Abstract
Objectives
Dose banding has been introduced to prevent waste caused by cancellation of the chemotherapeutic agents paclitaxel, carboplatin, docetaxel, gemcitabine, irinotecan and oxaliplatin. This could enhance the interchangeability of reconstituted chemotherapy, improving sustainability and cost-efficiency in healthcare. The aim of this project is to evaluate the impact of dose banding on the increase of reissuing admixtures and reduction of medication waste.
Methods
Dose bands were created, allowing a maximum deviation of 10% from the prescribed dose. These dose rounding rules were implemented in Hix 6.2 (ChipSoft BV). Drug waste (already prepared admixtures) and reissuing of admixtures were compared one year before and after implementation. Admixtures eligible for reissuing were identified, and reissued preparations were quantified as percentages of total eligible treatments. Economic benefits were calculated using product, material and preparation costs.
Results
Post-implementation, reissuing increased for all chemotherapies except carboplatin. The largest improvement was seen for oxaliplatin, where reissuing increased from 3.7% to 27.8%, which is an absolute increase of reissuing from 1 to 5 admixtures. Waste was reduced for all agents except carboplatin, with paclitaxel 80 mg/m2 showing the greatest reduction, an absolute reduction of already prepared admixtures from 90 to 23. The amount of waste was significantly reduced, resulting in yearly cost savings of approximately €5,200.
Conclusions
Dose banding enhances the reissuing of chemotherapeutic agents by increasing interchangeability. Broader implementation of this straightforward procedure could further improve reissuing and reduction of waste in chemotherapy preparation.
Key message
Introduction
In recent years, medication waste has become a significant concern in both primary and secondary care. At our large teaching hospital, over 20,000 unused doses of medication, including chemotherapy, are returned to the hospital pharmacy each month. 1 This growing issue has led to an increased awareness among healthcare providers about the need to combat medication waste.
Hospital pharmacists and providers develop initiatives to reduce waste and improve sustainability. Particularly in oncology, where costs of chemotherapy, immunotherapy, and oral oncolytic drugs are high, these efforts are worthwhile. At Isala Hospital, we treat around 5,200 oncology patients each year, which results in a large number of admixtures of chemotherapy and immunotherapy and thus administrations. Strategies like pooling, where a single vial is used for multiple patients, and clustering, in which admixtures of the same drug are prepared for multiple patients, are implemented in daily routine to minimize waste and improve resource efficiency.
In the Netherlands, several progressive initiatives have been introduced to address the challenges of giving patients optimal treatment and at the same time to do it in a sustainable way. For expensive immunotherapies like nivolumab and pembrolizumab, dose bands and fixed dosing regimens have been recommended to standardize treatment and reduce waste. 2 In addition, lower dosing options for drugs like pembrolizumab have been explored as potential cost-saving alternatives without compromising efficacy.2-5 Moreover, in the field of oral oncolytic drugs, therapeutic drug monitoring is a strategy to optimize drug exposure by adjusting doses based on individual pharmacokinetic profiles. Studies, such as those examining the pharmacokinetics of cobicistat boosted olaparib demonstrates the potential for personalized dosing strategies that reduce unnecessary drug exposure and costs. 6
These initiatives aim to reduce medication use, and thereby costs, by safely adjusting off-label dosages based on pharmacokinetic exposure. For classic, off-label, chemotherapy the focus is not on cost reduction but on the environmental aspect. In Table 1 an overview is given of comparable initiatives.7-15 The main difference between these initiatives and our study is the use of dose banding with a deviation of up to ±10% instead of ±5% of the calculated dose. Only one other study (Puisset et al.) applied a deviation of 10%, however, they only studied paclitaxel. 9 Additionally, in our study, we specifically measured the extent of chemotherapy reuse, whereas other studies tend to suggest that more reuse is theoretically possible, but do not measure this explicitly. Results of those studies were more focused on waste reduction.
Overview of all research done for dose banding.

Search terms: dose bands AND chemotherapy AND oncology
BSA: body surface area.
Aligned with the European Green Deal and sustainability efforts, there is growing interest in reusing unused medications. 16 In the case of chemotherapy, there is a risk that already reconstituted chemotherapy treatments are postponed or the dose is reduced due to the clinical status of the patient. Refinement of the process is necessary, as reissuing of the admixtures is hampered by the fact that most chemotherapy is dosed on body surface area (BSA), resulting in unique dosages for individual patients. This specific way of dosing opposes the interchangeability of chemotherapy agents as the likelihood of finding a patient with the same BSA is small. The introduction of standardized dose bands, based on BSA ranges, has been suggested to minimize medication waste and improve reissuing of chemotherapy agents.9,13
Through this research, we aim to demonstrate the added value of dose banding in reducing medication waste and increase reissuing of admixtures. Dose banding for immunotherapy is already common practice in our hospital. However, this approach had not yet been adopted for classic chemotherapeutic agents. Therefore, we initiated this study to examine the impact of introducing this new method. This study evaluates the effects on reissuing after the introduction of dose banding for classic chemotherapy at our large tertiary, training hospital in The Netherlands (Isala hospital, Zwolle)
Method
Design
At Isala Hospital, all different forms of cancers with both curative and palliative intent, with the exception of head and neck cancer, testicular cancer and sarcoma treated with curative intent. The most commonly treated cancers are breast- and lung cancer.
For this implementation study dose bands were created for all adult patients that were treated with one of the six most used chemotherapeutic agents of this hospital: paclitaxel 175 mg/m2 and 80 mg/m2, carboplatin, docetaxel, gemcitabine, irinotecan and oxaliplatin, irrespective of treatment intent. In children, dose banding was not applied. Introduction of these dose bands was considered standard of care in our hospital and as such was implemented as routine clinical care and not in a research setting, in addition to already existing strategies (see also Figure 1).
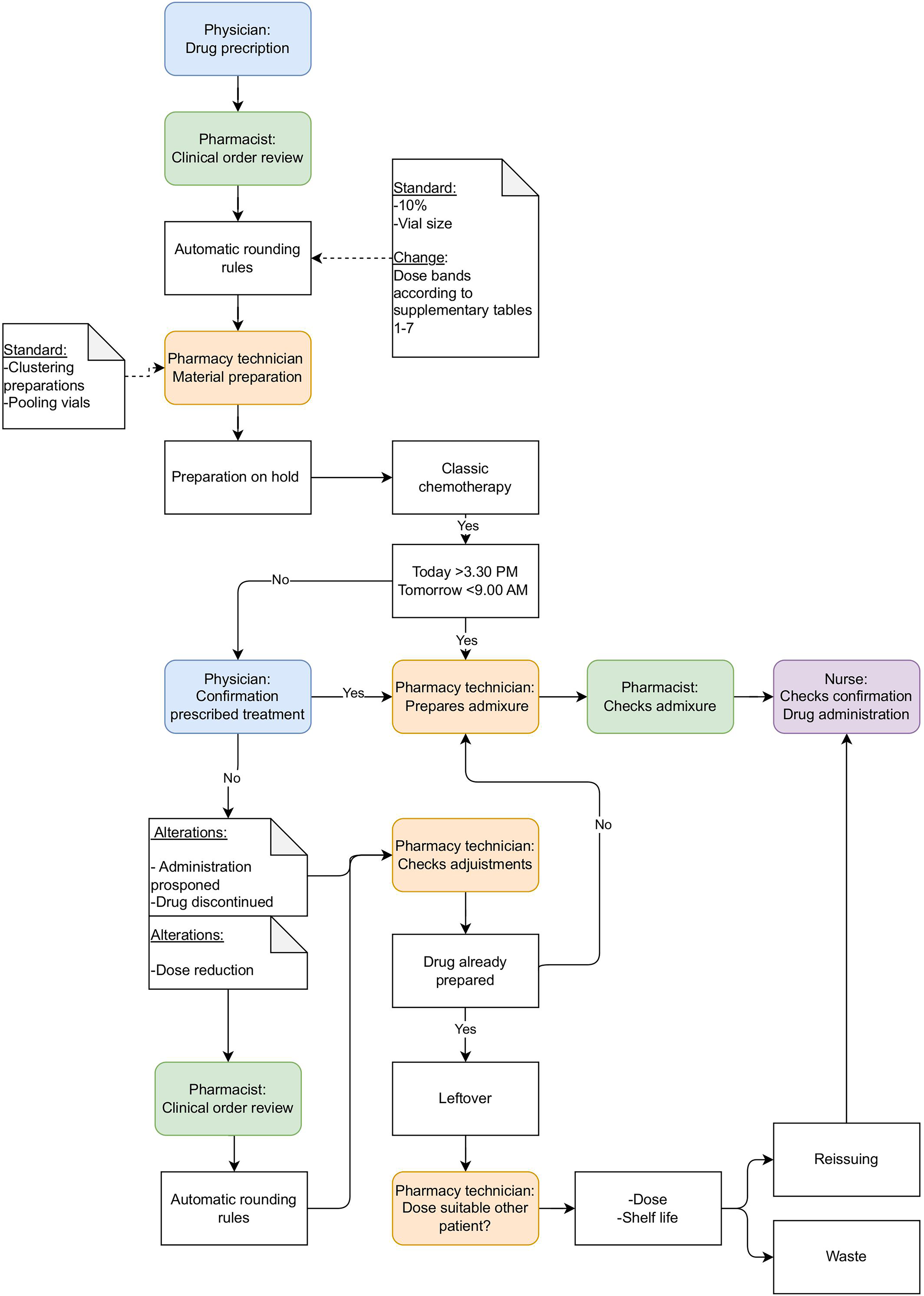
Chemotherapy workflow.
Dose bands were created based on the current rounding rules (Supplementary Table 2–8), vial size, and most prescribed dosages based on BSA. In consultation with the prescribers, the maximum deviation for dose rounding rules was already set at +/- 10% of the prescribed dose, if standard dosing based on BSA would have been used. Rounding of biologic and cytotoxic anticancer agents within 10% of the ordered dose is designated as acceptable for routine clinical care by the Hematology/Oncology Pharmacy Association in their 2017 position statement concerning dose rounding of biologic and cytotoxic anticancer agents. 17 Dose changes ≤10% are not expected to reduce the safety or effectiveness of therapy.17-20 For extreme BSAs on the lower and upper end no dose bands were created.
The dose bands were implemented in the drug preparation software (Hix 6.2, ChipSoft BV). When a dose was ordered by the physician, the nearest dose band was automatically chosen. Rounding was performed by the hospital pharmacist, who ensures the correct dose adjustment and adherence to the dose bands. For dosages falling outside these dose bands, the current default rounding rules were used, resulting in a unique dose based on BSA (excluded from this analysis). Dose calculation for carboplatin was based on the Calvert formula. 21 In Figure 1 we present the workflow of chemotherapy prescribing, preparation and administration in our hospital.
Reissuing of the drugs was compared the year before and the year after the implementation of dose bands. Dose bands were implemented at 01/04/2022. For these therapeutic agents the periods 01/04/2021–31/03/2022 and 01/04/2022–31/03/2023 were analyzed. Data related to administrations, such as the number of preparations, leftovers, unused and reissued admixtures were extracted from the drug preparation software (HIX6.2, Chipsoft BV).
A distinction is made between leftovers and waste. The following definitions were used:
Leftovers: This refers to chemotherapy preparations that were ready for administration but could not be administered because the patient was unable to receive treatment.
Waste: No suitable patient is found for reissuing, the leftover chemotherapy must be discarded and is then classified as waste.
Leftovers are already prepared, but unused admixtures. These admixtures can be reissued within the shelf life after reconstitution. Information from the SmPC was used to determine shelf life, supplemented with data from Stabilis (Supplementary Table 9). Reissued administrations are admixtures that are made for patient A, but administered to patient B. If there is no patient to whom the admixture can be administered to, the admixture is considered waste. The percentages of leftovers were based on the total number of admixtures of that particular drug. The percentages of reissuing were calculated based on the number of reissued admixtures and waste divided by the total of leftovers (Table 2). Increase of reissuing is calculated by division of the percentages reissued and wasted before and after dose banding.
Comparison of dosage variability and compliance with dose bands before and after implementing dose bands.

Data on costs were collected. For the calculation of the product costs of the chemotherapy agents the cheapest vial was used, based on purchasing costs. The medication and material costs used for the calculations in this article are shown in Supplementary Table 1. Finally, the preparation costs were calculated using standard costs of € 57,23 per treatment, which are the standard costs charged in the hospital for the preparation of chemotherapy by the pharmacy. Total costs are calculated and shown in Table 3.
Overview of all reissued and wasted treatments; one year before implementing dose bands compared to one year after implementing dose bands.
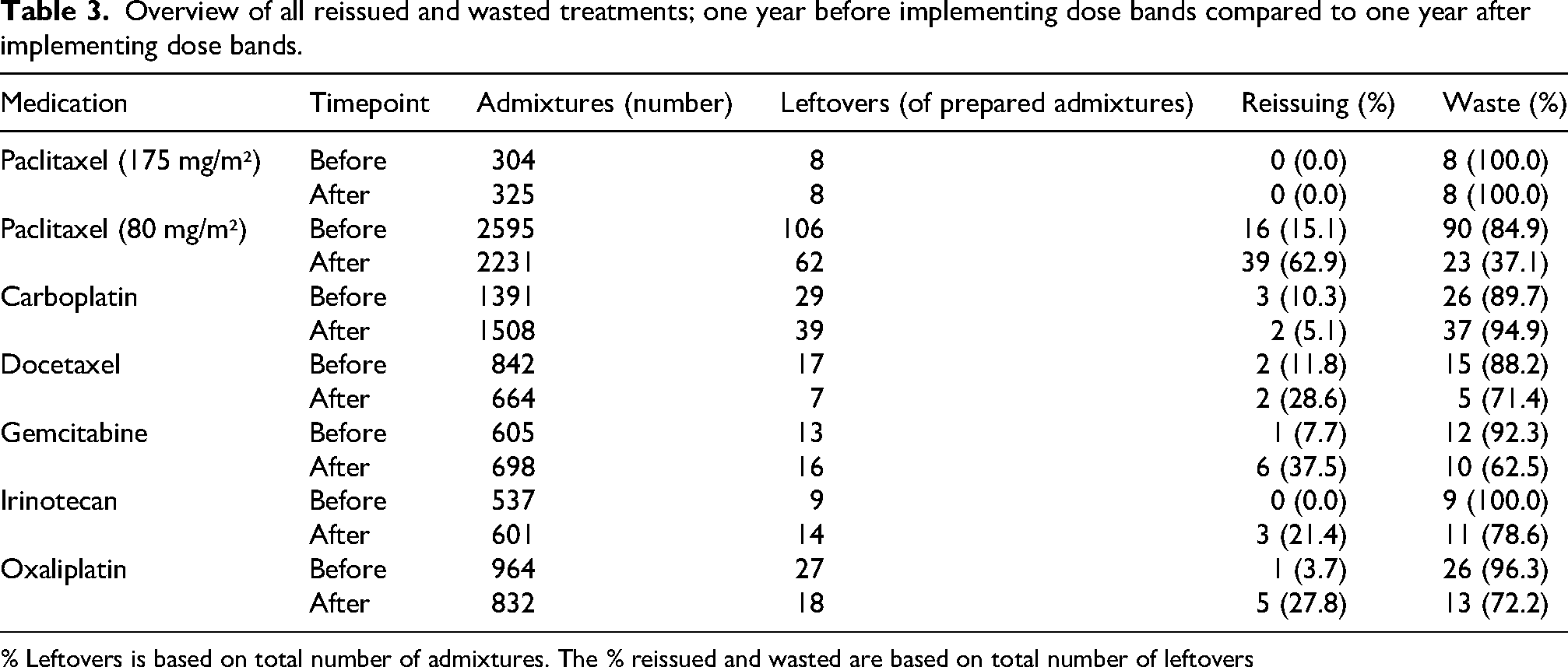
% Leftovers is based on total number of admixtures. The % reissued and wasted are based on total number of leftovers
Data of waste, reissuing and costs was analyzed using Microsoft Excel.
Results
Dose bands
The implementation of dose bands led to a change in the number of different dosages for the chemotherapy agents, mostly as expected a decrease. For example, paclitaxel 175 mg/m2 reduced from 41 to 27 different dosages and 80 mg/m2 from 73 to 34 dosages. Only for gemcitabine and irinotecan the number of different dosages stayed the same, respectively 12 and 29 dose bands. For all chemotherapy agents, more than 90% of the doses fell within the defined dose bands. These results are summarized in Table 2.
After the implementation of dose bands, reissuing increased for all chemotherapy agents, except for paclitaxel (175 mg/m2), for which there was no reuse before and after dose banding, and for carboplatin, for which there was a decrease (from 3 to 2) in reissuing (Table 3). Most reissuing was possible for paclitaxel (80 mg/m2) admixtures. Before dose banding 16 admixtures were reissued and after dose banding 39 admixtures were reissued, an increase from 15.1% to 62.9% (Table 3). Gemcitabine had an increased reissuing of 7.7% to 37.5% (1 to 6) and oxaliplatin an reissuing of 3.7% to 27.8% (1 to 5) (Table 3).
Before dose banding, paclitaxel (80 mg/m2) showed a waste of 90 admixtures (84.9%) and after dose banding only 23 admixtures (37.1%) were counted as waste (Table 3). However, not every chemotherapy showed reduction in waste. Paclitaxel (175 mg/m2) showed no difference in waste (8 admixtures before and after dose banding) and carboplatin showed a small increase in waste from 26 admixtures (89.7%) to 37 admixtures (94.9%) (Figure 2A/2B, Table 3).
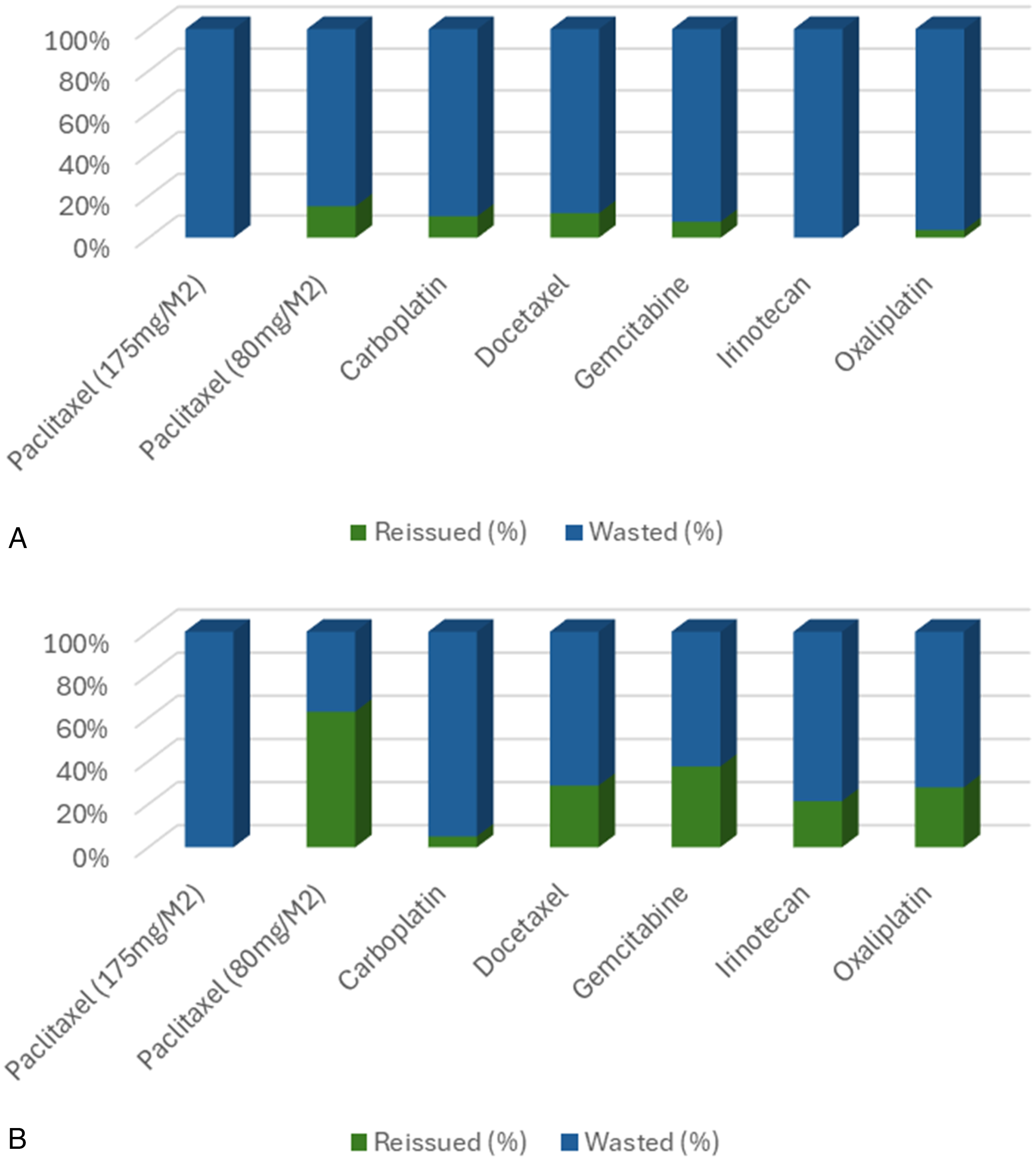
A+B: Percentages reissued and wasted chemotherapy before and after implementing dose bands.
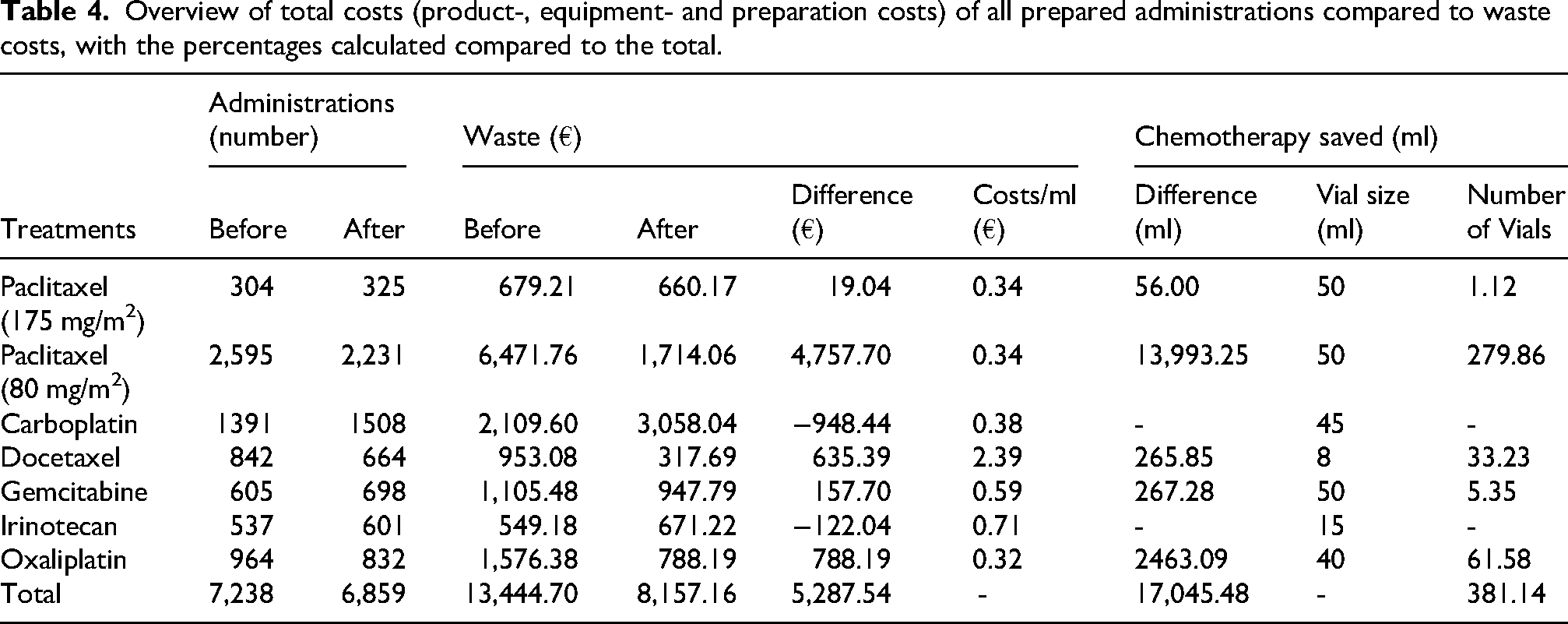
The total cost of all wasted chemotherapies reduced from €13,444.70 before dose banding to €8,157.16 after dose banding. In addition, more than 380 vials of chemotherapy were saved (Table 4). The largest reduction in costs was seen for paclitaxel (80 mg/m2), which had a decrease in costs of more than €4,700 (€6,471.76 to €1,714.06). This represents, for paclitaxel (80 mg/m2) alone, a waste reduction of almost 280 vials (Table 4).
Overview of total costs (product-, equipment- and preparation costs) of all prepared administrations compared to waste costs, with the percentages calculated compared to the total.
Discussion
This report shows promising results for the implementation of dose bands as a method to increase reissuing, and therefore decrease the number of leftovers, of chemotherapy admixtures. For almost every chemotherapy agent we found an increase in reissuing and a decrease in waste after the implementation of dose bands. Previous research, for example studies by Shah et al. and Fahey et al.,22,23 have demonstrated that dose banding for chemotherapeutic admixtures enhances patient safety, through the reduction of medication errors, reduction of costs, and promotion of more efficient working methods. Our approach might also result in reduction of workload through a more efficient working method. However, this was not measured due to the retrospective design.
The best result for minimization of waste was seen for paclitaxel (80 mg/m2), waste decreased from 90 to 23 admixtures. This was also the chemotherapeutic agent which was most frequently prescribed in our analysis. The number of treatments prepared and prescribed, of course, influences how often reissuing can take place. The likelihood of finding a patient within the same dose band, within the appropriate shelf life, is smaller when a chemotherapeutic agent is used less frequently. Smaller differences were seen for the other chemotherapy agents. For example, in the smallest treatment group (paclitaxel 175 mg/m2, 629 total treatments) no treatments could be reissued, compared to 55 treatments (1.1%) for the largest treatment group (paclitaxel 80 mg/m2, 4826 treatments). The highest increase of reissuing, when comparing before and after treatment, was shown for oxaliplatin admixtures (reissuing increased from 1 to 5 admixtures (3.7% to 27.8%) after dose banding)., which is the third largest group of admixtures.
Carboplatin has small numbers of leftovers. This is explained by the workflow of confirming carboplatin. Physicians often wait for the newest creatinine value to be determined, before confirmation of the dose. Therefore, carboplatin is often made after conformation, and thus only a few admixtures are made in advance. The low number of reissuing for carboplatin could be explained by the number of dose bands in the carboplatin group (≈70 compared to 8 for paclitaxel 80 mg/m2). Carboplatin doses are calculated using the Calvert formula combined with target area under the curves (AUC). 21 To stay within the 10% deviation, it is not possible to have less than ≈70 dose bands for carboplatin.
Based on the results of this study, we do not recommend the implementation of dose banding for carboplatin. Although the number of dose bands significantly reduced (from 100 to 69), this reduction, presumably due to dosing calculations using the Calvert formula, does not lead to an increase in reissuing and decrease in waste for this chemotherapy. In contrast to our findings, Kaestner et al. found positive effects of the introduction of dose bands for carboplatin. 24 However, differences in strategy regarding the target AUC, renal function ranges, maximum deviation in calculated dose, and overall dosing range probably explain the varying findings on the interchangeability of carboplatin.
The aim of implementing this new dosing strategy is to reduce the amount of leftover chemotherapy preparations. For gemcitabine and irinotecan, the absolute number of leftovers increased (Table 3). However, a larger proportion of the prepared doses were reused rather than discarded. Although the absolute amount of waste seems to have grown, the relative reuse of preparations increased significantly. This is partly due to a higher number of doses being prepared in the second year compared to the first. For gemcitabine, reuse rose from 7.7% to 37.5%, and for irinotecan from 0.0% to 21.4% (Table 3).
The saved costs, around €5,200 euros seem relatively small, however this is caused by the fact that in the Netherlands these are all generic off patent drugs. The impact of the number of vials, saving 381 vials, is significant. Depending on the medication prices in different countries this could have a larger financial impact. 25
This study was conducted in a tertiary hospital. To implement dose banding in other centers, it is important to consider several factors that may limit the generalizability.
First, a sufficient patient volume is essential to achieve the benefits of dose banding. The more patients there are, the greater the chance of reuse and, consequently, the reduction of waste. In addition, the hospital's workflow also plays a role. The time of preparation of the admixture in relation to the confirmation of treatment by the physician (see Figure 1), is of relevance and has impact on the leftovers that are unnecessarily prepared.
In conclusion, introduction of dose bands increases reissuing of chemotherapy by increasing interchangeability. We show that this approach ensures less medicine wastage, thereby reducing healthcare costs.
Supplemental Material
sj-docx-1-opp-10.1177_10781552251370560 - Supplemental material for Optimizing chemotherapy practices: The impact of dose banding on reissuing and waste reduction
Supplemental material, sj-docx-1-opp-10.1177_10781552251370560 for Optimizing chemotherapy practices: The impact of dose banding on reissuing and waste reduction by Alida Geertruida Catharina van Huët, Helle-Brit Fiebrich-Westra, Peder Nygård and Elise Joëlle Smolders in Journal of Oncology Pharmacy Practice
Footnotes
Patient consent for publication
Not applicable. No patient data were used. Only dosages prescribed were used when patient did not opt out for research.
Contributor statement
AH, HF, and PN were responsible for designing the study. AH collected the data and performed the analyses. AH, HF, and ES interpreted the results. AH drafted the initial manuscript, while HF and ES contributed to its revision and critical evaluation. All authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that supports the findings of this study are not available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
