Abstract
Introduction
Elranatamab, a bispecific antibody targeting B-cell maturation antigen (BCMA) and CD3, has demonstrated remarkable efficacy in relapsed or refractory multiple myeloma (RRMM). With the growing clinical use of BCMA-directed bispecific antibodies, their safety profile continues to evolve; however, progressive multifocal leukoencephalopathy (PML) has not been systematically described with elranatamab to date.
Case Report
A 67-year-old woman with RRMM achieved a complete response following elranatamab therapy. After the seventh treatment cycle, she developed neurological symptoms including dysarthria and gait disturbance. Brain magnetic resonance imaging revealed multifocal, non-enhancing white-matter lesions, and cerebrospinal fluid polymerase chain reaction confirmed JC virus infection, establishing the diagnosis of PML.
Management and Outcome
Elranatamab was discontinued immediately. Despite treatment with intravenous immunoglobulin, mirtazapine, and compassionate-use nivolumab, her neurological status progressively worsened, necessitating intubation and intensive care management.
Discussion
This case suggests a probable association between elranatamab therapy and JC virus reactivation leading to PML. The pathogenesis is likely multifactorial, reflecting both prior cumulative immunosuppression and elranatamab-induced plasma-cell depletion with resultant hypogammaglobulinemia. Clinicians should maintain vigilance for new or unexplained neurological manifestations in patients receiving BCMA-directed T-cell–redirecting therapies. Early neuroimaging and cerebrospinal fluid JC virus testing, combined with proactive immunoglobulin replacement and systematic pharmacovigilance, are essential for timely diagnosis and improved outcomes. Overall, this case highlights the need for early JC virus monitoring and awareness of delayed neuroinfectious complications associated with BCMA-targeted immunotherapies.
Keywords
Background
Elranatamab is a bispecific antibody targeting B-cell maturation antigen (BCMA) and CD3, designed to redirect cytotoxic T cells toward malignant plasma cells. In August 2023, it received FDA approval for the treatment of relapsed or refractory multiple myeloma (RRMM) based on the MagnetisMM-3 trial, which demonstrated an overall response rate (ORR) of approximately 61% in heavily pretreated patients. 1
Progressive multifocal leukoencephalopathy (PML) is a rare, demyelinating disease of the central nervous system caused by JC virus reactivation in immunocompromised hosts, most often described in association with immunosuppressive therapies. 2
The most common elranatamab-related adverse events include cytokine release syndrome (CRS), cytopenias, infections, and fatigue. 3 Opportunistic infections—particularly viral reactivations such as cytomegalovirus (CMV)—have been reported due to profound and prolonged immunosuppression.4,5 However, to date, PML associated with elranatamab therapy has not been reported in clinical trials or comprehensively characterized in the literature. Herein, we provide a detailed case description of PML temporally associated with elranatamab therapy.
Case presentation
A 67-year-old female patient was diagnosed with kappa light chain multiple myeloma in 2017 and had received seven prior lines of therapy, including immunomodulatory drugs (IMiDs), proteasome inhibitors (PIs), anti-CD38 monoclonal antibodies, alkylating agents, and autologous stem cell transplantation, before starting elranatamab treatment in February 2025. She had no prior exposure to BCMA-directed bispecific antibodies or CAR T-cell therapies. A complete response was achieved with elranatamab, and the treatment was well tolerated up to the 7th cycle. She had been receiving regular intravenous immunoglobulin (IVIG) (0.4 g/kg every 4 weeks) replacement during elranatamab therapy to manage hypogammaglobulinemia, maintaining IgG levels above 500 mg/dL.
After the 7th cycle of elranatamab, the patient was hospitalized with complaints of diarrhea and was diagnosed with CMV colitis, for which antiviral therapy was administered. At the time of CMV reactivation, she exhibited lymphopenia and neutropenia; however, both parameters improved following antiviral treatment. Despite a subsequent decrease in CMV viral load, the patient developed speech difficulty, psychomotor slowing, gait disturbance, and visual impairment. Within a few weeks, the patient's clinical condition progressed rapidly, culminating in neurological findings that included somnolence, muscle weakness graded 4/5, dysarthric speech, and a unilateral positive Babinski sign on the left.
Brain CT demonstrated hypodense areas adjacent to the posterior horn of the right lateral ventricle, in the splenium of the corpus callosum, left thalamus, bilateral parieto-occipital regions, and cerebellar peduncles. EEG revealed mild encephalopathic changes. Brain MRI revealed multifocal T2-FLAIR hyperintense lesions involving the temporal, left thalamic, and parieto-occipital regions without contrast enhancement (Figure 1). These findings were characteristic of PML. Cerebrospinal fluid (CSF) PCR testing for JC virus was positive, with a viral load of approximately 4.5 × 107 copies/mL (reference positive range: 1.2 × 103–1.2 × 108 copies/mL).
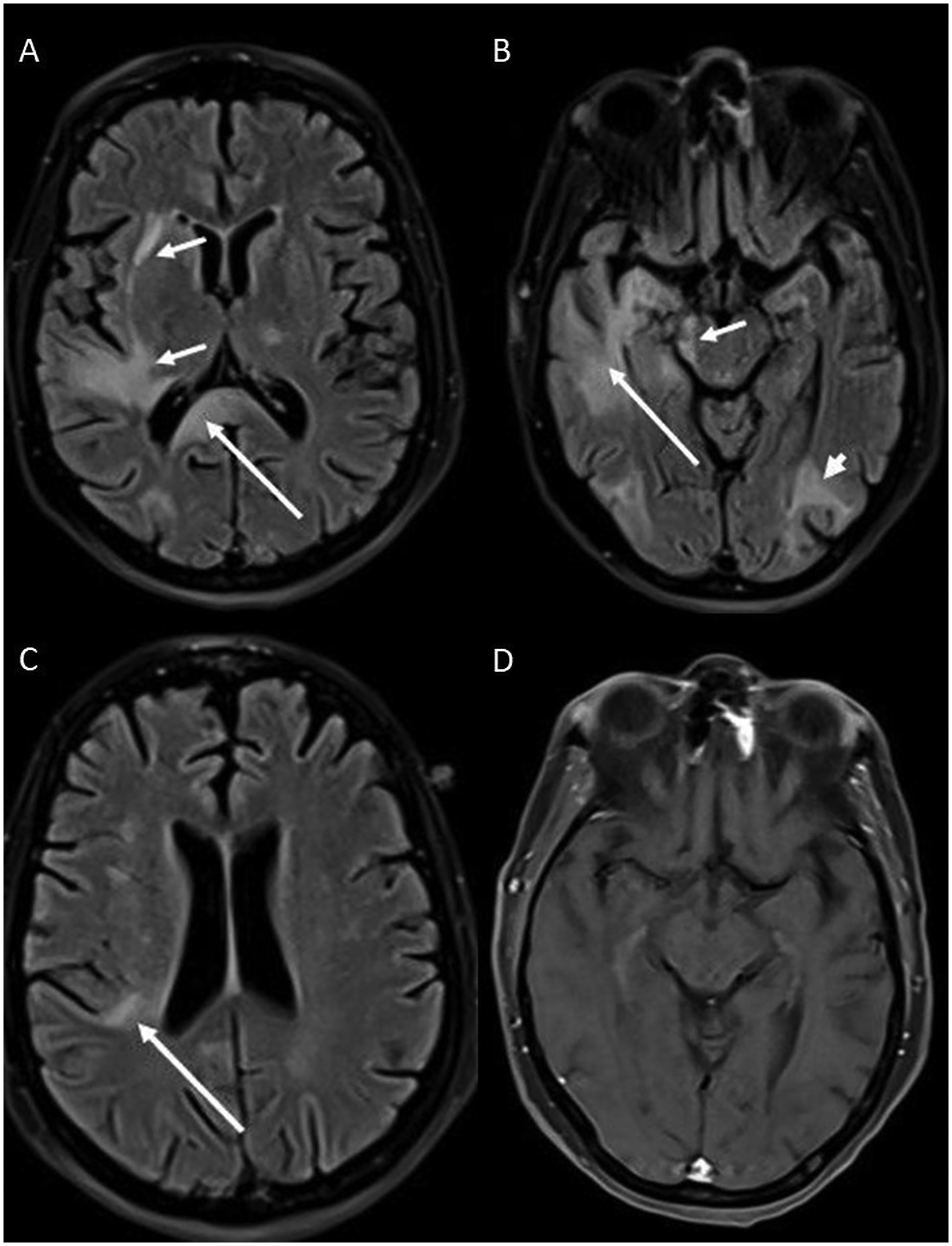
A. Transverse fluid-attenuated inversion recovery (FLAIR) MR image shows increased signal intensity within the splenium of the corpus callosum (long arrow), left lateral thalamus, and right temporal white matter and external capsule (short arrows). B. Transverse FLAIR MR image demonstrates hyperintense areas involving the right temporal lobe (long arrow), right cerebral peduncle (short arrow), and left occipital lobe (arrowhead). C. Transverse FLAIR MR image shows hyperintense area within the right posterior periventricular white matter (long arrow). D. Post-contrast T1-weighted MR image at the same level as Figure 1A shows no enhancement of the lesions.
Following discontinuation of elranatamab therapy, the patient was treated with mirtazapine and IVIG (2 g/kg over two days); however, no meaningful clinical improvement was observed. Subsequently, her level of consciousness progressively declined, necessitating intubation and intensive care management. In the absence of established therapeutic options for PML, nivolumab (3 mg/kg once every 2 weeks) was administered on a compassionate, off-label basis with the aim of restoring anti–JC virus immune surveillance via PD-1 pathway blockade, as previously reported in isolated case studies. Despite transient neurological stabilization, the patient exhibited no sustained recovery.
Discussion
Elranatamab, a bispecific T-cell engager targeting B-cell maturation antigen (BCMA) and CD3, represents an innovative therapeutic approach for relapsed or refractory multiple myeloma (RRMM). While its safety profile has been well characterized in pivotal MagnetisMM trials—primarily involving CRS, cytopenias, and infections—reports of PML remain extremely limited. To our knowledge, this case represents one of the first documented occurrences of PML associated with elranatamab therapy, thereby expanding the recognized spectrum of neurotoxicities linked to BCMA-directed immunotherapies.
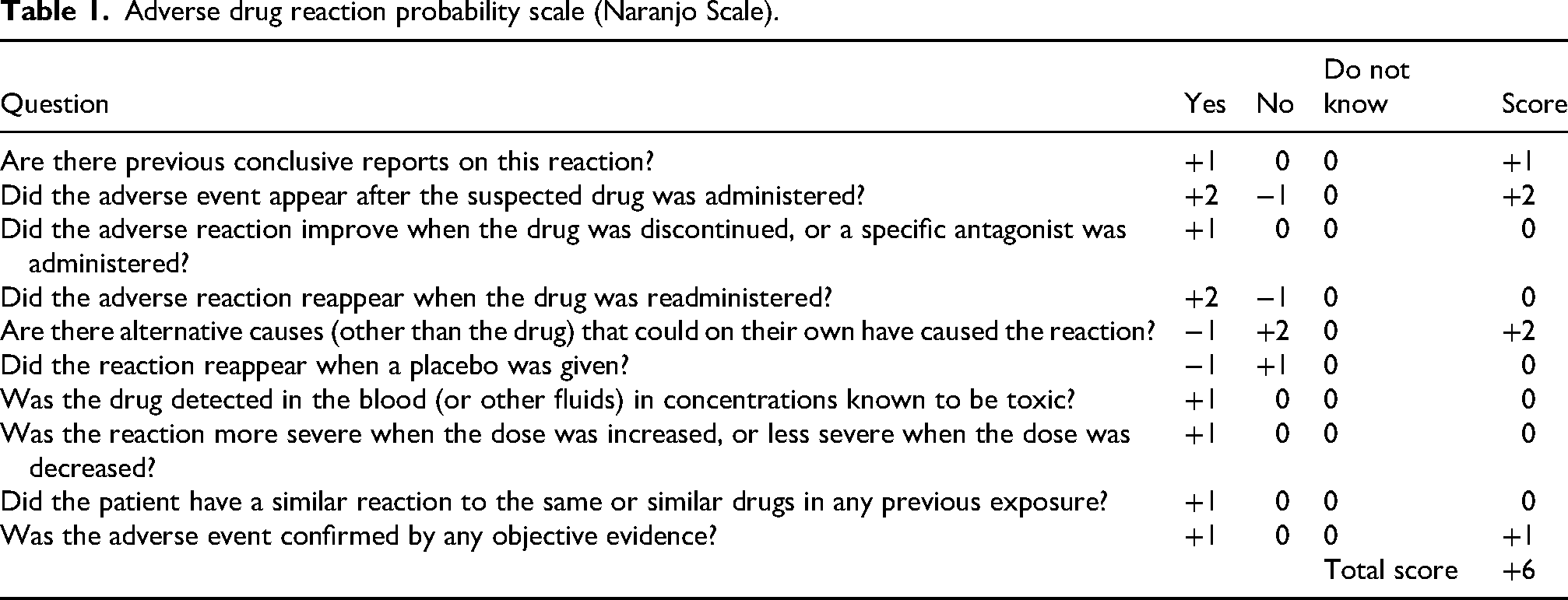
The development of PML in this patient likely reflects a multifactorial interplay between treatment-related and host-related factors. Prior exposure to multiple lines of therapy—including IMiDs, PIs, and anti-CD38 monoclonal antibodies—may have led to profound and sustained immunosuppression. Elranatamab itself may have further contributed by inducing T-cell exhaustion through continuous BCMA–CD3 engagement and causing hypogammaglobulinemia via depletion of normal plasma cells, thereby impairing immune surveillance against latent viruses such as JC virus. Although serum IgG levels were maintained above 500 mg/dL with regular intravenous immunoglobulin replacement, this likely reflects functional hypogammaglobulinemia, as endogenous antibody production and humoral immune competence may remain profoundly impaired following BCMA-directed plasma cell depletion. According to the Naranjo Adverse Drug Reaction Probability Scale, 6 the adverse drug reaction probability score was calculated as 6, indicating a “probable” relationship between elranatamab and the onset of PML (Table 1). However, because the mechanism appears to be indirect—mediated by elranatamab-induced immunosuppression facilitating JC virus reactivation rather than a direct toxic effect—the Naranjo scale may not fully capture the causal relationship.
Adverse drug reaction probability scale (Naranjo Scale).
Interestingly, the patient experienced CMV colitis prior to the onset of neurological symptoms, suggesting a state of marked cellular immunodeficiency. CMV reactivation may serve as an early warning sign of immune collapse and precede other opportunistic viral infections such as JC virus reactivation. This sequence reinforces the need for heightened infectious monitoring during bispecific antibody therapy.
Neurological complications associated with T-cell–redirecting therapies are increasingly recognized, particularly immune effector cell–associated neurotoxicity syndrome (ICANS). 7 ICANS typically arises within the first weeks of therapy, is cytokine-mediated, and responds well to immunosuppressive therapy such as corticosteroids or IL-6 blockade. In contrast, PML in our case manifested as a late-onset, progressive demyelinating disease caused by JC virus reactivation, where immunosuppressive therapy may aggravate the condition.
Therefore, distinguishing PML from ICANS or cytokine-mediated encephalopathies is critical. Radiologically, PML lesions are non-enhancing, asymmetric, and multifocal on T2-FLAIR sequences, whereas ICANS may show reversible edema or normal imaging. In this case, the positive JC virus PCR in CSF and non-enhancing white matter lesions firmly established the diagnosis of PML, underscoring the diagnostic value of early neuroimaging and CSF testing in atypical or delayed neurologic presentations. Thus, in patients with delayed or atypical neurotoxicity under BCMA-directed therapy, JC virus PCR testing should be incorporated early into diagnostic work-up.
Beyond elranatamab, several instances of JC virus reactivation and PML have been reported with other BCMA-targeted immunotherapies. Moreau et al. described a fatal case of PML associated with teclistamab, another bispecific BCMA–CD3 antibody. 8 Furthermore, BCMA-directed chimeric antigen receptor (CAR) T-cell therapies, such as idecabtagene vicleucel and ciltacabtagene autoleucel, have been linked to delayed-onset JC virus reactivation during long-term follow-up. 9 Collectively, these observations suggest a potential “class effect” of BCMA-directed immune modulation rather than a molecule-specific toxicity. This phenomenon may reflect a distinctive immunologic vulnerability arising from plasma cell depletion and impaired humoral immune surveillance.
There is no established antiviral or curative therapy for PML. Reported interventions—such as withdrawal of the implicated agent, IVIG, mirtazapine, or immune checkpoint inhibitors (nivolumab, pembrolizumab)—have yielded inconsistent results. 10 In this patient, despite administration of IVIG, mirtazapine, and nivolumab, neurological deterioration progressed, consistent with the generally poor prognosis observed in hematologic malignancy–associated PML.
This therapeutic limitation further emphasizes the importance of early recognition and accurate diagnosis. Unlike ICANS, where immunosuppression is beneficial, PML management requires immune reconstitution rather than further immune suppression; misdiagnosis may therefore have fatal consequences. Importantly, this case further underscores the need for ongoing studies to define the optimal duration and intensity of BCMA-targeted therapies, balancing sustained disease control against cumulative immunosuppression and late infectious complications. Notably, ongoing clinical studies are currently evaluating response-adapted and fixed-duration treatment strategies for BCMA-directed bispecific antibodies, aiming to mitigate cumulative immunosuppression while preserving durable disease control.
Conclusion
Given the increasing use of elranatamab and other BCMA-targeted bispecific antibodies, vigilance for atypical viral complications such as JC virus reactivation and PML is essential. Early neuroimaging and CSF PCR testing should be considered in patients presenting with new or unexplained neurological symptoms. Systematic pharmacovigilance, along with inclusion of JC virus monitoring in ongoing clinical trials, will be crucial to elucidate the true incidence, underlying mechanisms, and risk factors of elranatamab-associated PML. Collectively, these findings support continued investigation into risk-adapted treatment duration strategies for BCMA-directed immunotherapies.
Footnotes
Acknowledgments
The authors thank the clinical and nursing staff of the Hematology Department, as well as the intensive care and infectious disease teams, for their valuable support in patient management.
Consent for publication
Informed consent was obtained from the patient for the publication of this case report. Ethical approval was not required for this type of case report according to institutional guidelines. The authors affirm that patient identities have been protected and no identifiable information is included.
Author contributions
T.Ç. and U.I. contributed to conceptualization, data curation, drafting, project administration, and supervision.
Ü.A., M.Ç.B., S.A.K., and G.Y.Ü. participated in investigation, validation, and manuscript review.
K.K. and Ö.Ç. provided resources, visualization, and data curation.
O.S. supervised the study, revised the manuscript, and approved the final version.
All authors contributed to the preparation of this manuscript, reviewed the final version, and approved it for submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this case report are available from the corresponding author, U.I., upon reasonable request. Due to privacy and ethical restrictions, individual patient data are not publicly available.
