Abstract
Introduction
Patients with acute myeloid leukemia (AML) are at increased risk for fungal, viral, and bacterial infections as a result of hematopoietic and immune dysfunction from leukemia and myelosuppression from therapy. The incidence of bacterial infection is most common (approximately 75–80%), followed by fungal (7–33%) and viral (12–20%) infections.1,2 These rates are variable, depending on prophylaxis strategies and treatment regimens, with infections being a significant source of morbidity and mortality in patients with AML.1,3,4 Recommendations suggest utilization of antimicrobial prophylaxis: however the optimal agent, duration, and treatment for breakthrough infections remain ambiguous. Historically, induction therapy for AML has been 7 days of cytarabine with 3 days of anthracycline (7 + 3). 5 As the landscape for AML treatment evolves, recommendations for prophylaxis may need to adapt as novel regimens become more widely used.
A randomized controlled trial (RCT) from Germany published in 2007 showed that posaconazole was superior to itraconazole or fluconazole with significantly lower rates of invasive fungal infections, invasive Aspergillosis, and death in patients with AML or myelodysplastic syndrome (MDS) with prolonged neutropenia on induction chemotherapy. 6 There is otherwise a dearth of RCT data for fungal prophylaxis, and subsequent retrospective studies and meta-analyses have explored the benefit of prophylaxis,7,8 optimal agent,9–14 duration,15,16 and drug interactions. 17 However, given the risk of fungal infection in induction therapy for AML, the European Conference on Infections in Leukemia (ECIL) recommends prophylaxis with posaconazole. 18
Viral infection, namely with herpes simplex virus (HSV) and varicella zoster virus (VZV), is a common complication in patients receiving chemotherapy for AML. The incidence of clinical reactivation of HSV in patients with acute leukemia treated with intensive chemotherapy is approximately 37–68%. 19 The mechanism of clinical reactivation is likely linked to increased mucus membrane permeability and alterations in cell-mediated immunity from the underlying leukemia and immunosuppression. 20 For patients with known HSV / VZV seropositivity receiving induction therapy for AML, prophylaxis with a nucleoside-based antiviral (acyclovir, valacyclovir) is strongly recommended by a joint practice guideline from the American Society of Clinical Oncology (ASCO) and the Infectious Diseases Society of America (IDSA). 21 At present, however, there is insufficient randomized controlled trial data to establish a universally accepted viral prophylaxis indication or duration, 22 and uncertainty exists regarding prophylaxis with lower intensity therapy and novel regimens. In a multi-center prospective study, acyclovir did not significantly reduce the risk of infection in patients treated with azacitidine. 23
Bacterial prophylaxis is also often used in AML patients. A National Comprehensive Cancer Network (NCCN) guideline recommends fluoroquinolone prophylaxis for patients expected to have neutropenia for greater than 7 days. 24 The joint ASCO/IDSA guideline also recommends fluoroquinolones for AML and MDS. 21 The primary sources that inform these guidelines often combine outcomes of chemotherapy for solid tumors and hematological malignancies. Retrospective studies specific to AML have demonstrated that levofloxacin prophylaxis is associated with reduced bacterial infections, including studies that focus on 7 + 3 induction as well as areas known to have high fluoroquinolone resistance.25,26 The effect of antibacterial prophylaxis on overall mortality is unclear. Studies restricted to AML patients did not identify a mortality benefit.25,26 In contrast, a retrospective study in Germany demonstrated that bacterial prophylaxis was associated with an improvement in all-cause mortality for patients with hematological malignancies receiving high-dose chemotherapy. 27
Methods
Study design
A retrospective chart review was conducted for adult patients with either preexisting or newly diagnosed AML treated at Harbor-UCLA Medical Center in Torrance, CA, USA from January 1, 2014 to December 31, 2024. Patients with acute promyelocytic leukemia and patients who received best supportive care were excluded. Data collection included demographics, baseline clinical characteristics, antileukemic therapy, antimicrobial prophylaxis, infections, and survival.
Clinical parameters
Patients were classified as favorable, intermediate, or adverse risk according to the European LeukemiaNet (ELN) 2022 risk stratification. 22 Confirmed fungal, viral, or bacterial infection was defined by culture or laboratory-based identification of a pathogen with corresponding clinical manifestations. Probable infection was defined by clinical manifestations suggestive of an infection ± supportive radiographic findings in the absence of culture or laboratory-based identification of a pathogen.
Statistical analysis
Demographic information and ELN risk classification were compared between patients with and without prophylaxis using univariate analysis. We utilized Kaplan-Meier curves to depict overall survival, which is defined as time from diagnosis to death from any cause or date of last contact. Log rank test was used to compare curves, and hazard ratios (HR) were calculated using 95% confidence intervals (CI). For hazard ratio calculations, receiving prophylaxis (fungal, viral, bacterial) and no infection (fungal, viral, bacterial) were assigned as the interventional groups with no prophylaxis and positive infection history serving as controls, respectively. Z-test for comparing proportions was used to compare the probability of infection between groups. Statistical testing used a two-sided p-value <0.05 for significance.
Results
Demographics
Sixty-three patients with AML met inclusion criteria for the study. Median age at diagnosis was 53 years old (range 19–84). The cohort was composed of 32% favorable, 41% intermediate, and 27% adverse risk by ELN classification. There were no statistically significant differences in age, gender, race / ethnicity, or ELN risk classification between those who did and did not receive prophylaxis (fungal, viral, or bacterial) (Table 1).
Demographics and clinical characteristics.
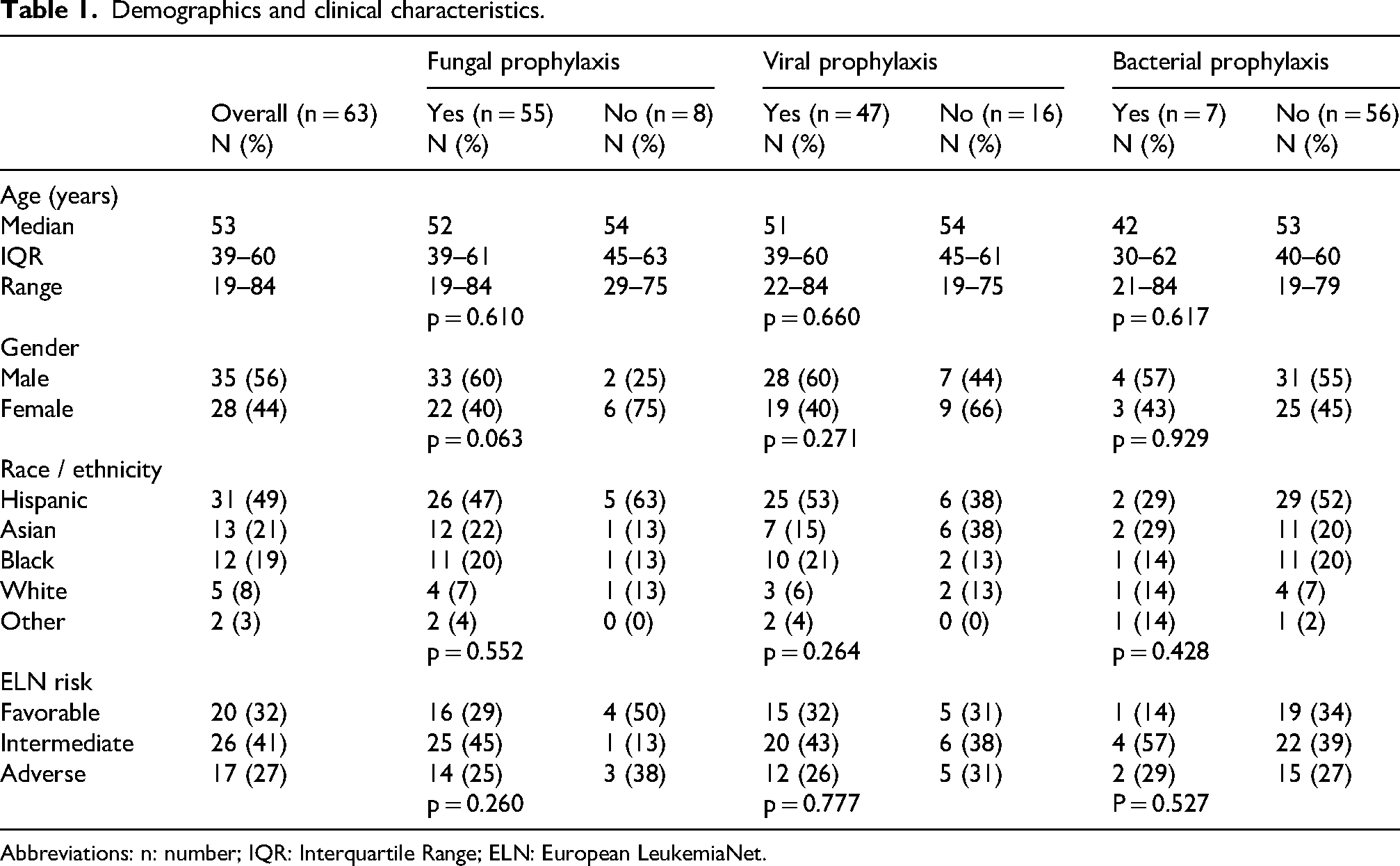
Abbreviations: n: number; IQR: Interquartile Range; ELN: European LeukemiaNet.
Induction therapy
The most common induction therapy was 7 + 3 (7 days of cytarabine plus 3 days of daunorubicin), which was used in 39 (61.9%) patients (Table 2). Other frequently used first-line regimens included azacitidine (11.1%), azacitidine + venetoclax (6.3%), and 7 + 3 + midostaurin (6.3%).
First-line therapies and infections during induction.

Abbreviations: N: number; 7 + 3: Seven days cytarabine and three days of daunorubicin.
Antimicrobial prophylaxis
Antifungal prophylaxis was utilized in 55 (87.3%) patients, most frequently posaconazole (Figure 1). Antiviral prophylaxis with acyclovir or valacyclovir was used in 47 (74.6%) patients. Prophylaxis against bacterial infections was utilized in 7 (11.1%) patients. There were 16 patients who switched from one antifungal agent to another and 5 patients who switched viral prophylactic drugs for reasons including toxicity, drug interactions, development of a breakthrough infection, treatment setting, and provider choice. There were no significant differences in the use antifungal, antiviral, or antibacterial prophylaxis between different regimens (Table 2).
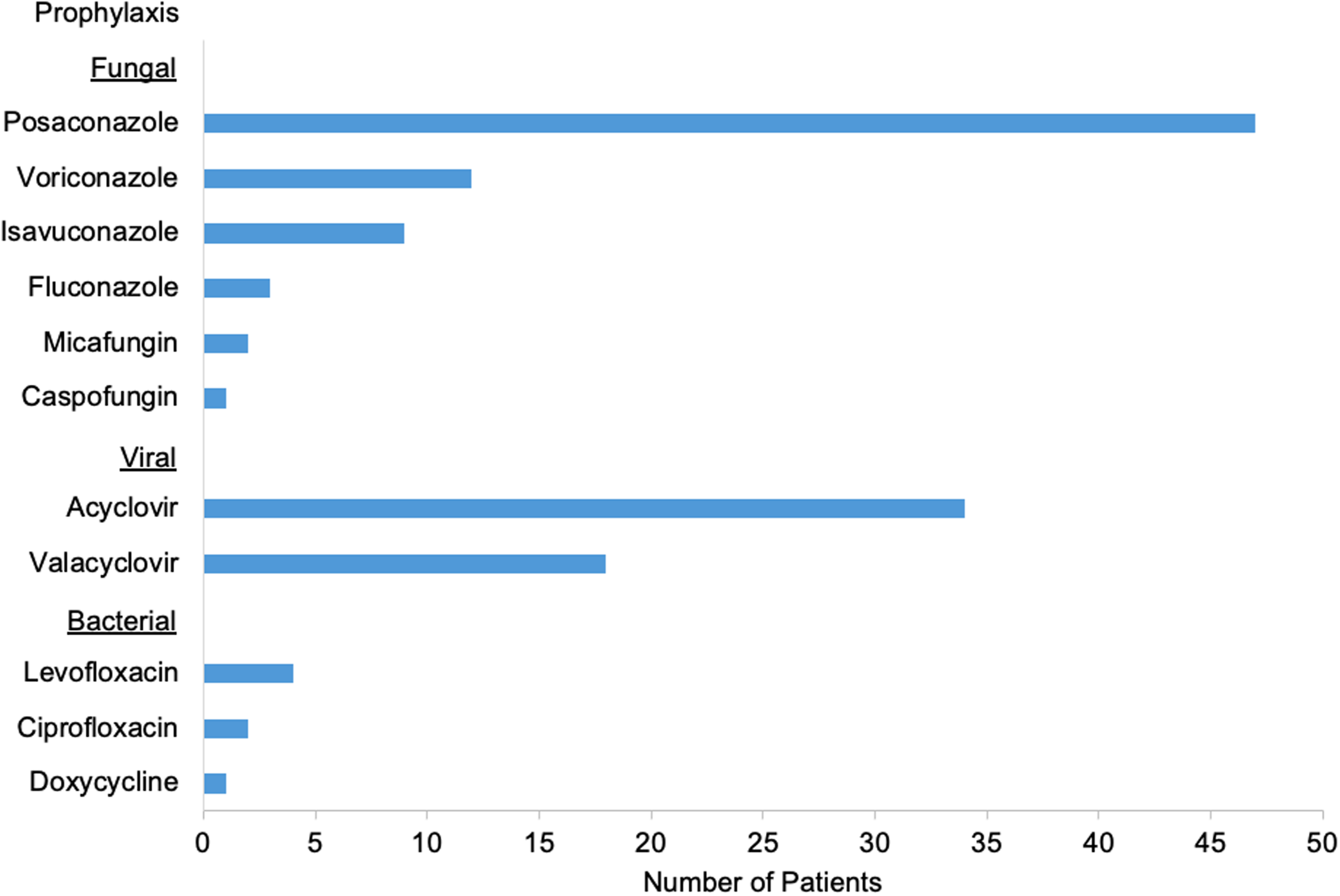
Antimicrobial agents used for prophylaxis. Posaconazole was most frequently used for fungal, Acyclovir for viral, and levofloxacin for bacterial prophylaxis.
Infections
Among the 4 most common regimens, azacitidine + venetoclax had the highest rate of fungal infection, which was significantly higher than 7 + 3 (50% vs. 5.1%, p = 0.003), azacitidine monotherapy (50% vs. 0%, p = 0.038), and all other regimens combined (50% vs. 3.4%, p < 0.001). There were no statistically significant differences in fungal infections during induction between 7 + 3 versus all other regimens (5.1% vs. 8.3%, p = 0.612), 7 + 3 versus hypomethylating agent (HMA) containing regimens (5.1% vs. 15.4%, p = 0.229), or HMA containing regimens versus all other regimens (15.4% vs. 4%, p = 0.134).
Six (9.5%) patients were diagnosed with a viral infection during induction. The risk of viral infection during induction was not significantly different between 7 + 3 versus azacitidine + venetoclax (7.7% vs. 0%, p = 0.562), 7 + 3 versus HMA containing regimens (7.7% vs. 7.7%, p = 1), 7 + 3 versus all other regimens (7.7% vs. 12.5%, p = 0.528), HMA containing regimens versus all other regimens (7.7% vs. 10%, p = 0.801), azacitidine + venetoclax versus all other regimens (0% vs. 10.2%, p = 0.503), or azacitidine + venetoclax versus azacitidine monotherapy (0% vs. 14.3%, p = 0.429).
Bacterial infections were diagnosed in 34 (54.0%) patients during induction. The rate of bacterial infection was significantly higher with 7 + 3 compared to azacitidine + venetoclax (64.1% vs. 0%, p = 0.014), HMA containing regimens (64.1% vs. 23.1%, p = 0.010), and all other regimens combined (64.1% vs. 37.5%, p = 0.040). Compared to all other regimens combined, azacitidine + venetoclax (0% vs. 57.6%, p = 0.025) and HMA containing regimens (23.1% vs. 62%, p = 0.012) had lower rates of bacterial infections.
Fungal infections
Across all lines of therapy, there were 16 fungal infections (6 confirmed, 10 probable) diagnosed across 14 (22.2%) patients including 4 infections during induction, 6 during consolidation, and 6 with second line therapy and beyond (Supplementary Table 1). The median absolute neutrophil count (ANC) at the time of fungal infection was 0.0 × 109/L. Across all lines of therapy, pneumonia (n = 11) was the most common type of fungal infection, and
The risk of fungal infection was 21.8% for patients who received antifungal prophylaxis and 25% for those who did not receive prophylaxis (p = 0.842). Stratifying by ELN risk classification, the rate of fungal infection was significantly higher in patients with adverse risk compared to patients with intermediate or favorable risk (41.2% vs. 15.2%, p = 0.028). There was no significant difference in fungal infections in patients <60 years old and patients aged 60 or older (20% vs. 27.8%, p = 0.503).
For patients with no diagnosed fungal infection, 6 and 12-month overall survival were 77% and 57%, compared to 55% and 39% in patients with a fungal infection (HR 0.64, 95% CI 0.22–1.51, p = 0.265) (Figure 2(a)). Six-month overall survival was 79% for patients who received antifungal prophylaxis and 88% for patients who did not receive prophylaxis (HR 1.73, 95% CI 0.53–5.62, p = 0.439) (Figure 2(b)).
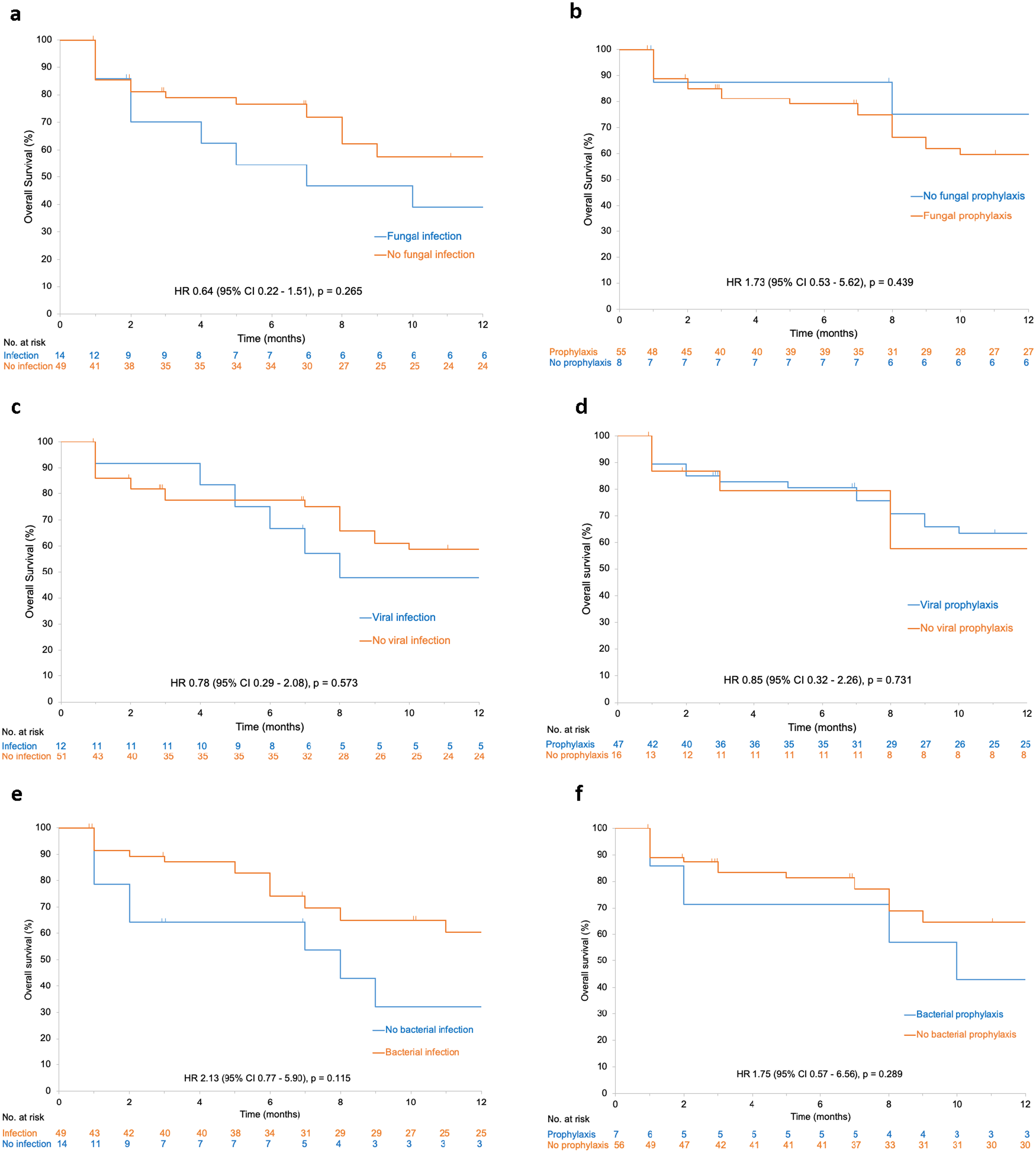
Kaplan-Meier curves for overall survival for patients with (a) no fungal infection vs. fungal infection; (b) fungal prophylaxis vs. no fungal prophylaxis; (c) no viral infection vs. viral infection; (d) viral prophylaxis vs. no viral prophylaxis; (e) no bacterial infection vs. bacterial infection; (f) bacterial prophylaxis vs. no bacterial prophylaxis.
Viral infections
There were 14 viral infections (10 confirmed, 4 probable) observed across 12 (19.0%) patients (6 induction, 2 consolidation, 6 s line and beyond). At the time of viral infection, the median ANC was 0.1 × 109/L. Upper respiratory infection (URI) was the most frequent type of infection (Supplementary Table 1). Coronavirus URI was diagnosed in 4 patients, including 1 patient with COVID-19. No cases of COVID-19 pneumonia or COVID-related deaths were observed. HSV was the most common viral pathogen with 2 polymerase chain reaction (PCR) confirmed cases of HSV type 2 esophageal and rectal ulcers and 3 cases of probable oropharyngeal HSV. Two patients were diagnosed with probable cutaneous VZV. Antiviral treatment with valacyclovir (n = 4) or acyclovir (n = 3) was initiated in cases of HSV or VZV, remdesivir was used for 1 patient with COVID-19, and the remaining patients with viral infections received supportive care.
The rate of viral infection was significantly lower in patients who received antiviral prophylaxis compared to patients with no prophylaxis (12.8% vs. 37.5%, p = 0.029). There were no statistically significant differences in the risk of viral infection for adverse risk versus intermediate or favorable risk (11.8% vs. 21.7%, p = 0.374) or patients <60 years old versus patients 60 years or older (22.2% vs. 11.1%, p = 0.313).
For patients without a viral infection, 6 and 12-month overall survival were 77% and 59%, compared to 75% and 48%, for patients with a viral infection (HR 0.78, 95% CI 0.29–2.08, p = 0.573) (Figure 2(c)). Six-month overall survival was 81% for patients who received antiviral prophylaxis and 79% for patients with no prophylaxis (HR 0.85, 95% CI 0.32–2.26, p = 0.731) (Figure 2(d)).
Bacterial infections
There were 120 bacterial infections (100 confirmed, 20 probable) diagnosed across 49 (77.8%) patients including 47 infections during induction, 28 during consolidation, and 45 with second line therapy and beyond (Supplementary Table 1). There were 27 patients with more than 1 bacterial infection, including 4 patients with 6 or more bacterial infections (maximum 9). The median ANC was 0.0 × 109/L at the time of bacterial infection. During induction, the most common types of bacterial infection were bacteremia (n = 14), urinary tract infection (n = 8), and skin / soft tissue infection (n = 8). Bacteremia accounted for 18/28 (64.3%) bacterial infections during consolidation. With second-line therapy and beyond, the most frequent infections were bacteremia (n = 27), urinary tract infection (n = 5), and pneumonia (n = 5). Across all lines of therapy, the most common infections were
Bacterial infections were diagnosed in 85.7% of patients who received antibacterial prophylaxis and 75% of patients without prophylaxis (p = 0.529). The risk of bacterial infection was 70.6% for adverse risk AML and 80.4% for intermediate or favorable risk (p = 0.407). There was no statistically significant difference in the rate of bacterial infection in patients <60 years old versus patients 60 years or older (82.2% vs. 66.7%, p = 0.180).
In patients with no diagnosed bacterial infection, 6 and 12-month overall survival were 65% and 32%, compared to 74% and 60% in patients with a bacterial infection (HR 2.13, 95% CI 0.77–5.90, p = 0.115) (Figure 2(e)). Six-month overall survival was 71% with antibacterial prophylaxis and 81% with no prophylaxis (HR 1.75, 95% CI 0.57–6.56, p = 0.289) (Figure 2(f)).
Discussion
Fungal
In this retrospective study of AML patients from a Los Angeles County hospital, there was variable use of antimicrobial prophylaxis. Most patients (87.3%) received fungal prophylaxis. Other retrospective studies report prophylaxis rates ranging from 75–100%.8,12,15,16 The rate of fungal infection was 6.3% during induction, which is comparable to prior observations of 5–15% in cohorts that received prophylaxis.6,14 Another study focusing on AML induction therapy without fungal prophylaxis demonstrated fungal infections at a rate of 13%. 28 22.7% of patients experienced an infection during consolidation. In a single-center retrospective study of 111 patients receiving high-dose cytarabine (HIDAC) consolidation, the rate of fungal infection was 3%. 2 The lung was the most common site of fungal infection with pneumonia occurring in 11 (73.3%) of patients. A similar finding was reported in Taiwan with 76% of newly diagnosed AML patients experienced fungal pneumonia. 29 Patients in our cohort that received azacytidine + venetoclax experienced fungal infections at 50%. Previously reported rates of HMA + venetoclax have reported rates ranging from 5.1–12.6%.15,30 This difference in fungal infections is likely related to the small number of patients that underwent azacytidine + venetoclax in our cohort.
Fungal infections were diagnosed in 21.8% of patients who received prophylaxis compared to 25% of patients without prophylaxis (p = 0.842). The lack of significant benefit may be attributable to the small sample size and confounding factors. Prior research seems to support the clinical benefits of prophylaxis including reduced infections, improved survival, and decreased costs.6,7,10
The recommendation to provide prophylaxis against mold for the duration of expected neutropenia for patients with AML is compatible with the results of the present study where the majority (56%) of fungal infections were mold and most infections (69%) occurred when patients were severely neutropenic. There was a significantly higher rate of fungal infections in adverse risk AML compared to intermediate or favorable risk (41.2% vs. 15.2%, p = 0.028), a finding which has been previously observed in AML patients treated with azacitidine + venetoclax. 8 With evidence suggesting differences in risk based on ELN risk category and a significant rate of fungal infection across all lines of therapy, more investigation is needed to risk stratify patients that may benefit from extended fungal prophylaxis.
Viral
In this cohort, viral prophylaxis was used in 74.6% of patients. There is a spectrum of reported antiviral prophylaxis frequencies from other retrospective studies ranging from 25–100%.28,31 The risk of viral infection was 9.5% during induction and 9.1% during consolidation. In comparison, prior studies have reported viral infection rates of 1.1% with 7 + 3 induction and 14% during HIDAC consolidation.2,12 In a comparative retrospective study, there was no difference in viral infections between high and low intensity induction chemotherapy throughout the 28-day course, 4 which aligns with our data which showed no difference in viral infections between regimens.
There was no difference in viral infection rate based on ELN risk classification in the present study, while previously published research has suggested increased risk of viral infection in patients with adverse risk AML. 32 While age itself is considered a risk factor for HSV reactivation, 22 there was no difference in viral infection risk based on age within our cohort.
Use of prophylaxis was associated with a significant reduction in viral infections (12.8% vs. 37.5%, p = 0.015). Prior studies have had mixed results with increased benefit noted in patients with baseline HSV / VZV seropositivity. In a randomized placebo-controlled trial of HSV-seropositive patients undergoing AML induction therapy, the acyclovir prophylaxis arm had 0% reactivation of HSV-related ulcers compared to 73.3% in the placebo group. 22 In a similarly designed RCT of HSV-seropositive patients, 67.5% of those receiving prophylaxis had reactivation infections, whereas 97.3% of those in the placebo group had an acute oral infection. 33 These studies have been crucial in informing ASCO/IDSA guidelines regarding antiviral prophylaxis; however, there are other studies that report no benefit in routine prophylaxis with acyclovir / valacyclovir.23,31 In a retrospective study of 85 patients receiving decitabine for AML or MDS, five out of eight culture-positive viral infections occurred in patients receiving antiviral prophylaxis. 31 The decision to use prophylaxis at our center was influenced by presence, degree, and expected duration of neutropenia, antileukemic therapy, and provider preference. Without routine assessment of baseline seropositivity, viral prophylaxis was associated with reduced viral infections, supporting its use in neutropenic AML patients.
Bacterial
The majority of patients (77.8%) experienced a bacterial infection. The largest portion of infections occurred during induction therapy, which is consistent with prior findings showing high rates of bacterial infection in early treatment. 4 Bacterial infections occurred at a significantly higher rate with 7 + 3 compared to HMA-containing regimens. A similar finding has been observed with increased bacterial infections associated with higher intensity chemotherapy for AML. 4 However, no bacterial infections were reported in our cohort that received azacytidine + venetoclax. A previous study of patients that received azacytidine or decitabine + venetoclax identified a bacterial infection rate of 33.6%. 30 Similar to our finding of fungal infections in the azacytidine + venetoclax cohort, the small sample size is likely responsible for the discordance from reported rates. A considerable risk of bacterial infection, particularly bacteremia, was observed with consolidation and later line therapy as well.
In the present study, antibacterial prophylaxis was not associated with a decrease in bacterial infections. Prior research regarding the effectiveness of prophylaxis in reducing bacterial infections is mixed.25,26,28 A study evaluating infection rates in AML and MDS patients receiving decitabine showed that fluoroquinolone prophylaxis had a non-significant association with lower rates of infection, 29 while other studies have reported positive results.25,26,28 Our study is limited by the small proportion of patients (11%) that received prophylaxis. With a high risk of bacterial infection observed across all lines of therapy and most cases (81%) occurring in patients with ANC < 0.5 × 109/L, fluoroquinolone prophylaxis during periods of severe neutropenia seems reasonable.
Patients that received bacterial prophylaxis had a lower overall survival than those that did not, though the results were not significant (Figure 2(f)). We acknowledge the limitations of the sample size in interpreting results, especially regarding outcomes in mortality based on bacterial prophylaxis. The sample size is likely influenced by selection bias and subsequent confounding of results, especially in the bacterial prophylaxis group with the lowest number of subjects. However, while the limited sample size likely contributed in our results, there is evidence that describes the lack of mortality benefit with prophylaxis. A recent meta-analysis from the ECIL determined that bacterial prophylaxis did not decrease mortality in hematologic cancers with neutropenia. The study addressed the concern of growing resistance rates and noted that their analysis evaluated settings with low to moderate resistance rates. 34 Interestingly, patients in our cohort with bacterial infection had a non-significant improved OS compared to patients without infection (Figure 2(e)). Bacterial infection remains a large cause of mortality, especially with multi-drug resistant organisms (MDRO).35,36 Our finding is not necessarily discordant with previous studies since only 11.1% received prophylaxis. Fluoroquinolones have been shown to decrease infection rates without improving mortality, 34 possibly hinting at MDRO having a stronger association with mortality over organisms covered by fluoroquinolones.
Limitations and future directions
As a result of the study's retrospective design and small sample size, a limitation of this study is lack of stratification for factors which may influence infections and survival. Given real-world patient heterogeneity and potential for confounding variables, the results should be interpreted cautiously. Findings should ideally by analyzed in prospective cohorts with larger sample size. Although some groups were small and the subject number was unbalanced across study arms, the chief objective of this analysis was effect size estimation and clinical direction rather than formal hypothesis testing. The decision to prescribe prophylaxis may be largely based on patient specific factors, regional microbial resistances, and other clinical factors that are not addressed by our analysis. Additional limitations include variable duration of prophylaxis and patients who switched prophylactic agents thereby preventing comparison of drugs. Future research should evaluate viral prophylaxis by stratifying seropositive versus seronegative patients along with broader efforts to study acute lymphoblastic leukemia (ALL) and MDS populations. It may be valuable to analyze cost-effectiveness of different antimicrobial agents. While placebo-controlled trials are impractical, head-to-head comparison of prophylactic agents could be feasible and impactful. Ongoing focus on prevention, early detection, and treatment of invasive infections in acute leukemia is essential.
Conclusion
In this single-center retrospective study of AML patients from a Los Angeles County hospital, antiviral prophylaxis demonstrated a benefit in reducing infections, though antifungal and antibacterial agents did not. There was no survival advantage associated with prophylaxis. Bacterial infections were significantly more common with 7 + 3. Patients with adverse risk AML were more susceptible to fungal infections. Our findings demonstrate that different AML therapies and ELN risk categories place patients at different levels of risk for developing infection. Understanding these distinctions may provide further clarity as to which patients would benefit from routine prophylaxis. Ultimately, these results are considered hypothesis-promoting and provide clinically informative estimates that support further investigation. As AML treatment becomes more personalized with novel or reduced intensity regimens, further investigation to describe infection risk and prophylaxis guidance is needed.
Supplemental Material
sj-docx-1-opp-10.1177_10781552261443919 - Supplemental material for Retrospective study of antimicrobial prophylaxis and infections in patients with acute myeloid leukemia
Supplemental material, sj-docx-1-opp-10.1177_10781552261443919 for Retrospective study of antimicrobial prophylaxis and infections in patients with acute myeloid leukemia by Jeffrey Jang, Timothy Nold, Joseph P Marshalek, Jessica Matthiesen and Sarah Tomassetti in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
None.
Author contributions
Jeffrey Jang: Conceptualization, Writing-Original draft. Timothy Nold: Conceptualization, Data curation, Writing-Original draft. Joseph P. Marshalek: Conceptualization, Data curation, Formal analysis, Data curation, Writing-Original draft. Jessica Matthiesen: Data curation. Sarah Tomassetti: Conceptualization, Writing-Review & Editing, Supervision. All authors reviewed and approved the final version of the manuscript.
Ethical approval
The study was performed in accordance with the ethical standards described in the Declaration of Helsinki 1975, revised Hong Kong 1989, and subsequent amendments. The Harbor-UCLA Medical Center Institutional Review Board (IRB) approved this study (IRB number 18CR-32681-01). The Harbor-UCLA Medical Center IRB granted an exemption from requiring written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data analyzed in the study is included in this article. Further inquiries regarding data can be directed to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
