Abstract
Introduction
Gonadotropin-releasing hormone (GnRH) agonists are the most commonly used form of androgen deprivation therapy (ADT) for advanced prostate cancer, often prescribed with radiotherapy or chemotherapy. This study examined national trends in the prescribing of injectable GnRH agonists in England from 2015 to 2024, by formulation type and demographic factors (age, ethnicity, and deprivation). We hypothesised that the use of longer-acting formulations has been increasing over time, specifically during the COVID-19 pandemic when access to face-to-face appointments was limited, and that variation over time and by demographics could inform future service delivery.
Methods
With the approval of NHS England, we conducted a cohort study using the OpenSAFELY-TPP database of 24 million adults. Monthly prescription counts and rates (per 100,000 men with prostate cancer) were visualised over time. Generalised linear models were used to estimate the impact of the COVID-19 pandemic.
Results
The cohort included 390,265 men with prostate cancer (mean age 69.8 years, SD 13.5). Overall, 1,535,725 prescriptions were issued to 208,010 participants (53%). Monthly prescription counts increased by 40%, from 11,787 in 2015 to 16,697 in 2024, while rates declined from 8453 to 7721. During 2020–2021, prescribing of 1- and 3-monthly formulations decreased, whereas 6-monthly formulations increased from 437 per month (245 per 100,000 men) in 2019 to 755 (349 per 100,000 men) in 2024, an excess of 29%.
Conclusions
Before the pandemic, 6-monthly formulations were rarely prescribed. Their uptake during the pandemic suggested a shift towards longer-acting formulations, reducing treatment burden. Declining GnRH rates may reflect earlier diagnosis and evolving treatment guidelines. Divergence between prescription counts and rates, and variation by demographic factors, reflected challenges faced by healthcare systems.
Keywords
Introduction
Prostate cancer is the second most common cancer in men, with nearly 1.5 million new cases diagnosed globally each year, 1 of which 300,000 are in the United States. 2 In the United Kingdom (UK), there are 50,000 new cases annually, and around 20% of these are diagnosed at the metastatic stage. 3 The National Institute for Health and Care Excellence (NICE) in the UK recommends androgen deprivation therapy (ADT) as the standard of care for high-risk localised disease in combination with radiation therapy, and for metastatic prostate cancer, alongside other treatments such as androgen receptor pathway inhibitors (ARPIs) and chemotherapy. 4 Treatment with gonadotropin-releasing hormone (GnRH) agonists is the most common form of ADT. Due to their flexibility and reversibility, GnRH agonists are preferred over surgical castration.
Castration levels of testosterone are typically achieved within a month of starting a GnRH agonist. However, an initial testosterone surge occurs, which requires management with an androgen receptor blocker to prevent exacerbation of prostate cancer symptoms and tumour flare. Additionally, GnRH agonists are administered via injection, necessitating regular healthcare appointments, which can be inconvenient and challenging, particularly for elderly or rural patients. The dosing frequency of GnRH agonists depends on the formulation. The most commonly prescribed formulations are the 3-monthly, followed by the 1-monthly options. Longer acting formulations are available, including those administered every six months, 5 and a new, once a year formulation which is not yet widely approved. 6
Despite their cost and capacity-saving advantages, 6-monthly formulations are rarely used in the UK. 7 Adapting clinical practice takes time. Primary care practitioners prescribe GnRH agonists based on specialist advice, local guidelines, and formularies, which may result in administrative delays in reviewing and implementing local agreements. The shift towards remote consultations during the COVID-19 pandemic led to a decrease in injection-based services, including the administration of GnRH agonists for prostate cancer, 7 musculoskeletal diseases 8 and haematology.9,10 GnRH agonist injections are generally not suitable for self-administration by patients. Before the pandemic, the injection of a GnRH agonist required a face-to-face appointment with a healthcare professional. Since then, some healthcare providers have adapted by training selected patients to self-inject GnRH agonists. 11 In addition, the introduction of new therapies, and updates to national guidelines including approvals of ARPIs such as enzalutamide, apalutamide, and abiraterone for metastatic prostate cancer,12–14 may have influenced prescribing trends.
Preliminary results from Barett et al. (2021), which included all primary care prescriptions dispensed in England between 2019 and 2020, showed a growing preference for longer-lasting 6-monthly formulations and a simultaneous decrease in 1-monthly formulations, linked to the COVID-19 pandemic. 7 Barett et al. (2021) focused on all prescriptions, not specifically on the prostate cancer population, and therefore could not account for the significant decrease in prostate cancer diagnoses which was observed during the first three years of the pandemic. 15 In this study, we investigated the prescribing trends of injectable GnRH agonists from 2015 to 2024, focusing specifically on men with prostate cancer. The objectives were to examine longitudinal trends overall and by formulation type, assess the impact of the COVID-19 pandemic, and explore differences by age, socioeconomic deprivation, and ethnicity. We hypothesised that the use of 6-monthly formulations increased over time, with shifts in prescribing associated with the COVID-19 pandemic, and that these trends varied by age, socioeconomic deprivation, and ethnicity. These stratifications were important because changes during the pandemic, as well as differences by age, socioeconomic deprivation, and ethnicity, have direct implications for healthcare quality and potential health inequities.
Materials and methods
Study purpose
We accessed data on primary care prescriptions of GnRH agonists in England, UK, from January 2015 to May 2024. We investigated changes over time and the impact of the COVID-19 pandemic on prescribing rates in men with prostate cancer, stratifying by formulation type and demographic factors.
Study design
This was a retrospective cohort study of men with prostate cancer using a large, nationally representative database. We conducted time-trend analysis to assess changes over time in monthly prescription counts and rates and applied interrupted time series analysis to investigate the impact of the COVID-19 pandemic.
Data source
We used the OpenSAFELY-The Phoenix Partnership (TPP) database comprising 24 million people registered with primary care practices that use TPP's SystmOne software. The dataset covered over 40% of England's population and was nationally representative. 16 Primary care records managed by the software provider, TPP, were linked to Secondary Uses Service hospital procedures and admissions data, and Office of National Statistics death data through the OpenSAFELY analytics platform. All data were stored and analysed securely using the OpenSAFELY platform. Data included pseudonymised data such as coded diagnoses, medications and physiological parameters. No free text data were available.
Study population and study dates
The study population were adult men registered with TPP practices between 1st January 2015 and 31st May 2024 and diagnosed with prostate cancer (prevalent and incident cases) of all cancer stages. Prostate cancer diagnosis was defined by a list of systematised nomenclature of medicine clinical terminology (SNOMED-CT) codes. The study entry date for each participant was either 1st January 2015, which was the start of the study period, or the date of prostate cancer diagnosis if it was after the 1st January 2015. The date of prostate cancer diagnosis was defined as the first time that a clinical code indicating prostate cancer was entered to a patient's healthcare record. The end of the follow-up was either deregistration, death or 31st May 2024, which was the end of the study period, whichever was the earliest. In the UK, the pandemic-related restrictions started in March 2020. Therefore, to analyse the effect of the COVID-19 pandemic, we used trends from before the pandemic (1st January 2015 to 29th February 2020), to predict the trends which would be expected during and after the pandemic (1st March 2020 to 31st May 2024), as if the pandemic had not occurred.
Outcome measures
We extracted primary care prescriptions for injectable GnRH agonists using the NHS Dictionary of Medicines and Devices codes. Code lists were compiled based on medication groups defined in the British National Formulary (BNF). As determined by the database set up, monthly prescription counts were extracted as the number of individuals with at least one prescription. Since it is unlikely that a patient would receive more than one prescription per month for an injectable GnRH agonist, the number of individuals approximated the number of prescriptions. The study cohort was stratified by demographics, including age (categorised as <65, 65–74, 75–84, and ≥85), ethnicity (Asian, black, white, mixed, Chinese, or other), and deprivation (by quintiles). Deprivation was defined using the Index of Multiple Deprivation (IMD). Prescriptions were stratified by formulation type, with 1-monthly, 3-monthly, and 6-monthly injections analysed separately.
Statistical analysis
The demographic characteristics of the study cohort were summarised using descriptive statistics. Counts were rounded to the nearest five preserving rules of statistical disclosure. For 2024, counts were calculated pro rata based on data from January to May 2024. Monthly prescription rates were calculated per 100,000 men with prostate cancer and visualised in longitudinal plots. The denominator included all prevalent and incident cases and was updated monthly, with new prostate cancer diagnoses (incident cases) added and men who died or deregistered from TPP practices removed. Thus, the denominator was recalculated each month, dynamically combining all living men with prostate cancer during that period. Rates were analysed alongside prescription counts to account for changes in the cohort size over time. Detailed changes in cohort size are reported elsewhere. 15 This was particularly important given the significant increase in prostate cancer incidence and prevalence reported in 2018, 17 and a drop reported from 2020 to 2023 due to the COVID-19 pandemic. 15 Average monthly prescription counts and rates were tabulated, and year-on-year changes in rates were reported with percentages. Generalised linear models (GLM) with a Gaussian distribution and identity link were used to conduct interrupted time series analyses. Pre-pandemic data (January 2015 to February 2020) were used to predict expected rates from March 2020 onward, had the pandemic not occurred. The model included a binary interruption variable (coded 0 before March 2020 and 1 after) and its interaction with time, allowing for an immediate level change at the onset of the pandemic and a change in slope from March 2020 onwards. Differences between observed and predicted rates were reported, with statistical significance assessed using 95% confidence intervals of the predicted values. Residual autocorrelation was assessed visually and using the Durbin–Watson (DW) diagnostic test. Mild positive autocorrelation was observed for trends in all injections combined (DW = 1.8, p = 0.099 for rates; DW = 1.7, p = 0.034 for counts) and for 3-monthly injections (DW = 1.9, p = 0.210 for rates; DW = 1.8, p = 0.099 for counts). Moderate positive autocorrelation was detected for 6-monthly injections (DW = 1.4 for rates and 1.5 for counts, p < 0.001), and strong for 1-monthly injections (DW = 0.5 for rates and 0.6 for counts, p < 0.001). For consistency, Newey–West robust standard errors were applied to models across all formulations. Seasonality was examined through visual inspection and residual autocorrelation diagnostics, and as no consistent annual pattern was observed, seasonal terms were not included.
Software and reproducibility
Data management was performed using Python 3.8, with analysis conducted in both Python and R. Code lists, along with the software for data management and analysis, were deposited in a GitHub repository github.com/opensafely/ADTinj and are available for review and reuse. We adhered to reporting guidelines for studies using observational, routinely collected health data. 18
Results
Study population
The study population comprised 390,265 men diagnosed with prostate cancer and registered with a TPP practice during the study period. The mean age at diagnosis was 69.8 years (standard deviation [SD] was 13.5). The age split was that 100,365 (26%) participants were <65 years old, 156,025 (40%) were 65 to 74, 106,045 (27%) were 75 to 84, and 27,835 (7%) were ≥85. Ethnicity information was missing for 75,175 (19%) participants. Of those for whom ethnicity was recorded, 300,065 (95%) were white, 6780 (2%) were black, 1610 (1%) were mixed, 5025 (2%) were Asian, and 1610 (1%) were Chinese.
In total, there were 1,544,100 prescriptions issued to 208,010 (53%) participants. The remaining 182,255 (47%) participants did not have prescription records during the study period. This group included prevalent cases who had completed treatment before January 2015 and were in a disease-free state during the study period, prevalent and incident cases under surveillance or watchful waiting, and individuals receiving ADT treatment outside of primary care. The following analysis focused on prescription trends among participants with a prescription record during the study period. By formulation type, there were 259,455 (17%) prescriptions for 1-monthly formulations, 1,229,310 (80%) for 3-monthly formulations, and 55,335 (3%) for 6-monthly formulations. The average number of prescriptions per month was 13,590 (SD 1699) across the study period and the average monthly rate was 7743 (SD 411).
Trends over time in prescription counts and rates
Overall, we observed a decreasing trend for prescription rates (Figure 1(A) to (D)) but an increasing trend in prescription counts across the study period (Figure 1(E) to (H)). The yearly count increased by 42% from 141,445 prescriptions in 2015 to 200,364 in 2024 (Table 1). The trend in rates was less linear than that of counts (Figure 1(A)). The highest rate was in 2015, with an average monthly rate of 8453 prescriptions per 100,000 men. This dropped by 12% to 7460 in 2017, increased by 6% to 7906 in 2019, and then dropped again after the start of the pandemic by 5% over the 2020 to 2022 period. The recovery from the pandemic was slow at 1% per year from 2023 onwards. In 2024, the average monthly rate was 7721 per 100,000 men.
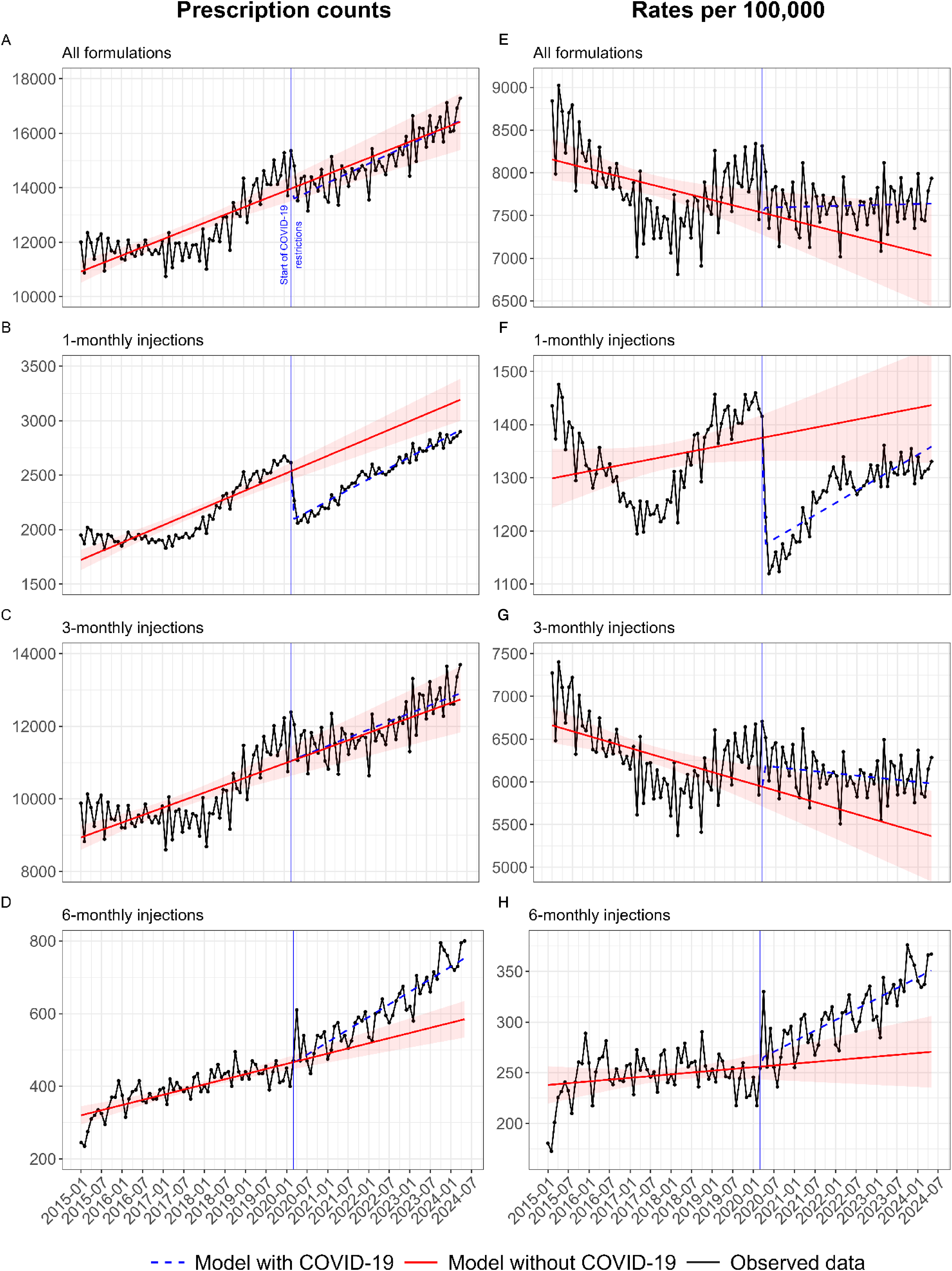
Trends in rates per 100,000 men with prostate cancer (A to D) and counts (E to H) for prescriptions of gonadotropin-releasing hormone (GnRH) agonist androgen deprivation therapy (ADT) in England,2015 to 2024. Observed values were shown with a black line and round markers, the trend after the COVID-19 pandemic was shown with a blue dashed line, and the modelled data representing values as if the pandemic had not occurred were shown with a red solid line and 95% confidence intervals (Cis). Figure (A) and (E), all formulations; B and F, 1-monthly formulations; C and G, 3-monthly formulations; and D and H, 6-monthly formulations.
Trends in average monthly counts and rates per 100,000 men with prostate cancer for prescriptions of gonadotropin-releasing hormone (GnRH) agonists in England, 2015 to 2024. Year-on-year changes in monthly rates were presented with percentages. Predicted rates for 2020 to 2024, estimated using general linear models based on 2015 to 2019 data, were presented with 95% confidence intervals (CIs). Differences between observed and predicted rates were presented with percentages. Statistical significance of the difference was estimated using the 95% CIs. Counts for 2024 were calculated pro rata based on data from January 2024 to May 2024.
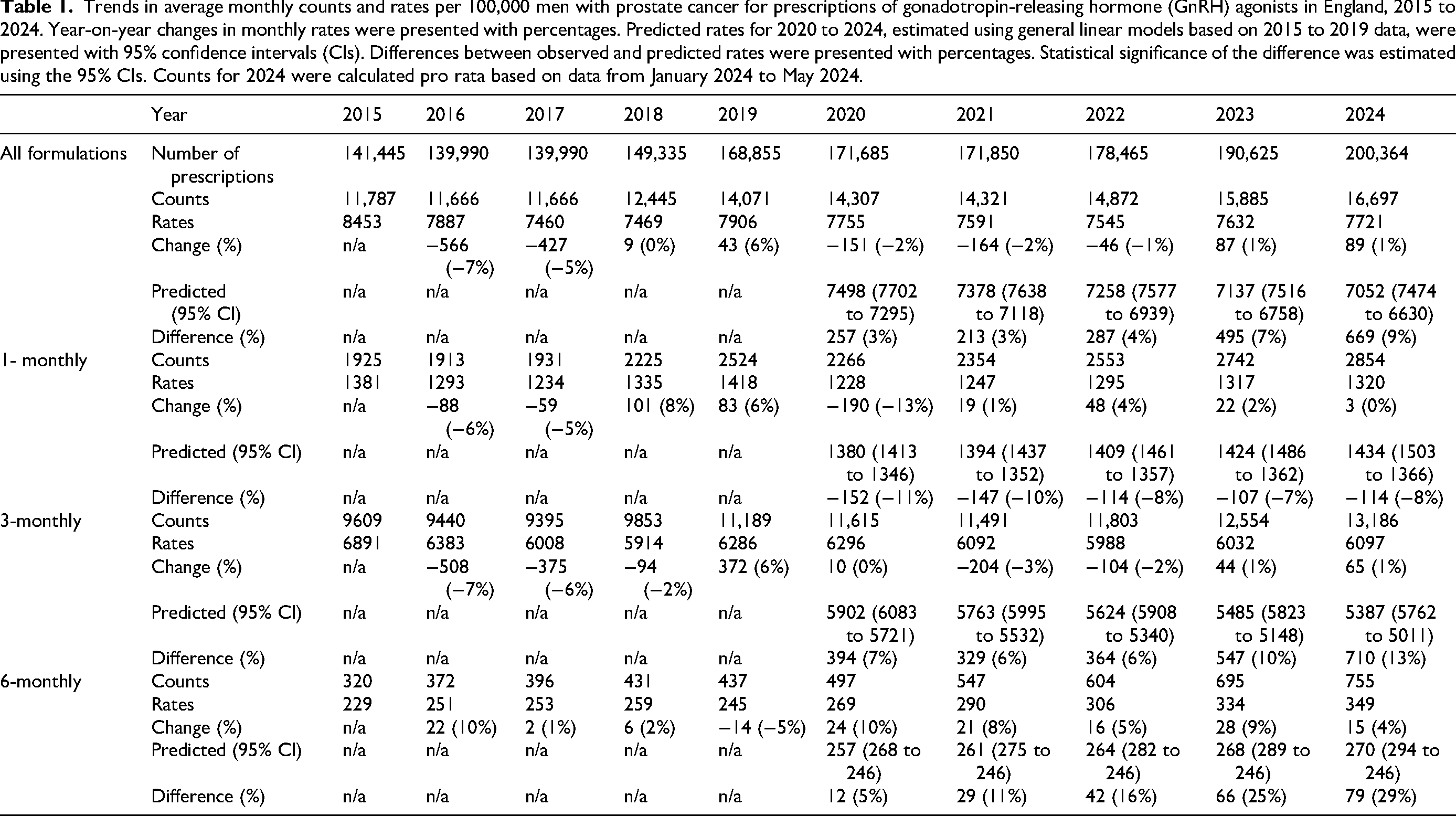
By formulation type, the trend in 3-monthly injections (Figure 1(C) and (G)) closely resembled the overall trend (Figure 1(A) and (E)), with 3-monthly injections accounting for 80% of all the prescriptions (Table 1). For 1-monthly injections, we first observed an 11% drop in rates from 1381 prescriptions per 100,000 men per month in 2015 to 1234 in 2017 (Figure 1(B)). Over 2018 and 2019, we observed a 14% increase, then a sharp drop at the start of the pandemic by 13%, and a recovery over the period of 2020 to 2022. In 2023 and 2024, the rates of 1-monthly injections settled at just over 1300 which was below the pre-pandemic rates. The rates of 6-monthly injections followed a unique trend (Figure 1(D)). While relatively stable from 2015 to 2019, at around 250 prescriptions per 100,000 men per month, from 2020 the rates started to increase by 5% to 10% per year, and for 2024 we recorded the highest average monthly rate of 349 prescriptions per 100,000 men (Table 1).
Frequency of prescribing by formulation type
Throughout the study period, 3-monthly formulations were the most commonly prescribed, accounting for 80% of all prescriptions. By the end of the study period, 3-monthly injections were prescribed at a rate of over 6000 per 100,000 men per month, while 1-monthly injections at a rate of over 1,300, and 6-monthly at a rate of around 350. When normalised to the number of months of treatment, this meant that prescriptions for 3-monthly injections provided 18,000 months of treatment per 100,000 men per month, 6-monthly provided 2100 months of treatment, and 1-monthly formulations provided the least, 1300 months of treatment per 100,000 men per month.
The effect of the COVID-19 pandemic
The increasing trend in the overall rate of injections, which we observed from 2018, was stalled by the pandemic (Figure 1(A)). The biggest drop due to the pandemic was observed for the 1-monthly formulations (Figure 1(B) and (F)). The rate of these injections dropped sharply in April 2020 by nearly 20%, from over 1400 prescriptions per 100,000 men per month to just over 1100. The observed average monthly rate for 1-monthly formulations in 2020 was 1,228, which was 11% lower than the predicted rate of 1380 (95% CI 1413 to 1346). Over 2020 and 2021, we observed a recovery in 1-monthly injections towards the pre-pandemic levels. However, by 2022 the recovery settled at just over 1300 and the pre-pandemic rates were not achieved again. By the end of the study period the observed rates were 8% lower than the predicted 1434 (95% CI 1503 to 1366). In contrast, we observed a steady increase for the 6-monthly formulations from the start of the pandemic (Figure 1(D) and (H)). The rates increased by 42% from the average of 245 in 2019 to 349 in 2024. The observed monthly rates in 2024 exceeded the predicted rates of 270 (95% CI 294 to 246), had the pandemic not occurred, by nearly 30%.
Differences by age, deprivation and ethnicity
There were notable differences in rates by age. The highest monthly rates were for men in the ≥85 age group (Figure 2(A)) with the average monthly rate across the study period of 12,209 prescriptions per 100,000 men 85 and older. This was more than double the rate of 4912 recorded for men <65 and nearly double than the rate of 6479 recorded for men in the 65 to 74 age group. The average monthly rate across the study period for the 75 to 84 age group was 8096. Over time, the differences in rates between the age groups were getting smaller. This was mainly due to the decreasing rates in prescribing for older men. In 2015, the difference in monthly rates between <65 and ≥85 was over 9,000, while in 2024, it decreased to under 5000. In 2015, the rates were 4,967, 6,602, 9262 and 14,176 for <65, 65 to 74, 75 to 84 and ≥85 age groups respectively. In 2024, the rates were 5,520, 7,104, 7864 and 10,315 and represented 11% increase, 8% increase, 23% decrease and 27% decrease in rates for each age group respectively.
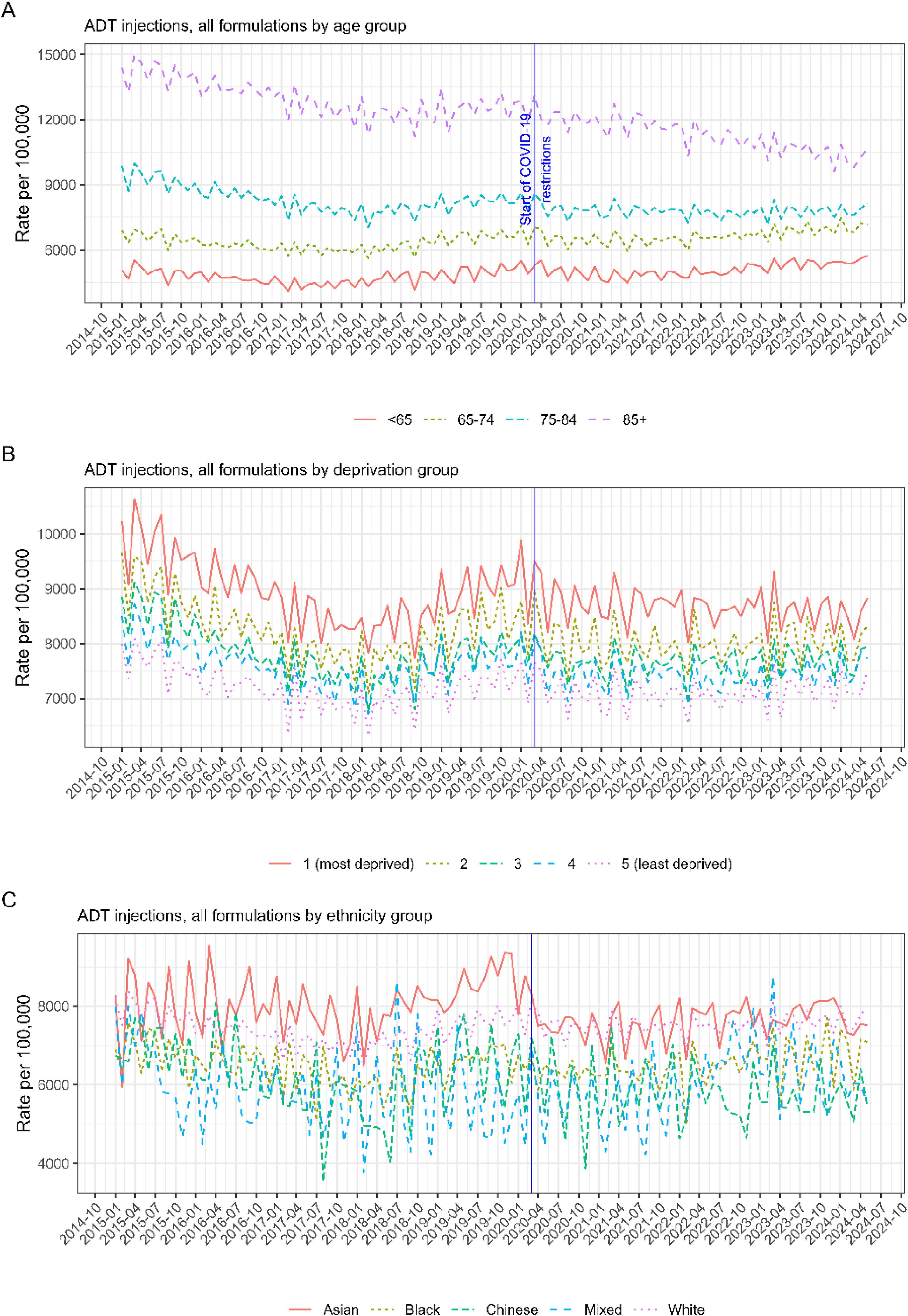
Trends rates per 100,000 men with prostate cancer for prescriptions of gonadotropin-releasing hormone (GnRH) agonist androgen deprivation therapy (ADT), January 2015 to May 2024, stratified by demographic factors. Figure A by age, B by index of multiple deprivation, and C by ethnicity.
Similarly to age, we observed an ordered trend by deprivation and ethnicity. The highest rates were in the IMD quintile 1 group (the most deprived people) and the lowest rates were in the IMD quintile 5 group (the least deprived people). The difference between deprivation groups remained relatively stable across the study period. In 2015, the average monthly rates were, 9,792, 9,006, 8,572, 8123 and 7673 for IMD groups quintile 1 to 5 respectively, and the difference between IMD quintile 1 and 5 was 22%. In 2024, the average monthly rates were 8,547, 8,050, 7,752, 7615 and 7256 (representing 13%, 11%, 9%, 6% and 5% decreases from 2015) and the difference between IMD 1 and 5 was 15%. By ethnicity, the highest average monthly rate of 7860 prescriptions per 100,000 was observed for Asian men, followed by the rate of 7475 for white men. For black and Chinese men, we observed monthly rates of 6497 and 6035 respectively.
Discussion
Summary of findings
From the start of the COVID-19 pandemic, we observed increasing rates of 6-monthly injections alongside a sharp decline in 1-monthly formulations. We interpret these trends as reflecting a shift toward longer-acting formulations, potentially linked to reduced face-to-face appointments within the NHS during the pandemic. Our findings are consistent with those of Barrett et al. (2021), who also reported a growing trend for 6-monthly formulations and a decline in 1-monthly formulations during the same period. 7
While 3-monthly formulations remained the most frequently prescribed throughout the study period, 6-monthly formulations were prescribed to the smallest proportion of men. However, the shift observed during the pandemic led to 6-monthly injections surpassing 1-monthly injections in terms of total months of treatment provided. These findings should be interpreted in the context of treatment duration, as the increasing use of 6-monthly injections was likely associated with fewer prescriptions, lower prescribing rates, and reduced healthcare appointments, but a greater total number of months of treatment delivered.
Overall, we found that while the crude number of prescriptions increased over the study period, prescribing rates declined. The U-shaped trend, with a dip in 2017 and 2018, may reflect rising demand, particularly when considered alongside the 23% increase in prostate cancer incidence in 2018. 15 However, it is important to note that the observed decline in rates may be explained by changes in clinical guidelines, the introduction of new treatments and increased diagnosis of lower-stage cancers. 19 Since the overall decline in rates was most pronounced among older men, this trend may reflect changing preferences in this patient group, and an increasing number of men opting for active surveillance, particularly for low-risk disease, and watchful waiting, in line with NICE recommendations to reduce treatment side effects and maintain quality of life.4,20
Additional explanations for the reduction in rates include the increased adoption of combination systemic therapies in the advanced setting, as well as substitution with oral ADT agents, all of which may alter prescribing patterns of injectable GnRH agonists. 20 It is important to note that this study included only primary care prescribing, and the findings should be interpreted within this context. Secondary care prescribing data were not accessible, and trends did not include ARPIs such as enzalutamide, apalutamide, and abiraterone, 13 immunotherapy, 21 and oral GnRH antagonists such as relugolix, which is increasingly used as an alternative to injectable GnRH agonists, particularly when avoidance of testosterone flare or reduction in cardiovascular risk is a consideration. 22 It was not possible to investigate treatment switches to alternative ADT regimens or changes in prescribing pathways to hospital-based care, including to ADT combination regimens (e.g., with dexamethasone). Therefore, an important consideration when interpreting these findings is that declining rates may reflect therapeutic substitution rather than a reduction in treatment.
Strengths and limitations
A key strength of this study was the size and completeness of the OpenSAFELY dataset. 23 OpenSAFELY-TPP covers over 40% of the population in England, providing access to comprehensive patient records, including data from primary care, secondary care, and mortality. With the dataset capturing a substantial proportion of prostate cancer patients in the country, our findings are likely to be representative of the population in England, enhancing their relevance to national policy and clinical practice. The analysis enables insights into variations across demographic groups. Future studies in other parts of the UK and internationally are needed to understand the global context.
By reporting prescribing rates alongside counts, we were able to interpret our findings in the context of changing prostate cancer incidence and prevalence in England.15,24 This was important because GnRH agonists are also used for other indications. Using rates allowed us to better link prescribing patterns specifically to prostate cancer management. Another strength was the wide time window. We analysed a 10-year period (2015 to 2024), which was essential for modelling and interpreting complex, non-linear trends. OpenSAFELY provided access to near real-time data, and to our knowledge, this is the only study reporting trends in rates of injectable GnRH agonists in England over this period. Importantly, analyses were conducted within the secure OpenSAFELY platform, where data remain on the secure servers and are not transferred externally, safeguarding patient privacy. All analytics code and code lists were shared openly, allowing for transparency, reproducibility, and validation of the study.
In the UK, there are no national datasets of patient-level secondary care prescribing. Although we had access to the SUS dataset, a limitation of this study was the lack of hospital prescribing data. As a result, we were unable to investigate prescribing rates for ARPIs, which are not prescribed in primary care, but are an increasingly important ADT option for men with prostate cancer. Although GnRH agonists are typically prescribed in primary care in the UK, service delivery has changed over time and may vary by region. While the trends presented are expected to approximate national trends, a proportion of injectable GnRH agonists, especially in the earlier years of the study, could have been prescribed in hospitals. Therefore, the study may underrepresent total treatment, with some of the observed variation reflecting differences in prescribing models and the speed of their adoption.
Monthly rates were calculated using the number of patients with at least one prescription. Since patients typically receive no more than one injectable GnRH agonist prescription per month, this count closely approximates the number of prescriptions. To enhance the reliability of our findings, we validated the results using publicly available prescribing data from OpenPrescribing.net. 25
Implications for practice
Injection-based services, including GnRH agonist administration, were particularly affected by the COVID-19 pandemic due to their reliance on face-to-face appointments, which became increasingly limited.7–9 Six-monthly formulations offer important advantages, including greater patient convenience and reduced drug and healthcare costs due to fewer injections. 26 Traditionally, 3-monthly formulations have been the most commonly used, with 6-monthly options rarely prescribed. It is possible that the pandemic prompted a shift in clinical practice, increasing familiarity with and preference for longer-acting formulations. Future research is needed to confirm whether this trend persists.
For oncology pharmacy practice, the shift towards longer-acting GnRH agonist formulations highlighted the important role of national and local formularies in supporting these complex transitions between formulation types and ensuring cost-effectiveness by balancing drug costs with administration burden and service capacity. Although GnRH agonists are not routinely suitable for patient self-administration, some providers introduced self-injection training during the pandemic. 11 As longer acting and potentially self-administered formulations become more widely adopted, pharmacists will play a key role in assessing patient suitability, providing training on safe handling and injection technique, and monitoring adverse effects. During the pandemic, pharmacists were central to counselling clinicians and patients on injection frequency, coordinating care between primary and secondary settings, and ensuring continuity of supply when access to healthcare services was limited. Beyond cost savings, the reduced need for appointments also strengthens system resilience in preparation for future pandemics.
Reducing inequalities in cancer treatment remains a key priority. While health status, comorbidity, frailty, and cancer stage are central to treatment decisions in prostate cancer, we observed systematic differences in GnRH agonist prescribing by demographic factors such as age, deprivation, and ethnicity. Older men, as well as Asian and white men from more deprived backgrounds, had higher prescribing rates, suggesting they were more likely to receive a GnRH agonist than younger, black, or Chinese men from less deprived areas. We hypothesise that this may reflect differences in disease stage at diagnosis, or differences in treatment preferences, suggesting that some groups were more likely to present with advanced prostate cancer, or less likely to choose radical treatment and ADT.27,28 Although these findings are consistent with the National Prostate Cancer Audit,13,29 without information on cancer stage, comorbidities, frailty, or patient preferences, it is difficult to determine whether the observed differences by age, deprivation, and ethnicity reflect inequities or appropriate clinical variation. It is also possible that individuals from less deprived backgrounds sought treatment in private, non-NHS facilities. Further research is needed to explore the underlying drivers of these demographic variations and their implications for equity in prostate cancer care.
Conclusions
Prescribing of injectable GnRH agonists for prostate cancer in England has changed notably over the past decade. Although the total number of prescriptions increased, prescribing rates declined, suggesting shifts in treatment patterns alongside rising patient numbers and the emergence of new ADT therapies. We observed an accelerated shift towards longer-acting formulations, particularly 6-monthly injections, reflecting adaptation to reduced face-to-face care and evolving clinical practice. These findings highlight important changes in service delivery and treatment provision within prostate cancer care.
Footnotes
Acknowledgements
We are very grateful for all the support received from the TPP Technical Operations team throughout this work, and for generous assistance from the information governance and database teams at NHS England and the NHS England Transformation Directorate. RK was supported by a Cancer Research UK Career Establishment Award (RCCCEA-Nov24/100001). The project was supported by the NIHR Imperial Biomedical Research Centre (BRC). The views expressed are those of the authors and not necessarily those of the NIHR, NHS England, UK Health Security Agency (UKHSA), the Department of Health and Social Care, or other funders. Funders had no role in the study design, collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Membership of the OpenSAFELY Collaborative: Alex J. Walker, Brian MacKenna, Peter Inglesby, Christopher T. Rentsch, Helen J. Curtis, Caroline E. Morton, Jessica Morley, Amir Mehrkar, Seb Bacon, George Hickman, Chris Bates, Richard Croker, David Evans, Tom Ward, Jonathan Cockburn, Simon Davy, Krishnan Bhaskaran, Anna Schultze, Elizabeth J. Williamson, William J. Hulme, Helen I. McDonald, Laurie Tomlinson, Rohini Mathur, Rosalind M. Eggo, Kevin Wing, Angel Y. S. Wong, Harriet Forbes, John Tazare, John Parry, Frank Hester, Sam Harper, Ian J. Douglas, Stephen J. W. Evans, Liam Smeeth & Ben Goldacre.
Ethical approval and information governance
NHS England is the data controller of the NHS England OpenSAFELY COVID-19 Service; TPP is the data processor; all study authors using OpenSAFELY have the approval of NHS England. 30 This implementation of OpenSAFELY is hosted within the TPP environment which is accredited to the ISO 27001 information security standard and is NHS IG Toolkit compliant. 31
Patient data have been pseudonymised for analysis and linkage using industry standard cryptographic hashing techniques; all pseudonymised datasets transmitted for linkage onto OpenSAFELY are encrypted; access to the NHS England OpenSAFELY COVID-19 service is via a virtual private network (VPN) connection; the researchers hold contracts with NHS England and only access the platform to initiate database queries and statistical models; all database activity is logged; only aggregate statistical outputs leave the platform environment following best practice for anonymisation of results such as statistical disclosure control for low cell counts. 32
The service adheres to the obligations of the UK General Data Protection Regulation (UK GDPR) and the Data Protection Act 2018. The service previously operated under notices initially issued in February 2020 by the Secretary of State under Regulation 3(4) of the Health Service (Control of Patient Information) Regulations 2002 (COPI Regulations), which required organisations to process confidential patient information for COVID-19 purposes; this set aside the requirement for patient consent. 33 As of 1 July 2023, the Secretary of State has requested that NHS England continue to operate the Service under the COVID-19 Directions 2020. 34 In some cases of data sharing, the common law duty of confidence is met using, for example, patient consent or support from the Health Research Authority Confidentiality Advisory Group. 35
Taken together, these provide the legal bases to link patient datasets using the service. GP practices, which provide access to the primary care data, are required to share relevant health information to support the public health response to the pandemic and have been informed of how the service operates.
The study was approved by the Health Research Authority (Research Ethics Committee reference 20/LO/0651) and the London School of Hygiene and Tropical Medicine (London, UK) Ethics Board (reference 21863).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
GS, CW, MR, and AL were responsible for developing the study proposal. GS, CW, AC, and AL were responsible for data curation and analysis. All authors contributed to data interpretation and manuscript preparation. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The OpenSAFELY platform is principally funded by grants from: NHS England [2023–2025]; The Wellcome Trust (222097/Z/20/Z) [2020–2024]; MRC (MR/V015737/1) [2020–2021]. Additional contributions to OpenSAFELY have been funded by grants from: MRC via the National Core Study programme, Longitudinal Health and Wellbeing strand (MC_PC_20030, MC_PC_20059) [2020–2022] and the Data and Connectivity strand (MC_PC_20058) [2021–2022]; NIHR and MRC via the CONVALESCENCE programme (COV-LT-0009, MC_PC_20051) [2021–2024]; NHS England via the Primary Care Medicines Analytics Unit [2021–2024].
AL was funded by MRC (MR/W021390/1) as part of the postdoctoral fellowship undertaken at the Bennett Institute, University of Oxford.
Declaration of conflicting interest
AM is a former employee and interim CMO of NHS Digital, a member of RCGP health informatics group and the NHS Digital GP data Professional Advisory Group. BMK is employed as a pharmacist by NHS England and seconded to the Bennett Institute and is a trustee of a charity ICAP. JA has received honoraria from J&J Innovative Medicine, Bayer, Accord, Astellas, AstraZeneca and Merck, he is the UK CI of the PROTEUS study (NCT03767244).
Data availability
Access to the underlying identifiable and potentially re-identifiable pseudonymised electronic health record data is tightly governed by various legislative and regulatory frameworks and restricted by best practice. The data in the NHS England OpenSAFELY COVID-19 service is drawn from General Practice data across England where TPP is the data processor. TPP developers initiate an automated process to create pseudonymised records in the core OpenSAFELY database, which are copies of key structured data tables in the identifiable records. These pseudonymised records are linked onto key external data resources that have also been pseudonymised via SHA-512 one-way hashing of NHS numbers using a shared salt. University of Oxford, Bennett Institute for Applied Data Science developers and PIs, who hold contracts with NHS England, have access to the OpenSAFELY pseudonymised data tables to develop the OpenSAFELY tools. These tools in turn enable researchers with OpenSAFELY data access agreements to write and execute code for data management and data analysis without direct access to the underlying raw pseudonymised patient data, and to review the outputs of this code. All code for the full data management pipeline — from raw data to completed results for this analysis — and for the OpenSAFELY platform as a whole is available for review at github.com/OpenSAFELY. The data management and analysis code for this paper was led by AL and contributed to by CA.
Data sharing
Detailed pseudonymised patient data are potentially re-identifiable and therefore not shared. OpenSAFELY provides a secure software interface allowing the analysis of pseudonymised electronic healthcare records of patients from England in near real-time within the TPP's highly secure data centre, avoiding the need for large volumes of patient data to be transferred from where they are managed. This, in addition to other technical and organisational controls, minimises any risk of patient re-identification. All code (including code lists used to define the variables in this study) was shared online via ![]() .
.
