Abstract
Introduction
This study aimed to reduce hypoglycemia by 25% in patients receiving intravenous insulin therapy for hyperkalemia through a new multimodal treatment order set. Historically, the hypoglycemia rate with the previous single-agent order set was 12.1%. Given the high-risk nature of the oncology population, even one episode of hypoglycemia can increase morbidity and negatively impact clinical outcomes.
Methods
Between October 1st and November 30th, 2023, educational materials on the new order set were presented to advanced practice practitioners, hospital physician leadership, and nursing groups. Postintervention data were collected retrospectively from December 1st, 2023, to January 22nd, 2024, using electronic health records of patients who received intravenous insulin and dextrose 50% (D50%) per the new order set. A manual chart review assessed compliance and the rate of hypoglycemia (blood glucose <70 mg/dL). Insulin dosing compliance followed weight-based dosing (0.1 units/kg, max 10 units), and D50% dosing was based on baseline point-of-care (POC) BG levels (25G if POC BG 140-250 mg/dL and 50G if <140 mg/dL).
Results
Of 125 encounters reviewed, 91 (73%) received intravenous insulin. The hypoglycemia rate was 6%, with no severe hypoglycemia (<40 mg/dL). The median baseline POC BG, creatinine clearance, and patient weight were 132.5 mg/dL, 51 mL/min, and 73 kg, respectively. All insulin doses were compliant, and 81% of D50% doses adhered to guidelines.
Conclusion
The new multimodal treatment order set led to a 45.5% reduction in hypoglycemia. Provider education and standardized protocols were key in enhancing patient safety during hyperkalemia management.
Background
Hyperkalemia, defined as potassium levels greater than 5.1 mEq/L, is often asymptomatic but can also lead to life-threatening cardiac abnormalities. Many medications are used to treat hyperkalemia, with insulin being a common choice in hospitalized patients. Intravenous insulin administration can rapidly correct hyperkalemia by shifting potassium intracellularly, but carries the inherent risk of hypoglycemia. The risk of hypoglycemia (<70 mg/dL) and severe hypoglycemia (<40 mg/dL) is associated with increased morbidity and mortality.1,2 This risk can be heightened in hospitalized or critically ill patients, as these patients are more likely to have risk factors of hypoglycemia, which can lead to severe hypoglycemia with insulin treatment. 3 Key patient-specific risk factors of hypoglycemia include a low baseline BG, renal impairment, and no history of diabetes mellitus.4,5 Current strategies to mitigate these risks may include measuring baseline BG, implementing weight-based insulin dosing, and monitoring post-insulin point-of-care (POC) BG. 6 In a single-center retrospective study comparing the effects of standard flat insulin doses of 10 units to weight-based insulin dosing at 0.1 units per kg body weight with a maximum of 10 units, the weight-based approach reduced hypoglycemic events by 15.2%. 6 In March 2019, an inpatient order set was implemented at the University of Texas MD Anderson Cancer Center (MDACC) for management of hyperkalemia, which included a standard intravenous insulin dose of 10 units with a linked 25-gram dextrose 50% (D50%) order as a single treatment option. In February of 2022, additional updates were made to this order set based on reported safety events due to hypoglycemia with the use of this order set. These updates included the addition of weight-based (0.1 units/kg) intravenous regular insulin with a maximum dose of 10 units per treatment, along with guidance to the ordering provider on key risk factors for patients at higher risk of hypoglycemia. Higher doses of D50% were also added for patients with lower baseline POC BG levels. Despite these updates, reported incidents of hypoglycemia-related safety events continued to occur, and rates of hypoglycemia from hyperkalemia treatment with insulin were 12.1%, with 20% of the hypoglycemia patients experiencing severe hypoglycemia. After various iterations of hyperkalemia treatment order sets, it was recognized that creation of a multimodal hyperkalemia treatment order set was needed to offer providers treatment options other than insulin in patients with high risk of hypoglycemia. Strategies to prevent hypoglycemia included the development of a multimodal hyperkalemia treatment order set and provider education to key stakeholders prior to order set implementation. For this reason, this quality improvement project's intervention incorporated education to key stakeholders ahead of the go-live date for the new multimodal inpatient order set for the treatment of hyperkalemia (Table 1). We aimed to decrease the hypoglycemia rate in patients who received insulin by at least 25% through the new order set by February 2024.
New MDACC Institutional Order Set for ICU and Floor Use.

Materials and methods
This project was reviewed and approved by the institution's Quality Improvement Assessment Board (QIAB). Pre-intervention educational materials were created by key stakeholders within the Division of Pharmacy between October 2023 and November 2023. A multimodal treatment approach was added to the new institutional order set consisting of treatment options to shift potassium intracellularly or eliminate potassium (Table 1). Education was targeted toward leadership and providers. Providers included physicians, advanced practice providers (APPs), clinical pharmacy specialists and nurses. Pre-intervention material consisted of live presentations and written communication via emails for further awareness of order set changes. Post-intervention data were collected retrospectively utilizing electronic health records of all patient encounters in which providers selected insulin and D50% through the new institutional hyperkalemia order set from December 1st, 2023, through January 22nd, 2024. Key data points collected included baseline creatinine (Cr), baseline BG, baseline potassium, patient weight, POC BG value immediately before insulin administration, dose of insulin, dose of D50%, post-insulin potassium, and hourly POC BG values for the first four hours after insulin administration. Creatinine clearance (CrCl) was calculated using the baseline patient data (Table 2). A manual chart review was conducted to assess order set compliance, identify if insulin was appropriately administered and determine the rate of hypoglycemia for orders in which insulin and D50% were administered. The rate of hypoglycemia for patients who received insulin from the new hyperkalemia order set was compared to the institution's historical rate of 12.1%. Descriptive statistics were used for analysis in this study.
Baseline characteristics.

*Refers to value at the time hyperkalemia was identified
δRefers to hypoglycemia risk factors
Results
We identified 125 patient encounters in which intravenous insulin was ordered through the new inpatient hyperkalemia order set between December 1st, 2023, and January 22nd, 2024. Table 2 shows baseline characteristics and risk factors for hypoglycemia at baseline for the patients included. Of the 125 encounters, insulin was administered in 91 (73%) encounters. Six (6%) of these 91 encounters resulted in hypoglycemia. This was a 45.5% decrease from the institution's historical baseline of 12.1%. The hypoglycemia group in the previous institutional order set had a median post-insulin POC BG of 95 mg/dL (33–329 mg/dL) as compared to 81 mg/dL (44–188 mg/dL) in the new order set. Table 3 shows the median of each hourly post-insulin POC BG readings. In the hypoglycemic group, the median POC BG was hypoglycemic beginning at hour 2. This persisted through hour 3 before rising above 70 mg/dL in hour 4. The ranges included hypoglycemic events in all four hours monitored. There were no POC BG readings less than 40 mg/dL observed with use of the new order set. The hypoglycemia group had a lower average baseline BG (111 mg/dL) and CrCl (50 mL/min) compared to the no hypoglycemia group BG (162 mg/dL) and CrCl (62 mL/min) (Table 2). Additionally, the hypoglycemia group also had a higher percentage of patients that did not have diabetes compared to the no hypoglycemia group (83% vs 54%). Average baseline potassium level in the hypoglycemia group (5.7 mEq/L) was lower than in the no hypoglycemia group (5.9 mEq/L). Both groups had the same post-insulin average potassium level of 5 mEq/L at follow up (Table 4). Furthermore, the manual chart review determined that 100% of the insulin doses ordered through the order set were compliant. The D50% doses for these encounters, however, showed 81% compliance.
Median hourly post-insulin POC blood glucose readings.

Comparison of baseline and post-insulin potassium levels.

*There were 3 instances in which follow up potassium level was not provided
Discussion
To our knowledge, this is the first study at a major cancer center evaluating the rate of hypoglycemia induced from intravenous insulin treatment for hyperkalemia. Patients with cancer are often frail, with poor nutritional status and many comorbidities that can increase the risk of hypoglycemia. The management of hyperkalemia with insulin at MDACC highlights the critical balance between efficacy and safety in treating potentially life-threatening electrolyte disturbances. The reduction in hypoglycemia rates from 12.1% to 6% aligns with the improvements seen in previous studies that employed similar strategies. The baseline characteristics and identified risk factors in our patient cohort were consistent with those reported in other studies.3,5–7,9 Patients without a history of diabetes, lower baseline BG and compromised renal function, were more susceptible to hypoglycemia. This reinforces the need for vigilant BG monitoring and tailored insulin dosing in at-risk populations.
Our intervention included the development and implementation of a new multimodal order set with a comprehensive educational campaign, which contributed to the improved compliance with subsequent reduction in hypoglycemia rates. This multidisciplinary approach, targeting physicians, APPs, clinical pharmacy specialists, and nurses, aimed to employ a cohesive understanding and application of the updated order set. These adjustments were critical in addressing the primary predictors of hypoglycemia identified in prior studies, particularly the administration of insulin doses greater than 0.1 units/kg and pre-insulin BG levels below 140 mg/dL.6–8 Additionally, we emphasized the importance of early and frequent POC BG monitoring within the first four hours post-insulin administration to promptly identify and manage hypoglycemic events. Although the median POC BG readings appear to be increasing by hour 4, two hypoglycemic readings were still observed. This suggests that BG monitoring beyond 4 h may be warranted for patients receiving insulin, particularly in those at higher risk for hypoglycemia. In the no hypoglycemia group, the number of missing readings increased with each hourly measurement (Table 3), which may have affected the final hourly values and contributed to the lack of difference between the median baseline and post-insulin POC BG readings.
Pre-Insulin POC glucose and dose of D50% given.
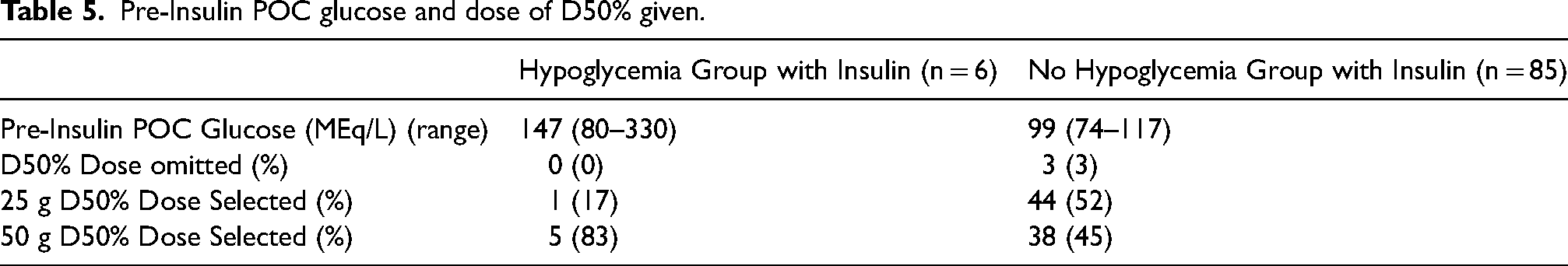
Our study revealed that 6% of insulin administrations still resulted in hypoglycemia, however there were no instances of severe hypoglycemia (BG < 40 mg/dL).However, it is important to note that in the hypoglycemia group's single noncompliant D50% dose, 12.5 g was given instead of the appropriate dose of 50 g based on their pre-insulin POC BG value of 127 mg/dL. In that specific encounter, 25 g was incorrectly selected, but 12.5 g of D50% was given. The residual risk of severe hypoglycemia, along with deviations from the order set's guidance on D50% dosing highlights areas for further education to front-line staff and optimization. This includes considering alternative intravenous insulin dosing strategies for patients with renal dysfunction, post-insulin POC BG follows ups for greater than four hours and identifying potential safety measures in the front-line to ensure the appropriate D50% dose is given8–10
This quality improvement project underscores the necessity of continuous evaluation and iteration of clinical protocols to enhance patient safety. The conclusions of this study are limited by the relatively small patient population. For the 34 encounters in which insulin was ordered but not administered, there is no clear documentation as to why insulin was not given. It is possible that the no insulin group trended towards insulin avoidance because the population was at a higher risk of hypoglycemia. Future efforts should include ongoing provider education and continued assessment of order set compliance to further increase order set compliance rates. Further investigation could be explored in patients who received overlapping doses of insulin and potentially exploring implementing provider alerts. Additionally, expanding the study to include more patients could provide more robust data and facilitate the development of universally applicable guidelines for the safe administration of insulin in hyperkalemia management.
Conclusion
Incorporating education when implementing a new hyperkalemia order set for inpatient use proved effective in decreasing hypoglycemia rates with intravenous insulin administration. With several optimization efforts of the order set and persisting reported safety events, it was determined that incorporating education ahead of the updated order set's implementation would be an impactful intervention. The new hyperkalemia order set offers expanded options for providers in the treatment of hyperkalemia, which allows an opportunity to utilize a different treatment and avoids the use of insulin in patients with an elevated risk of hypoglycemia. The order set also added provider guidance built directly into the order set to highlight risk factors for hypoglycemia. In conclusion, our study highlights the critical role of standardized insulin dosing and vigilant BG monitoring in reducing hypoglycemia risk among hyperkalemic patients. Through continuous improvement and multidisciplinary collaboration, treatment protocols can be optimized to ensure both the efficacy and safety of hyperkalemia management in diverse patient populations.
Footnotes
Author contributions
All authors have made substantial contributions to the design, analysis, and interpretation of this work. All authors have contributed to the drafting, reviewing, and approval of this work and agree to be accountable for all aspects of this work's accuracy and integrity.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
