye symptoms in MGYSS, MGS, NIBUTf, NIBUTav, and meibomian gland dropout score (bottom) for patients with post-laser corneal refractive surgery dry eyes in short term.
Introduction
With the widespread use of mobile devices in recent years, the number of dry eye patients has been increasing annually, and there is a trend of younger onset age.1,2 Especially for Asian populations, evaporative dry eye and meibomian gland dysfunction (MGD) are the most common types, with an incidence rate reaching up to 80%.3–5 Among these individuals, myopia patients are particularly common. 6 However, corneal refractive surgery used for myopia correction has been shown to induce dry eye symptoms,7,8 further exacerbating the dry eye symptoms in patients who already had dry eye. 9 The postoperative recovery period is long, 10 and in severe cases, it can significantly affect surgical outcomes and postoperative visual quality,11,12 even leading to serious psychological issues in patients.13,14
There are various treatment options available for dry eye currently. 15 Recently, intense pulsed light (IPL) has gained significant attention among clinicians as a noninvasive, relatively safe, fast-acting, and effective treatment method.16,17 Optimized IPL is a technique that optimizes the pulse number and peak of IPL and is widely used in clinical practice.18–21 The principle behind IPL in treating MGD and the related dry eye primarily involves broad-spectrum noncoherent light’s photothermal effect, thermal radiation effect, bactericidal and anti-inflammatory effects, light modulation, and restoration of local hypoxic conditions. 22 These actions help improve the microenvironment of the meibomian glands and ocular surface, reduce inflammatory factors in ocular tissues, enhance tear film stability, alleviate lid margin inflammation, and ultimately treat MGD and MGD-related dry eye, alleviating symptoms and signs of ocular surface diseases. 23 Previous studies have demonstrated that IPL can promote the alleviation of dry eye symptoms in patients after surgery. 24 However, IPL treatment alone can only soften the meibum within the meibomian glands, while combining it with meibomian gland massage can promote the opening of meibomian gland orifices, expel problematic lipids, and improve the secretion function of the meibomian glands. Previous studies have shown that IPL combined with MGX can treat chalazion.25–28 Therefore, for patients with preexisting MGD undergoing refractive surgery, combining IPL treatment and meibomian gland expression (MGX) will greatly enhance postoperative safety, reduce the risk of exacerbating dry eye, provide immediate relief for patients’ dry eye symptoms, and prevent patients from suffering from ocular surface microenvironment disruption caused by dry eye and a prolonged postoperative recovery period.
The aim of the present study was to investigate whether the systematic management of perioperative dry eye using IPL treatment and MGX in patients with preexisting dry eye or MGD before and after refractive surgery can rapidly improve postoperative dry eye symptoms and signs. We checked dry eye-related symptoms at 1 week and 1 month postoperatively to evaluate the immediate effect of IPL treatment and MGX on improving dry eye symptoms in patients.
Methods
Study and patients
This prospective, self-controlled, single-center study aimed at evaluating the safety and efficacy of using IPL treatment and MGX to treat lipid abnormality or mixed-type dry eye during the perioperative period of refractive surgery, and its effects on postoperative refractive surgery and ocular surface microenvironment. The trial was conducted at the Eye Refractive Center of the Xi’an People’s Hospital (Fourth Hospital of Xi’an City). This study received approval from the Ethics Committee of Xi’an People’s Hospital for the project titled “A Study on the Effects of Intense Pulsed Light on Post-LASIK Dry Eye Following Femtosecond Laser Flap Creation” (ID: 20180048). The research was conducted in accordance with the Declaration of Helsinki, as revised in 2013. All participants were adequately informed and provided written consent.
Patients were recruited between March 2021 and March 2023, and written informed consent was obtained from all participants.
The following patients were included:
Patients with lipid abnormality or mixed-type dry eye (noninvasive tear breakup time (NIBUT) <10 s, or Schirmer I test (ST) (without anesthesia) Patients had type IV or below Fitzpatrick skin. Patients could complete the treatment plan and follow-up requirements. No severe systemic diseases.
The following patients were excluded:
Age <18 years. Patients with insufficient dry eye disease (DED) aqueous solution. Patients with ocular diseases. Patients unwilling to participate in any form of intervention. Patients with contraindications for corneal refractive surgery.
Finally, 68 patients were included in the study.
Sample size calculation
We utilized G*Power (version 3.1.9.7) to determine the required sample size for paired-samples analyses (consistent with our self-controlled study design, comparing preoperative and postoperative outcomes within the same participants). The analysis parameters were set as follows: Tail option = one-tailed, significance level (α) = 0.05, and statistical power (1−β) = 0.80 (a commonly accepted threshold in clinical research to minimize type II error).
We used data from a well-matched published study 29 that investigated the same primary outcome measure—average noninvasive breakup time (NIBUTav)—in patients undergoing refractive surgery (the same population as our study). This study reported NIBUTav values of 11.24 ± 5.90 (preoperative) and 9.02 ± 4.86 (1 month postoperative), with a statistically significant difference (P = 0.027). Based on this, we calculated an effect size coefficient of 0.41, and through this comprehensive analysis, we finally estimated an initial sample size of 67 participants.
Ocular surface workup
For all eligible patients, IPL therapy combined with MGX was performed at 2 different time points: 7 ± 2 days before corneal refractive surgery and 7 ± 2 days after surgery. For detailed timeline, refer to Fig. 1. Before each treatment and 30 ± 4 days after surgery, routine ophthalmic examinations and ocular surface evaluations were conducted. All examinations were performed by the same physician. The examination parameters included best-corrected (for data of 1 week before surgery)/uncorrected distance (for data of 1 week/month after surgery) visual acuity, intraocular pressure (IOP), ST, meibomian gland assessment [including meibomian gland expressibility score (MGYSS) and meibum yielding secretion score (MGS)], SPEED, tear meniscus height (TMH), first noninvasive breakup time (NIBUTf), NIBUTav, redness scan (R-scan), meibomian gland dropout score (upper and bottom), average LLT (Avg LLT), and partial blinks (PB). Evaluation was done using the LipiView® ocular surface interferometer (TEARSCIENCE, USA) and Oculus Keratograph 5 M (Oculus, Germany).
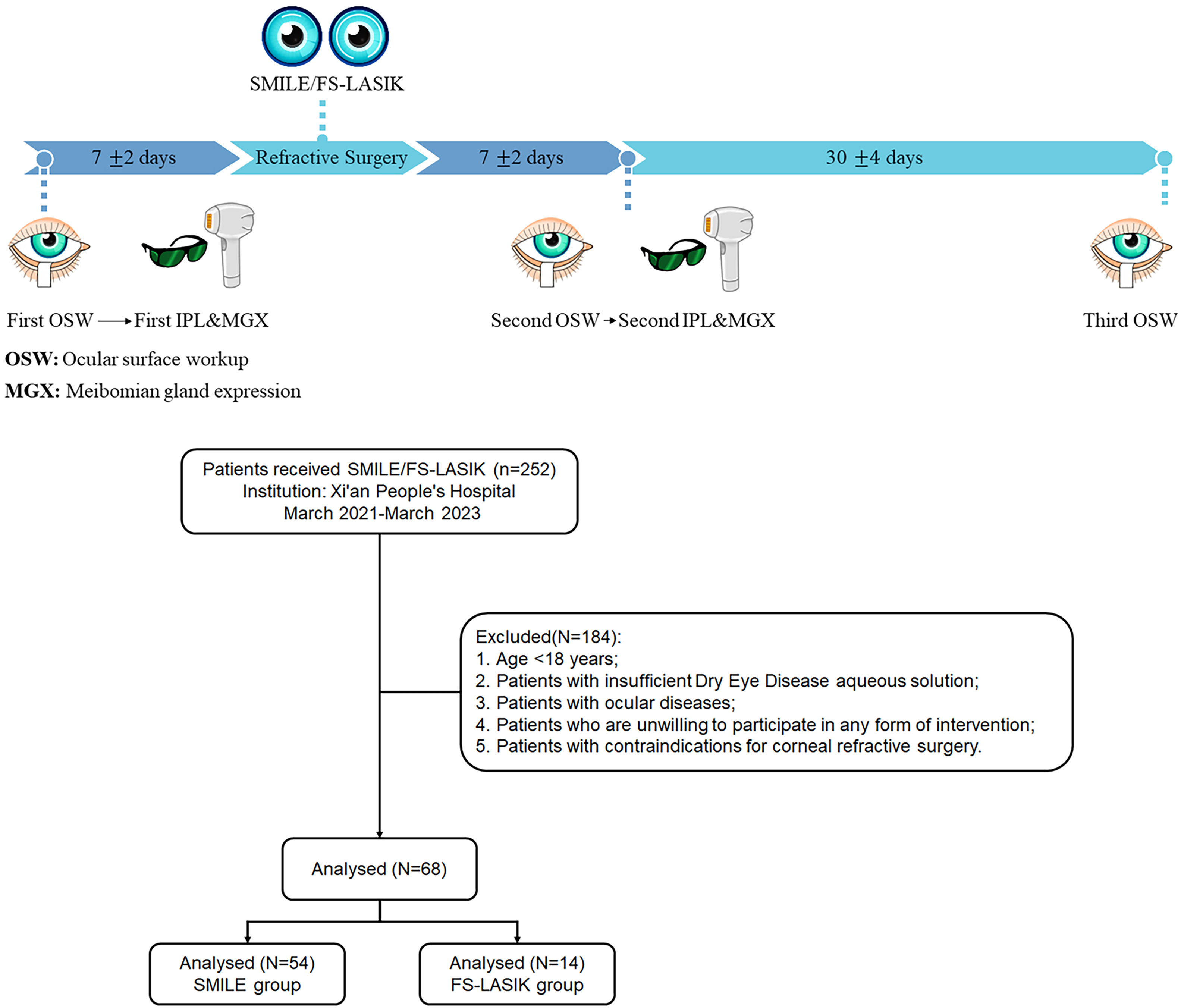
Timeline of SMILE/FS-AF surgeries, 2 rounds of intense pulsed light (IPL) treatment combined with meibomian gland expression (MGX) and 3 rounds of ocular surface workup. SMILE, small-incision lenticule extraction.
Visual acuity was measured using the Chinese standard logarithmic visual acuity chart and then converted to logMAR (logarithm of the minimum angle of resolution) standards.
Noncontact tonometry was used to measure IOP, with 3 measurements taken for each eye and the average recorded.
The ST was performed to evaluate the secretion function of the main lacrimal gland (physiological secretion). Without the use of topical anesthesia, the test was conducted in a quiet, dark room with a consistent room temperature. An ST strip (5 mm × 35 mm, Tianjin Jingming) was folded at the designated line and placed at the junction of the lower eyelid and the outer one-third of the conjunctival sac. The patients were instructed to gently close their eyes. After 5 min, the wet length of the yellow-stained area on the strip was measured, and the tear secretion value was recorded based on the length. A value >10 mm indicated normal secretion, 5–10 mm indicated low tear secretion, and <5 mm indicated insufficient tear secretion.
The SPEED questionnaire 30 was administered by the same physician to assess dry eye symptoms. It consisted of 12 items, with a total score ranging from 0 to 28. A SPEED score of 0 indicated no symptoms, a score of 1–9 indicated mild-to-moderate symptoms, and a score of ≥10 indicated severe symptoms.
The meibomian gland evaluation was performed by an experienced clinical physician using a slit lamp. The evaluation included 2 aspects:
MGYSS: Meibomian gland evaluator (MGE) was used for this assessment. The MGE applied constant pressure (0.8–1.2 g/mm2), simulating the pressure exerted by blinking. The examiner observed the opening of 5 glands on the nasal side to the center of the lower eyelid and assessed the condition and type of meibum secretion from each opening. The scoring criteria were as follows: 0 points for all 5 glands expressing secretion upon eyelid compression, 1 point for 3 or 4 glands expressing secretion, 2 points for 1 or 2 glands expressing secretion, and 3 points for no glands expressing secretion. A score of 0 indicated normal, while a score of 1 or higher indicated abnormalities. MGS: A total of 8 meibomian glands on the nasal side to the center of the lower eyelid were evaluated. The scoring criteria were as follows: 0 points for clear and transparent secretion, 1 point for cloudy secretion, 2 points for turbid granular secretion, and 3 points for thick, toothpaste-like secretion. The scores of the 8 glands were summed, with 0 points indicating normal and 1 point or higher indicating abnormalities.
LipiView ocular surface interferometer was used to quantitatively measure the thickness of the tear film lipid layer (LLT). It automatically analyzed the distribution of LLT and calculated the Avg LLT. The instrument also recorded blink frequency and incomplete blink count (PB). The LLT values obtained by the instrument were expressed in interferometric color units (ICU), where 1 ICU equals 1 nm. The conformance factor (CF) represented the quality of the data, with CF <0.7 indicating the need for retesting to ensure accuracy.
The ocular surface was examined using Oculus Keratograph 5 M (Oculus, Germany) for TMH, NIBUTav, and NIBUTf, and to detect meibomian gland loss using infrared photography. The scoring criteria for meibomian gland loss 31 were as follows: 0 points for no loss, 1 point for ≤1/3 loss, 2 points for >1/3 to ≤2/3 loss, and 3 points for ≥2/3 loss. Record the sum of upper and lower eyelid scores (0–6 points).
IPL treatment
The optimized IPL used is the M22 Optima IPL device (Lumenis, Israel). Before each treatment, patients must refrain from using any skincare or cosmetic products and keep their facial skin clean and dry. Before treatment, patients should wash their face with water and wear eye protection and then apply 5–10 mm of medical coupling agent evenly to the facial and nasal areas below the lower eyelid. Set the energy level to 10–14.0 J/cm2, with a pulse light wavelength of 590 nm. The physician should wear protective goggles during the treatment.
Selection of treatment sites: Start emitting light from the patient’s temple on 1 side and irradiate along the lower eyelid, as close to the eyelid margin as possible. Treat the area below the cheekbones, face, and nose until reaching the temple on the opposite side. Each treatment spot should overlap by no more than 10%. Repeat the procedure twice, avoiding areas with excessive hair growth.
MGX should be performed immediately after each IPL treatment. After instilling 1 to 2 drops of proparacaine hydrochloride eye drops (Alcon, Belgium) in both eyes and wiping secretions with a sterile cotton swab, the physician used eyelid forceps to perform compressive massage on the upper and lower eyelids of both eyes of the patient under direct visualization with a slit lamp. This technique helps express problematic meibum from the meibomian glands and facilitates the clearance of blocked meibomian gland orifices. The same physician should perform both IPL treatment and MGX. IPL treatment and MGX should be performed once before laser corneal refractive surgery and 7 ± 2 days after surgery.
All patients underwent laser corneal refractive surgery performed by the same ophthalmic surgeon (J.D.). All surgeries were performed under local anesthesia, including small incision lenticule extraction (SMILE) or femtosecond laser-assisted in situ keratomileusis (FS-LASIK). The surgeries were successful without any complications. The postoperative medication regimen included the following eye drops: fluorometholone eye drops 1 mg/mL (Aifulong, Allergan, Dublin, Ireland) 3 times a day for 1 month, gatifloxacin ophthalmic gel 3 mg/g (Diyou, Sinqi, Shenyang, China) 4 times a day for 7 days, followed by discontinuation, and polyethylene glycol (Systane, Alcon, Fort Worth, USA) 3 times a day for 30 days.
Outcomes
The endpoints were the changes in visual acuity, IOP, ST, MGYSS and MGS, SPEED, TMH, NIBUTf, NIBUTav, R-scan, meibomian gland dropout score (upper and bottom), Avg LLT, and PB from 1 week before refractive surgery to 1 month after surgery.
Statistical analyses and model development
All statistical analyses included in this study were conducted using Python software version 3.9.1. Paired-sample t-test was used to evaluate the significance of changes in various indicators between different time points (1. A week before surgery vs 1 week after surgery; 2. A week before surgery vs 1 month after surgery; 3. A week after surgery vs 1 month after surgery). False discovery rate (FDR) correction was performed with the multiple tests function from Python statsmodels package. FDR-adjusted P-value <0.05 was considered statistically significant.
Results
IPL treatment combined with MGX effectively improves dry eye symptoms for patients with post-laser corneal refractive surgery dry eyes
Overall, 68 patients (136 eyes) fulfilled the study criteria and were included. Fifty-four patients (108 eyes) received SMILE, and 14 patients (28 eyes) received FS-LASIK. The characteristics of all participants are shown in Table 1.
Characteristics of the Participants
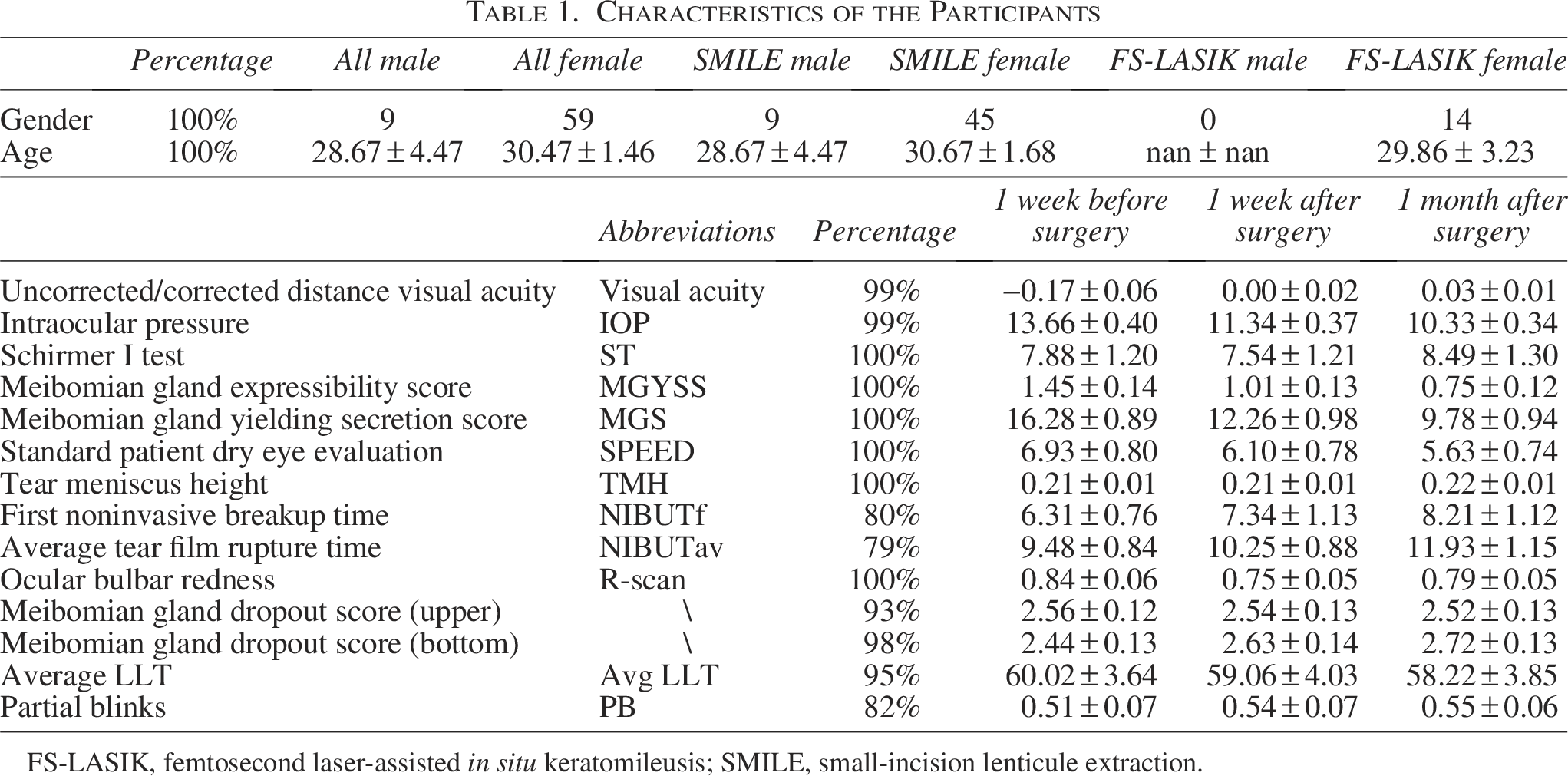

FS-LASIK, femtosecond laser-assisted in situ keratomileusis; SMILE, small-incision lenticule extraction.
Overall, paired-sample t-test demonstrated significant changes on uncorrected/corrected distance visual acuity, IOP, MGYSS, MGS, NIBUTf, NIBUTav, R-scan, and meibomian gland dropout score (bottom) (Table 2, Fig. 2, Supplementary Figs. S1, S2, S4, S5, S8, S9, S10, and S12, FDR-adjusted P < 0.05). Among them, uncorrected/corrected distance visual acuity, IOP, MGYSS, and MGS showed significant changes as early as 1 week after surgery and kept significant changes with the same trend between 1 week and 1 month after surgery (Supplementary Figs. S1, S2, S4, and S5).
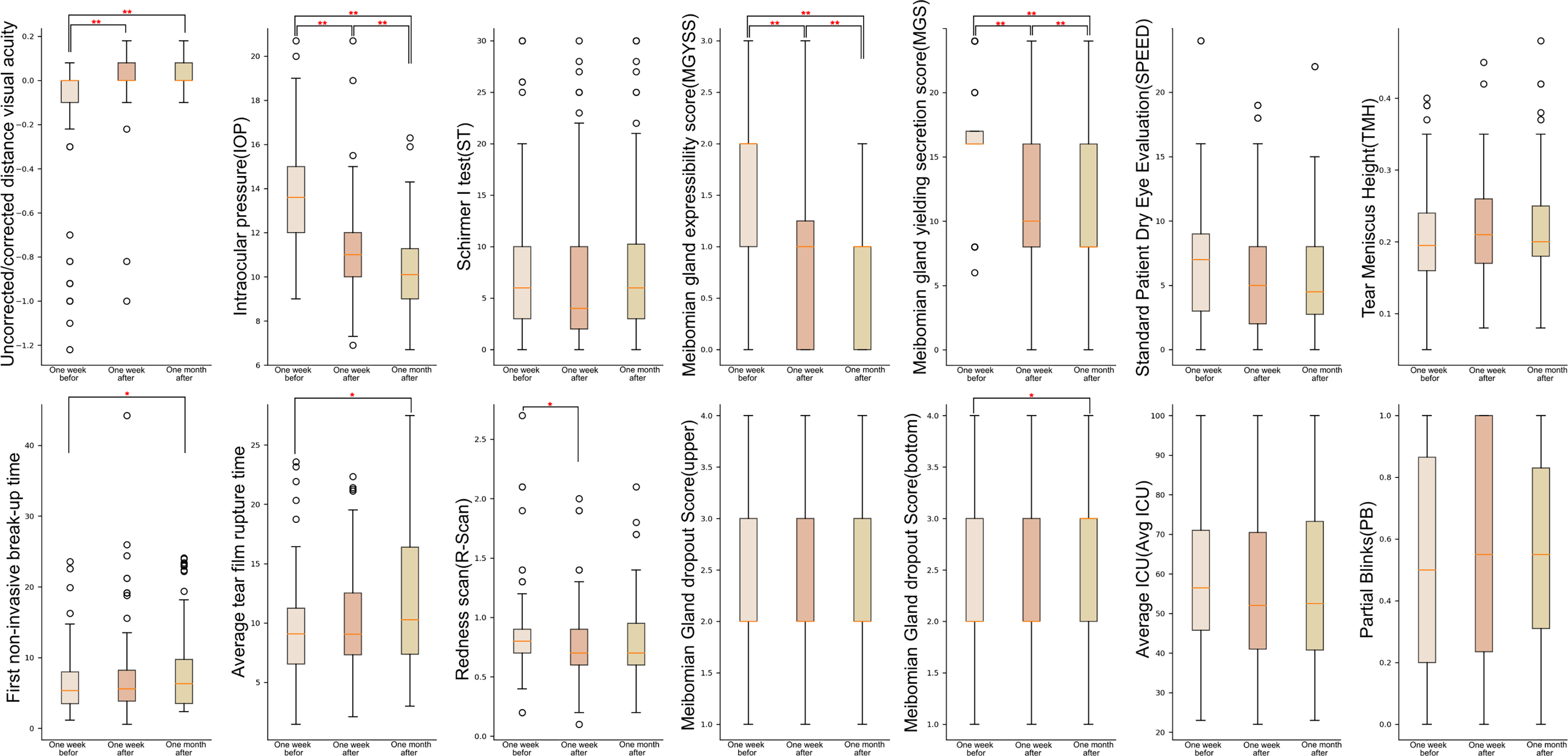
Boxplot of preoperative and postoperative changes in 14 indicators (*: FDR-adjusted P-value <0.05; **: FDR-adjusted P-value <0.01). FDR, false discovery rate.
Paired t-Test of Patients Before Surgery, at 1 Week Postsurgery, and at 1 Month Postsurgery (FDR-Adjusted P-value)
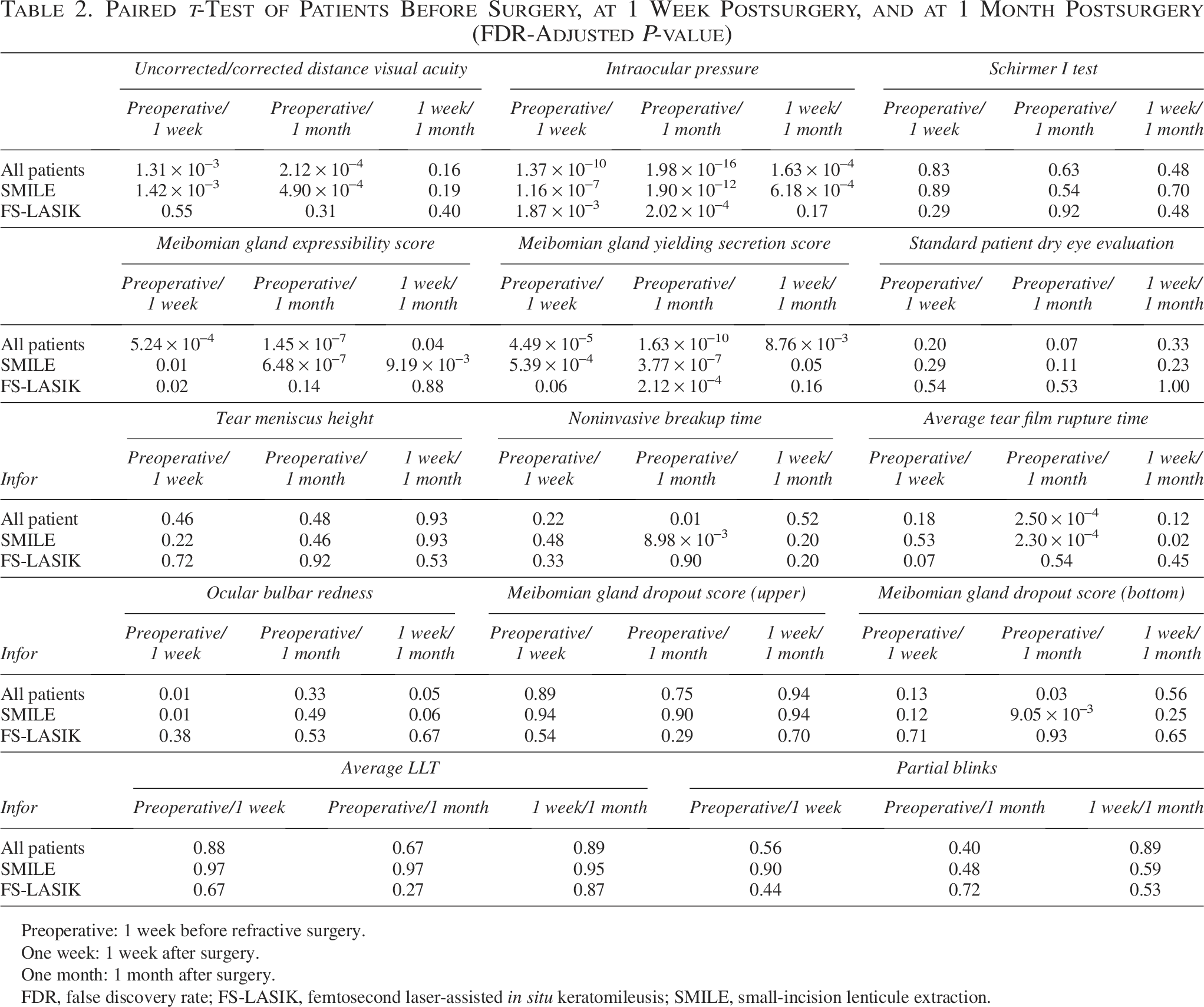




Preoperative: 1 week before refractive surgery.
One week: 1 week after surgery.
One month: 1 month after surgery.
FDR, false discovery rate; FS-LASIK, femtosecond laser-assisted in situ keratomileusis; SMILE, small-incision lenticule extraction.
NIBUTf, NIBUTav, and the meibomian gland dropout score (bottom) only showed a significant increase 1 month after surgery compared with 1 week before surgery (Supplementary Figs. S8, S9, and S12). R-scan was only observed to have a significant decrease at 1 week after surgery and showed a slight improvement after 1 month but not significant (Supplementary Fig. S10). Conversely, no significant changes were observed in ST, TMH, SPEED, meibomian gland dropout score (upper), Avg LLT (average lipid layer thickness), and PB (partial blink) before and after surgery.
All these significant changes can be observed in patients who received SMILE treatment. However, only the significant changes of IOP, MGYSS, and MGS could be observed in patients who received FS-LASIK (Supplementary Figs. S2, S4, and S5).
Comparative analysis between patients of SMILE group and those of FS-LASIK group
We conducted a comparative analysis of ocular surface indicators between patients of SMILE group and those of FS-LASIK group at 3 time points to evaluate the efficacy differences of IPL combined with MGX during postoperative recovery after both types of refractive surgery. At baseline, there were no significant differences observed among all ocular surface indicators. The results from t-tests revealed significant differences in MGYSS, MGS, average tear film rupture time, meibomian gland dropout score (upper and bottom), and average LLT between the 2 surgical groups (Table 3). After adjusting for age and gender variables, multivariate logistic regression analysis indicated that the FS-LASIK group only exhibited superior therapeutic outcomes (lower score) compared with the SMILE group in MGYSS at 1 week after surgery (Fig. 3, Table 3).
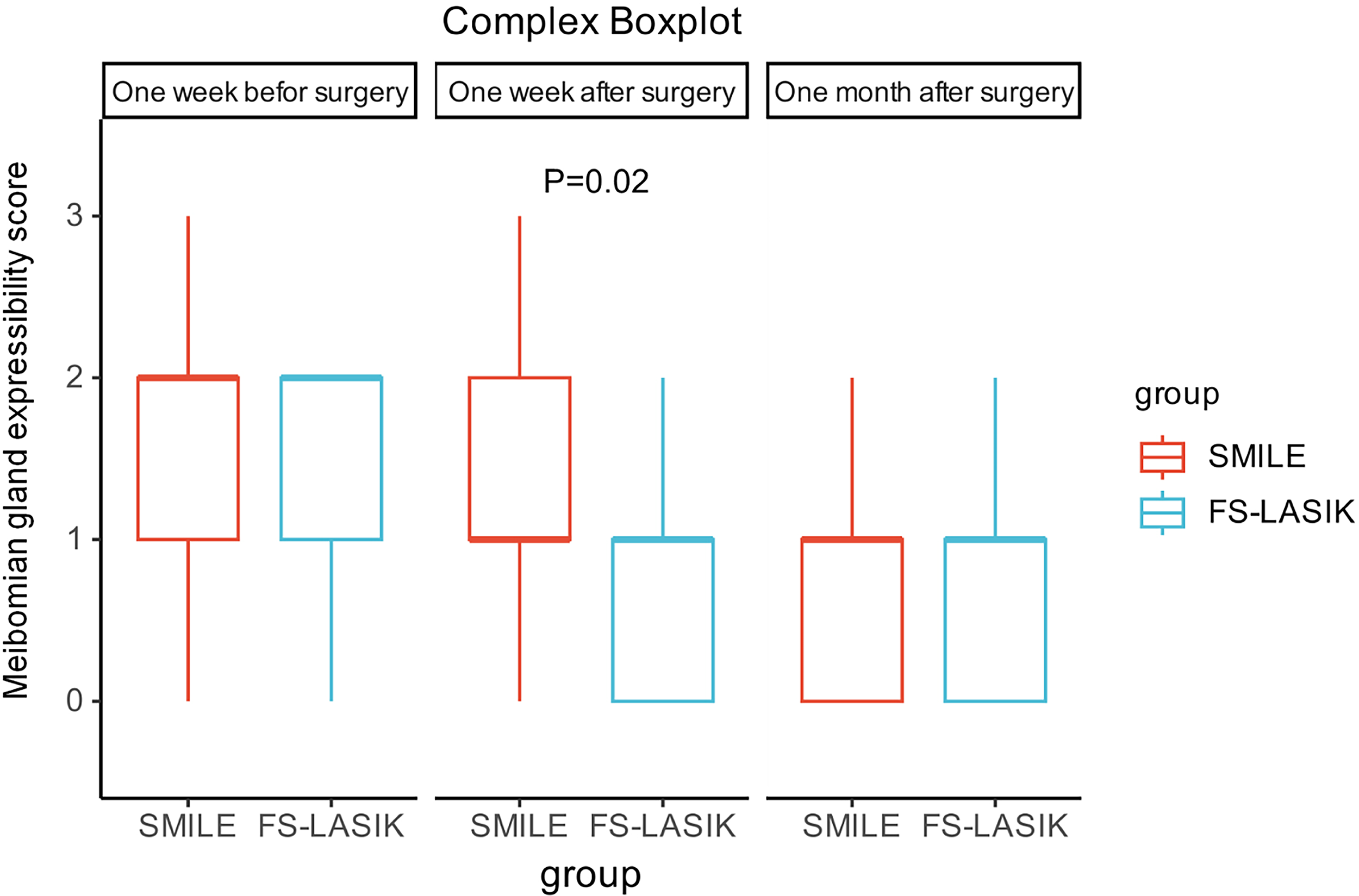
Boxplot of meibomian gland expressibility score (MGYSS) between patients of small-incision lenticule extraction group and those of femtosecond laser-assisted in situ keratomileusis (FS-LASIK) before surgery, 1 week postsurgery, and 1 month postsurgery.
Comparative Analysis Between Patients of Small-Incision Lenticule Extraction Group and Those of Femtosecond Laser-Assisted In Situ Keratomileusis Before Surgery, 1 Week Postsurgery, and 1 Month Postsurgery
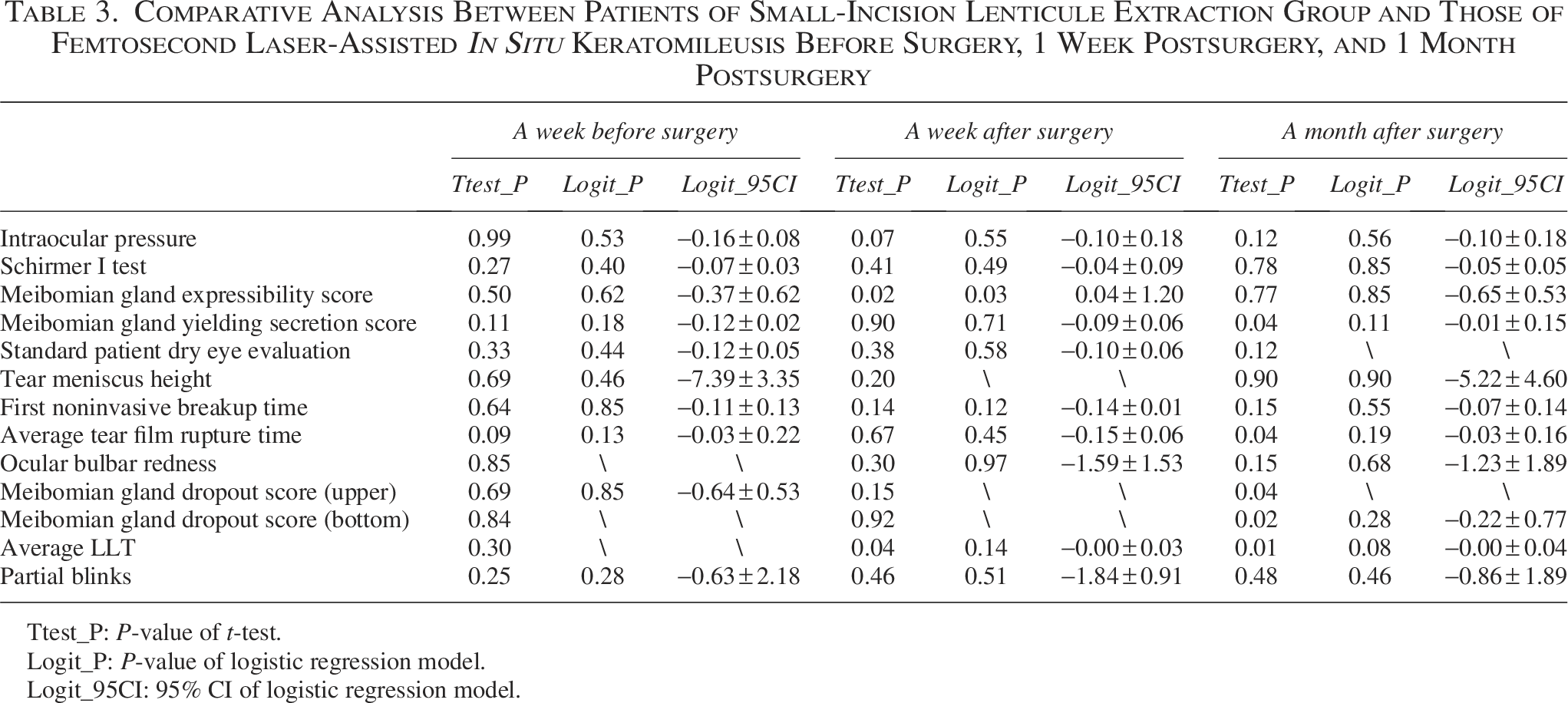
Ttest_P: P-value of t-test.
Logit_P: P-value of logistic regression model.
Logit_95CI: 95% CI of logistic regression model.
Discussion
This is the first study to use IPL technology combined with MGX for prophylactic intervention in mild-to-moderate evaporative DED and MGD in post-laser corneal refractive surgery dry eyes. Improving outcomes of refractive surgery in terms of ocular comfort in the postoperative course represents a primary concern for both patients and clinicians. Currently, a common clinical concern is that refractive surgery may lead to postoperative dry eye or exacerbate preexisting dry eye. For patients with mild-to-moderate evaporative dry eye or MGD, there is an increased risk of exacerbating dry eye symptoms after surgery.32,33 Increasing evidence suggests that optimizing ocular surface status in patients with preexisting DED is a crucial step to achieve the desired postoperative outcomes and avoid complications, particularly in patients undergoing cataract surgery. 34 Similarly, studies in corneal refractive surgery have demonstrated that appropriate preoperative treatment of dry eye is significantly superior to postoperative treatment. 35
This study was conducted on 68 patients (136 eyes) with preexisting mild-to-moderate evaporative dry eye or MGD, who underwent laser corneal refractive surgery. These patients received 2 sessions of IPL treatment combined with MGX before surgery and 1 week after surgery. By comparing the dry eye indicators of patients 1 week and 1 month after surgery with those before surgery, we observed significant improvements in MGYSS, MGS, NIBUTf, and NIBUTav of dry eye symptoms in patients who received IPL treatment combined with MGX before and after refractive surgery.
One week postsurgery, we observed a significant decrease in the R-scan score. However, due to the administration of both glucocorticoids and antibiotics in the postoperative period, it is currently challenging to ascertain whether this notable reduction in the R-scan score is attributable to the effects of IPL therapy—given that IPL technology possesses inherent anti-inflammatory properties—or if it results from the anti-inflammatory responses elicited by the medications. Following 1 week after surgery, there was a reduction in the frequency of antibiotic and glucocorticoid use, which may correlate with a gradual increase in the R-scan score. At 1 month postsurgery, no significant difference was noted in the R-scan score compared with preoperative values. Based on these findings, we recommend a combined approach utilizing IPL therapy alongside anti-inflammatory medication following refractive surgery to more reliably mitigate the risk of developing dry eye syndrome subsequent to such procedures.
Compared with the use of IPL therapy alone, MGX has been shown to facilitate the opening of meibomian gland orifices, expel problematic lipids, and enhance the secretory function of meibomian glands. In our study, we observed that MGX significantly improved various indicators related to meibomian glands—such as MGYSS and MGS—within just 1 week postoperation. Conversely, results from Martinez-Hergueta MC et al., 24 utilizing IPL alone, indicated that significant changes in upper and lower infrared meibography were only evident 3 months after treatment. Furthermore, the combination of IPL and MGX demonstrated improvements in NIBUTf and NIBUTav within 1 month. Notably, no enhancement in NIBUTf was recorded with IPL therapy alone. 24 These findings suggest that integrating IPL with meibomian gland massage can effectively expedite relief from dry eye symptoms following refractive surgery.
Dry eye is one of the most common complications after refractive surgery. According to the TFOS DEWS II Definition and Classification Subcommittee, DED is a multifactorial disorder in which the dysregulation of tear film homeostasis constitutes the core pathophysiological concept. 36 According to the Dry Eye Syndrome Preferred Practice Pattern 37 (2023 updates) from the American Academy of Ophthalmology, IPL therapy and warm compresses combined with meibomian gland massage have been recommended for patients with moderate DED to improve their meibomian gland secretion function. Building on this, we suggest combining ocular massage after IPL therapy to promote the secretion of problematic lipids, thereby further enhancing the therapeutic effect.
Among the currently popular surgical methods, SMILE has a lower incidence of postoperative dry eye, while FS-LASIK had a reported incidence rate of 36%–75% for dry eye. 38 It has been reported that 80% of SMILE patients do not require any tear supplements at 6 months postoperatively, compared with 57% of LASIK patients. 39 To alleviate postoperative dry eye symptoms, SMILE is the preferred surgical method, which resulted in the patient cohort consisting mostly of individuals who received SMILE surgery. In the study, only significant changes in MGYSS were observed at 1 week after surgery when comparing the FS-AF group with the SMILE group. This suggests that the combination of IPL and MGX may facilitate a more rapid recovery of meibomian gland-related factors in the FS-AF group compared with the SMILE group in the short term. However, over an extended period, both patient groups are likely to achieve a comparable level of recovery.
Overall, this study highlights the potential benefits of IPL therapy combined with MGX as a prophylactic intervention for evaporative dry eye and MGD in patients undergoing laser corneal refractive surgery. We conducted an initial evaluation of providing 1 session of IPL therapy combined with MGX before refractive surgery, followed by another additional IPL therapy combined with MGX 1 week after surgery. Previous studies have mainly focused on treating dry eye symptoms after refractive surgery or using a prophylactic thermal pulsation therapy approach with lip flow before surgery. In our study, we took advantage of the favorable 2-week treatment duration of IPL and implemented a comprehensive treatment approach involving both MGX and IPL treatments, which were administered both preoperatively and postoperatively during the perioperative period of refractive surgery. This approach meets the timely surgical needs of refractive surgery patients while ensuring the safety of the surgery and postoperative recovery, making it a highly suitable treatment option for clinical needs.
This study has several limitations. The single-center design with a relatively small sample size, compounded by the absence of a control group not receiving OPT therapy based on current findings, may restrict the generalizability of the results. Data from diagnoses made by a single surgeon may also introduce personal bias. Furthermore, as the study cohort exclusively comprised Asian participants, the findings require validation in diverse ethnic populations to account for variations in skin pigmentation and potential race-specific differences in laser treatment parameters and efficacy. Further research will be conducted to assess the cost-effectiveness and determine whether it is reasonable to use IPL for every patient undergoing laser refractive surgery or to focus treatment on patients at higher preoperative risk (with preexisting MGD and mild ocular surface diseases) for better outcomes.
Summary
In summary, we evaluated the therapeutic and protective effects of using optimal IPL therapy combined with MGX before and after surgery on post-laser corneal refractive surgery dry eyes. We found that this kind of combination therapy could effectively improve ocular discomfort and dry eye symptoms in IOP, MGYSS, MGS, NIBUTf, NIBUTav, R-scan, and meibomian gland dropout score (bottom) for patients with post-laser corneal refractive surgery dry eyes. By using this effective and convenient treatment approach, it is possible to save time and costs while providing a safer treatment option for dry eye patients who have received refractive surgery.
Authors’ Contributions
J.D. and Y.W. designed the entire project and composed the article. Y.R. analyzed the data. Z.Y. was in charge of the illustrations. Y.L., X.-M.W., and J.S. collected the data. J.L., S.-S.W., J.-R.G., and Q.-N.H. reviewed and modified the article.
Footnotes
Acknowledgment
The authors deeply appreciate the data support provided by the Xi’an People’s Hospital.
Author Disclosure Statement
There is no conflict of interest within this article.
Funding Information
This work was supported by the Key Research and Development Program of Shaanxi Province (No. 2023-YBSF-586), the Natural Science Basic Research Plan of Shaanxi Province of China (No. 2017JM8043), the Health Research Project of Shaanxi Province (No. 2020YB11), and the Science Research of Xi’an Fourth Hospital of Shaanxi Province of China (No. FZ-4).
Consent for Publication
All authors agreed to publish this article.
