Abstract
Introduction
Although many backcountry first aid kits contain antibiotic ointment, the supply can be quickly exhausted if a patient has extensive wounds or if there are multiple patients.
Methods
We assessed the antibacterial properties of bark extract from four North American woody plant species known to native Missourians as medicinal plants (Quercus macrocarpa, Salix humilis, Pinus echinata, and Hamamelis vernalis). We tested their antimicrobial properties, with the disc diffusion technique, against four common pathogenic bacterial species: Klebsiella pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus, and Enterobacter aerogenes (now known as Klebsiella aerogenes).
Results
We report evidence of antibacterial activity of bark extract from all four plant species.
Conclusions
Our results confirm that traditional uses of these species may be useful in fighting infection and could be especially useful in a wilderness setting when modern antibiotics are exhausted.
Introduction
Indigenous knowledge of herbal medicines is a common first step in new drug discovery. In North America, thousands of plant species have been historically used by Native peoples to treat a multitude of diseases and other ailments such as bacterial infection.1,2 There exist historical accounts of the medicinal use of Prairie Willow (Salix humilis) by the Cherokee, Menominee, and Meskwaki people; Bur Oak (Quercus macrocarpa) by the Chippewa and Iroquois people; Shortleaf Pine (Pinus echinata) by the Rappahannock people; and Witch Hazel (Hamamelis virginiana) by the Cherokee and Iroquois people to treat illnesses commonly associated with bacterial infections such as diarrhea, pneumonia, and sore throats as well as their topical use to treat open wounds and as a disinfectant.1,3 Confirmation of the antibiotic properties of plant extracts is often a first step in the discovery of new pharmaceuticals, but confirmation of traditional use may also prove to be especially valuable in situations where commercial antibiotics are unavailable or limited.
Methods
Procedures
The plant species used were selected according to three criteria: (1) the species was native to the Missouri region, (2) there existed historical evidence of its medicinal use at either the species or genus level, and (3) the species was represented by a minimum number of three trees in the Washington University Arboretum. The bacterial species utilized for the disk diffusion assay were selected due to their ability to infect wounds and their availability as stock cultures within the Washington University facilities. Bark extraction was done in diluted ethanol (Fisher Scientific Decon Labs ethanol, 200 proof [100%], USP; catalog number 04-355-223). The disk diffusion assay was set up using standard Petri dishes (VWR Petri Dish size 100 × 15 mm; catalog number 25384-302) and Mueller Hinton media (BD Difco Mueller Hinton Power; catalog Number DF0757-17-6). Diffusion disks (Thermo Scientific Remel Blank Paper Disk; Catalog number R55054) were applied to the inoculated Petri dishes immediately after extract application.
Sample Collection
Twigs of approximately 1 cm in diameter were collected from Q. macrocarpa, S. humilis, P. echinata, and H. vernalis. Samples were taken from three separate plants of each species. Any buds (S. humilis) or flowers (H. vernalis) were removed prior to bark collection. Twigs were stripped of their bark using a vegetable peeler, and the bark samples were cut into short strips approximately 8–10 cm long to facilitate processing. The samples were then stored in open paper bags and allowed to dry at 30 °C and 20% humidity for 4 d. 4
Extraction
After drying, bark samples were crudely ground with a commercial spice grinder, resulting in a heterogeneous solid mixture of bark powder and short, thin fibers. All ground samples were extracted by reflux boiling in 25 mL 70% ethanol for 6 h at 80 °C. The extract was then vacuum filtered to remove bark fibers and debris. The resulting filtrate was evaporated at 50 °C using a rotary evaporator to obtain solid extracts.5,6 The dried extracts were stored in glass vials at −80 °C until application to the culture plates.
Plating
Bacterial cultures of Enterobacter aerogenes, Klebsiella pneumoniae, Staphylococcus aureus, and Pseudomonas aeruginosa were diluted to a 0.5 McFarland standard using a 0.85% NaCl saline solution. For each bacterial species, two Mueller-Hinton Agar plates were inoculated using the spread plate method with a sterile cotton swab, dipped once into the diluted culture. 5 Three replicates, each of two different extracts, were tested per plate. The commercially available triple antibiotic Neosporin, which contains neomycin sulfate, polymyxin B, and bacitracin zinc, and the broad-spectrum antibiotic tetracycline were used as positive controls. Disks saturated with water served as a negative control.
Disk Diffusion Assay
Extracts were resuspended in water to a concentration of 10 mg/mL. Sterile 5 mm diffusion disks were saturated with 50uL of bark extract before being placed onto the inoculated plates. 5 The Neosporin control was tested via the disk diffusion method as well as direct application to the inoculated plate. Plates were allowed to grow at 37 °C for 17 h and then observed for the presence of zones of inhibition. Any zones of inhibition (ZoIs) were measured with a ruler, and the diameter of the zone was recorded in millimeters (mm).
Statistics
Statistical comparison of the effectiveness of each extract was limited due to each bacteria-extract interaction being described by only three samples in the case of H. vernalis, S. humilis, and P. echinata or two samples in the case of Q. macrocarpa. Instead, a mean calculation of the three ZoIs, corresponding to Samples A, B, and C of each plant species (B and C for Q. macrocarpa) was made for each extract-bacteria pair and presented alongside its standard deviation.
Results
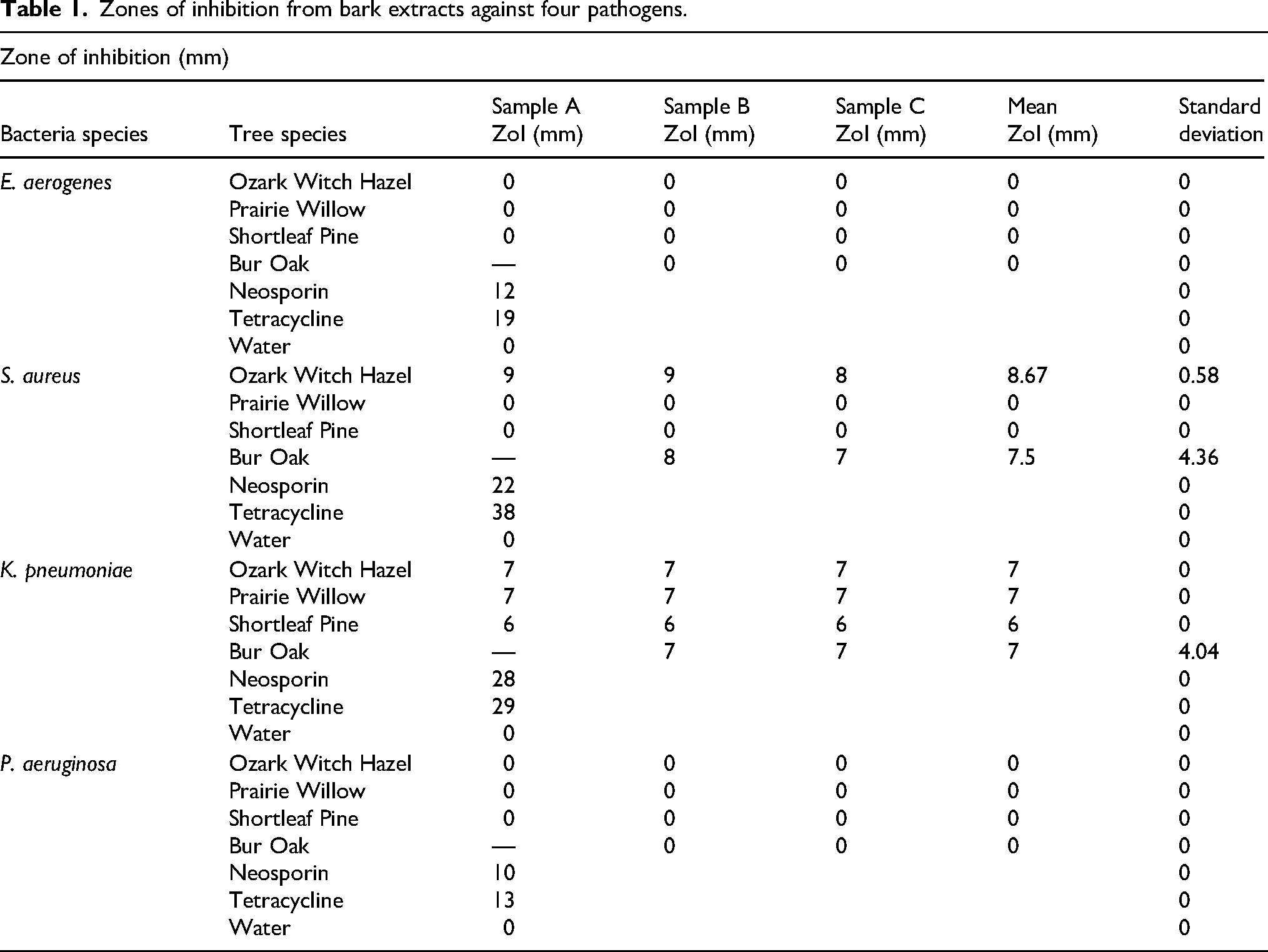
Bark extracts from all four tree species exhibited some antimicrobial effect, but none were as effective as Neosporin (Table 1). One sample from Bur Oak was not included in the analysis due to a misidentification of the tree. Since bacterial resistance and susceptibility to antibiotics is dependent on the concentration of the active compound and we were not seeking to identify or quantify the exact compound(s) responsible, the presence or absence of ZoIs was used to determine if each tested extract had antibacterial activity against the target bacterial species. E. aerogenes and P. aeruginosa exhibited no response to treatment with any of the four bark extracts. S. aureus was inhibited by Ozark Witch Hazel (8.67 mm) and Bur Oak (7.5 mm). K. pneumoniae was susceptible to all extracts with a mean zone of inhibition of 7 mm for Ozark Witch Hazel, Prairie Willow, and Bur Oak and 6 mm for Shortleaf Pine.
Zones of inhibition from bark extracts against four pathogens.
Discussion
We examined the antimicrobial activity of bark extract from four native Missouri woody plant species for which there was evidence of historical medicinal use by Native American peoples. We demonstrated antibacterial activity by bark extracts of S. humilis, Q. macrocarpa, P. echinata, and H. vernalis against K. pneumoniae as well as antibacterial activity of Q. macrocarpa and H. vernalis extracts against S. aureus. None of these bark extracts were as effective as Neosporin and only Neosporin was effective against E. aerogenes or P. aeruginosa. However, the fact that all had antibiotic properties supports the hypothesis that indigenous use of these species in medicinal poultices and teas was based on real efficacy.
Many traditional European herbal medicines were selected by Galen's Doctrine of Signatures, which matched the superficial appearance of a plant to the affected body part. 7 In contrast, Native American herbal traditions appear to have been based more on efficacy than doctrine. 1 Decades ago, McCutcheon tested the antibiotic and antimycobacterial activity of dozens of traditional Native American herbal remedies.8,9 They found that extracts from 82 of 96 plant species used in Native American traditional medicine did, in fact, have antibiotic activity. 8 Among these, 75 were active against S. aureus and 46 were active against P. aeruginosa. 9 Furthermore, 13 of the 19 species that were found to be active against Mycobacterium tuberculosis were traditionally used to treat tuberculosis by native people. 8 Hence, it is not surprising that our results support the effectiveness of traditional native Missouri bark extracts against some bacterial species.
In the absence of modern antibiotics, traditional teas made from these barks are likely to have real therapeutic value against K. pneumoniae, and a traditional poultice of Q. macrocarpa or H. vernalis is likely to help fight or avoid infection of superficial wounds. We are not advocating the use of these barks in place of antibiotic pharmaceuticals but call attention to their real antimicrobial activity and potential value in remote emergency situations when traditional pharmaceuticals are not available.
Conclusions
All of the tree bark extracts studied were found to have some antibacterial properties. While the zones of inhibition for the experimental bacteria-extract treatment pairs that did produce regions of inhibited bacterial growth do not match even the lowest observed susceptibility breakpoints for the species—15 mm for Staphylococcus spp. treated with Gentamicin and 9 mm for K. pneumoniae treated with Cefpodoxime—the presence of the ZoIs reflects some antimicrobial activity against these bacterial species. 10 As prior studies have found, the degree of antimicrobial activity varies both by the extraction method and in the concentration used for treatment. Understanding the influence of seasonality on extract efficacy, the impact of concentration, and the identification of other susceptible bacterial species will be necessary steps in the continued investigation of these species as useful sources of antibiotic treatments. Additionally, owing to the use of specialized laboratory equipment in the present study, the extraction protocol used here would not be repeatable in a wilderness setting. However, extraction in boiling water would be more in accord with traditional usage and better serve in resource-scarce environments.
Although our research involved a small sample size (three representatives from each of the chosen woody plant species) and one set of trials against the selected bacterial species, it supports the general hypothesis that many traditional indigenous medications are, in fact, biologically effective. Additionally, our results suggest that traditional poultices from the bark of these four woody plant species may offer antibiotic support when commercial pharmaceuticals are unavailable.
Footnotes
Acknowledgments
Thank you to Dr. Kevin Moeller and members of the Moeller Lab within the Washington University in St. Louis Department of Chemistry for sharing their laboratory space as well as offering guidance as we planned and conducted our experiment, to the Jeanette Goldfarb Plant Growth Facility's horticulturists for allowing us to use their growth chambers, and to the Washington University Arboretum for inspiring and supporting our research. We would like to especially voice our appreciation for the invaluable guidance, support, and effort of our coauthor Dr. Stan Braude (deceased), Professor of Biology and Curator of the Washington University Arboretum. His long and impressive history as a researcher, educator, and mentor made it an honor to work alongside him. We hope this work can contribute to his legacy in the fields of wilderness medicine and conservation biology.
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
