Abstract
Objective:
To analyze the differential expression of inflammatory proteins in the tear fluid of patients with polypoidal choroidal vasculopathy (PCV) or neovascular age-related macular degeneration (nAMD).
Methods:
A total of 19 patients with PCV, 17 patients with nAMD, and 18 normal controls (NC) aged ≥50 years were enrolled. Tear samples were collected, and the expression levels of 92 inflammatory proteins were quantified using Olink technology. Differentially expressed proteins (DEPs) among the groups were analyzed, with particular attention to consistency with previous findings from aqueous humor studies.
Results:
Olink analysis revealed extensive DEPs among PCV, nAMD, and NC groups. Compared with NC, the PCV group exhibited significant upregulation of VEGFA, Interleukin (IL) -18, IL-1α, IL-8, IL-7, Monocyte chemotactic protein (MCP)-2, and Neurturin (NRTN), along with downregulation of Tumor necrosis factor (TNF) and IL-10. The nAMD group showed a more pronounced pro-inflammatory profile, with upregulation of VEGFA, IL-18, IL-8, IL-1α, IL-7, Fibroblast growth factor (FGF)-19, MCP-1, Matrix metalloproteinase (MMP)-10, NRTN, Stem cell factor (SCF), Osteoprotegerin (OPG), Eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1), MMP-1, MCP-2, Fms-related tyrosine kinase 3 ligand (Flt3L), Tumor Necrosis Factor - like Weak Inducer of Apoptosis (TWEAK), Caspase (CASP)-8, and Tumor necrosis factor receptor superfamily member 9 (TNFRSF9), and downregulation of IL-10 and IL-12β. Comparison between PCV and nAMD indicated that IL-12β and Hepatocyte growth factor (HGF) were specifically upregulated in PCV, whereas SCF, VEGFA, Flt3L, OPG, MCP-1, T-cell surface glycoprotein CD8α (CD8α), Cystatin D (CST5), MMP-1, TNFRSF9, Transforming growth factor-α (TGF-α), 4E-BP1, and CASP-8 were significantly downregulated in PCV relative to nAMD. Boxplot analysis confirmed that Flt3L, IL-18, IL-1α, IL-7, IL-8, MCP-1, MMP-1, OPG, SCF, and VEGFA were specifically elevated in nAMD compared with both PCV and NC groups, while IL-10 was specifically suppressed in PCV.
Conclusions:
Tear fluid analysis represents a feasible and noninvasive approach to investigate the pathogenesis of PCV and nAMD.
Keywords
Introduction
Polypoidal choroidal vasculopathy (PCV) is an exudative choroidal-retinal disease characterized by orange-red nodular lesions in the fundus and the presence of branching vascular networks (BVN) and terminal polypoidal lesions on indocyanine green angiography (ICGA). 1 In the past, PCV was widely recognized as an independent disease; however, some scholars have proposed that it may be classified as a subtype of type 1 macular neovascularization (MNV) in neovascular age-related macular degeneration (nAMD). 2 Despite this, PCV differs from nAMD in terms of epidemiology, genetic heterogeneity, and treatment response. 3 Although the pathogenesis is still under in-depth exploration, some scholars still support the classification of PCV as the pachychoroid spectrum. 4 The first-line treatment for PCV is intravitreal injection of anti-vascular endothelial growth factor (VEGF) agents, as in nAMD, and may be combined with photodynamic therapy (PDT). 1 However, the actual therapeutic effect is suboptimal, and repeated intravitreal injections carry potential treatment risks. 5 Verteporfin, the key agent required for PDT, remains in global shortage. To develop effective and targeted therapies, it is essential to further elucidate the PCV pathogenesis. Previously, many studies have investigated differences in aqueous humor and vitreous cytokine profiles between PCV and nAMD.6–8 However, as the collection of aqueous or vitreous humor is an invasive procedure with potential risks of surgical complications and infection, it is not feasible as a routine diagnostic method. 9 Tear analysis has attracted considerable attention in ophthalmic research.10,11 In addition to reflecting the health of the ocular surface, tears play a critical role in the clinical monitoring and evaluation of intraocular diseases.12–15 Tear samples can be collected through noninvasive methods such as microcapillary tubes or Schirmer test strips. 16 Previous studies have demonstrated that tear VEGF levels may serve as noninvasive biomarkers for detecting AMD. 8 However, no studies have investigated tear biomarkers in patients with PCV. This study aims to compare the levels of cellular inflammatory factors in the tears of patients with PCV or nAMD, with the goal of providing a noninvasive approach for further exploring the pathogenesis of these two diseases.
Methods
Subjects
Tear samples were collected from 19 patients with PCV (PCV group), 17 patients with nAMD (nAMD group), and 18 normal controls (NC group) aged ≥50 years. All participants provided written informed consent. The study protocol was approved by the Ethics Committee of Hainan General Hospital (Approval No.: 2025-155). All enrolled PCV and nAMD patients underwent standardized ophthalmic assessments, including best-corrected visual acuity, slit-lamp biomicroscope, and fundus examination under mydriasis. Inclusion required: (1) recent onset of visual impairment; (2) optical coherence tomography (OCT) revealed active macular lesions, evidenced by the presence of intraretinal/subretinal fluid, hemorrhage, or edema; 17 (3) fundus fluorescein angiography and ICGA findings confirmed the diagnosis, meeting internationally accepted criteria.18,19 Specifically, PCV patients consistently exhibited BVNs and polyps on ICGA, sharp-peaked pigment epithelial detachment, and circular sub-retinal pigment epithelium lesions on OCT, plus funduscopic orange-red nodules. 20 Exclusion criteria encompassed: (1) moderate-to-severe dry eye confirmed by ocular surface disease index, 21 positive corneal fluorescein staining, or abnormal tear film breakup time; (2) active ocular surface diseases; (3) concomitant any other fundus pathologies; (4) any prior treatment for fundus disease; (5) systemic comorbidities such as diabetes mellitus or hypertension; (6) presence of macular atrophy or fibrotic scarring, regardless of accompanying macular edema or hemorrhage.
Tear collection and testing
Approximately 10 μL of tear fluid was collected from participants in all three groups using a microcapillary tube placed gently in the inferior fornix conjunctival sac, avoiding cornea contact and leveraging capillary action. The samples were subsequently transferred to sterile 0.5 mL Eppendorf tubes and stored at −80°C until further analysis.
Proteomic analysis
Tear proteomic profiling was conducted by Lianchuan Biotechnology (Hangzhou, China) using the Olink® Target Metabolism panel (Olink Proteomics, Sweden, https://olink.com/products/olink-target-96). Detection was performed based on Olink proximity extension assay technology. This method employs pairs of antibodies equipped with complementary DNA oligonucleotides that bind simultaneously to the target protein. Upon binding, the oligonucleotides hybridize, forming a double-stranded DNA barcode that is quantified via microfluidic quantitative PCR (qPCR). The amount of amplification products is directly proportional to the initial concentration of the target protein.
Bioinformatics analysis
Raw data were processed and normalized using Olink Signature software (version 1.5.3.0) to generate a normalized protein expression (NPX) matrix. Protein annotation was conducted by matching panel targets to publicly available databases, including UniProt (https://www.uniprot.org/), Gene Ontology, and the Kyoto Encyclopedia of Genes and Genomes. Differentially expressed proteins (DEPs) were identified using a t-test on log2-transformed NPX values, with significance defined as p < 0.05 after adjusting for multiple comparisons via the Benjamini–Hochberg method. Data visualization and functional enrichment analysis were performed in R software (version 4.4.3), with the ggplot2 package used to generate figures.
Statistical analysis
Statistical analysis was performed using SPSS software (version 27.0). Normality of data distribution was assessed by the Kolmogorov–Smirnov test. Normally distributed data are presented as mean ± standard deviation (SD). Homogeneity of variances was verified using appropriate tests. A one-way analysis of variance (ANOVA) was used to compare differences among the three groups, followed by Bonferroni’s post hoc tests for multiple pairwise comparisons. A two-sided p-value <0.05 was considered statistically significant.
Results
Clinical data comparison of the three groups
There were 19 PCV patients, 17 nAMD patients, and 18 NC patients. There were no significant intergroup differences in mean age or sex distribution (p > 0.05, Table 1).
Clinical Characteristics of the Three Groups

nAMD, neovascular age‐related macular degeneration; NC, normal control; PCV, polypoidal choroidal vasculopathy.
Differential expression analysis of inflammatory proteins in tear samples
Through the Oink analysis, we compared the expression levels of 92 inflammatory proteins among the PCV group, the nAMD group and the NC group, 89 inflammatory proteins displayed significant differential expression among three groups (Fig. 1A). The heatmap illustrates the expression patterns of a subset of proteins that were significantly altered (p < 0.05) in the three-group comparisons, revealing distinct clusters of DEPs among the groups (Fig. 1B). Volcano plots were generated to visualize DEPs between the different groups (Fig. 2). The analysis used thresholds of |log2(fold change) |>1and an adjusted p < 0.05. Compared the NC group, the PCV group showed notable upregulation of VEGFA, Interleukin (IL)-18, IL-1α, IL-8, IL-7, Monocyte chemotactic protein (MCP)-2, and Neurturin (NRTN), and downregulation of Tumor necrosis factor (TNF) and IL-10 (Fig. 2A). The nAMD group exhibited significant upregulation of VEGFA, IL-18, IL-8, IL-1α, IL-7, Fibroblast growth factor (FGF)-19, MCP-1, Matrix metalloproteinase (MMP)-10, NRTN, Stem cell factor (SCF), Osteoprotegerin (OPG), Eukaryotic translation initiation factor 4E-binding protein 1 (4E-BP1), MMP-1, MCP-2, Fms-related tyrosine kinase 3 ligand (Flt3L), Tumor Necrosis Factor - like Weak Inducer of Apoptosis (TWEAK), Caspase (CASP)-8, and Tumor necrosis factor receptor superfamily member 9 (TNFRSF9), along with downregulation of IL-10 and IL-12β (Fig. 2B). When comparing the PCV group with the nAMD group, IL-12β and Hepatocyte growth factor (HGF) were significantly upregulated in PCV group, whereas SCF, VEGFA, Flt3L, OPG, MCP-1, T-cell surface glycoprotein CD8α (CD8α), Cystatin D (CST5), MMP-1, TNFRSF9, Transforming growth factor-α (TGF-α), 4E-BP1, and CASP-8 were significantly downregulated (Fig. 2C). Figure 3 depicts the expression levels of the DEPs across the three groups using boxplot analysis. Compared to both the PCV and the NC groups, the nAMD group exhibited elevated expression of Flt3L, IL-18, IL-1α, IL-7, IL-8, MCP-1, MMP-1, OPG, SCF, and VEGFA. In contrast, the PCV group showed decreased expression of IL-10 relative to both the nAMD and NC groups. Direct comparison between the nAMD and PCV groups revealed significantly elevated levels of IL-12β and HGF in the PCV group (p < 0.001), whereas Flt3L and SCF were markedly upregulated in the nAMD group (p < 0.0001).
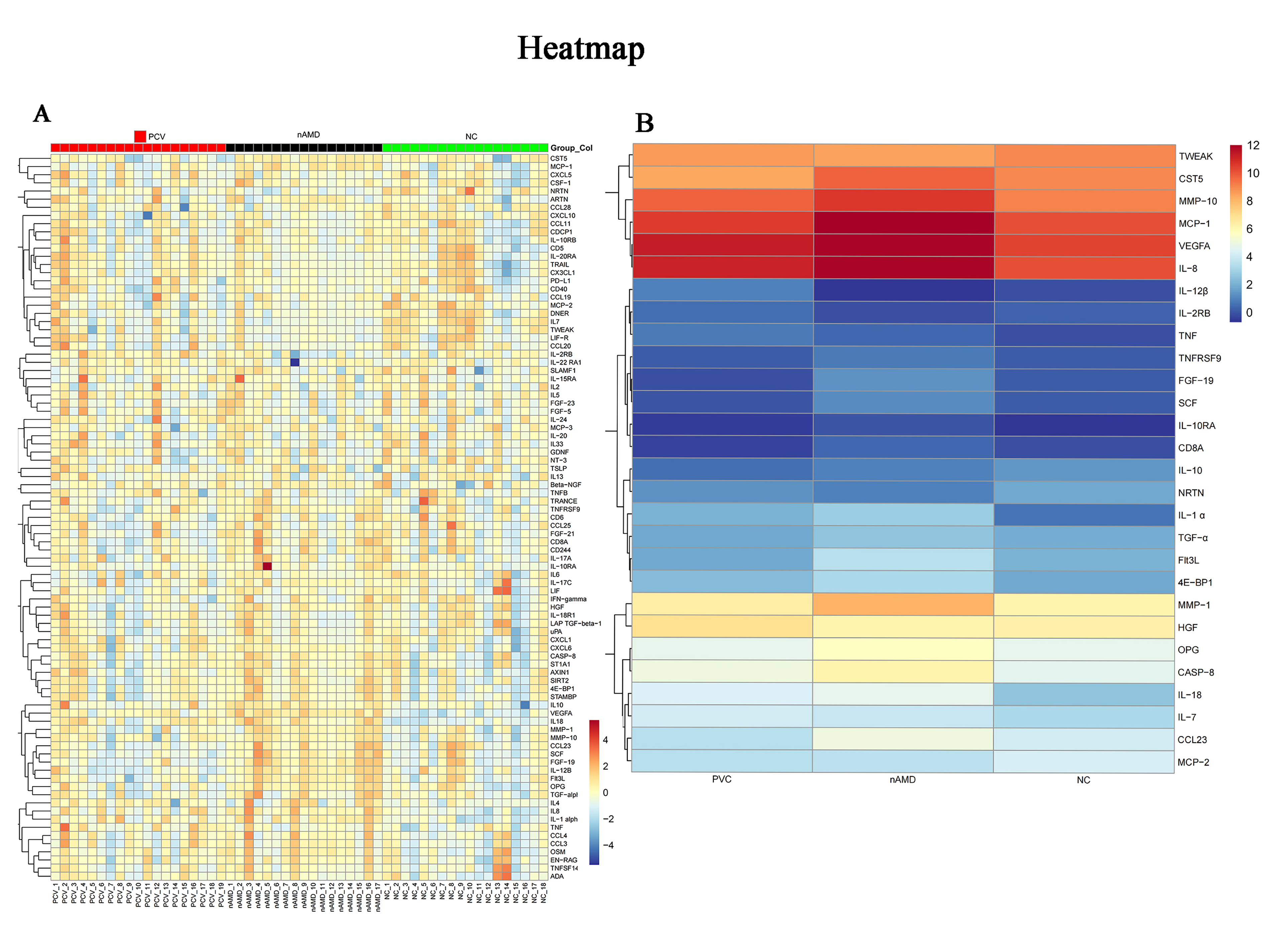
Trend protein hotspot map of the three patient groups.
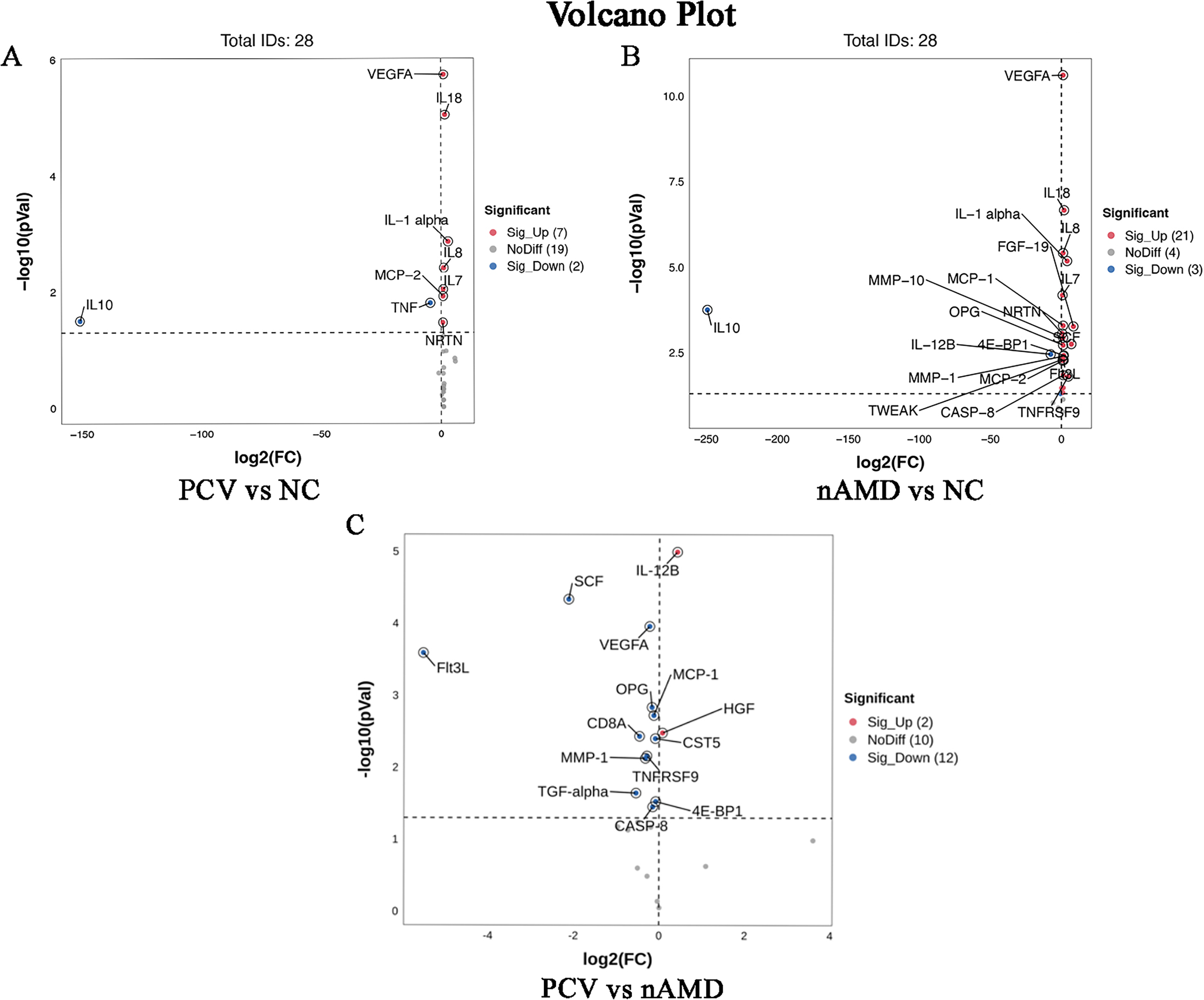
Volcano plots showing differentially expressed trend proteins in pairwise comparisons among the three patient groups.
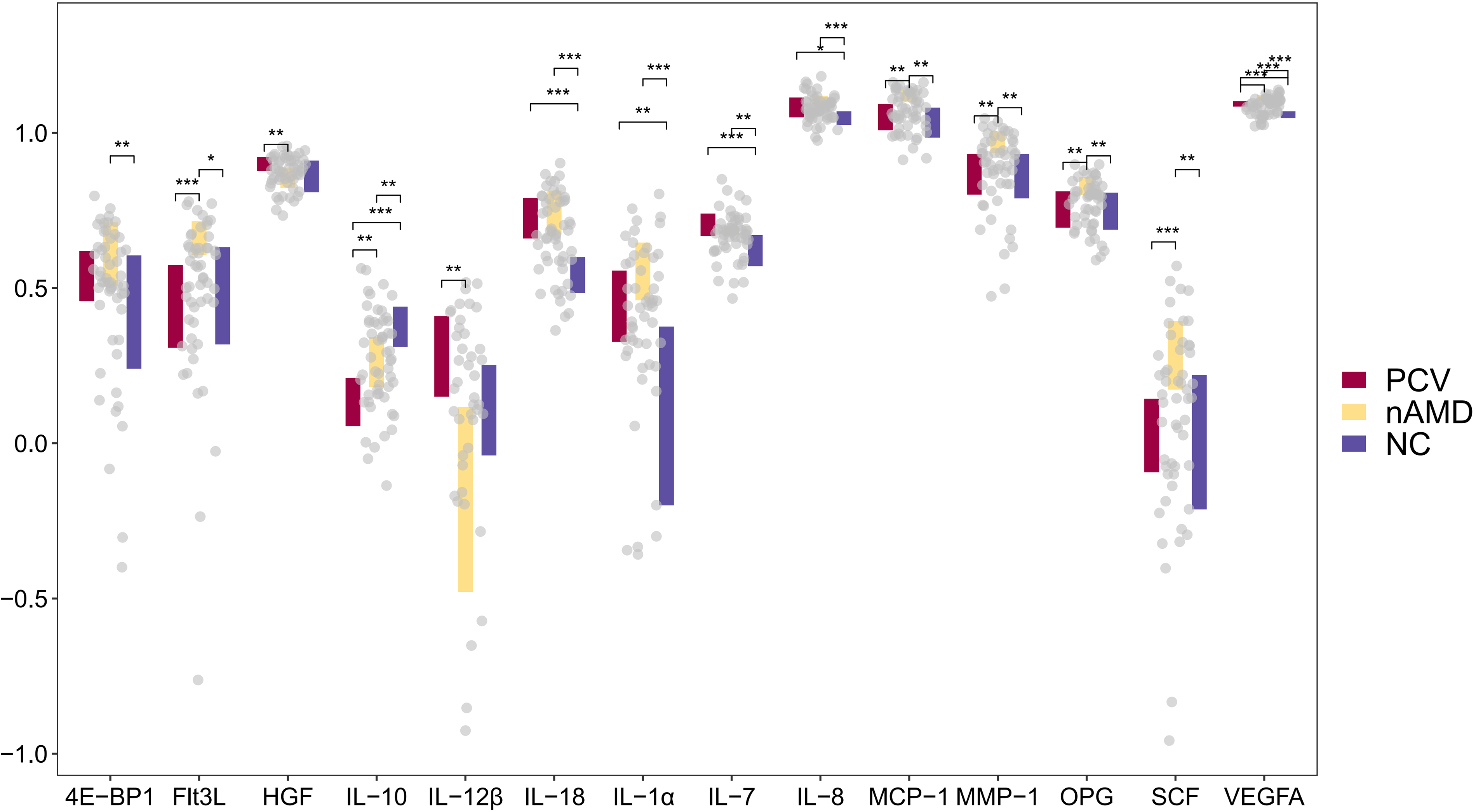
Boxplots comparing expression of 14 significant proteins. *p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
Tears have emerged as a promising source of biomolecular markers. Previous studies have identified 481 proteins in tears and 191 proteins in aqueous humor, with 82 proteins shared between the two fluids. 22 Tear fluid-based molecular biomarkers show significant potential, offering value not only for diagnosing ocular diseases but also as auxiliary tools for detecting systemic conditions such as multiple sclerosis and Parkinson’s disease. 13 An important advantage of tear sampling is its convenience and repeatability, especially compared to more invasive procedures such as cerebrospinal fluid or aqueous humor collection. However, tear composition is influenced by several confounding factors, including smoking, age, sex, ethnicity, and environmental influences. 13 In this study, we analyzed 92 target proteins and identified those with significantly altered expression compared to the NC group. Although certain DEPs showed notable trends, their association with PCV or nAMD has not been conclusively established in the existing literature. Further investigation and long-term follow-up studies are needed to clarify their potential roles.
Compared to the NC group, both the PCV and nAMD groups showed upregulation of VEGFA, IL-18, IL-8, IL-1α, IL-7, and MCP-2 in tear fluid. Among these, IL-18, IL-8, IL-1α, and IL-7 showed expression trends consistent with previous reports from aqueous humor or serum studies. 23 In contrast, MCP-2 has not been previously linked to PCV or nAMD. As a member of the CC chemokine family, MCP-2 is known to recruit mast cells, eosinophils, basophils, monocytes, T cells, and NK cells to inflammation sites. It activates inflammatory cells and enhances the release of proinflammatory mediators, thereby amplifying the inflammatory response. 24 This finding further underscores the importance of inflammatory mechanisms in the pathogenesis and progression of both nAMD and PCV. 25 VEGFA is a central molecule in studies of MNV. As a major angiogenic factor, VEGF exists in several isoforms and serves as a key regulator of angiogenesis, promoting endothelial cell proliferation and neovascular formation. 26 Most clinically used anti-VEGF agents specifically target VEGFA. 27 In this study, elevated VEGFA levels were detected in the tear fluid of both PCV and nAMD groups, aligning with previous findings in tear and aqueous humor studies,8,28 and reinforcing the role of VEGF-mediated angiogenesis in the pathogenesis of both diseases. Notably, VEGFA upregulation was more pronounced in the nAMD group, consistent with an earlier ELISA-based study reporting higher VEGF concentrations in the aqueous humor of nAMD patients compared to those with PCV. 28 Genetic, proteomic, and imaging studies, as well as differential responses to anti-VEGF therapy, have revealed substantial distinctions between PCV and nAMD. 29 Clinically, many treatments effective for AMD demonstrate limited efficacy in PCV patients. 30 These findings suggest that monitoring VEGFA levels may help inform optimal treatment timing in the future. In contrast, IL-10 was downregulated in both the PCV and nAMD groups compared to NC, with a more marked reduction in PCV. IL-10 is a multifunctional cytokine with a potent immunosuppressive role, including inhibition of pro-inflammatory cytokine production and antigen presentation. Its expression is regulated by various metabolic and signaling pathways, which may be dysregulated in inflammatory conditions, leading to suppressed IL-10 secretion. While elevated tear IL-10 levels have been reported in uveitis, 31 the downregulation observed here implies a distinct, yet unclear, regulatory mechanism in PCV and nAMD that merits further investigation.
Compared to the nAMD group, the PCV group showed significant upregulation of IL-12β and HGF. Although IL-12β in tears has not been previously associated with ocular diseases, it is known to play an independent and critical role in early immune response. 32 HGF regulates retinal pigment epithelial barrier function 33 and influences diverse biological processes, including cell proliferation, tissue repair, and angiogenesis. 34 The co-upregulation of IL-12β and HGF in PCV patients may not be coincidental, suggesting potential synergistic effects. Growing evidence supports the involvement of inflammation in PCV pathogenesis. 35 We hypothesize that IL-12β may initiate activation that alters the ocular microenvironment, thereby modulating HGF expression and function. In turn, HGF may affect inflammation through angiogenesis and reparative mechanisms, possibly forming a feedback loop with IL-12β in PCV development. Conversely, Flt3L and SCF were significantly upregulated in the nAMD group compared to the PCV group. To our knowledge, no previous studies have linked Flt3L to nAMD, making this a novel finding. Flt3L supports the proliferation, differentiation, and survival of hematopoietic cells, 36 and elevated levels of Flt3L have been associated with autoimmune and inflammatory diseases such as rheumatoid arthritis, Sjögren’s syndrome, and systemic lupus erythematosus. 37 SCF(c-kit ligand) is an important regulator of cell growth and survival. 38 A study by Choi et al. 39 reported elevated SCF in the aqueous humor of nAMD patients, with levels correlating positively with VEGFA. Our tear fluid data also showed concurrent elevations of VEGFA and SCF in the nAMD group compared to both PCV and NC, supporting this correlation and suggesting a common regulatory pathway. Notably, the nAMD group exhibited marked upregulation of MCP-1, MMP-1, and OPG in tear fluid relative to the other group. MCP-1 upregulation aligns with previous studies, as it is one of the most well-characterized chemokines in AMD pathophysiology. 23 It plays a key role in monocytes/macrophages recruitment and extravasation. 40 Previous Luminex-based aqueous humor analyses suggested MCP-1 as a potential biomarker for the angiogenic phase of AMD. 41 Yamada et al. 42 further demonstrated that VEGF upregulates MCP-1 expression. The parallel increase in VEGFA and MCP-1 in the nAMD group supports the feasibility of tear biomarker detection for this disease. MMP-1, a matrix metalloproteinase, participates in tissue remodeling, wound healing, angiogenesis, and inflammation by degrading extracellular matrix (ECM) components. 43 In ocular tissues, MMP-1-mediated collagen remodeling contributes to both homeostasis and pathological processes. 44
Compared to PCV, nAMD is more frequently associated with fibrosis. 45 During nAMD progression, choroidal neovascularization and macular leakage trigger pathological responses leading to fibrotic scar formation. As a key enzyme in ECM degradation, MMP-1 is activated during this process to facilitate neovascularization and tissue remodeling, which may explain its elevated levels in nAMD patient tears. OPG, a glycoprotein in the TNF receptor superfamily, exhibits anti-apoptotic effects and has been implicated in vascular diseases development. 46 Previous studies reported elevated serum OPG in nAMD patients and proposed that OPG may act as a protective factor or a secondary response within the disease process. 47 The increased tear OPG levels observed in nAMD patients may reflect a compensatory mechanism aimed at suppressing pathological angiogenesis. OPG might inhibit TNF-induced apoptosis and reduce pro-fibrotic cytokines release, thereby attenuating fibrotic progression.
Current anti-VEGF therapies demonstrate suboptimal efficacy in PCV, highlighting the need to explore more cost-effective and targeted therapeutic strategies. In our previous studies, we administered periocular injections of triamcinolone acetonide to PCV patients with subretinal hemorrhage and observed superior therapeutic outcomes compared to anti-VEGF treatment. 48 The effectiveness of conventional anti-inflammatory agents partially supports the hypothesis that inflammatory processes play a significant role in the pathogenesis of PCV. However, further investigation into the underlying etiology and molecular mechanisms is essential to establish a stronger foundation for future treatment strategies.
Conclusion
This study focused on the analysis of baseline data from patients with PCV and nAMD. An exploratory, broad-spectrum screening strategy was used to qualitatively evaluate a wide array of cytokines in tear fluid, aiming to identify factors with notable expression trends. The results provide a foundation for future targeted quantitative studies. We suggest that tear fluid holds great potential as a valuable tool for investigating diseases such as PCV and nAMD. Variations in tear composition may not only reflect underlying disease mechanisms but could also offer a convenient and noninvasive biomarker for dynamically monitoring disease activity and optimizing the timing of re-treatment during long-term anti-VEGF therapy.
Authors’ Contributions
Y.C. and S.Z. contributed equally to both the research and the manuscript writing; Q.Z. and S.C.were responsible for data collection; H.D. and W.Y. performed the data analysis; K.Z. designed the research, reviewed the manuscript, and is the corresponding author. All authors have read and approved the final version of the manuscript.
Footnotes
Author Disclosure Statement
The authors declare that they have no conflicts of interest.
Funding Information
This study was supported by Hainan Provincial Key R&D Program (Social Development Project; Grant No. ZDYF2022SHFZ124; granted to: K.Z.) and Hainan General Hospital 530 Talent Development Project (Grant No.2021MSXM06; granted to K.Z.).
