Abstract
Purpose
External contamination of hazardous drug vials poses significant risks of environmental contamination and occupational exposure. This study evaluated the effectiveness of Vial Protect Pack shrink-tack labels (VPP I and II) in preventing surface contamination on vials containing doxorubicin, paclitaxel, and gemcitabine.
Methods
A total of 150 vials from 14 production lots were tested: 60 doxorubicin vials (10 mg, 6 lots) with VPP I, 60 paclitaxel vials (30 mg/5 mL, 5 lots) with VPP II, and 30 gemcitabine vials (200 mg/5 mL, 3 lots) with VPP II. External vial surfaces were sampled using standardized wipe protocols and analyzed by liquid chromatography tandem mass spectrometry.
Results
All 150 vials showed contamination levels below the analytical detection limits, indicating no measurable drug residues on external vial surfaces across all drugs and production lots tested. No lot-to-lot variation was observed.
Conclusion
VPP shrink-tack labels maintained external contamination below analytical detection limits on all tested vials, demonstrating their effectiveness as an engineering control to reduce occupational exposure. Incorporating vials with validated contamination-control technology into hospital procurement policies may enhance healthcare worker protection and support safer compounding practices.
Keywords
Introduction
Hazardous drugs may cause adverse health effects in workers such as pharmacists, pharmacy technicians, and nurses handling these drugs. Safety guidelines have been developed and implemented to reduce the potential risk of exposure.1,2
In hospital pharmacy where hazardous drugs are compounded, there are two critical steps that may cause environmental contamination and potential exposure: the compounding process where drugs are transferred from a vial into a syringe, infusion bag or infusion pump, and external contamination of drug vials entering the compounding process. While closed-system drug transfer devices (CSTDs) can minimize contamination during the compounding process, the issue of pre-existing external vial contamination remains a significant challenge requiring attention at the source.
External vial contamination poses a particular risk because the external surface of drug vials may be contaminated with the drug itself, creating a risk for spread of contamination into the environment and for contamination on prepared products even if the drugs are compounded in an isolator or Biological Safety Cabinet. A review (1990–2018) of 24 published studies has shown that the external surfaces of 56% of 4248 vials sampled tested positive for traces of drugs. 3 Some recent studies show the same contamination problem indicating this issue is not solved yet and still of concern.4–8 Current practices such as wiping vials before use, are time-consuming, generate hazardous waste, and may not completely remove contamination.
The objective of this study was to evaluate the effectiveness of two types of Vial Protect Pack shrink-tack labels (VPP I and VPP II) in preventing external surface contamination on hazardous drug vials. Specifically, we measured contamination levels on the external surfaces of doxorubicin, paclitaxel, and gemcitabine vials wrapped in these protective labels to determine whether they could provide vials with minimal to nondetectable contamination for healthcare workers.
Materials and methods
The study was performed by Exposure Control Sweden AB (Bohus-Björkö, Sweden).
Vial Protect Pack shrink tack labels
There are two different Vial Protect Pack shrink tack labels: VPP I and VPP II (IL Pharma Packaging Co, Ltd Aichi, Japan, https://group-il.com/en/) (Figure 1). For type VPP I, the vial is covered by a plastic base protector plate and a shrink tack label, which stretches from the aluminum stopper to the base, minimizing the risk of breakage and spillage. For type VPP II, the vial is covered with a plastic cup (protector) and a shrink tack label, which stretches from the aluminum stopper to the base. This type offers additional handling comfort and safety compared to type VPP I.
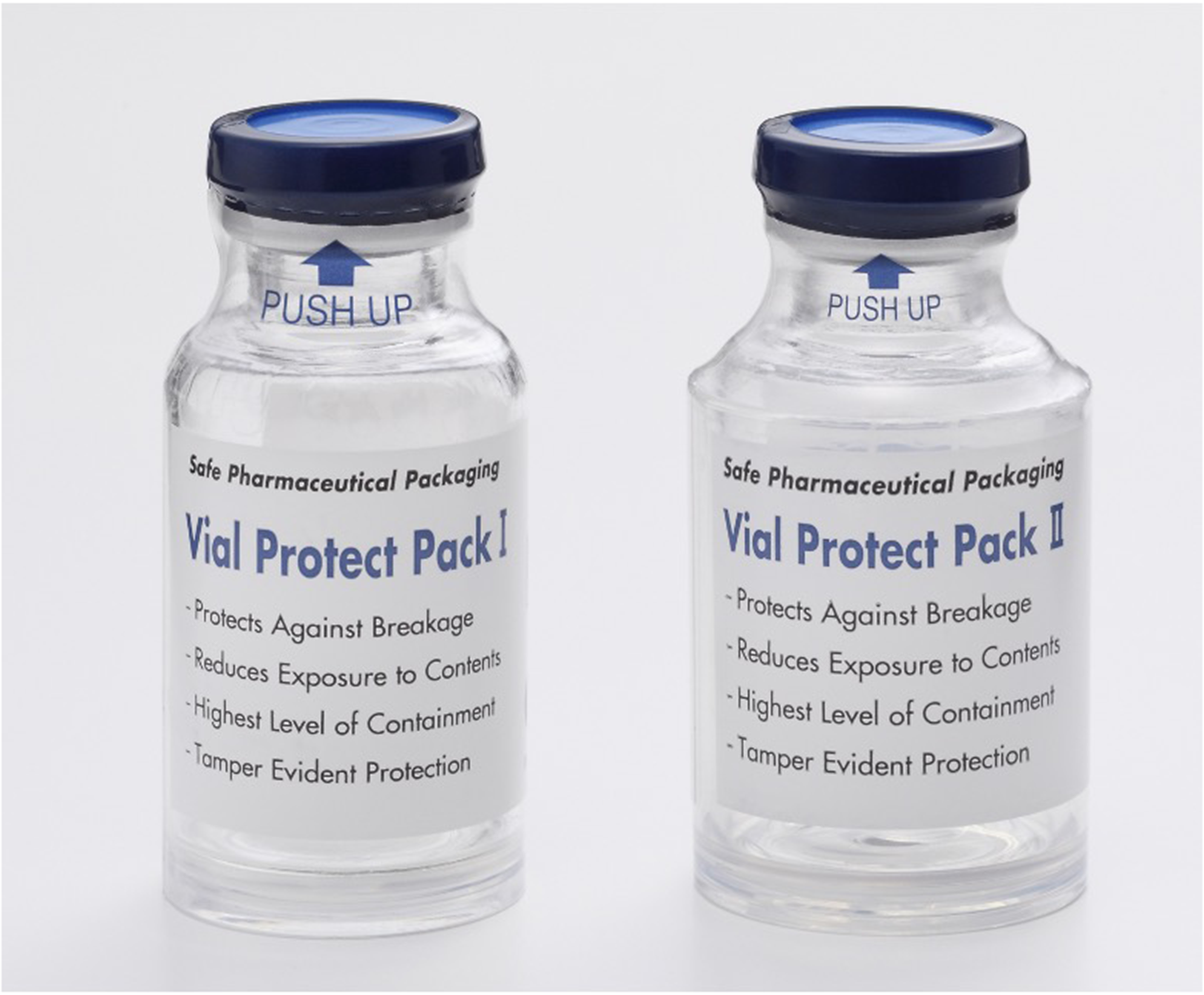
Vial Protect Pack shrink tack labels VPP I (left) and VPP II (right).
Hazardous drug vials tested
The external surface of 150 vials were tested:
- 60 Doxorubicin vials (Doxorubicin Hydrochloride for Injection 10 mg, Nippon Kayaku Co., Ltd), 6 lots of 10 vials wrapped in VPP I (Figure 2). - 60 Paclitaxel vials (Paclitaxel for Injection 30 mg/5 mL, Nippon Kayaku Co., Ltd), 5 lots of 12 vials) wrapped in VPP II (Figure 3). - 30 Gemcitabine vials (Gemcitabine for I.V. Infusion 200 mg/5 mL, Nippon Kayaku Co., Ltd), 3 lots of 10 vials wrapped in VPP II (Figure 4).
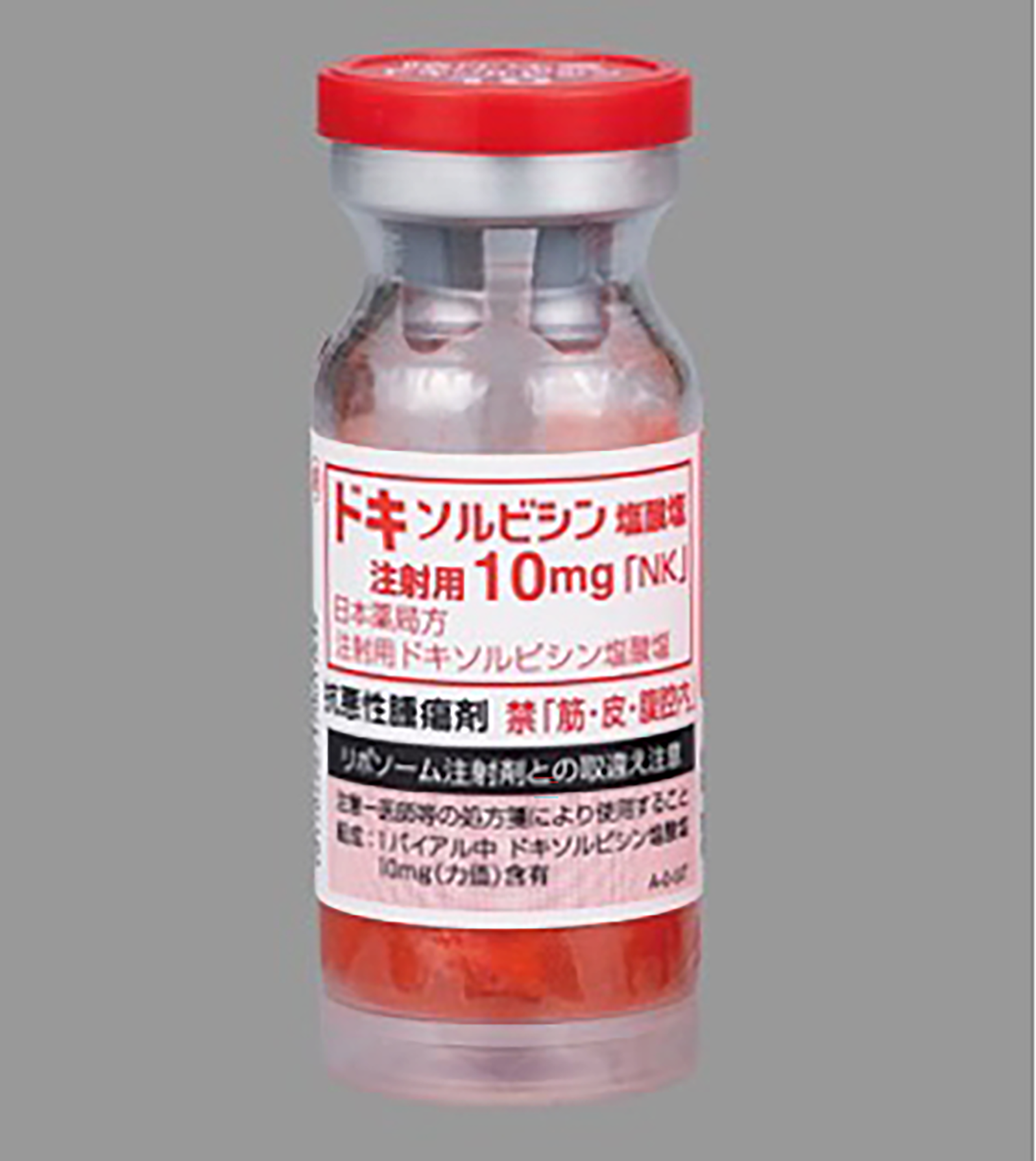
Doxorubicin vial (10 mg) wrapped in VPP I.
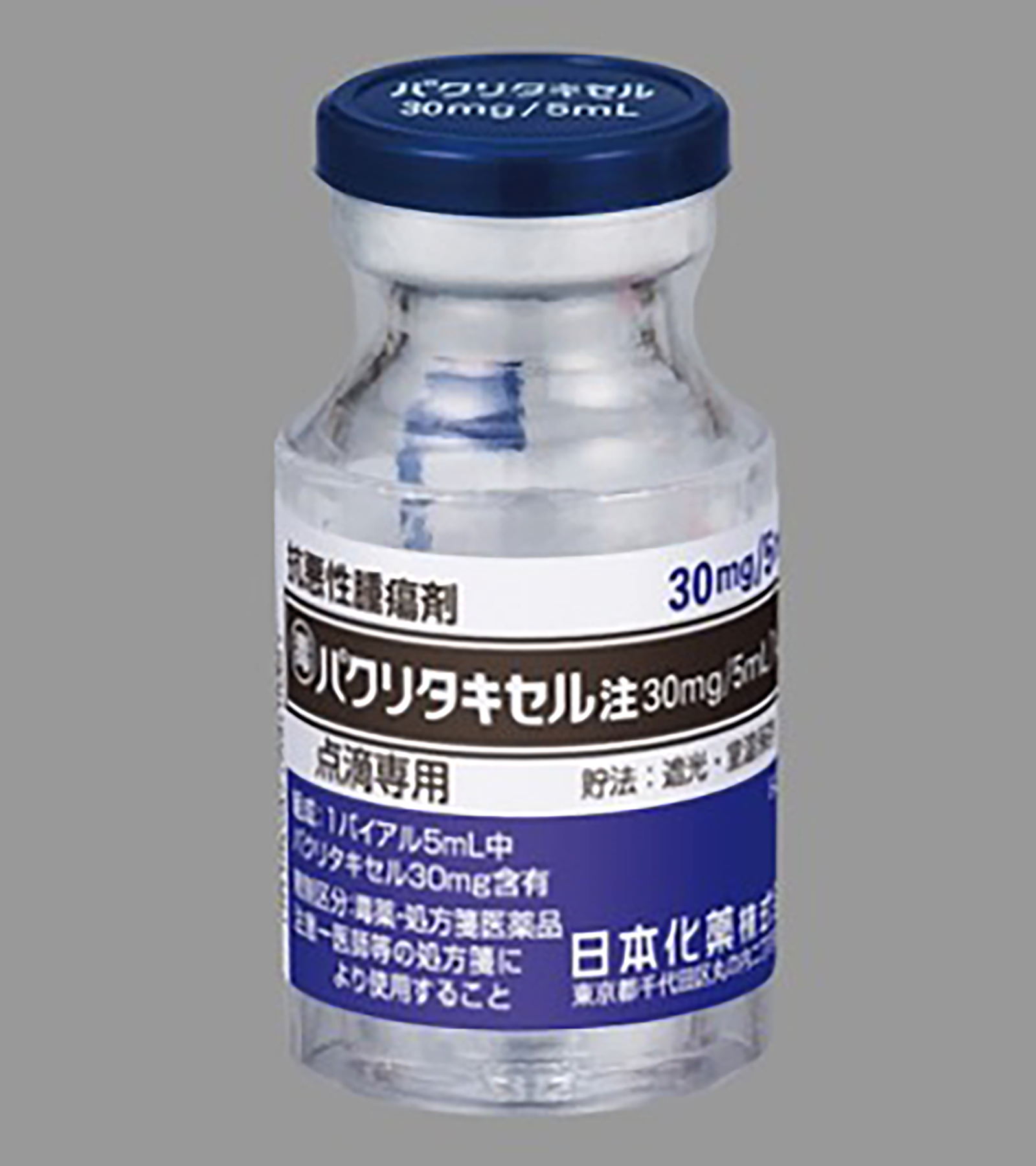
Paclitaxel vial (30 mg/5 mL) wrapped in VPP II.
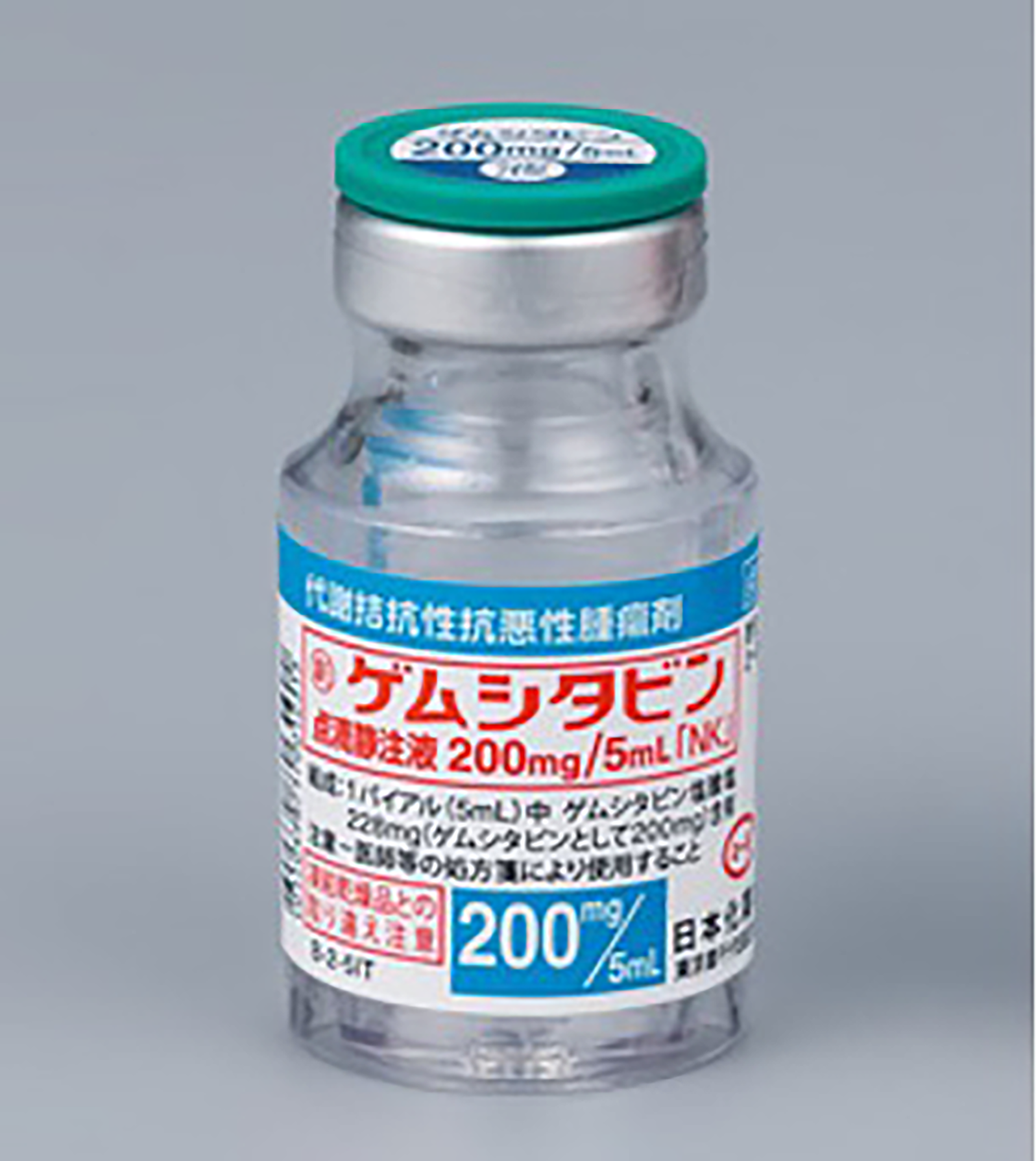
Gemcitabine vial (200 mg/5 mL) wrapped in VPP II.
The external surface of the vials was measured with a caliper, and the surface areas were calculated to express the contamination in ng/cm2. The surface areas were calculated by measuring the diameter and height of five separate parts of each drug vial and adding them together (flip-off cap, neck, shoulder, body, and bottom). The diameter and height varied for the five separate parts and between the three vials. All characteristics are presented in Table 1.
Doxorubicin, paclitaxel, and gemcitabine on the external surface of 150 drug vials.

ND: Not Detected (doxorubicin < 2 ng/mL extract; paclitaxel < 1 ng/mL; gemcitabine < 0.1 ng/mL extract).
Wipe sampling procedure
Cyto Wipe Kits from Exposure Control Sweden AB were used for surface wipe sampling (www.exposurecontrol.net). 9 Formic acid (0.1%, 10 mL) was dripped on a tissue (45 cm × 24 cm) and the entire vial, including the bottom and plastic flip-off cap, was wiped. A single tissue was used for each vial. After sampling, the tissue was placed in a container and 90 mL 0.1% formic acid was added. Samples were extracted by shaking for 60 min.
Liquid chromatography with tandem mass spectrometry analysis
The wipe samples were analyzed immediately after sampling and extraction. 1 mL extract was used. Doxorubicin, gemcitabine, and paclitaxel were analysed using liquid chromatography with tandem mass spectrometry (LC-MS/MS). Details of the analytical method and equipment used have recently been published. 9 The detection limit is 0.1 ng/mL for gemcitabine, 1 ng/mL for paclitaxel, and 2 ng/mL for doxorubicin.
Results
The results of the contamination analysis on the 150 VPP-wrapped vials are presented in Table 1. All vials showed contamination levels below the analytical detection limits: <2 ng/mL (5.6 ng/cm2) for doxorubicin, <1 ng/mL (2.3 ng/cm2) for paclitaxel, and <0.1 ng/mL (0.2 ng/cm2) for gemcitabine, indicating no measurable contamination was found on the external surfaces of the vials. The contamination per unit area (cm2) was calculated assuming 100% recovery and wipe efficiency.
Discussion
This study demonstrates that VPP shrink-tack labels completely prevented detectable external contamination on 150 hazardous drug vials across three different drugs and 14 production lots. The 100% success rate (0/150 contaminated vials) contrasts sharply with historical data showing contamination rates of 56% in unwrapped vials, indicating that VPP technology is an effective engineering control for source contamination. 3
The economic implications of these findings warrant consideration. Although initial implementation of VPP technology requires investment from manufacturers, downstream savings may be substantial. Healthcare facilities currently spend considerable resources on vial cleaning procedures (estimated at 5–10 min per preparation) as well as on hazardous waste disposal, and potential worker compensation claims from occupational exposures. Eliminating external contamination at the manufacturing level shifts the economic burden upstream while potentially reducing overall healthcare system costs.
These findings align with current pharmaceutical safety guidelines, including ISOPP Standards for the Safe Handling of Cytotoxics, which emphasize the hierarchy of controls and prioritize engineering controls over administrative measures and personal protective equipment. 1 VPP technology embodies this principle by preventing contamination at the source, rather than relying on downstream mitigation. This preventive approach is particularly valuable given the variability and incomplete effectiveness of manual decontamination procedures.
Several limitations should be acknowledged. First, unwrapped vials from the same manufacturers were not included in the study for comparison indicating it is not possible to determine whether the non-detectable contamination is due to the VPP technology or reflects clean manufacturing processes. Second, wipe recovery efficiency was not evaluated. Laboratory validation studies have shown >80% recovery rates for similar protocols but efficiency on VPP-wrapped vials may differ. 9 Even under a conservative assumption of 50% recovery, the absence of detectable contamination suggests true contamination levels were negligible. Third, only three drugs were tested, although they represent diverse chemical classes (anthracyclines, taxanes, and antimetabolites) with distinct physicochemical properties. The consistent results across these compounds suggest the findings may be generalizable to other hazardous drugs.
Integration with existing safety infrastructure is another important consideration. Compatibility testing have been started to validate proper seal integrity with VPP-wrapped vials. This could ensure that both critical contamination pathways (external vial contamination and drug transfer procedures) can be controlled simultaneously without compromising effectiveness. In addition, research should be expanded in testing a broader range of hazardous drugs, including newer immunotherapies and targeted therapies such as Antibody Drug Conjugates (ADCs).
Conclusion
The study demonstrates that VPP technology can realize external vial contamination at nondetectable levels. Hospitals are encouraged to procure clean vials to prevent environmental contamination and protect healthcare workers from occupational exposure. These findings support the adoption of validated contamination-control technology at the manufacturing level as a proactive strategy to enhance safety in healthcare settings.
Footnotes
Acknowledgements
Parts of the study have been presented as a poster during the ISOPP Conference in Vancouver, Canada, April 3–6, 2025.
Author contributions
Both authors designed the study; PS collected the data and performed the analysis; both authors interpreted the results; PS drafted the manuscript. Both authors reviewed and approved the final version of the manuscript.
Funding
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
