Abstract
Objective
Trastuzumab deruxtecan (T-DXd) and sacituzumab govitecan (SG) are antibody-drug conjugates (ADCs) that are associated with clinically significant antineoplastic-induced nausea and vomiting (AINV), for which prophylaxis and management strategies vary in clinical practice. Integration of available evidence, guideline recommendations, and expert clinical experience is required to develop practical, expert-informed strategies and approaches for the prevention and management of AINV in patients receiving these agents.
Data Sources
A systematized literature review of PubMed publications from 2020 to 2024 was conducted to identify evidence regarding AINV management in patients receiving T-DXd or SG. Search terms were developed in consultation with a multidisciplinary Canadian expert panel, including medical oncologists and oncology pharmacists from academic and tertiary cancer centers, and included terms related to nausea, vomiting, emesis, and supportive care, as well as specific ADC agents and antiemetic therapies. Findings were contextualized using local and international guidelines. Expert clinical consensus was achieved through two virtual meetings and two rounds of structured group discussions using a modified Nominal Group Technique.
Data Summary
The search identified 1229 records, with 32 articles relevant to ADCs. The Canadian expert panel used this evidence, supplemented with non-ADC chemotherapy studies where ADC-specific data were limited, to inform practical clinical strategies. These included prophylactic, combination-based antiemetic strategies tailored to emetogenic risk; steroid-sparing and individualized approaches based on patient response and tolerability; early escalation or modification of therapy for persistent symptoms; structured patient education on symptom recognition, medication adherence and tools for symptom monitoring for acute and delayed AINV.
Conclusions
We provide expert consensus recommendations for practical approaches for the management of AINV in patients receiving T-DXd or SG, including an antiemetic algorithm and tools for structured symptom monitoring. These approaches are intended to support consistent implementation of AINV management strategies in diverse oncology settings.
Introduction
Antibody-drug conjugates (ADCs), which combine a cytotoxic payload and a targeted monoclonal antibody, are increasingly used to treat a range of solid tumors. 1 However, these agents can be associated with antineoplastic-induced nausea and vomiting (AINV), one of the most feared and distressing adverse effects, negatively impact patients’ health-related quality of life. 2
Efforts to classify the emetogenicity of ADCs have been complicated by limited reporting of specific nausea and vomiting data in clinical trials. 3 Unlike oral agents, which carry a continuous emetogenic risk over the duration of therapy due to repeated administration,3,4 emetic events for intravenous (IV) agents typically occur over three phases: the acute phase, occurring within 24 h of administration; the delayed phase, occurring from 24 to 120 h; and the long-delayed or persistent phase, occurring beyond 120 h.5,6
Among ADCs, trastuzumab deruxtecan (T-DXd), a HER2-targeted ADC with a topoisomerase 1 inhibitor payload, 7 is associated with both acute and delayed AINV. T-DXd can directly stimulate the peripheral gastrointestinal (GI) tract, causing nausea and vomiting shortly after administration, and may also have a secondary effect on the chemo-emetic area in the brain/central nervous system, contributing to delayed AINV. 8 In clinical trials, high rates of nausea and vomiting have been observed: in DESTINY-Breast01 (participants with heavily pretreated HER2 + metastatic breast cancer), 77.7% experienced any-grade nausea (7.6% Grade ≥3) and 45.7% experienced any-grade vomiting (4.3% Grade ≥3) 9 ; in DESTINY-Breast03 (that established T-DXd as standard-of-care for patients with HER2 + metastatic breast cancer in the second-line setting), 77% experienced any-grade nausea (7% Grade ≥3) and 52% experience any-grade vomiting (2% Grade ≥3). 10 Given the expanding use of T-DXd across HER2-low and ultra-low metastatic breast cancer,11,12 and other solid tumors, 13 there is a growing need to establish consistent, evidence-informed strategies for the prevention and management of AINV across institutions.
Similarly, sacituzumab govitecan (SG), a Trop-2–targeted ADC with a topoisomerase 1 inhibitor payload, has been associated with clinically significant nausea and vomiting. In the ASCENT trial (which established SG as standard-of-care for patients with metastatic triple-negative breast cancer), 57% of participants treated with SG experienced any-grade nausea (approximately 3% Grade ≥3) and 29% experienced any-grade vomiting (approximately 2% Grade ≥3) compared with 26% of participants receiving physician's-choice chemotherapy who experienced any-grade nausea (<1% Grade ≥3) and 10% who experienced any-grade vomiting (<1% Grade ≥3). 14 In the TROPICS-02 trial (participants with HR+/HER2– metastatic breast cancer), 59% of participants receiving SG experienced any-grade nausea versus 35% in the chemotherapy group. 15 While overall health-related quality of life improved in domains such as physical functioning, fatigue, and pain, scores for nausea and vomiting declined from baseline highlighting the persistent burden of AINV for patients receiving SG. 16
While management strategies for the treatment and prevention of AINV have been proposed, 17 there remain a number of unmet needs, including patient adherence to sometimes complex AINV regimens, and challenges managing delayed AINV. 18
Guideline-based management of AINV for ADCs remains challenging, in part because of variability in how these agents are classified across major antiemetic guidelines. Specifically, T-DXd is classified as highly emetogenic in the antiemesis clinical practice guidelines published by the National Comprehensive Cancer Network (NCCN Version 2.2025), 19 moderately emetogenic according to the 2023 Multinational Association of Supportive Care in Cancer (MASCC) and European Society for Medical Oncology (ESMO) guideline update for the prevention of chemotherapy- and radiotherapy-induced nausea and vomiting, 4 and moderately emetogenic in the 2020 American Society of Clinical Oncology (ASCO) antiemetic update. 20 SG is classified as highly emetogenic by NCCN and moderately emetogenic by MASCC/ESMO.4,19 The NCCN guidelines, which are updated more frequently, also include newer ADCs such as datopotamab deruxtecan, classified as highly emetogenic 19 ; however, detailed evaluation of this agent is beyond the scope of the present review, which focuses on ADCs with established clinical experience.
Survey data from a personal practice assessment provided by STA Healthcare Communications (unpublished, 2023), involving 46 healthcare professionals from Canada, Brazil, and Argentina, indicated that most Canadian respondents, with representation across the most populous provinces, use the Cancer Care Ontario Guidelines to inform institutional protocols for AINV management, with ASCO cited as the second most commonly used framework and NCCN as the third. In the same unpublished assessment, healthcare professionals also identified challenges in recognizing patients at increased risk of CINV with moderately emetogenic regimens and in supporting patient adherence to antiemetic protocols, highlighting practical barriers to effective AINV prevention.
Despite reliance on established guidance, evidence from published registries suggests that adherence to antiemetic guidelines in routine clinical practice remains suboptimal.21,22 For example, the Nausea/Emesis Registry in Oncology (NERO), a multicenter European registry involving 35 sites, reported in 2022 that guideline-consistent AINV prophylaxis was achieved in only 23% of patients. In that study, guideline-consistent AINV prophylaxis was defined according to the MASCC/ESMO 2016 antiemetric guidelines and included patients receiving highly emetogenic chemotherapy (HEC), moderately emetogenic chemotherapy (MEC), and carboplatin-based regimens across a range of tumor types, with breast cancer accounting for 46% of cases. Similarly, the Pan European Emesis Registry (PEER) reported guideline-consistent prophylaxis rates of 29% during the 2009–2010 study period, 22 while the INSPIRE prospective observational study conducted across four oncology practice networks in the United States reported modestly higher adherence at 57.3%. 23 Collectively, these findings demonstrate persistent gaps between guideline recommendations and real-world practice. International expert groups have highlighted the need for practical strategies to manage AINV associated with ADCs, emphasizing prophylaxis, monitoring, and adherence to improve symptom control and patient quality of life.24,25
Building on these international efforts, this systematized review integrates published evidence, guideline recommendations, and Canadian clinical experience to provide expert-informed, practical considerations for the management of AINV in patients receiving T-DXd or SG. By addressing both acute and delayed phases and focusing on strategies to support patient adherence and structured symptom monitoring, these considerations aim to promote consistent implementation across Canadian oncology practice while remaining broadly applicable to other settings.
Methods
Study design
This consensus study was developed in accordance with the Accurate Consensus Reporting Document (ACCORD) guidelines. 26 It employed a systematized review methodology, incorporating a structured literature search, predefined eligibility criteria, and transparent study selection, while allowing flexibility in evidence synthesis. 27
A systematized approach was selected given that AINV outcomes are typically reported as secondary safety endpoints within clinical trials. The objective of this study was to contextualize available evidence and translate it into practical management strategies, integrating clinical studies, existing guideline recommendations, and expert clinical experience, particularly in areas where guideline recommendations are based on extrapolation rather than direct evidence. Given the anticipated reliance on indirect evidence, formal risk-of-bias assessment and quantitative synthesis were not performed, as they would not meaningfully inform the development of practical management strategies. The systematized review was complemented by a structured expert consensus process to support interpretation of findings and development of expert-informed strategies for healthcare providers and patients.
Expert panel and consensus process
The study was guided by a multidisciplinary panel of medical oncologists and oncology pharmacists affiliated with academic cancer centers and tertiary hospitals across major Canadian population centers. The panel collaboratively defined the scope of the review and the key topics of interest during initial virtual discussions. The authors served as both the steering committee and the consensus panel, with logistical and coordination support provided independently by STA Healthcare Communications. Panel members hold senior academic appointments, leadership roles in national training and certification bodies, editorial positions in peer-reviewed journals, and active involvement in national and international clinical trials groups, guideline committees, and patient advocacy organizations. Together, the panel has extensive clinical and research experience across major Canadian population centers. Drawing on this multidisciplinary and geographically diverse expertise, the panel identified the research question, interpreted the literature, and developed evidence-informed management strategies and resources for nausea and vomiting.
Consensus was achieved through two additional virtual meetings and two rounds of discussion using a modified Nominal Group Technique. 28 The Nominal Group Technique facilitated structured discussion and ensured equitable participation, with each panel member contributing ideas independently, followed by group discussion and prioritization to achieve agreement on the strategies and resources.
Research question development
The panel formulated the following research question to guide the review: What is the optimal strategy for the prevention and management of AINV in patients receiving ADCs, specifically T-DXd and SG?
Panel members also considered existing guidance and literature during question development, acknowledging that relevant strategies or resources not retrieved in the literature search could be incorporated during the consensus process.
Information sources and search strategy
A systematized literature review was conducted in PubMed in November 2024 to support the consensus process. The search was restricted to English-language publications, focused on oncology, and limited to newer ADCs approved in Canada with high emetogenic potential, specifically T-DXd and SG. The search included publications from January 2020 to November 2024. Key search terms were defined in consultation with the expert panel (Table 1), and additional primary literature referenced in the guidelines and other publications identified through this search was also considered. Two authors independently screened all records, resolving discrepancies through discussion.
Keywords used to identify PubMed records.

English-language records.
Data synthesis and evidence integration
Eligible articles were included if they addressed nausea and vomiting management in adult patients receiving newer ADCs, involved antiemetic drugs authorized for use in Canada, and reported relevant data. No quantitative analyses or formal risk-of-bias assessments were conducted, consistent with systematized review methodology. Evidence from the literature was synthesized narratively, and strategies and resources for nausea and vomiting management were developed iteratively using expert judgment through the modified Nominal Group Technique. The panel also retained flexibility to incorporate additional references identified during guidance review or through clinical experience.
Reporting
To ensure transparency of the literature identification and selection process, a Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)-style flow diagram was developed. While formal PRISMA reporting is not required for systematized reviews, this diagram provides a visual summary of the search strategy, screening, and inclusion process used to support the consensus development.
Results
The systematized search identified 1229 records, of which 1099 records remained after removal of duplicates. These 1099 records were screened by title and abstract, resulting in the exclusion of 968. The full texts of 131 articles were assessed for eligibility, and 32 records met the inclusion criteria and were included in the narrative synthesis (Figure 1). The included reports comprised clinical trials6,16,29–34; systematic reviews or meta-analyses35–38; retrospective or real-world studies39–43; guidelines or expert consensus4,25,44,45; regulatory reviews46–49; narrative reviews24,50–54; and one case series. 55 Clinical trial data, regulatory summaries, and the case series were limited to T-DXd and SG; other records addressed antiemetic prophylaxis and management across anticancer therapies and were included based on their applicability to these ADCs.
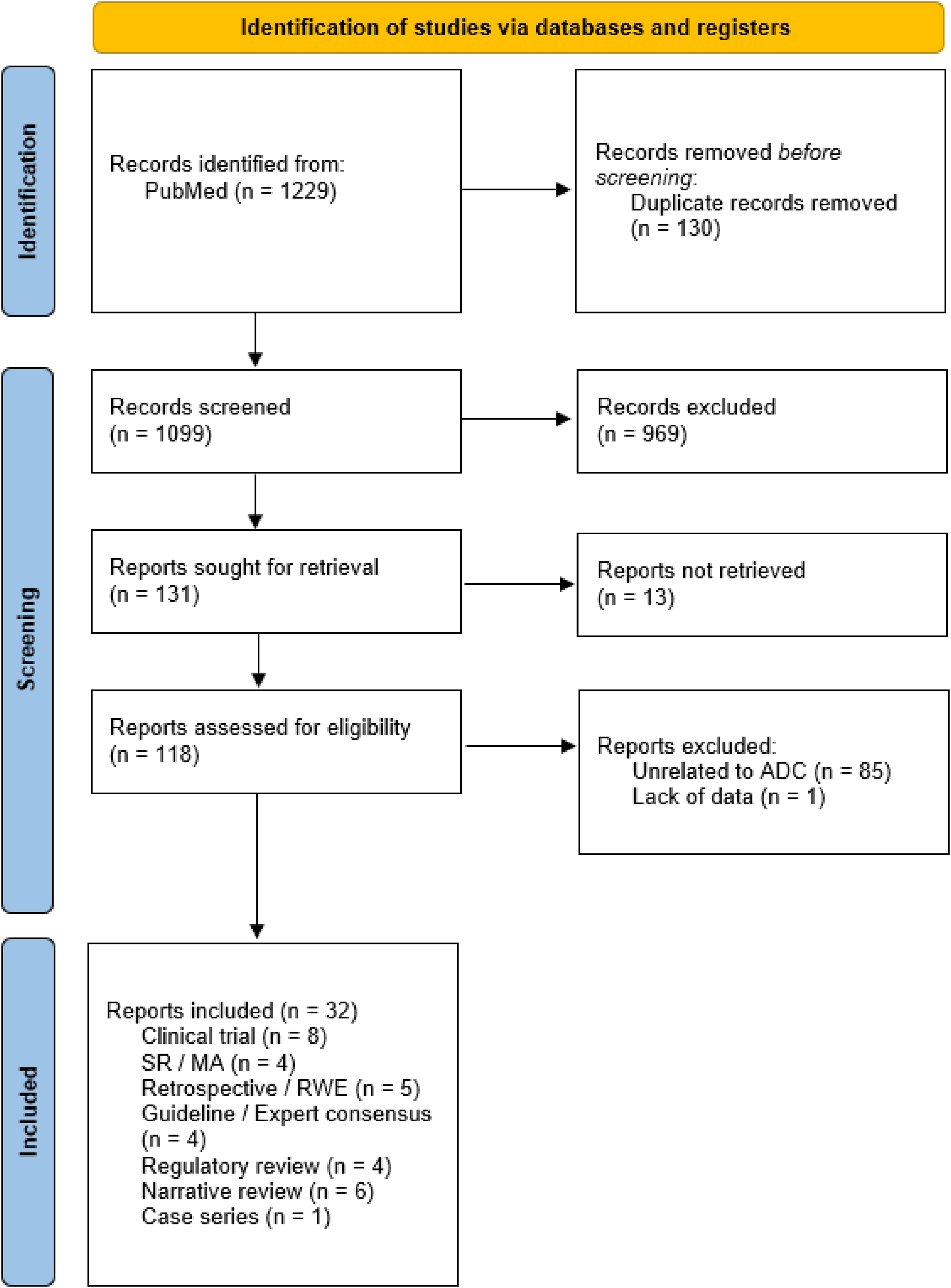
PRISMA flow diagram.
The systematized literature search retrieved ASCO, NCCN, and MASCC/ESMO guidelines.4,20,56 In addition, the multidisciplinary expert panel supplemented the review with Canadian guidance from Cancer Care Ontario 57 and BC Cancer, 58 which were considered during panel discussions and interpretation of the evidence.
Algorithm for the management of AINV
Based on themes discussed during the planning of the consensus process and drawing on ASCO, BC Cancer, Cancer Care Ontario, MASCC/ESMO, and NCCN guidelines,4,19,20,57,58 an algorithm was developed (Figure 2) to support early identification of patients at risk of nausea and vomiting, as well as the prevention and ongoing management of these symptoms in patients receiving T-DXd or SG.
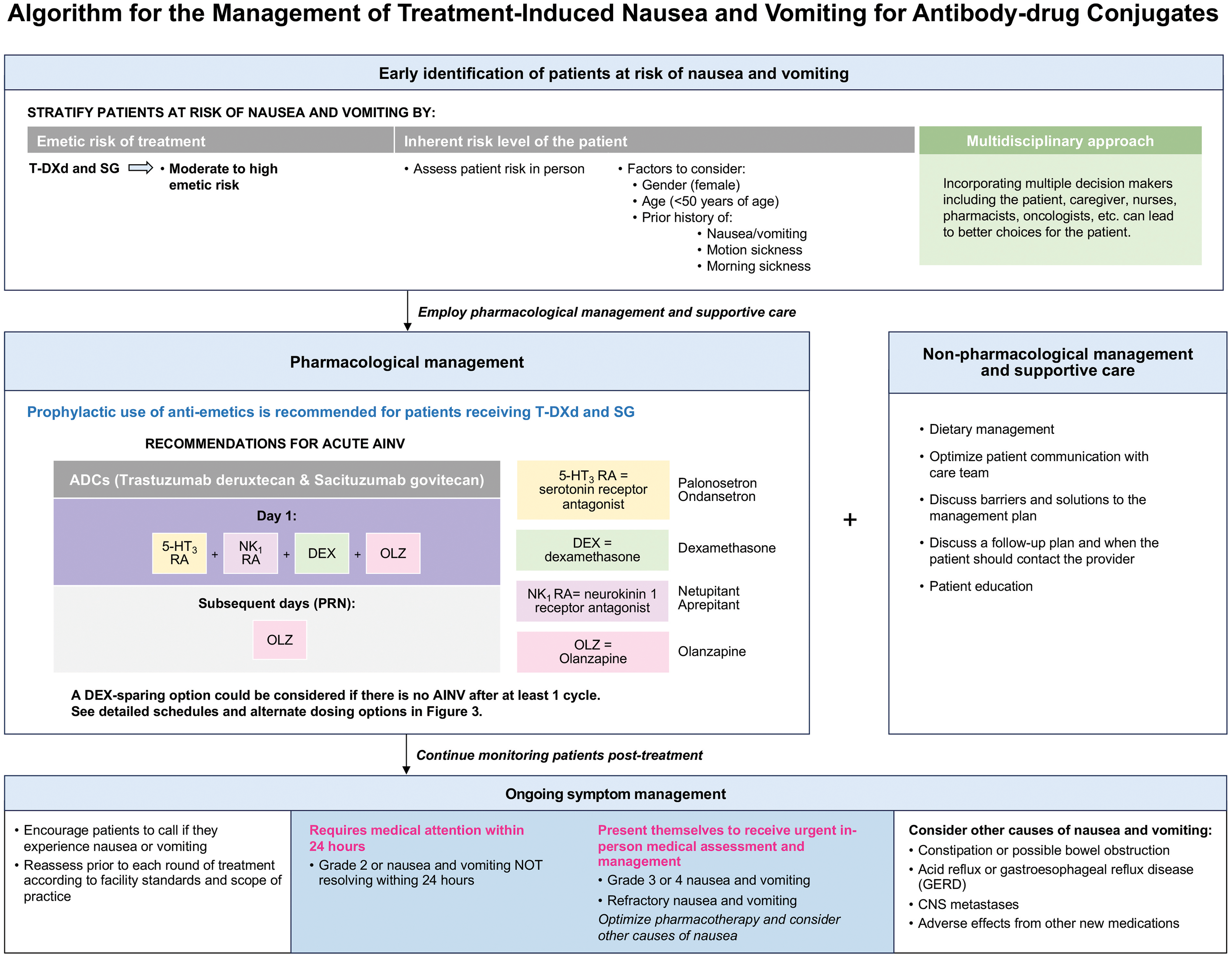
Algorithm for the management of AINV for antibody-drug conjugates.
The development of this algorithm was informed by evidence demonstrating a high burden of gastrointestinal toxicity with these ADCs. In pooled analyses of clinical trial data, nausea has been reported in approximately 75% of patients receiving T-DXd and 66% receiving SG, while vomiting occurs in 45% and 44% of patients, respectively. 37 Retrospective and real-world studies report lower rates of nausea and vomiting than those observed in pivotal clinical trials, including 32% all-grade nausea with T-DXd 40 ; 51% all-grade nausea and 13% all-grade vomiting with SG, 39 and 61% all-grade nausea and 22% vomiting with T-DXd. 41 The expert panel considered that these differences likely reflect limitations inherent to retrospective data collection and differences in routine clinical practice.
Taken together with the consistently high incidence of AINV observed in clinical trials and its potential impact on treatment tolerability, the panel identified a need for structured approaches to the early identification, prevention, and management of nausea and vomiting in patients receiving T-DXd or SG. Accordingly, the algorithm incorporates both pharmacologic and non-pharmacologic strategies, including dietary guidance, proactive patient education, structured discussions of potential barriers to symptom management, clear follow-up plans, and guidance on when patients should contact the care team. Emphasis is placed on engagement of the broader multidisciplinary care team, rather than reliance on the oncologist alone, to support ongoing symptom assessment and management throughout treatment.
Healthcare professionals may underestimate emetic risk, which can further complicate symptom control, based on the expert clinical experience of the panel. In addition, limited ADC-specific data on AINV risk factors necessitate extrapolation from established chemotherapy-induced nausea and vomiting risk factors, including female gender, younger age, a history of motion sickness or morning sickness, and prior episodes of AINV. 58 By integrating guidelines, published evidence, and Canadian clinical experience, the algorithm is intended to support consistent identification and management of AINV in routine practice.
Pharmacological management
T-DXd and SG are classified as highly emetogenic in the NCCN guidelines, whereas MASCC/ESMO and ASCO classify T-DXd as moderately emetogenic and SG as moderately emetogenic where listed.4,19,20 Within the MASCC/ESMO guidelines, the emetogenic risk of T-DXd and SG is considered comparable to that of carboplatin administered at an AUC ≥5, for which a three-drug antiemetic regimen is recommended. 4 For adults receiving moderately or highly emetogenic therapy, current guidelines generally recommend combination antiemetic prophylaxis incorporating a 5-hydroxytryptamine (5-HT3, serotonin) receptor antagonist (RA), dexamethasone, a neurokinin 1 (NK1) RA, and olanzapine.4,19,20 These recommendations are largely derived from studies of cytotoxic chemotherapy rather than ADC-specific trials.
ADC-specific evidence supports these principles. A phase 2 study in patients with metastatic breast cancer treated with T-DXd demonstrated that the addition of olanzapine to a 5-HT3 RA and dexamethasone reduced emetic events during the delayed and persistent phases. 6 Similarly, a randomized phase 2 study comparing granisetron plus dexamethasone versus granisetron, dexamethasone, and aprepitant in patients receiving T-DXd found higher complete response rates in the triple combination arm during the overall phase (70% vs 36.8%; p = 0.019). 30 In a real-world retrospective analysis, the use of a fixed combination of netupitant and palonosetron improved vomiting control in the first cycle and, when escalated in the second cycle for patients with persistent symptoms, significantly reduced both nausea and vomiting. 42 Similarly, treating T-DXd as a high-risk emetic regimen in a hospital-based community setting decreased nausea and vomiting and allowed uninterrupted therapy. 55 Collectively, these studies reinforce that ADC-specific prophylaxis may require triple therapy—including NK1 RA and olanzapine—particularly for high-risk patients.
Consistent with this approach, and as summarized in Figure 2, the expert panel used the literature retrieved in this review together with their clinical experience to develop practical examples of antiemetic prophylaxis. The panel supports the use of a four-drug regimen consisting of a 5-HT3 RA, an NK1 RA, dexamethasone, and olanzapine on Day 1. Continuation of olanzapine on subsequent days is guided by clinical judgment as illustrated in Figure 3. In patients who do not experience nausea or vomiting following at least one treatment cycle, dexamethasone-sparing strategies may be considered.
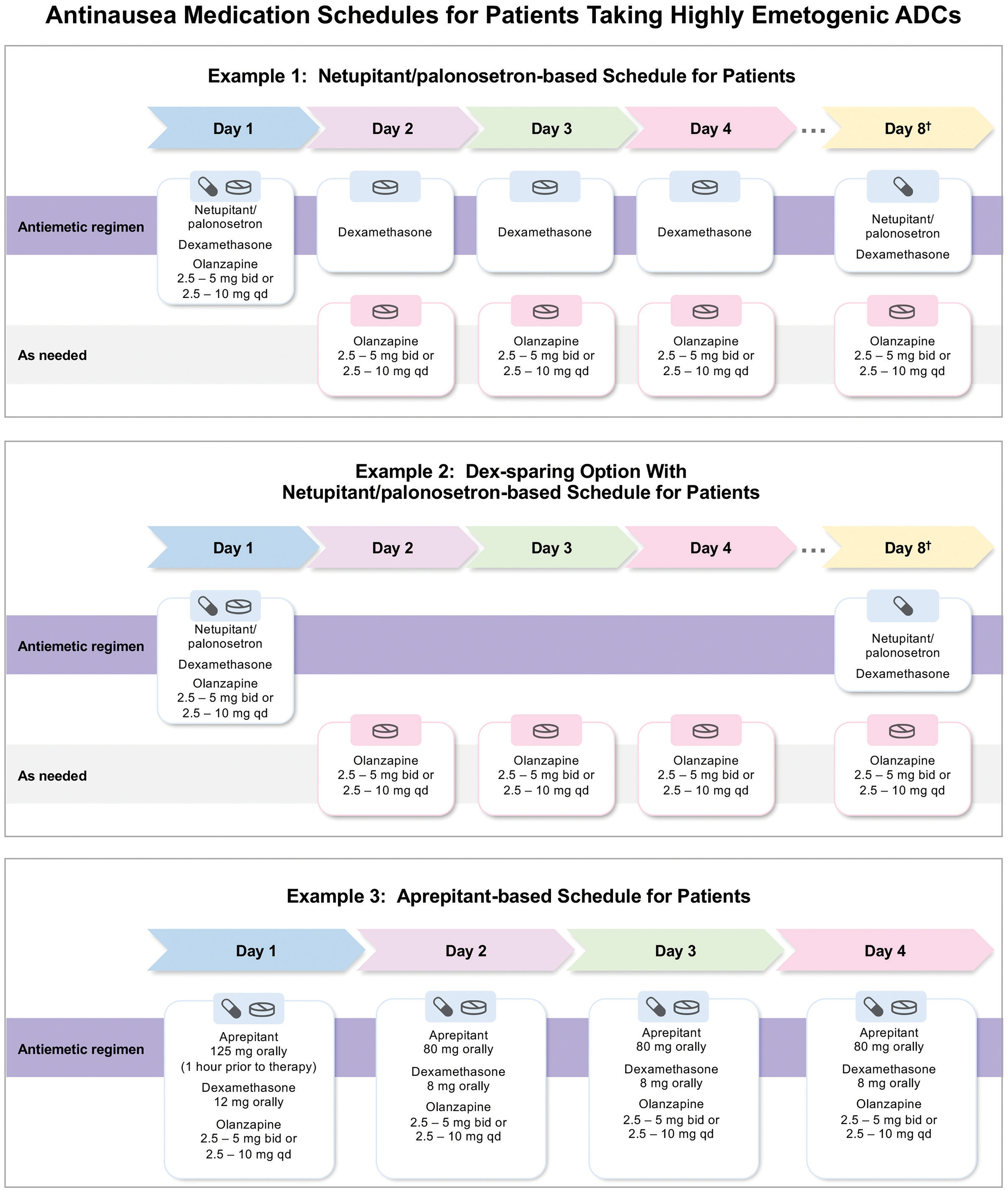
Antinausea medication schedules for patients taking highly emetogenic ADCs. Schematic representation of scheduled and as-needed antiemetic regimens agreed upon by the expert panel for patients receiving ADCs. Ongoing therapy is guided by clinical judgment and patient response, with consideration of de-escalation in later cycles.
Figure 3 illustrates how these principles may be implemented in clinical practice. These include NK1 RA–based regimens using netupitant/palonosetron (NEPA) in combination with dexamethasone and olanzapine on Day 1, with continuation of dexamethasone and/or olanzapine through Days 2–4 as indicated, as well as a dexamethasone-sparing option for selected patients. A separate example of an aprepitant-based regimen, administered over Days 1–4 in combination with dexamethasone and olanzapine, is also shown. For Cycles 2 and onward, de-escalation of the antiemetic regimen may be considered based on individual patient tolerance and symptom control. 53 While Figure 3 is intended as a resource for healthcare professionals, the panel notes that it could also be adapted to develop patient-facing educational materials to support understanding and adherence to antiemetic regimens.
Delayed and persistent AINV
The pattern of nausea and vomiting can vary between patients, with events occurring one week or more after each infusion, extending into the delayed emetic phase. 53 Population pharmacokinetic analyses of SG indicate that the ADC may exhibit target-mediated disposition, whereby binding to the target antigen contributes to non-linear clearance and prolonged systemic exposure, potentially extending the risk of delayed and persistent nausea and vomiting.33,47 The half-life of SG and its SN-38 payload are approximately 16–18 h, with most payload released within 3 days, which may further contribute to delayed symptoms. 54 These pharmacokinetic properties may contribute to delayed and persistent nausea and vomiting in patients receiving ADCs. 53
Based on these observations, and consistent with current guideline recommendations that support consideration of alternative antiemetic strategies for delayed symptoms when standard prophylaxis is insufficient,4,19 the expert panel reached consensus that olanzapine may be considered on an as-needed basis for the management of delayed AINV, including use beyond Day 4 and up to Day 8 in selected regimens when symptoms persist despite guideline-recommended prophylaxis. Dosing may be individualized from 2.5–10.0 mg once daily or 2.5–5.0 mg twice daily according to patient need (Figure 3).
Non-Pharmacologic management
Some guidelines for AINV management, including the BC Cancer Management Guidelines, also include non-pharmacologic strategies, such as consuming small bland meals and maintaining adequate hydration through sufficient fluid intake. 58 Where feasible, involvement of a nutritionist as part of the multidisciplinary care team may further support symptom management.
Patient education is another important factor influencing outcomes and safety. Education may include encouraging patients to monitor the onset and frequency of nausea and emesis, as well as fluid intake. In addition, patients benefit from clear guidance on when to seek additional medical attention. 58
Advances in supportive care have allowed healthcare professionals to adapt strategies to minimize the incidence and impact of adverse effects on patients. 50 These non-pharmacologic elements were considered by the expert panel and are incorporated into the clinical care pathway summarized in Figure 1, which integrates dietary guidance, patient education, proactive symptom monitoring, and multidisciplinary follow-up alongside pharmacologic approaches.
Monitoring of AINV
The expert panel considered the use of structured tools to support ongoing monitoring of AINV in patients treated with T-DXd or SG. While validated instruments such as the MASCC Antiemesis Tool (MAT) are available, 59 the panel also recognized that standardized questionnaires are not consistently integrated into routine oncology workflows across institutions.
Based on institutional practice at the Centre Hospitalier de l'Université de Montréal (CHUM), a structured set of questions has been used successfully to track patient-reported symptoms and antiemetic use. This provides a practical approach that supports consistent assessment and timely identification of persistent or refractory nausea. The panel shares this example to illustrate a feasible, real-world strategy for structured monitoring, while acknowledging that alternative validated questionnaires may also be used according to local practice (Supplementary Figure 1).
Based on the panel's clinical experience, patients may be asked to complete symptom questionnaires daily to reduce recall bias and provide a more accurate record of nausea and vomiting, thereby supporting follow-up and ongoing management.
The panel also emphasized the importance of clear instructions for antiemetic administration in routine practice. Institutionally prepared schedules were identified as helpful in supporting adherence to dosing, timing, and as-needed instructions, while accounting for patient literacy and primary language. The example schedules presented in Figure 3 reflect institutional practices used by panel members.
When adjustments to the antiemetic regimen are required, the multidisciplinary care team may consider additional agents (e.g., olanzapine, an NK1 RA, metoclopramide, or prochlorperazine) that have not previously been administered to the individual patient. Based on clinical experience, metoclopramide or prochlorperazine should be used with caution when coadministered with olanzapine due to the potential for adverse effects, including tardive dyskinesia and other extrapyramidal symptoms.
Steroid-sparing
As no ADC-specific studies evaluating steroid-sparing antiemetic strategies were identified in the literature review, the expert panel considered a dexamethasone-sparing approach based on their clinical experience and on evidence from non-ADC populations across various cancer types. These studies were identified by the panel as informative when considering antiemetic management in the absence of ADC-specific data.
In breast cancer, three studies demonstrated that a 1-day dexamethasone regimen was non-inferior to a 3-day regimen in patients treated with anthracycline-based chemotherapy. 4 In a non-inferiority study of cisplatin-treated patients receiving NEPA, reducing dexamethasone from a multiday regimen (12 mg once on Day 1 and 4 mg twice daily on Days 2–4) to a single 12-mg dose on Day 1 resulted in no significant changes in quality of life, as assessed by the Functional Living Index-Emesis and the proportion of patient achieving no impact on daily life score. 60 In addition, a retrospective real-world study in patients receiving NEPA plus a single dose of dexamethasone as AINV prophylaxis for HEC across various cancer types reported complete response rates of 95.7% in the acute phase and 98.6% in the delayed phase during the first cycle of chemotherapy, with 100% complete response observed from the third treatment cycle. 61
Given that control of nausea and vomiting in the first cycle predicts subsequent AINV incidence, 62 the panel considered that a dexamethasone-reduced regimen may be appropriate to consider in later cycles for patients who experience no AINV in the first treatment cycle.
Based on both available evidence and clinical experience, the panel did not support a steroid-sparing approach using aprepitant, as results in this setting have been inconclusive. A randomized trial failed to demonstrate non-inferiority of a dexamethasone-sparing regimen with aprepitant or fosaprepitant plus palonosetron compared with a 3-day dexamethasone regimen in patients treated with cisplatin (overall complete response rates: 57.8% with single-dose dexamethasone vs 66.7% with 3-day dexamethasone). 63
Before reducing dexamethasone therapy, the panel emphasized the importance of considering additional clinical indications for corticosteroid use within a given treatment regimen, such as prophylaxis to reduce risk of acute infusion or cutaneous reactions.
ADC dose reductions
The expert panel emphasized that early recognition, prevention, and effective management of AINV should be prioritized before any consideration of ADC dose reduction. Optimized supportive care, including guideline-informed antiemetic strategies, patient education, and structured symptom monitoring, is critical to maintain treatment tolerability and adherence. Dose reduction of the ADC should be reserved as a last resort and considered only when all available interventions have been implemented and patients continue to experience unacceptable nausea or vomiting. Decisions regarding dose modification should be made collaboratively with the patient, taking into account symptom burden, treatment goals, and quality of life, and guided by the product label and clinical judgment.7,64
Discussion
This manuscript provides expert clinical perspectives from a Canadian context, along with practical antiemetic algorithms, and structured approaches to symptom monitoring designed for implementation in real-world oncology settings. This contrasts with recent review articles,5,65,66 which describe ADC-related nausea and vomiting and supportive care principles conceptually. By combining evidence with practical clinical strategies, these approaches can directly support clinical decision-making and patient care.
ADCs, such as T-DXd and SG represent an important advance in cancer therapy but introduce distinct challenges for supportive care, including the prevention and management of AINV. Supportive care has been recognized as a core component of oncology practice, encompassing the prevention and management of treatment-related adverse effects, the involvement of a multiprofessional team, and the integration of patient education and monitoring to improve quality of life across the cancer continuum. 66 The pharmacokinetic properties of these agents (e.g., prolonged half-life, target-mediated drug disposition, and delayed payload release) appear to extend emetogenic risk beyond the acute phase traditionally addressed by existing guidelines.33,47,67 As a result, delayed and persistent AINV has emerged as a clinically relevant concern in patients receiving these agents, for which specific guidance remains limited. Safety analyses from the phase 3 ASCENT trial of SG demonstrated that nausea and vomiting frequently occurred well into the delayed and persistent phases, with a median time to first nausea event of 8 days and a median time to first vomiting event of 24.5 days. 68 These findings were considered by the panel alongside observations from a prospective cohort study in patients with breast cancer treated with NCCN-classified HEC or MEC regimens, in which delayed nausea (59.4%) was more frequent than acute nausea (44.9%), although acute-phase symptoms were more severe based on visual analog scale scores. 69 Healthcare providers in that study underestimated the incidence of delayed nausea and vomiting, underscoring the potential for delayed symptoms to be overlooked in clinical practice. Recent narrative reviews emphasize that with emerging ADCs, persistent nausea and vomiting is increasingly recognized, and prophylactic antiemetic strategies (particularly fixed-combination agents such as NEPA) may be effective in controlling these extended symptoms. 5 Collectively, these data informed the panel's assessment of delayed-phase risk and the need for structured monitoring and prophylactic strategies in ADC-treated patients.
The panel used these contextual data, together with their clinical experience, to develop practical considerations for AINV management in patients receiving T-DXd or SG. These included prophylactic, combination-based antiemetic strategies and structured approaches to symptom monitoring to facilitate early identification of delayed or refractory symptoms. Farhat et al. (2023) highlight that optimal toxicity management in ADC therapy is essential to maintain dose intensity and avoid unnecessary treatment interruptions, supporting the integration of proactive antiemetic planning into routine practice. 65 Multidisciplinary perspectives, including oncology pharmacy expertise and representation from multiple regions across Canada, strengthened the applicability of these approaches and helped ensure that they could be adapted to different institutional contexts.
Evidence from HEC chemotherapy populations further supported the inclusion of olanzapine in antiemetic regimens. Randomized studies of patients receiving anthracycline- or platinum-based chemotherapy for early stage breast cancer demonstrated that adding olanzapine to a triple therapy regimen significantly improved nausea control and health-related quality of life without unexpected toxicities. 70 Similarly, another study in patients representing a range of solid tumors scheduled to receive HEC compared placebo with olanzapine in combination with dexamethasone, aprepitant or fosaprepitant, and a 5-HT3 RA, and found substantially higher rates of no chemotherapy-induced nausea and vomiting in the olanzapine arm across acute, delayed, and overall periods. 71 The panel considered these findings in the context of ADC therapy to support multidrug strategies targeting delayed-phase AINV.
Since the literature search did not retrieve any studies directly comparing NEPA- and aprepitant-based regimens in patients treated with T-DXd or SG, the panel drew on evidence from non-ADC chemotherapy populations to inform clinical decision-making. In patients with breast cancer treated with cyclophosphamide and doxorubicin, a greater proportion of those receiving NEPA achieved complete response (defined as no vomiting and no use of rescue therapy) compared with those receiving aprepitant, ondansetron and dexamethasone. 72 A meta-analysis of patient populations with diverse solid tumors receiving various antineoplastic regimens found that NEPA plus dexamethasone was more effective at preventing AINV than other NK1 RA–based regimens combined with a 5-HT3 antagonist and dexamethasone. 73 In a pooled analysis of cisplatin-treated patients diagnosed with lung, head and neck, or ovarian cancer, use of NEPA- and aprepitant-based regimens led to similar complete response rates during the acute phase; however, significantly higher rates of no significant nausea were observed with NEPA during the delayed phase (81.5% vs. 76.4%; p = 0.031). 74 Additional trials, including head-to-head comparisons of single-dose NEPA plus dexamethasone versus a 3-day aprepitant/5-HT3 RA/dexamethasone regimen in patients receiving MEC or HEC regimens for solid tumors, demonstrated numerically higher complete response rates across the acute and delayed phases with NEPA.75,76
Steroid-sparing strategies were discussed by the panel, particularly to minimize cumulative corticosteroid exposure across multiple treatment cycles. Evidence from non-ADC chemotherapy studies suggested that dexamethasone reduction or de-escalation may be feasible in patients who achieve good symptom control during initial cycles.4,60–63
Although derived from non-ADC chemotherapy populations, these findings provide rationale for both the inclusion of olanzapine and the use of NEPA- or aprepitant-based regimens in ADC-treated patients, particularly for addressing delayed-phase nausea and vomiting. Together, the evidence supports the potential benefits of four-drug strategies and individualized approaches, including dexamethasone-sparing or cycle-to-cycle de-escalation, to optimize efficacy and tolerability. Prospective studies are needed to confirm these observations specifically in patients receiving ADC therapy.
The panel noted that the prolonged and non-linear pharmacokinetics of ADCs likely contribute to delayed and persistent AINV, highlighting the need for proactive, systematic approaches to symptom monitoring, patient education, and adherence support. Although validated tools exist, 59 their integration into routine practice is variable, and institution-specific strategies (e.g., daily symptom tracking and clear antiemetic instructions) can help identify persistent or delayed symptoms and support timely interventions. Despite these approaches, the panel highlighted ongoing challenges in access and equity: patient and provider awareness of available supportive services remains limited, and socioeconomic, geographic, ethnic, and language barriers can further hinder implementation. In Canada, interprovincial variations in the access to cancer therapy and supportive medications, compounded by the lack of comprehensive national drug coverage, contribute to disparities in care.77,78 While some validated tools are available in multiple languages, patient-facing educational materials remain inconsistent, particularly for languages beyond English and French. Together, these observations underscore the need for practical, adaptable strategies that integrate monitoring, education, and adherence support within diverse oncology settings.
Economic considerations may also influence antiemetic strategy selection, although evidence specific to ADC-treated populations is lacking. Analyses in non-ADC chemotherapy populations suggest that NEPA-based regimens may reduce overall healthcare costs and improve quality-adjusted outcomes compared with aprepitant-based combinations, driven primarily by reduced AINV-related healthcare utilization and, in some cases, lower drug acquisition costs.79–82
AINV can also affect family caregivers, adding to the overall burden of care. 83 Recognizing caregiver impact when planning symptom management may help ensure comprehensive support and could inform the broader integration of antiemetic strategies in clinical practice.
Given that more indications for ADCs are expected across tumor types in oncology, these Canadian expert perspectives aim to support AINV management alongside the ASCO, NCCN, and MASCC/ESMO guidelines, with consideration of agents for delayed-phase prophylaxis in the context of ADC pharmacokinetics. Emphasizing prophylactic AINV management is important to avoid issues with treatment adherence and anticipatory AINV. 24 The broader impact of ADCs on healthcare delivery is promising, with the potential for targeted delivery to expand into therapeutic areas beyond oncology. 67 As research in this field continues to expand, 84 future trials would benefit from prospectively evaluating optimal strategies for managing adverse effects and the impact of the pharmacological profiles of emerging therapies on these events. The practical approaches presented, including antiemetic algorithms and structured monitoring strategies, integrate expert clinical experience, ADC-specific evidence, and the principles of supportive oncology care, and are intended to support consistent implementation of AINV management in practice.
Supplemental Material
sj-docx-1-opp-10.1177_10781552261441533 - Supplemental material for Practical management of antineoplastic-induced nausea and vomiting from antibody–drug conjugates: Insights from a Canadian expert panel
Supplemental material, sj-docx-1-opp-10.1177_10781552261441533 for Practical management of antineoplastic-induced nausea and vomiting from antibody–drug conjugates: Insights from a Canadian expert panel by Katarzyna J Jerzak, Stephanie Snow, Jan-Willem Henning, Nathaniel Bouganim and Lysanne Besse in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
Editorial support was provided by STA Healthcare Communications (Montréal, Canada) with funding from Knight Therapeutics.
Author contributions
KJJ served as corresponding author, provided leadership and conceptualization. All authors contributed to the writing of the manuscript, reviewed drafts, provided comments and approved of the final version taking responsibility for its accuracy. LB also shared some institutional resources which were pivotal in developing ideas and figures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this publication was provided by Knight Therapeutics Inc. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KJJ has been a consultant, speaker, or advisory board member for Amgen, AstraZeneca, Apo Biologix, Daiichi Sankyo, Eli Lilly, Esai, Genomic Health, Gilead Sciences, Knight Therapeutics, Merck, Myriad Genetics, Novartis, Organon, Pfizer, Roche, and Viatris; has received research funding from AstraZeneca, Eli Lilly, and Pfizer; has received support for attending meetings or travel from AstraZeneca and Daiichi Sankyo; and has received drug supply from Pfizer and Viatris for an investigator initiated clinical trial. SS has been a consultant, speaker, and/or advisory board member for Amgen, Astellas, AstraZeneca, Bayer, Beigene, BMS, Boehringer Ingelheim, Daiichi Sankyo, Janssen, Knight, Lilly, Merck, MSD, Novartis, Pfizer, Roche, Sanofi, Taiho and Takeda; has received institutional paid research funding from Amgen, Astellas, Arcus, AstraZeneca, BMS, GSK, Merck, Novartis and Sanofi. J-WH has received research grants from Pfizer, Astra-Zeneca, Novartis, CIHR, and BioCanRx; speaker honorarium from Amgen, Pfizer, AstraZeneca, Roche, Novartis, Eisai, Seagen, Knight, Sanofi, Bayer, Eli-Lily, the University of Toronto, and Oncology Education; and was a medical advisory consultant for Amgen, Pfizer, AstraZeneca, Roche, Novartis, Eisai, Seagen, Knight, Sanofi, Bayer, and Eli-Lily. NB has received speaker honorarium from Amgen, Pfizer, AstraZeneca, Roche, Novartis, Knight, Eli-Lily, and Merck. LB has received speaker honorarium from Apotex/Apobiologix, Pfizer, AstraZeneca, Roche, Novartis, Eisai, Seagen, Knight, Sanofi, Bayer, and Eli-Lily; and was an advisory consultant for Amgen, Knight, Pfizer, AstraZeneca, Roche, Novartis, Eisai, Seagen, Knight, Sanofi, Bayer, and Eli-Lily.
Data sharing statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Supplemental material
Supplemental material for this article is available online.
