Abstract
Introduction
Infectious complications are a leading cause of morbidity and mortality in pediatric leukemia, particularly during intensive chemotherapy. Febrile neutropenia (FN), bacteremia, and fungemia are common and potentially life-threatening. This study evaluated the effectiveness of antimicrobial prophylaxis in preventing infectious complications in pediatric patients with leukemia.
Methods
A retrospective, matched chart review was conducted involving 182 pediatric patients (aged 1–18 years) diagnosed with leukemia. Patients were stratified based on receipt of antimicrobial prophylaxis, Levofloxacin, Caspofungin, Fluconazole, or a combination (n = 40), versus no prophylaxis (n = 64). Primary outcomes included the number of FN episodes, bacteremia, and fungemia.
Results
Patients who received antimicrobial prophylaxis experienced significantly fewer FN episodes and bacteremia events compared to those who did not receive prophylaxis. No cases of increased antimicrobial resistance were observed in the prophylaxis group. Rates of fungemia were low in both groups.
Conclusions
Antimicrobial prophylaxis during high-risk phases of chemotherapy is associated with reduced FN and bacteremia in pediatric leukemia patients. These findings support its implementation as a preventative strategy in HR patients to reduce infectious complications without increasing antimicrobial resistance.
Introduction
Leukemia is the most common pediatric cancer, accounting for approximately 30% of all childhood malignancies, with acute lymphoblastic leukemia (ALL) and acute myeloid leukemia (AML) representing the most prevalent subtypes.1,2 While survival outcomes have markedly improved over the past decades due to risk-adapted chemotherapy and supportive care, infectious complications continue to be a leading cause of morbidity and treatment delays in this population.3,4
Febrile neutropenia (FN), bacteremia, and invasive fungal disease (IFD) are frequent and potentially life-threatening sequelae of myelosuppressive chemotherapy. Both standard-risk (SR) and high-risk (HR) leukemia patients experience treatment-induced immunosuppression. However, the risk is particularly pronounced in HR subgroups such as patients with AML, T-cell ALL, Philadelphia chromosome-positive ALL, and Down syndrome-associated ALL due to prolonged neutropenia, central line use, and mucosal barrier injury.5–7 In AML patients, antibacterial and antifungal prophylaxis has become standard of care in many centers, based on evidence from randomized trials and guidelines demonstrating reduced incidence of bloodstream infections and IFD without significantly increasing antimicrobial resistance.8–10
However, in pediatric ALL, especially HR de novo cases, the role of routine antimicrobial prophylaxis remains less clearly defined. Although recent studies such as those by Alexander et al. and Wolf et al. have explored levofloxacin prophylaxis in children with ALL during induction, findings remain mixed, and uptake has been variable.11,12 Moreover, concerns persist regarding the off-label use of fluoroquinolones in children, particularly due to potential musculoskeletal toxicity and regulatory restrictions in some jurisdictions. 13
Similarly, the optimal antifungal agent for prophylaxis in immunocompromised children remains under debate. While fluconazole has been widely used, it offers no protection against molds such as Aspergillus. Caspofungin, an echinocandin with broad-spectrum activity, has shown promise in pediatric AML patients, 14 but its role in HR ALL has not been rigorously evaluated outside of selected cohorts.
Given the ongoing uncertainties and variability in practice, there is a need to better characterize the utility of prophylactic antimicrobial strategies in pediatric patients with HR leukemia, particularly beyond AML and relapsed ALL. This study aimed to assess the effectiveness of antimicrobial prophylaxis, including levofloxacin and either fluconazole or caspofungin, in reducing episodes of FN, bacteremia, and fungemia in a real-world cohort of pediatric patients with leukemia. Unlike previous studies limited to relapsed ALL or AML cohorts, we included both SR and HR patients with de novo leukemia. Our goal was to evaluate the impact of prophylaxis across a broader risk spectrum to better inform its potential role in contemporary pediatric oncology supportive care.
Materials and methods
We performed a descriptive historical matched chart review of 182 patients diagnosed with leukemia at IWK Health, Nova Scotia, Canada, aged 1–18 years old. Of the 182 charts initially screened, 104 patients met the inclusion criteria for matched comparison. Patients were matched based on age group (±2 years), sex, and leukemia subtype (HR or SR). Matching was conducted retrospectively using a 1:1 manual selection from available charts. The non-prophylaxis group included patients diagnosed from 2003–2023, whereas patients in the prophylaxis group included the years of 2018–2023, when prophylaxis with antibiotics and antifungals were first introduced in de novo ALL at the IWK. Our protocol, described in supplemental information, highlights the phases/ cycles of therapy in which antifungal and antibiotic prophylaxis were administered for specific populations and diagnoses. Given the relative stability of the COG trials over the past 20 years, the intensive phases of chemotherapy have not significantly changed. Patients who had received prior hematopoietic stem cell transplantation, blinatumomab, or CAR T-cell therapy were excluded. The patients were separated into two groups based on whether they received antimicrobial (antibiotic, antifungal or both) prophylaxis during their treatment (prophylaxis group, n = 40) or whether they did not receive prophylaxis (control group, n = 64) (Tables 1,2). Antibiotic prophylaxis included levofloxacin 10 mg/kg PO/IV twice daily (maximum 500 mg/day) for children 6 months and greater to less than 5 years. Children 5 years and older received 10 mg/kg PO/IV once daily (maximum 750 mg/day). Antifungal prophylaxis consisted either of caspofungin 50 mg/m2/day IV once daily (max 50 mg) or fluconazole 6–12 mg/kg (max 400 mg). Prophylaxis was given when the absolute neutrophil count approached 200/uL.
Number of high risk and standard risk patients receiving the following antifungal agent(s).
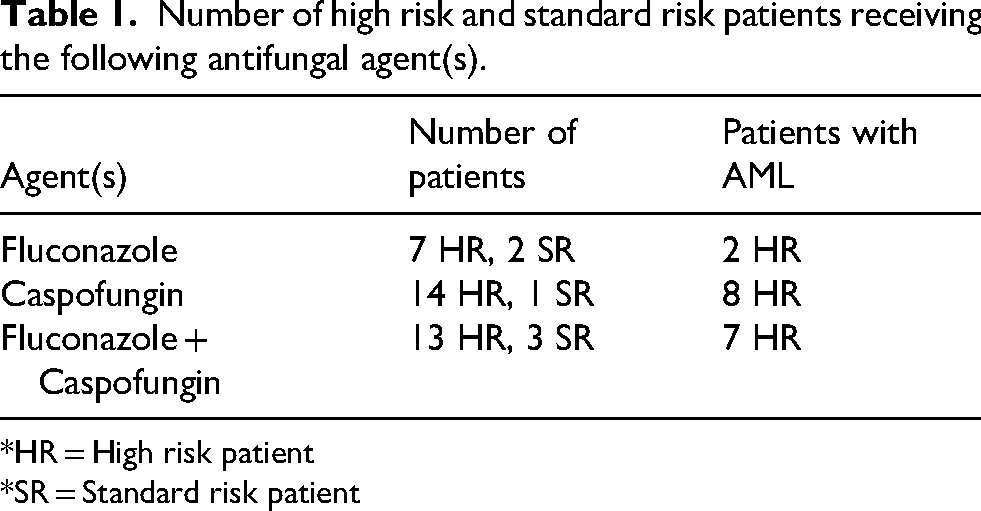
*HR = High risk patient
*SR = Standard risk patient
Number of high risk and standard risk patients receiving the antibacterial prophylaxis with Levofloxacin vs no antibacterial prophylaxis.
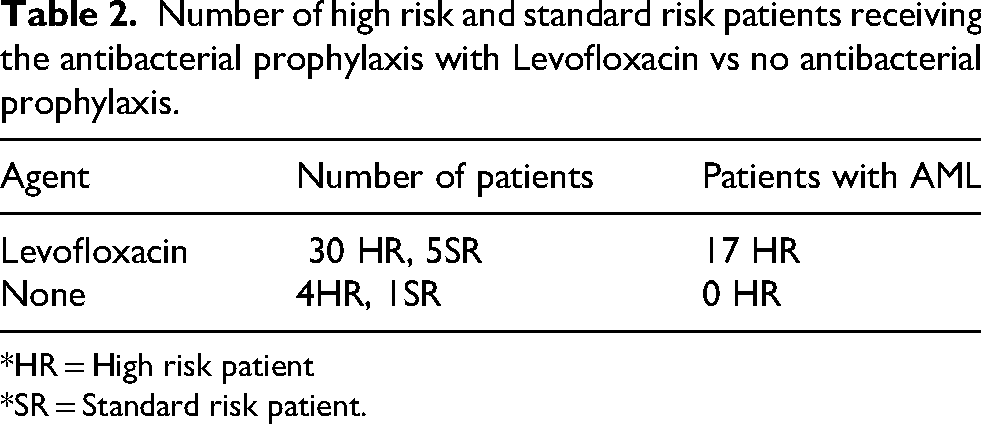
*HR = High risk patient
*SR = Standard risk patient.
The two groups were further separated based on whether they were deemed to be standard risk (SR) or high risk (HR). HR ALL patients were defined according to NCI classification as WBC greater than 50.0 × 109 cells/L at time of diagnosis, 10 years of age and older, or a diagnosis of, HR B cell ALL, T cell ALL and Philadelphia positive ALL. In addition, patients with Down syndrome-associated ALL who are noted to have increased treatment toxicity and infectious complications were included under HR. AML patients were also included as they are at significant risk of infectious death during treatment. As the published evidence supports antimicrobial prophylaxis for patients with AML, we ran separate analysis both with and without AML patients to isolate the effect in ALL patients.
Data was collected from the electronic medical record and clinic charts. Data on age at diagnosis, sex, and diagnosis were recorded for each patient. The number of episodes of FN, bacteremia, and fungemia, were analyzed for each patient. Episodes of FN, bacteremias, and fungemias were assessed between the time frame of diagnosis to the completion of intensive therapy. Bacteremia and fungemia were defined as any positive culture for bacteria or fungus in the blood. If a patient had more than one bacteremia, this was captured as a separate infection (Table 3). Finally, bacterial and fungal infections other than bloodstream infections that developed during therapy were reviewed as well. These measures were then compared between groups, separating based on patients’ leukemia risk, and whether they received antimicrobial prophylaxis or not (Table 4).
Comparison between high-risk leukemia patients receiving prophylactic Levofloxacin, Caspofungin/ Fluconazole, or both and their matched control that did not receive prophylaxis, for episodes of febrile neutropenia, bacteremia, and fungemia data.
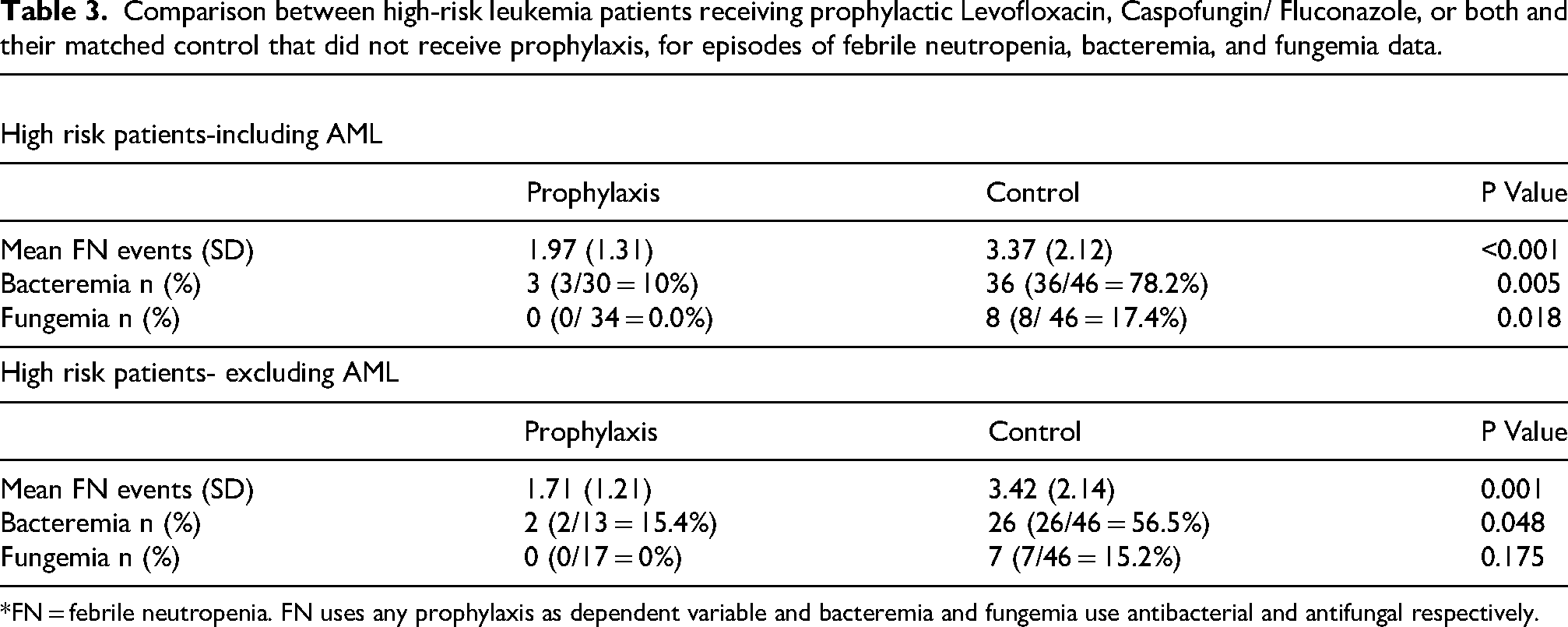
*FN = febrile neutropenia. FN uses any prophylaxis as dependent variable and bacteremia and fungemia use antibacterial and antifungal respectively.
Patients separated by risk and prophylaxis vs control and the infections experienced.

Statistical analysis
Statistical analysis was performed using SPSS version 28.0. Mann-Whitney and Chi-squared tests were performed with a p-value of ≤0.05 was considered significant. When analyzing the effect of prophylaxis on infections, whether patients received antibacterial prophylaxis was used as the grouping variable for bacterial infections and antifungal prophylaxis was used for fungal infections. FN events were grouped based on whether they received any prophylaxis (antibacterial or antifungal).
Results
Of the charts reviewed, 104 patients met the inclusion criteria of having leukemia in the past 20 years, presented in Table 5. In the control group (n = 64), 46 patients were HR and 18 were SR. In the prophylaxis group (n = 40), 34 patients were HR and 6 were SR. In the HR prophylaxis group, all patients received at least one antifungal (n = 34), and 30 patients received Levofloxacin (Tables 1,2).
Mean age, sex, diagnosis, and disease risk in patients receiving prophylaxis compared to patients who did not receive prophylaxis.
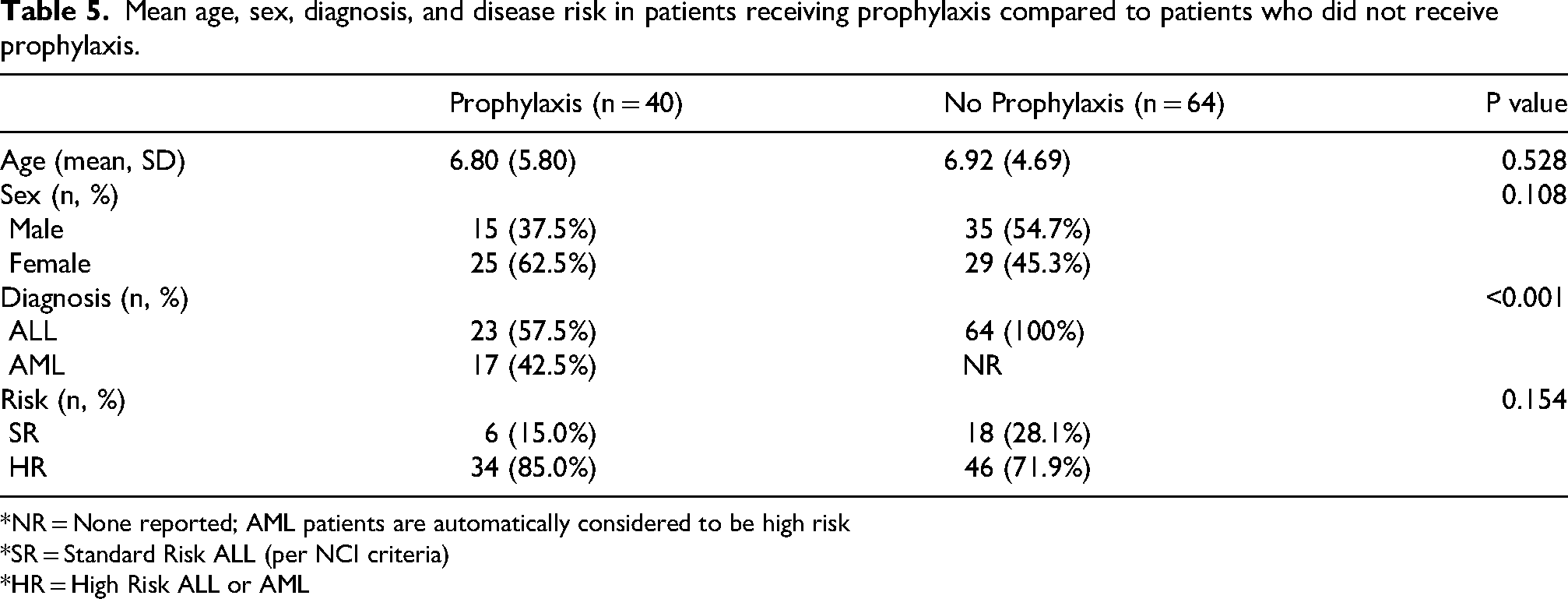
*NR = None reported; AML patients are automatically considered to be high risk
*SR = Standard Risk ALL (per NCI criteria)
*HR = High Risk ALL or AML
Febrile neutropenia
As presented in Table 3, the mean number of FN episodes, bacteremias, and fungemias between HR patients who received prophylaxis were compared to the control group. When including AML patients, the frequency of FN episodes in the prophylaxis group (1.97
Blood-borne infections
In terms of bacteremias, in the HR risk group when including AML patients, 3/30 (10%) patients on antibacterial prophylaxis had an infection, whereas 36/46 (78%) patients in the control group had an infection (p = 0.005). When AML patients were excluded from the HR group, the results remained significant where 2/13 (15.4%) patients on antibacterial prophylaxis had an infection, whereas 26/46 (57%) patients not on antibacterial prophylaxis had an infection (p = 0.048). In terms of fungemias, when including AML patients in the HR group, patients on antifungal prophylaxis had significantly fewer episodes of fungemia: 0/34 (0%), whereas 8/46 (17%) patients in the control group episodes of fungemias (p = 0.018). When excluding AML patients, the results were no longer statistically significant for HR patients whereby, 0/17 (0%) patients on antifungal prophylaxis had an infection, and 7/46 (15%) patients in the control group had an infection (p = 0.175). In comparison, the SR prophylaxis group did not show significant differences for fungemias (p = 0.187), but it was significant for bacteremias (p = 0.006). The majority of bacteremias in patients within the control group were equally split between gram-positive and gram-negative bacteria, whereas patients receiving prophylaxis were mainly infected with gram-negative bacteria. None of the control or prophylaxis patients in the study were resistant to levofloxacin. The fungal infections captured in this study were aspergilla and candida species. It is also important to note that all blood-borne infections occurred after the onset of antimicrobial prophylaxis. In addition to blood-borne infections, there were some other infections noted in the control and prophylaxis group patients. Notably, among HR patients, there were 12 cases of Clostridium difficile reported in the prophylaxis group and 24 in the control group (Table 4). Rates were therefore not higher in the prophylaxis cohort.
Adverse events
In patients who received antifungal prophylaxis with Fluconazole, 2 patients (5.1%) experienced mild elevated liver transaminases. There were no adverse events attributed to levofloxacin or caspofungin prophylaxis. None of the patients in this cohort had to discontinue antimicrobial prophylaxis due to an adverse event. None of the bacterial isolates cultured from prophylaxis patients showed resistance to levofloxacin.
Discussion
The literature supports the use of fluoroquinolones and caspofungin prophylaxis in patients with AML for the prevention of FN, bacteremia, and fungemia, however; there is a paucity of information in de novo ALL patients.9,10 Our study therefore extended the sample population beyond AML to include children diagnosed with; T-ALL, B-ALL, AML, biphenotypic acute leukemia, Down-syndrome ALL, relapsed ALL, and Philadelphia positive ALL.
The evidence for antimicrobial prophylaxis is varies by leukemia subtype. For example, in patients with AML, antibacterial and antifungal prophylaxis is considered standard of care due to high rates of infectious complications.5,6 In contrast, recommendations for de novo ALL, remain inconsistent, with limited prospective data to guide current practices.8,12 In our historical matched chart review, antimicrobial prophylaxis in HR ALL group was associated with significantly fewer episodes of FN and bacteremia. Notably, among the HR ALL patients who received antifungal prophylaxis, there were no episodes of fungemia. Although this reduction did not reach statistical significance when excluding AML patients, the absence of fungal bloodstream infections in the prophylaxis groups remains clinically meaningful given the small sample size.
Our finding of a predominance of gram-negative bacteremia in the prophylaxis group is consistent with trends in pediatric oncology, where levofloxacin prophylaxis may shift the microbial profile of bloodstream infections. 6 Importantly, our local microbiology data showed no evidence of fluoroquinolone resistance within the gram-negative isolates, consistent with reports that levofloxacin prophylaxis in this setting does not substantially drive resistance when used appropriately. 12 In addition, the rate of Clostridium difficile infections was not higher in the prophylaxis group, addressing a common safety concern associated with antibacterial prophylaxis in this vulnerable population.
The majority of studies analyzing the efficacy of antibacterial and antifungal prophylaxis have specifically focused on HR pediatric patients with leukemia specifically relapsed ALL patients, and AML patients.8,10,13 By including both HR and SR patients in our analysis, we contribute real-world data from a broader leukemia population. In our cohort, we had 6 SR patients who received both antifungal and antibiotic prophylaxis. Due to the small sample size, we found no differences between the SR prophylaxis group and SR non-prophylaxis group in the episodes of FN, and fungemias, but was significant for bacteremias.
This study has several limitations. As a retrospective chart review, some infections may not have been documented. Additionally, we were unable to perform statistical analysis on pulmonary fungal infections. Given that Aspergillus is the predominant fungus causing pneumonia in our population, and both caspofungin and fluconazole have limited activity against molds,14,15 the true impact of antifungal prophylaxis on pulmonary fungal disease may be underrepresented.
While levofloxacin and caspofungin are not formally approved for prophylaxis in children in many jurisdictions, their use is supported by existing literature in high-risk hematologic populations.9,10 Levofloxacin was well-tolerated in our cohort, with no observed musculoskeletal adverse events. Nevertheless, we acknowledge concerns regarding long-term cartilage toxicity,13,15 and off-label use and emphasize the need for ongoing pharmacovigilance and larger safety studies.
Our findings support the implementation of antifungal and antibiotic prophylaxis in certain high-risk pediatric leukemia patients during intensive phases of chemotherapy treatment in order to prevent treatment related morbidities and life-threatening infections. Our data also shows that antimicrobial prophylaxis reduces hospital admissions by reducing the incidence of febrile neutropenia and serious infections in immunocompromised individuals without increasing resistance.
Supplemental Material
sj-docx-1-opp-10.1177_10781552251388113 - Supplemental material for Antimicrobial prophylaxis in pediatric patients with leukemia: Reducing incidence of febrile neutropenia episodes and bloodborne infections
Supplemental material, sj-docx-1-opp-10.1177_10781552251388113 for Antimicrobial prophylaxis in pediatric patients with leukemia: Reducing incidence of febrile neutropenia episodes and bloodborne infections by Rachel Creighton, Zara Forbrigger, Jeannette Comeau, Stephanie Villeneuve, Ketan Kulkarni and Tamara MacDonald in Journal of Oncology Pharmacy Practice
Footnotes
Author contributions
RC: collected all data, wrote the draft, made all tables and figures, applied corrections for final manuscript. TM: designed the study, edited the manuscript. ZF: ran the analysis, edited the manuscript. JC: edited the manuscript. SV: edited the manuscript. KK: designed the study, gave guidance on all steps of the study, edited the manuscript.
Declaration of interest
The authors report there are no competing interests to declare
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
