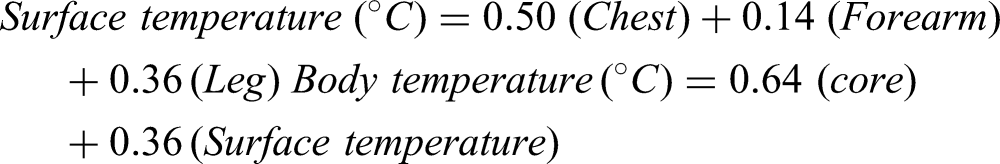Abstract
Introduction
Capsaicin, a chili pepper extract, can stimulate increased skin blood flow (SkBF) with a perceived warming sensation on application areas. Larger surface area application may exert a more systemic thermoregulatory response. Capsaicin could assist with maintaining heat transport to the distal extremities, minimizing cold weather injury risk. However, the thermoregulatory and perceptual impact of topical capsaicin cream application prior to exercise in the cold is unknown.
Methods
Following application of either a 0.1% capsaicin or control cream to the upper and lower extremities (10 g total, ∼40–50% body surface area), 11 participants in shorts and a t-shirt were exposed to 30 min of cold (0 °C, 40% relative humidity). Exposures comprised of 5 min seated rest, 20 min walking (1.6 m·s−1, 5% grade), and 5 min seated rest. Temperature (skin, core), SkBF, skin conductivity, heart rate, thermal sensation, and thermal comfort were measured throughout.
Results
The capsaicin treatment did not differ from the control treatment in skin temperature (treatment mean: 30.0 ± 2.5, 30.1 ± 2.4 °C, respectively, p = 0.655), core temperature (treatment mean: 37.3 ± 0.5, 37.4 ± 0.4 °C, respectively, p = 0.113), SkBF (treatment mean: −8.4 ± 10.0, −11.1 ± 10.7 A.U., respectively, p = 0.492), skin conductivity (treatment mean: −0.7 ± 5.1, 0.4 ± 6.4 µS, respectively, p = 0.651), or heart rate (treatment mean: 83 ± 29, 85 ± 28 beats·minute−1, respectively, p = 0.234). The capsaicin and control treatments also did not differ in thermal sensation (p = 0.521) and thermal comfort (p = 0.982), with perceptual outcomes corresponding with feeling “cool” and “just uncomfortable,” respectively.
Conclusions
0.1% topical capsaicin application to exposed limbs prior to walking in a cold environment does not alter whole-body thermoregulation or thermal perception.
Keywords
Introduction
During cold exposures, vessels in the skin vasoconstrict and decrease peripheral shell blood flow to minimize the heat loss gradient and protect core temperature. 1 However, reduced peripheral shell blood flow linearly reduces heat transport to the distal extremities,2,3 which can contribute to cold weather injury risk. Cold exposures can thus create a conflict at the distal extremities when protecting core temperature becomes imperative. Targeting and exploring cold weather countermeasures that help maintain or increase skin blood flow (SkBF) could help mitigate cold weather injury risk by maintaining heat transport to the distal extremities. Recent works have begun examining how pharmaceuticals can influence autonomic thermoregulation and perception.
Capsaicin, a chili pepper extract4–6 in some over-the-counter analgesics, is thought to influence autonomic thermoregulation by inducing vasodilation and SkBF1,7 on application areas through activation of a thermoreceptor, transient receptor potential vanilloid 1 (TRPV1). 6 TRPV1 is activated by painfully hot skin temperatures >45 °C.1,7,8 However, capsaicin uniquely sensitizes and lowers the neuronal/discharge activation threshold of TRPV1 to lower less painful skin temperatures. 9 In contrast to the cold environment scope of the present work, capsaicin patches and creams are largely explored to enhance convective or evaporative heat dissipation in hot environments by sensitizing heat thermoreception.10–12 Local capsaicin application with local heat is successful at provoking vasodilation and increased SkBF in comparison to a control.13,14 However, attempts to replicate local capsaicin effects at the whole-body level in ambient heat with patches11,12 or large topical coverage areas 10 varies, demonstrating successful vasodilation and increased SkBF as well as no effect in comparison to a control. These equivocal findings may be explained by a cutaneous vasodilatory ceiling for heat dissipation. 15 The combined thermoregulatory needs of exercise in a hot ambient environment might be indistinguishable from any capsaicin-induced changes in thermoregulation. The isolated influence of capsaicin on thermoregulation may be evident in a cold environment where there is no vasodilatory or SkBF ceiling, and any possible capsaicin-induced TRPV1 activation could be distinctly observed.
Regardless of thermoregulatory outcome, what remains common between the local and whole-body levels is a perceived heat/burning sensation on application areas.10–14 To temper capsaicin-induced heat/burning, some have used capsaicin and local cooling in tandem,16–19 wherein cooling capsaicin-treated skin alleviates heat/burning sensations and reduces cold sensitivity/perception.16–19 Capsaicin appears to inhibit cold peripheral thermoreception, but it remains unclear if cold desensitization translates to the whole body with vasodilation and increased SkBF. Recent works examining opposing pharmacological and environmental stimuli (ie, pharmacological menthol cooling in hot environments) have found that whole-body thermoregulation can be altered despite opposing afferent signals. 20 Using this rationale, it is conceivable that a heat signal from capsaicin could prevail during a cold exposure. Capsaicin use during cold exposure could help maintain heat transport to the distal extremities via vasodilation and SkBF while also reducing cold perception. The overall outcome would help minimize cold weather injury risk with more comfortable environmental perception.
The thermoregulatory and perceptual outcomes of capsaicin application in ambient cold environments with exercise are yet to be assessed. A possible consequence is a conflicting cold and capsaicin-induced response. Vasoconstriction and decreased SkBF are protective in the cold, and deliberately imposing a pharmacologically opposed heat stimulus could inadvertently decrease body temperature. Yet, superimposing capsaicin use in the cold with exercise could take advantage of metabolic heat production to assist with core temperature maintenance. Thus, cold environments with capsaicin could be physiologically tolerated. Therefore, the purpose of this work is to determine the thermoregulatory and perceptual outcomes due to topical capsaicin application to the limbs during acute exercise in a novel cold environment. This work will help direct potential ensuing capsaicin uses in cold environments. We hypothesize that topical capsaicin application prior to a cold exposure will assist in maintaining/increasing SkBF and skin temperature while still permitting metabolic heat production from exercise to maintain core temperature in comparison to a control. We further hypothesize that topical capsaicin will improve thermal perception and thermal comfort as compared to a control.
Methods
Preliminary Testing
The University of Montana's Institutional Review Board approved all ethics and procedures (IRB# 31-32), and all participants provided written informed consent. Participants (n = 11, 8 males, 3 females, 29 ± 7 y, 77.2 ± 13.7 kg, 177.1 ± 8.1 cm, 17.3 ± 8.1% body fat) without capsaicin allergies and who did not habitually use capsaicin were recruited from the surrounding university area and cleared for physical activity using guidelines established by the American College of Sports Medicine. 21 Prior to experimental testing, a small amount of 0.1% capsaicin cream (CVS, Woonsocket, RI) was applied to each participant’s dorsal forearm (∼5 cm × ∼5 cm) and monitored for 15 min at ambient temperature for rash, hives, or severe/abnormal burning sensations. Sex-specific body composition was then derived from skinfolds measured at the chest, midaxillary, triceps, subscapular, abdomen, suprailiac, and thigh using Harpenden skinfold calipers. 21
Experimental Testing
Two treatments were completed in a single-blind, randomized, counterbalanced, and repeated measures crossover design. The capsaicin and control cream (CVS, Woonsocket, RI) were from the same national pharmacy chain and similar in color, texture, and smell (lack of). The highest available over-the-counter capsaicin concentration was selected. The present dose was chosen in relation to the minimum amount and concentration required (1 g, 0.025%) for limb coverage and sensation on treatment areas. 10 Both creams were preweighed into four 2.5 g portions totaling 9.98 ± 0.02 g. Treatments were separated by ≥72 h to account for capsaicin carryover effects.
Each experimental visit required a minimum 3 h fast while abstaining from alcohol, caffeine, and strenuous exercise during the preceding 24 h. Experimental visits began with a nude body weight prior to instrumentation with a self-inserted (12–15 cm) rectal thermistor, 3 skin thermistors, heart rate chest strap, SkBF probe, and finger electrodes. Participants wore shorts, a t-shirt, socks, and shoes for each trial. Measurements began with 5 min of seated baseline data at ambient temperature (23.9 ± 0.6 °C, 21.9 ± 2.8% relative humidity). A gloved researcher then applied 2.5 g of cream to each limb from shoulder to wrist and mid-thigh to ankle around but not under each sensor. The distance without cream to the center of the temperature and SkBF sensors on the limbs was ∼1.3 cm, wherein capsaicin can elicit an effect up to 4 cm beyond the coverage area. 13 Cream covered an estimated 40–50% total body surface area. 22 There were no immediate sensational differences (heat, burning) between treatments upon application. Limbs were chosen as the application sites to assess if SkBF and, thus, heat transport could be tracked upstream of the hands and feet where cold weather injuries commonly occur. 23 Following cream application, participants entered an environmental chamber (Tescor Inc., Warminster, PA) set to 0 °C and 40% relative humidity (0.8 ± 0.3 °C, 61.7 ± 9.9% relative humidity) for an additional 5 min seated rest (initial cold), 20 min of walking at 1.6 m·s−1 and 5% grade on a motorized treadmill (Woodway, Waukesha, WI, USA), and a final 5 min seated rest (final cold). Participants marked their perceived thermal sensation and thermal comfort during all rest periods and exercise on two 20 cm continuous visual analog scales.24,25 The thermal sensation questionnaire asks for perception of the ambient temperature with 9 equally spaced prompts ranging from very cold to very hot. The thermal comfort questionnaire asks for perceived satisfaction of the ambient environment with 6 equally spaced prompts ranging from very uncomfortable to very comfortable. Experimental visits concluded with a nude body weight. Heart rate, skin temperature, core temperature, SkBF, and skin conductivity were collected at 1000 Hz throughout and integrated into an analog-to-digital converter (ADInstruments, Sydney, Australia).
Heart Rate and Temperatures
Heart rate signals were transmitted from a chest strap (Polar, Kempele, Finland) to a receiver (Polar, Kempele, Finland) at a cyclic threshold detection of 0.85 volts. Core (Physitemp, Clifton, NJ) and skin thermistor (Physitemp, Clifton, NJ) measurements were digitized via pods (ADInstruments, Sydney, Australia). Heart rate and temperature were analyzed as 5 min averages during rest and two 10 min averages during exercise. Skin temperature was measured on the left side of each participant at three sites: chest ∼8–10 cm from the midline between the sternum and xiphoid process, forearm on the extensor digitorum midline, and calf on the tibialis anterior in line with the lower gastrocnemius head.
26
Surface temperature was calculated using a weighted aggregate of all 3 skin sites, and body temperature was calculated using a weighted aggregate of surface and core temperatures via the following Burton equations
27
:
Skin Blood Flow and Skin Conductivity
SkBF was measured using a standard surface probe (ADInstruments, Sydney, Australia) and single-channel laser Doppler flowmeter (ADInstruments, Sydney, Australia). SkBF is a relative measurement calculated as the change from baseline. Laser Doppler flowmetry measures can be impaired by potential differences in regional/spatial microvasculature. 28 To ensure consistent regional/spatial microvasculature, the probe was placed on the flattest part of the forearm distal (<3 cm) to the forearm thermistor. This consistency is further evinced via post-hoc paired t-test analysis for baseline SkBF flux (Microsoft Excel, Redmond, WA). The capsaicin and control treatments did not differ in baseline SkBF flux (53.0 ± 9.9 AU, 54.9 ± 10.2 AU, respectively, p = 0.566). Thus, baseline flux values suggest a suitable probe placement for which to calculate the relative change in SkBF. Laser Doppler flowmetry probes are also sensitive to movement artifact 28 from exercise. For this reason, resting data alone was analyzed as the change from baseline in 5 min averages. Data was collected using a low pass 50 Hz filter and 5 s rolling average according to manufacturer specifications.
Skin conductivity was measured from finger electrodes (ADInstruments, Sydney, Australia) Velcro strapped to the index and middle fingers’ proximal phalanx palmar surface via an amplifier (ADInstruments Sydney, Australia). Skin conductivity is quantified from moisture produced beneath reference electrodes as a relative measurement of sweat gland activity/output29–34 from preceding physiological stimuli. Higher conductivity suggests greater sweat gland activity in microsiemens (µS). We have previously used the presented skin conductivity technique 20 to estimate sweat gland activity, but other permutations have been used in humans.35–37 The skin conductivity amplifier was zeroed during each treatment prior to the baseline period. Resting periods were averaged into 5 min segments and exercise was averaged into two 10 min segments for analysis as the change from baseline.
Statistical Analysis
A repeated measures 2-way analysis of variance (treatment × time) was used to assess differences in all dependent variables. Sphericity was evaluated using Mauchly's test of sphericity and corrected using the Huynh-Feldt procedure when appropriate. Post-hoc comparisons were completed with a Fisher's protected least significant difference procedure. Analysis of variance was completed using SPSS (IBM, Armonk, NY). Statistical significance was set at a type I probability error of < 5% (p < 0.05). Data are presented as mean ± SD.
Results
Body Weight
Body weight decreases did not differ between the capsaicin and control treatments (p = 0.093). There was, however, an overall decrease in body weight from pre-exercise (grand mean: 77.8 ± 13.4 kg) to post-exercise (grand mean: 77.6 ± 13.4 kg, p < 0.001) that amounted to 0.11 ± 0.10 kg and 0.14 ± 0.12%.
Heart Rate
Heart rate did not differ between the capsaicin and control treatments (p = 0.234). Heart rate decreased from baseline (grand mean: 70 ± 17 beats·minute−1) upon initial cold exposure (grand mean: 65 ± 17 beats·minute−1, p = 0.001), increased from baseline due to exercise (grand mean 0–10 min: 105 ± 25, beats·minute−1, grand mean 10–20 min: 107 ± 28, beats·minute−1, p < 0.001), and recovered to baseline levels during the final cold period (grand mean: 71 ± 22, beats·minute−1, p = 0.627). Resting heart rate upon initial cold exposure was lower than the final cold period (p < 0.001). Heart rate did not differ during exercise (p = 0.061). Both resting cold periods were lower than exercise (p < 0.001).
Temperatures
Chest and forearm temperature did not differ between the capsaicin and control treatments (p = 0.849: Figure 1A and p = 0.792: Figure 1B, respectively). Chest and forearm temperature decreased from baseline upon initial cold exposure that persisted into the final resting cold period (p < 0.001: Figure 1A, 1B, respectively). Calf temperature did not differ between the capsaicin and control treatments (p = 0.848, Figure 1C). Calf temperature decreased from baseline upon initial cold exposure which persisted into the final resting cold period (p < 0.001, Figure 1C). However, calf temperature did slightly increase due to exercise when min 0–10 and 10–20 are compared (p = 0.022). This elevation in calf temperature remained when exercise min 0–10 and 10–20 were compared to the final cold period (p = 0.002, p = 0.007, respectively, Figure 1C). Surface temperature did not differ between the capsaicin and control treatments (p = 0.655, Figure 1D). Surface temperature derived from the Burton equation 27 using weighted chest, forearm, and calf temperatures decreased from baseline upon initial cold exposure which persisted into the final resting cold period (p < 0.001, Figure 1D).
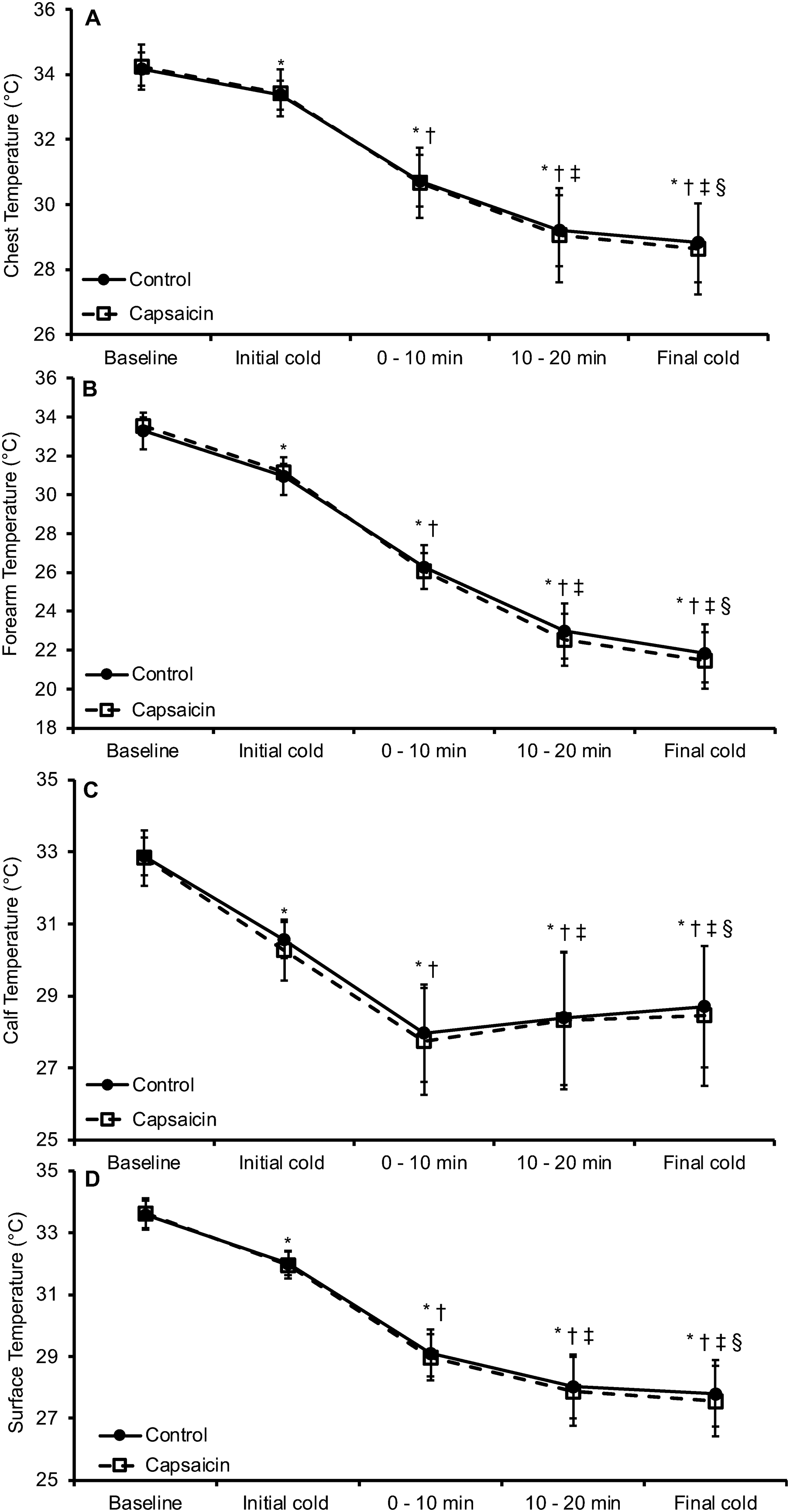
A, chest; B, forearm; and C, calf temperatures during two seated 5-min pre-exercise periods (baseline, initial cold), 20 min of walking, and seated 5-min post-exercise period (final cold). D, surface temperature was derived from an aggregate weighted calculation of chest, forearm, and calf temperatures. Topical capsaicin or a control cream was applied between the baseline and initial cold periods. *p < 0.05 from baseline, †p < 0.05 from initial cold, ‡p < 0.05 from min 0–10, §p < 0.05 from min 10–20. Data presented as mean ± SD.
Core temperature did not differ between the capsaicin and control treatments (p = 0.113, Figure 2A). Core temperature decreased from baseline upon initial cold exposure (p < 0.001), recovered to baseline levels due to exercise (0–10 min: p = 0.144, min 10–20: p = 0.079), and was elevated above baseline during the final cold period (p < 0.001, Figure 2A). Core temperature upon initial cold exposure was lower than the final cold period (p = 0.001, Figure 2A). Core temperature increased due to exercise when min 0–10 was compared to min 10–20 (p < 0.001, Figure 2A). Core temperature upon initial cold exposure was lower than exercise (min 0–10: p = 0.041, min 10–20: p < 0.001, Figure 2A). Core temperature during the final cold period was higher than exercise (p = 0.001, Figure 2A). Body temperature did not differ between the capsaicin and control treatments (p = 0.190, Figure 2B). Body temperature as derived from the Burton equation 27 using weighted surface and core temperatures decreased from baseline upon initial cold exposure which persisted until min 10–20 of exercise (p < 0.001), wherein body temperature stabilized and did not differ from the final cold period (p = 0.108, Figure 2B).
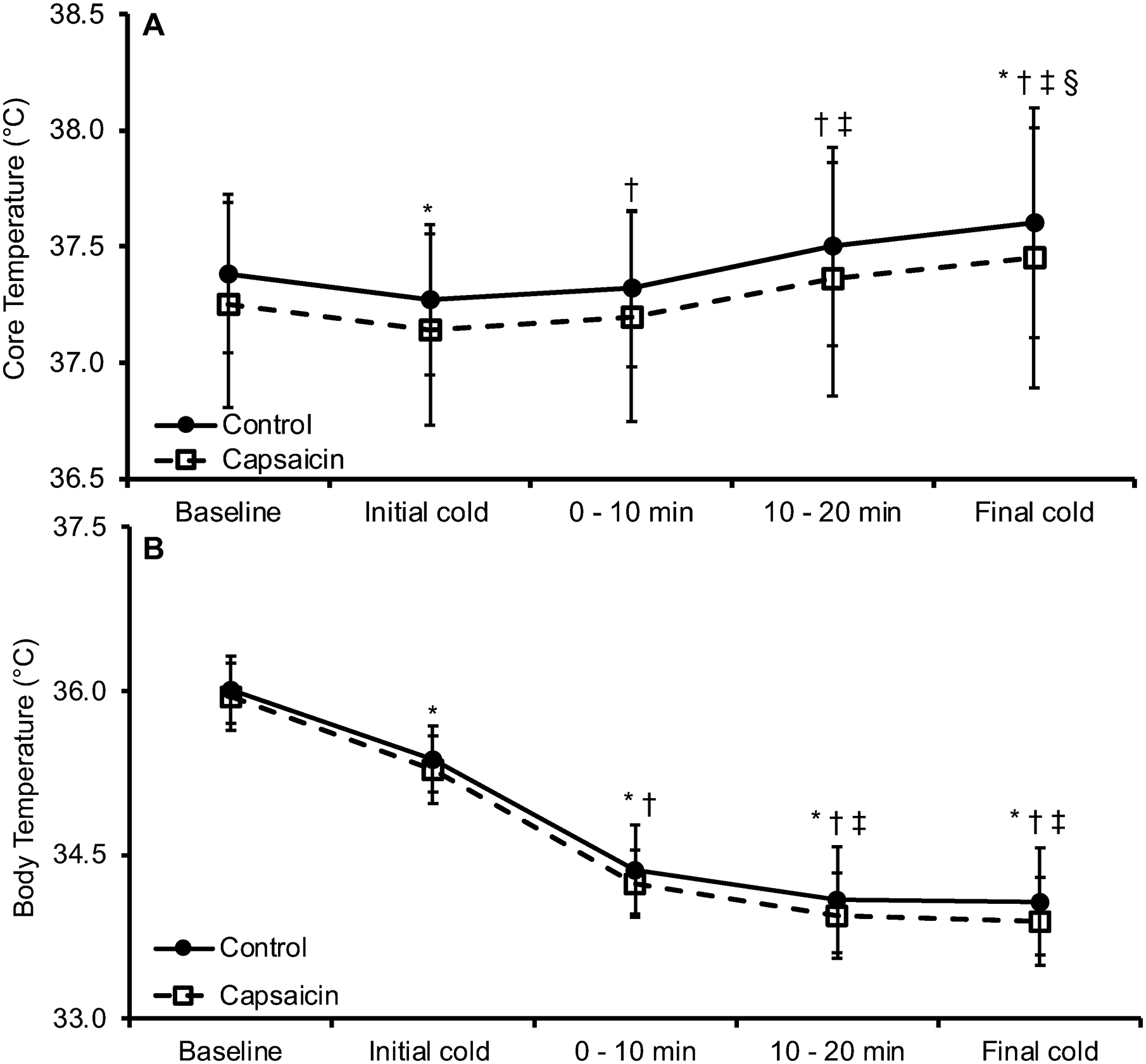
A, core temperature during two seated 5-min pre-exercise periods (baseline, initial cold), 20 min of walking, and a seated 5-min post-exercise period (final cold). B, body temperature was derived from an aggregate calculation of surface (Figure 1D), and A, core temperatures. Topical capsaicin or a control cream was applied between the baseline and initial cold periods. *p < 0.05 from baseline, †p < 0.05 from initial cold, ‡p < 0.05 from min 0–10, §p < 0.05 from min 10–20. Data presented as mean ± SD.
Skin Blood Flow and Skin Conductivity
SkBF did not differ between the capsaicin and control treatments (p = 0.492). SkBF decreased from baseline (set to 0 AU) upon initial cold exposure (grand mean: −7.1 ± 11.3 AU, p = 0.012) that persisted into the final cold period (grand mean: −12.4 ± 8,7 AU, p < 0.001). SkBF decreases approached significance when comparing the initial cold exposure to the final cold period (p = 0.055).
Skin conductivity did not differ between the capsaicin and control treatments (p = 0.651, Figure 3). Skin conductivity was not different from baseline (set to 0 µS) upon initial cold exposure (p = 0.130), during exercise (min 0–10: p = 0.655, min 10–20: p = 0.518), or the final cold period (p = 0.195). Skin conductivity decreases approached significance when comparing the initial cold exposure to exercise (min 0–10: p = 0.080, min 10–20: p = 0.055) and was significantly decreased when compared to the final cold period (p = 0.009, Figure 3). Skin conductivity during exercise approached significant decreases when min 0–10 is compared to min 10–20 (p = 0.058). Skin conductivity during the final cold period was lower than exercise min 0–10 (p = 0.006) but not exercise min 10–20 (p = 0.176, Figure 3).
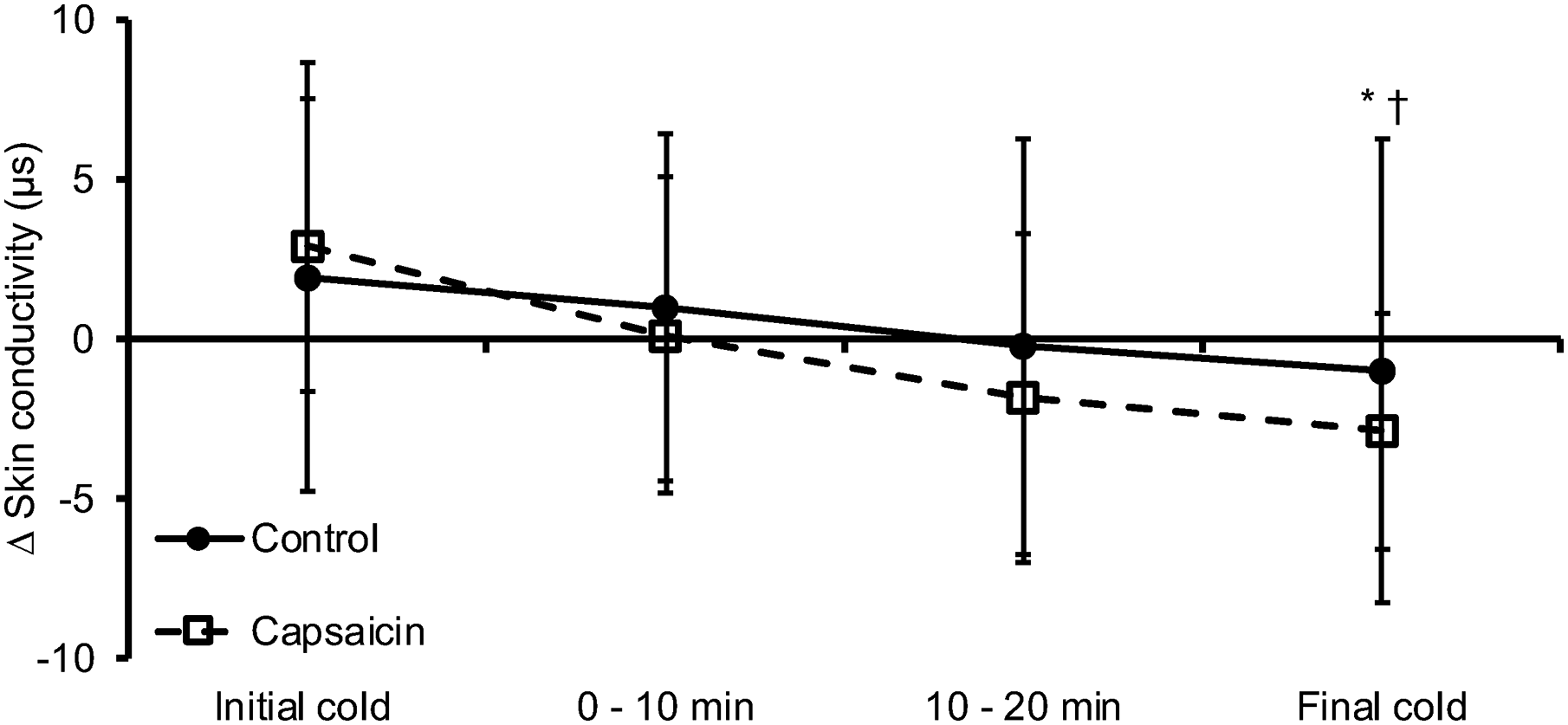
Change in skin conductivity from a 5-min seated baseline period (set to 0 AU) during rest in the cold (initial cold), 20 min of exercise, and a seated 5-min post-exercise period (final cold). Topical application capsaicin or control cream were applied prior to the initial cold period. *p < 0.05 initial cold, †p < 0.05 from min 0–10. Data presented as mean ± SD.
Thermal Sensation and Thermal Comfort
Thermal sensation did not differ between the capsaicin and control treatments (p = 0.521). Thermal sensation decreased from baseline (grand mean: 104.2 ± 17.0 mm) during the initial cold exposure (grand mean: 38.9 ± 17.1 mm, p < 0.001), exercise (grand mean min 0–10: 38.0 ± 16.2 mm, grand mean min 10–20: 34.4 ± 15.6 mm, p < 0.001), and the final cold period (grand mean: 35.1 ± 18.4 mm, p < 0.001). Thermal sensation did not change once participants entered the cold chamber (p > 0.05). Thermal sensation values began at “neutral” during baseline before cold exposure, wherein values corresponded with becoming “cold.”
Thermal comfort did not differ between the capsaicin and control treatments at any point (p = 0.982). Thermal comfort decreased from baseline (grand mean: 140.2 ± 25.3 mm) during the initial cold exposure (grand mean: 66.9 ± 18.9 mm, p < 0.001), exercise (grand mean min 0–10: 63.7 ± 20.3 mm, grand mean min 10–20: 54.5 ± 23.0 mm, p < 0.001), and the final cold period (grand mean: 49.8 ± 24.0 mm, p < 0.001). Thermal comfort decreased from the initial cold exposure to the final cold period (p = 0.033). Thermal comfort decreased during exercise from min 0–10 to 10–20 (P = 0.021). Thermal comfort did not differ from initial cold exposure due to exercise (min 0–10: p = 0.651, min 10–20: p = 0.148). Thermal comfort during the final cold period was lower than exercise min 0–10 (p = 0.033) but not different than exercise min 10–20 (p = 0.441). Thermal comfort values began at “comfortable” during baseline, decreasing to “just uncomfortable” and “uncomfortable” throughout the cold exposure.
Discussion
The purpose of this work was to determine the whole-body thermoregulatory and thermal perception outcomes of 0.1% topical capsaicin application to exposed limbs prior to walking in a cold environment. We targeted capsaicin as a possible cold weather countermeasure that maintains heat transport via vasodilation and SkBF to the extremities with a concurrent reduction in cold perception that still permits metabolic heat production from exercise to maintain core temperature. We demonstrated no capsaicin-induced thermoregulatory or perceptual effect in the cold. The only outcomes exhibited were due to the cold environment: skin and body temperature decrease, SkBF decrease, skin conductivity maintenance/decrease, cold-induced bradycardia, perceived ambient cold, and perceived thermal discomfort. Based on these findings, topical capsaicin has minimal whole-body thermoregulatory and perceptual impact during exercise in the ambient cold with exposed skin.
Any potential capsaicin-induced effects may have been abolished by the marked reduction in skin temperature. Capsaicin's burning/heat sensation on the skin can be amplified with external heat. The additive, synergistic, and/or hyperalgesic capsaicin effect suggests some interaction between external heat or at least warmer skin temperatures. Skin temperature may, therefore, be more crucial in mediating capsaicin's effect than core temperature. This is evinced by the more consistent vasodilation and SkBF increases seen with local capsaicin and heating but not ambient heat, where the latter could result in a core temperature increase. Capsaicin's effect could become evident under insulative clothing. Here, with exposed skin, the metabolic heat of exercise and the cold environment's feed-forward stimulus 38 to decrease SkBF, we observed a marked skin temperature decrease across treatments with a core temperature maintenance within −0.06 to +0.21 °C of baseline. A commonality among works that do demonstrate a capsaicin-induced vasodilatory effect and SkBF maintenance/increase occurs with skin temperatures >33 °C at the local13,14 and whole-body levels.11,12 Cutaneous vasoconstriction and SkBF decreases are thought to begin at skin temperatures <33–35 °C 39 and are considered complete at skin temperatures <31 °C.3,40 These skin temperature thresholds were breached, with baseline beginning at 33.6 ± 0.5 °C before decreasing to 27.7 ± 1.1 °C during the final resting cold period.
Lower skin temperatures also tend to display lower skin conductivity/sweat gland activity estimates. 41 Though sweat production and SkBF are not always 1:1, 42 capsaicin did not influence SkBF, indicating no maintained and/or additional heat transport from the upper extremities to the skin conductivity finger measurement site. Blood pressure was unmeasured presently, but a capsaicin effect on vasodilation, SkBF, and potentially skin conductivity in the cold could be additionally described by coupling SkBF and blood pressure for cutaneous vascular conductance. However, since treatments did not differ in heart rate and skin temperature, cold exposure was likely the sole variable capable of altering mean arterial pressure via peripheral vascular resistance. Indeed, the cold caused a transient fall in heart rate upon chamber entrance most attributable to a cold-induced bradycardic response due to a periphery to core blood flow redistribution.43,44 Even as heart rate and core body temperature increased due to the exercise demands, there was no additive increase in skin conductivity due to capsaicin. This finding was confirmed by minimal decreases in body weight and, thus, sweat loss.
Capsaicin-treated skin displays desensitization to local cooling16–19 as TRPV1 activation can reciprocally inhibit cold thermoreception. 45 However, these spatial reductions in cold sensitivity on capsaicin-treated areas have been observed using differing capsaicin concentrations and post-treatment strategies than the present. Local reductions in cold sensitivity are demonstrated following 15–30 min treatment periods in temperate testing environments with presumably homeostatic skin temperature ranges (>33 °C) in addition to higher capsaicin concentrations (0.6–8%) than presently used.16–19 Albeit, higher capsaicin concentrations than the 0.1% presently used are not typically available over the counter. Moreover, the cold sensitivity testing is completed with direct contact probes smaller than the treated area16–19 as opposed to an ambient cold-driven reduction in skin temperature. Regardless, capsaicin's effect may only extend up to 4 cm beyond treated areas, 13 which would suggest that regional effects might not translate systemically when exposed to cold environments. Here, capsaicin covered ∼40–50% total body surface area 22 to try and elicit a systemic alteration in environmental perception. Across treatments, the final cold period was perceived as “cold” and “uncomfortable.”
Limitations
The intent of this work was to begin at the furthest edge of potential capsaicin use to steer subsequent possibilities. Skin temperature maintenance or even priming/activating capsaicin's effect with an external heat load to induce amplified heat/burning prior to cold exposure might be required for capsaicin to be a cold weather countermeasure. Alternatively, deliberate capsaicin application to commonly cold weather–injured areas (ie, hands, feet, and nose) and measurement of skin temperature and SkBF at these sites could be an additional cold weather countermeasure avenue. Accordingly, we cannot disqualify possible effects under insulative clothing or even locally at the hands and feet under gloves/socks. Justification for this comes from participant reports of increased heat/burning at conclusion of their capsaicin trials during de-instrumentation and rewarming to the temperate laboratory environment. Participants also reported heating/burning surges when showering, wearing more clothing, and during exercise. During these conditions, skin temperatures were likely elevated above cutaneous vasoconstriction thresholds of 33–35 °C. 39 Although the over-the-counter capsaicin cream used was not independently verified at a 0.1% concentration, the burning sensation reports do confirm the presence of capsaicin.
Conclusions
The aim of this work was to determine the whole-body thermoregulatory and perceptual impact of topical capsaicin application on exposed limbs prior to walking in the cold. There was no effect due to capsaicin on skin or core temperature, heart rate, SkBF, skin conductivity, thermal sensation, or thermal comfort. Topical capsaicin may not alter whole-body thermoregulation or thermal perception when skin is openly exposed to cold environments. The effectiveness of capsaicin under insulative clothing where skin temperature is better maintained cannot be disqualified.
Footnotes
Acknowledgments
The authors thank Dr. Mark McGlynn and Walter Hailes for their technical expertise, assistance, and insightful manuscript revisions. The authors further extend gratitude to the participants for their time and efforts.
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare no competing interests, financial or otherwise. All methodology and ethics were approved by the University of Montana Institutional Review Board (IRB # 31-32) in accordance with the Declaration of Helsinki. All participants provided written, informed consent prior to participation. Data has not been deposited into any public repositories but may be available upon request from the corresponding author.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.