Abstract
Introduction
Due to the complex needs, paediatric oncology patients require specialized pharmaceutical care provided within an interdisciplinary setting.
Objective
To implement a Pharmaceutical Care Model at the Paediatric-Adolescent Oncology Unit (PAOU) at Sir Anthony Mamo Oncology Centre, Malta, focusing on individualised pharmaceutical care issues (PCIs) based on patient specific needs.
Methods
A tool consisting of a novel PCI classification system based on the Pharmaceutical Care Network Europe Foundation classification for drug related problems and the DOCUMENT system was developed to document and classify PCIs. The tool was presented in tabular format with eleven PCI categories, category description, PCIs, pharmaceutical interventions, and outcomes. Following ethics approval, the pharmacist attended consultant-led ward rounds where patient files, treatment charts and chemotherapy prescriptions were reviewed to identify PCIs. Identified PCIs were discussed with the healthcare team, pharmaceutical interventions were proposed, and outcomes were recorded.
Results
Validation and practicality testing demonstrated tool relevance, robustness, and practicality for use at the PAOU. Over 8 months, 545 PCIs were identified during 325 pharmaceutical care sessions. Most PCIs featured in the counselling (26.97%), drug selection (23.67%), dose selection (19.27%) and monitoring (15.41%) categories. The majority (95% n = 516) of the proposed pharmaceutical interventions were accepted and implemented by the healthcare professionals or the parents.
Conclusion
This study highlights the pharmacist's contribution to paediatric oncology care through a pharmaceutical care model focused on PCIs and patient specific needs. The high acceptance rate of pharmaceutical interventions demonstrates the model's quality, the clinical pharmacist's professional competence, effective interdisciplinary collaboration, and integration within the healthcare team.
Introduction
The pharmacotherapeutic management of cancer represents a high-risk treatment modality, often comprising of complex medication regimens that are fraught with a multitude of patient safety issues, including serious medication errors.1–4 The consequences of these safety issues may pose serious health risks to the patients with substantial morbidity and mortality.5–7
Children and adolescents suffering from cancer are a vulnerable patient group with special pharmaceutical care needs.8,9 Drug therapy and disease management of cancer in this patient cohort is challenging for several reasons. They exhibit the typical attributes of cancer patients, necessitating the administration of multiple medications including chemotherapy, supportive care agents and medications for the management of co-morbidities. 9 They also possess the inherent characteristics of paediatric patients,2,9–11 including distinctive pharmacokinetics and pharmacodynamics that are affected by substantial variations in size from infancy to adolescence and the associated developing physiological processes.12,13 In the child suffering from cancer, the distinguishing pharmacological characteristics result in varying levels of exposures and clearance of chemotherapy and associated medications and hence in difficulty to predict efficacy and toxicity. They also lead to the need for frequent computation of individualised dosages and manipulation of drug formulations to meet the need for smaller doses.9,10,14 A significant proportion of drugs is often administered off-label, implying less clear guidance on dosing, potential side effects and contraindications, which may jeopardise medication safety for the children.10,15,16 The overall medication management process from selection and prescribing to monitoring, evaluation and education is complex in this population. 1
When a pharmaceutical care model is in place, clinical pharmacists work in collaboration with the patient and the patient's healthcare professionals (HCPs) to design, implement and monitor a pharmacotherapeutic plan which is tailor-made to each individual patient's requirements and optimizes patient's therapeutic outcomes. 17 In paediatric oncology, the therapeutic outcomes are to cure a patient's disease, if possible, to eliminate or reduce patient's symptoms, to arrest or slow disease progression and to prevent disease or its symptoms namely mortality, adverse effects, severe organ toxicity and drug resistance. 6 In oncology, some adverse effects require dose reductions and could even necessitate drug interruption. 16 This implies that therapeutic success strongly relates to the extent of therapy-associated toxicity. 16 Clinical pharmacists play a crucial role in ensuring that drugs are utilised to their maximum therapeutic efficacy while minimising severe adverse effects.6,18–23
Problems identified by the clinical pharmacist during the provision of pharmaceutical care are referred to as pharmaceutical care issues (PCIs).24,25 PCIs cover a wider spectrum of care issues when compared to drug-related problems (DRPs). DRPs are defined as events or circumstances involving drug therapy that actually or potentially interfere with desired health outcomes.13,23,26 PCIs include potential and actual DRPs as well as patients’ need for information, support and other pharmaceutical services. 25 Development of an optimum pharmacotherapeutic plan involves the clinical pharmacist making an assessment of the paediatric patient's drug related needs, identifying PCIs, proposing ways to solve and prevent the identified PCIs, and making follow-ups and medication monitoring to evaluate outcomes. 27 Subsequently, this ensures the safe and optimum use of medications, improving or maintaining the paediatric patient's health-related quality of life.6,28–32 The aim of the study was to implement a Pharmaceutical Care Model at a Paediatric-Adolescent Cancer Unit at an oncology hospital setting.
Methods
Settings and patients
The research was implemented at a pilot stage for a period of 8 months at the Paediatric-Adolescent Oncology Unit (PAOU) at Sir Anthony Mamo Oncology Centre (SAMOC), Malta. The PAOU is a 14-bed ward specialising in the treatment of various forms of cancer, such as leukaemia, lymphoma, solid tumours, and brain and spinal cord tumours, in children and adolescents. Patients are either admitted as in-patients or visit as day-cases. Day-case patients come in for scheduled appointments every week, either for medical review exclusively or for both medical review and chemotherapy treatment. At the time of the study, the interdisciplinary healthcare team was made up of consultant paediatric oncologists, resident specialists, higher specialist trainees, nurses, a social worker, a psychologist, a speech therapist, a physiotherapist, a nutritionist, and a teacher. There was no clinical pharmacist directly integrated within the interdisciplinary health care team to contribute to cancer management in this patient group. The development of pharmaceutical services aimed to complement the existing interprofessional services following a gap finding analysis to establish needs from the clinical pharmacist in this setting. 33
Development of tool
Prior to the implementation of the clinical pharmacy service at the PAOU, a tool intended for use by the clinical pharmacist during the provision of the service was developed, validated and tested for practicality. The tool consisted of a novel PCI classification system based on the Pharmaceutical Care Network Europe (PCNE) Foundation classification for drug related problems version 8 and the DOCUMENT system.34,35 The purpose of the tool was to have a PCI classification system which would provide a standardised and consistent documentation and classification of PCIs and pharmaceutical interventions to be proposed by the pharmacist-researcher, as well as the availability of a system which was robust enough to account for all the PCIs identified during the research.
The PCI classification system (Appendix 1) was presented in tabular format. It consisted of five columns with the following headings respectively: category of PCI, category description, PCI, pharmaceutical intervention/s to resolve the PCI and outcome. The tool covered eleven PCI categories, namely, drug selection, dose selection, dosage regimen selection, duration of treatment, unwanted drug effects, dispensing, compliance, drug administration, monitoring, counselling and seamless care for transitioning patients across care scenarios. Categories were discrete and unambiguous. A short description of every category was included to ensure that each category is comprehensible, to ease interpretation and assist accurate classification of PCIs. Each category encompassed between one and ten subcategories or PCIs. One or more pharmaceutical interventions to be proposed by the pharmacist to resolve the PCIs were determined and included in the classification system. The outcome refers to the status of the pharmaceutical intervention proposed. Three possible outcomes were determined and included in the classification system, namely, accepted and implemented, accepted and not implemented, and not accepted. Only one outcome was possible per pharmaceutical intervention proposed.
Validation of tool
A panel of experts consisting of a drug information pharmacist working within a hospital setting and a community pharmacist validated the classification system. The experts were invited to assess the tool in terms of content validity and appropriateness of the categories of the PCIs, PCIs, pharmaceutical intervention(s) and outcomes. The assessment was carried out in a qualitative manner where comments put forward were discussed. The comments of the expert panel were taken into consideration and the classification system was updated accordingly.
Practicality testing of tool
The updated version of the tool was subjected to a test for practicality and ability for it to be applied consistently by different pharmacists. Ten scenarios were selected from the dataset whereby each scenario described one to three PCIs that occurred in the paediatric oncology unit. Two hospital pharmacists and one community pharmacist classified the PCIs using the developed classification system. The pharmacists were reminded about the importance of classifying each respective issue under a single sub-category. Subsequently the classification by the three pharmacists was compared.
Ethics approval and data collection
Following ethics approval by the University Research Ethics Committee (UREC), the PAOU was attended by the pharmacist-researcher three times per week. Ward rounds and comprehensive reviews of patients’ files, treatment charts and chemotherapy prescriptions were undertaken for identification of PCIs. Validation of treatment charts and chemotherapy prescriptions focused on confirmation of the correct medication dose according to the weight or body surface area of the patient, the correct medication regimen, the suitability of the drug and its indication, dose adjustments recommended in the literature according to patient characteristics, the absence of contraindications and clinically significant drug interactions, conformity with the therapeutic protocol and compliance with pharmacotechnical parameters (compatible diluent, infusion volume and concentration of the solution). The identified PCIs were discussed with the physicians, nurses or parents respectively and subsequently, pharmaceutical interventions were proposed to solve them. The outcome of the proposed interventions was recorded. Other pharmaceutical services which were required to support the ward service were also developed.
Results
Validity and practicality of tool
Following validation of the classification system for content and appropriateness of the categories of the PCIs, PCIs, pharmaceutical intervention(s) and outcomes, a total of 43 suggestions for additions, omissions or changes were put forward by the expert panel and incorporated in the final version of the tool. Out of the total number of suggestions (N = 43), 42 were related to the PCI categories, PCIs and respective pharmaceutical interventions. The remaining suggestion was to put forward a definition for the terms pharmaceutical care issue, pharmaceutical intervention and outcome respectively. Subsequently, for the practicality testing, the total number of PCIs identified by the pharmacist researcher on the ward (control) was 22. Upon comparing the results of the three pharmacists with the control, they identified 20, 20 and 19 PCIs, respectively, in the same manner as the control. Overall, 89% of the PCIs were identified and classified in the same manner by the three pharmacists, implying that the classification system can be followed by trained pharmacists with minimal variation in the classification of PCIs identified.
Pharmaceutical care issues
A total of 545 PCIs were identified during 325 pharmaceutical care sessions provided over 8 months. For the purpose of this research, a pharmaceutical care session is defined as the pharmaceutical service for an individual in-patient or day-case patient within one day capturing file review, discussion with HCPs and ward round. The most common PCI categories were counselling [26.97% (N = 147)], drug selection [23.67% (N = 129)], dose selection [19.27% (N = 105)] and monitoring [15.41% (N = 84)]. Other categories included unwanted drug effects [4.59% (n = 25)], drug administration [3.85% (n = 21)], dosage regimen selection [2.57% (n = 14)], seamless care [2.57% (n = 14)], dispensing [0.73% (n = 4)] and duration of treatment [0.37% (n = 2)]. The distribution of the PCIs identified during the research is illustrated in Figure 1.
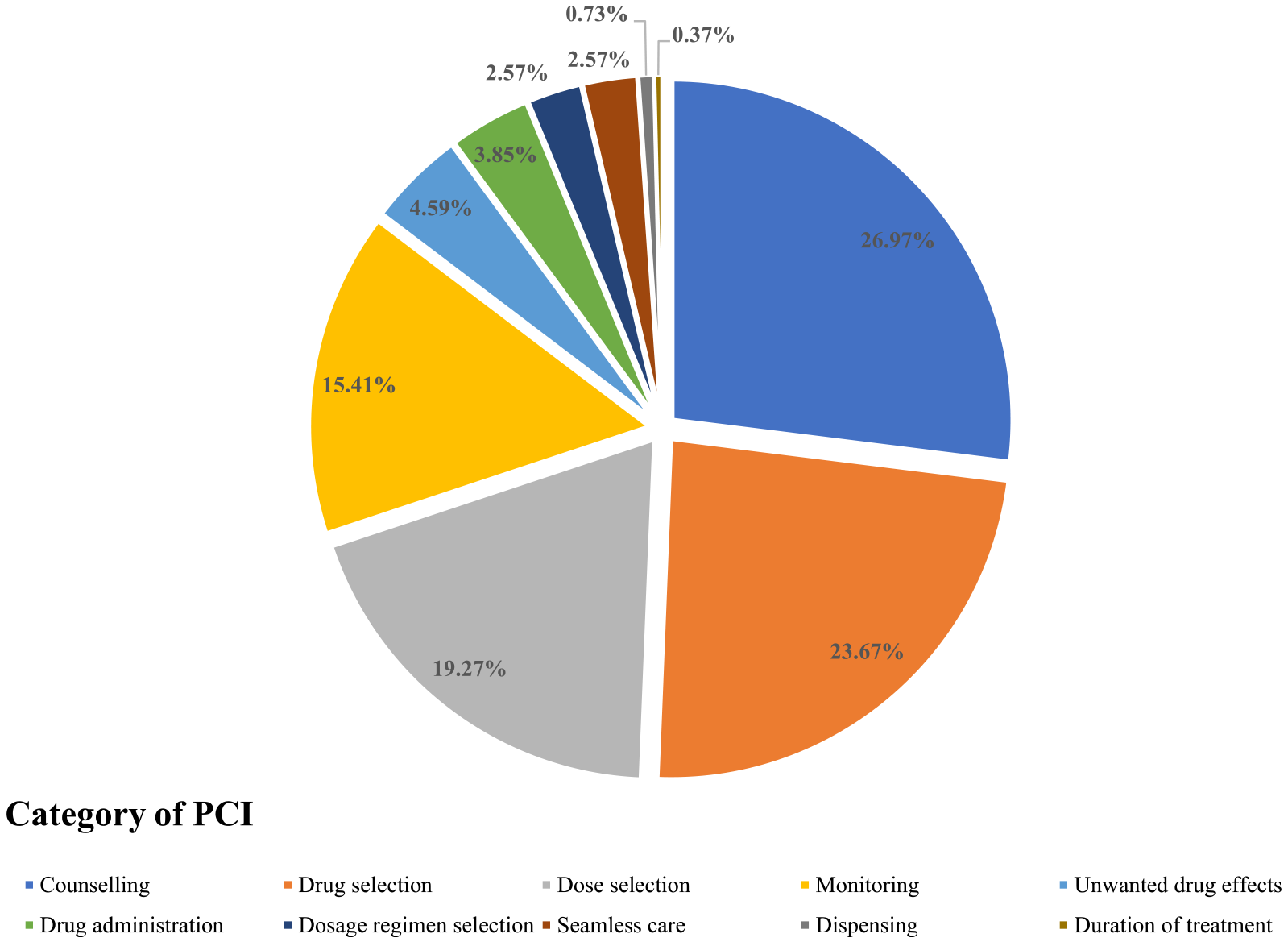
Categories under which the identified PCIs (N = 545) were classified.
The most common single PCIs identified were counselling need to parents or legal guardians (n = 147), monitoring need (n = 84), no indication for drug or indication no longer apparent (n = 55), under dose for patient's age, weight and indication and/or severity (n = 47), over dose for patient's age, weight and indication and/or severity’ (n = 44) and no drug treatment despite existing indication requiring management or prevention (n = 35). Other PCIs included adverse drug reaction or side effect (n = 25), seamless care (n = 14), drug interaction (n = 10), inappropriate route (n = 10), dosage regimen too frequent (n = 8), inappropriate dosage form (n = 7), non-adherence to protocol or guidelines (n = 6), inappropriate infusion rate (n = 3), ineffective drug (no improvement in therapeutic outcomes despite being on the medication) (n = 4), dosage regimen not frequent enough (n = 3), wrong drug dilution (n = 3), contraindication (n = 2), duration of treatment too short (n = 2), need additional drug to properly manage a condition (n = 2), too many drugs prescribed for indication (n = 1), wrong drug dispensed (n = 1), prescribed drug not available in the required strength (n = 1), prescribed drug not available in required dosage form (n = 1) and prescribed drug not available at all in the service (n = 1).
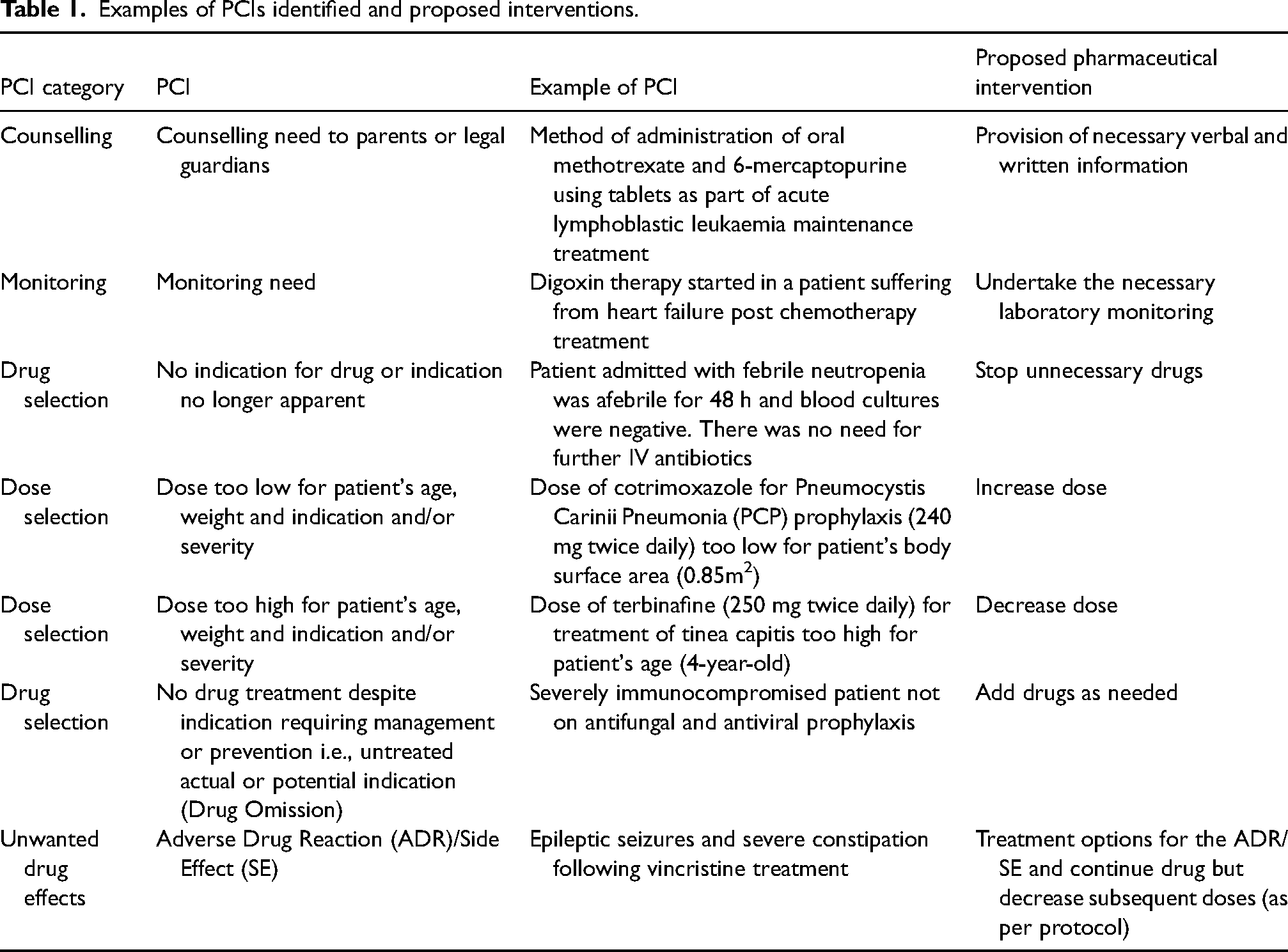
Actions or recommendations put forward by the pharmacist to solve the PCIs included verbal and written counselling, monitoring of the necessary laboratory and non-laboratory parameters, treatment discontinuations, drug dosage adjustments, drug additions, changing the method of drug administration, dosage regimen adjustments and changes in duration of treatment. Examples of PCIs and the respective pharmaceutical interventions proposed are included in Table 1. Out of the total number of pharmaceutical interventions proposed, 95% (n = 516) were accepted and implemented by the healthcare professionals or the parents. The remaining 5% were discussed with the physicians and not accepted due to concerns that the patients were in the palliative stage.
Examples of PCIs identified and proposed interventions.
Other services provided to complete the ward-based pharmacy service
In addition to the PCIs, other pharmaceutical services provided to support the ward service included dosage calculations, medication information to nurses and physicians, and guiding physicians and nurses in identifying and filling appropriate pharmacy related forms to ensure patient access to medications. Liaison with other sections of the hospital, mostly the dispensary and the compounding sections to ensure availability of the treatment according to patient needs and patient entitlement units, preparation of chemotherapy flowsheets and proformas and participation in research studies were also undertaken (Table 2).
Pharmaceutical services provided to complete the ward service during the 8 month study period.

Discussion
Novel classification system for identified PCIs
The developed tool consisting of an easy-to-use PCI classification system enables standardisation and consistency in the way identified pharmaceutical care issues are classified and documented. It also serves as a guidance tool with regards to the pharmaceutical interventions or recommendations to be proposed for every pharmaceutical care issue. Practicality testing demonstrated that the innovative PCI classification system was relevant, robust and practical for use, making the model feasible to run at the PAOU.
Validation of the interventions of the pharmacist through PCIs
The physical presence of the pharmacist at the PAOU enabled the provision of several direct patient care activities, unique to the skill set of a pharmacist. One of the primary activities was the identification of PCIs and putting forward suitable pharmaceutical interventions to solve them. The most common PCI category identified during the study period was the counselling category, with ‘Counselling need to parents’ being the most common PCI. The result aligns with findings from a study by Randolph et al. (2016), 36 where patient counselling constituted the highest percentage of interventions. Receiving a cancer diagnosis is profoundly distressing. The provision of consistent information regarding the complex pharmacotherapy involved helps improve learning and understanding, adherence, health outcomes and satisfaction. 37 In the local research there were instances where information about medications was requested by the parents. There were other instances where information about medications was provided simply because the pharmacist felt it necessary to share it with the parents such as at the time of initiation of a drug or complex chemotherapy regimen, when there was a change in drug therapy and prior to discharge from hospital. Counselling by the pharmacist captured the reasons for use, administration, side effects and their management, handling, storage and drug interactions.
The second most common PCI category pertained to drug selection, with the majority involving ‘No indication for drug or indication no longer apparent’ and ‘No drug treatment despite existing indication requiring management or prevention i.e., untreated actual or potential indication’. Examples of no indication for drug or indication no longer apparent were those involving failure to stop intravenous antibiotics when no longer indicated such as after patients admitted with febrile neutropenia would have been afebrile for 48 h and blood cultures would be negative (recommendation as per protocol). Prolonged use of antibiotics may increase the risk of adverse effects whilst increasing the risks of drug resistance. A case example of no drug treatment despite existing indication requiring management or prevention i.e., untreated actual or potential indication was that of a severely immunocompromised child needing to be started on antifungal and antiviral prophylaxis. The issue was discussed with the clinician and initially the drug was not started due to the concern that the patient was in a palliative stage. Eventually, the patient developed herpes zoster amongst other multiple bacterial mixed infections and received the relevant treatment. These scenarios highlight the importance of the pharmacist as the drug expert exerting the pharmaceutical advice for the benefit of the patient.
The third most common PCI category was the dose selection category. The most common PCIs which featured under this category were ‘dose too low for patient's age, weight and indication and/or severity’ and ‘dose too high for patient's age, weight and indication and/or severity’. A case example of dose too low was for a patient suffering from acute lymphoblastic leukaemia (ALL). Patients suffering from leukaemia need to be on prophylactic co-trimoxazole for pneumocystis carinii pneumonia (PCP) prophylaxis. Doses vary according to body surface area and are specified in the protocol. In this case, the child had put on weight, the body surface area had changed but the dose of co-trimoxazole had not been increased accordingly. PCP is a very serious opportunistic infection in immunocompromised patients and was the most common cause of death amongst children receiving chemotherapy prior to the inclusion of PCP prophylaxis. 38 Ensuring that the right dose of co-trimoxazole is prescribed is crucial as otherwise potentially serious consequences can occur. Children often experience fluctuations in weight due to their growth, and this variability might be the reason for “dose too low” being the most prevalent PCI associated with dose selection. A case example of dose too high was that of a child suffering from ALL who was in the maintenance phase of leukaemia treatment. The patient had lost weight and consequently the body surface area had decreased. The patient had come for review at the PAOU and upon calculating the doses of 6-mercaptopurine and methotrexate, it was noted that the doses were too high based on the new body surface area. Implications of high doses of chemotherapy are increased risks of haematological toxicity and subsequently interruption of doses. Dose interruptions decrease the rate of therapeutic success and can result in treatment failure and relapse. The presence of a pharmacist on the ward to ensure an optimal therapeutic plan, limiting these side effects and subsequently achieving optimal treatment outcomes is of utmost importance. In paediatrics, medication dosing errors are one of the most frequent prescribing errors.10,39 These PCIs may have potentially serious consequences, either via enhanced toxicity or impaired disease control.1,16,40,41 As shown in this research, pharmacists play a key role by ensuring that drug doses are appropriate, oftentimes, requiring adjustment of doses based on changes in weight.1,32,42,43 Their intervention has a major impact on detecting and reducing dosing errors.1,8,10,44–48
The fourth most common PCI category was the monitoring category related to laboratory and non-laboratory parameters. A case example of laboratory monitoring was that of a child who was on digoxin therapy. Laboratory results following request for monitoring of electrolytes, in particular magnesium, potassium and calcium showed persistently low magnesium levels. Subsequently, magnesium supplementation was started to prevent potential digoxin toxicity due to hypomagnesaemia. A case example of non-laboratory monitoring was that of a patient who was on high dose steroids. The issue of the need for monitoring of fasting and post- prandial blood glucose was raised upon steroid treatment initiation. On one occasion, the random blood glucose result was 11 mmol/L. As a result, fast acting soluble insulin was prescribed as an ‘as required medication’. The two cases described signified that pharmacists play a significant role in detecting drug-related problems. 29
The clinical pharmacist: an asset to patients?
Published literature describing the integration and impact of clinical pharmacy services within in-patient and out-patient haematology, oncology and transplant settings demonstrates that clinical pharmacists bring forth specialised expertise and competencies concerning medications and their significance in the context of cancer treatment, rendering them indispensable members of interdisciplinary cancer care teams. 4 Through collaboration with other HCPs, clinical pharmacists occupy a prime position to deliver top-tier, evidence-based pharmaceutical care to young cancer patients, including initial and subsequent treatment phases, supportive care and survivorship.1,2,8,11,23,49–52 They can help to optimize drug therapy administered to children through the identification of PCIs.1,11,13,15,23,42,53 These key outlooks are comparable to the outcomes of this research conducted within the speciality of paediatric oncology. In this research, the pharmacist was adept at identifying and managing PCIs, with acceptance rates of pharmacist-initiated interventions for paediatric oncology patients being 95%. The remaining 5% were discussed with the physicians and not accepted due to concerns that the patients were in the palliative stage. The high acceptance rate may be viewed as a qualitative indicator of the effectiveness of the pharmacist at the PAOU. It indicates physician confidence in the pharmacists’ recommendations and the establishment of a good working relationship with the medical team as well as the parents which is the basis of successful clinical practice.
Conclusion
At the end of this research, one can reflect on the relevant contribution of the clinical pharmacist at ward level within the interdisciplinary healthcare team which was made possible through the implementation of an innovative pharmaceutical care model focusing on pharmaceutical care issues individualised according to each patient specific needs. Pharmaceutical care issues including drug related problems are frequent and may result in reduced quality of life, and even morbidity and mortality. Clinical pharmacists can effectively identify and prevent clinically significant drug-related problems and physicians, nurses and parents acknowledge and act on the clinical pharmacist's suggestions for interventions to the drug-related problems.
Supplemental Material
sj-docx-1-opp-10.1177_10781552251350553 - Supplemental material for Implementation of a pharmaceutical care model within paediatric oncology
Supplemental material, sj-docx-1-opp-10.1177_10781552251350553 for Implementation of a pharmaceutical care model within paediatric oncology by Sephorah Falzon, Nathalie Galea, Jennifer T Pham, Louise Grech and Lilian M Azzopardi in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
This paper is dedicated to the memory of Dr Victor Calvagna, who was the Lead Consultant Paediatric Oncologist at the PAOU at the time of the study, and whose contribution and mentorship was crucial for the successful completion of the research. His invaluable humane and professionalism towards these patients, colleagues and students will be remembered.
Author contributions
S Falzon: Conceptualisation, Methodology, Validation, Formal analysis, Investigation, Data Curation, Writing- Original Draft, Visualization, Project administration.
N Galea: Conceptualisation, Writing - Review & Editing.
JT Pham: Conceptualisation, Writing - Review & Editing.
L Grech: Conceptualisation, Writing - Review & Editing, Visualization, Supervision.
LM Azzopardi: Conceptualisation, Writing - Review & Editing, Visualization, Supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
