Abstract
Introduction
Active cancer and body mass index (BMI) greater than 30 kg/m2 have been shown to increase venous thromboembolism (VTE) risk in hospitalized patients. Optimal dosing strategies in this population remain uncertain. This study evaluated the incidence of healthcare associated VTE (HA-VTE) in patients with active malignancy and obesity, comparing adjusted versus standard dose enoxaparin for VTE prophylaxis.
Methods
This multicenter, retrospective, cohort study compared enoxaparin 40 mg subcutaneously (SC) daily with adjusted enoxaparin dosing (> 40 mg SC daily) for VTE prophylaxis in hospitalized adult patients with a BMI greater than 30 kg/m2 who received cancer therapy between January 1, 2020, and August 17, 2023. The primary outcome was incidence of HA-VTE. Secondary outcomes included VTE type and bleeding events. Descriptive statistics were used for all outcomes.
Results
A total of 330 patients were included, with a median BMI of 33.7 kg/m² (IQR 31.6–37.9 kg/m²). Standard dose enoxaparin was administered to 300 patients, while 30 received adjusted doses. HA-VTE occurred in 13 patients in the standard dose group, with no HA-VTE events in the adjusted group (p = 0.245). In the standard dose group, 4 patients had minor bleeds, and 2 had clinically relevant non-major bleeds. One minor bleed occurred in the adjusted group (p = 0.490).
Conclusions
HA-VTE was more frequent in the standard dosing group, though the small sample size limits the clinical significance. Patients on adjusted doses did not have increased bleeding events. Further research is needed to determine optimal thromboprophylaxis strategies for cancer patients with a BMI over 30 kg/m².
Introduction
Hospital-associated venous thromboembolism (HA-VTE) poses significant and often preventable health risks, such as prolonged hospital stay and death in hospitalized patients. 1 While VTE prophylaxis in hospitalized patients is standard of care, select risk factors, such as obesity and cancer, can complicate management. Previous literature has demonstrated that active cancer and obesity (defined by the World Health Organization as a body mass index (BMI) greater than 30 kg/m2) can increase the odds of venous thromboembolism (VTE) in hospitalized patients by 1.96 and 1.12 times, respectively. 1 While standard dose pharmacologic thromboprophylaxis has been shown to reduce the rate of HA-VTE from 3.44% to 0.06% in medical inpatients, optimal dosing strategies amongst patients with obesity and concomitant cancer remain uncertain. 2
The National Comprehensive Cancer Network (NCCN) guidelines suggest enoxaparin 40 mg subcutaneously (SC) every 12 h or 0.5 mg/kg SC daily for VTE prophylaxis in oncology patients with BMI > 40 kg/m2. However, this recommendation is extrapolated from studies of patients who have experienced trauma or undergone bariatric surgery. 3 Data for anticoagulation management in patients with BMI > 30 kg/m2 is even less clear with existing studies utilizing a dose of enoxaparin 40 mg every 12 h and focusing on a cut off of BMI > 40 kg/m2; no clear guideline recommendations exist in this patient population.4–6
Our study aimed to evaluate the incidence of HA-VTE in patients with active cancer and obesity to optimize thromboprophylaxis strategies in this unique population. We hypothesized that adjusted dosing (i.e., doses of greater than 40 mg once daily) of enoxaparin for VTE prophylaxis in patients with obesity and cancer may be associated with a lower incidence of HA-VTE without increased bleeding rates compared to standard dosing of enoxaparin.
Methods
Trial design
This was a multi-center, retrospective, cohort study of patients admitted to the five University of California medical centers (Davis, Irvine, Los Angeles, San Diego, and San Francisco) between January 1st, 2020, and August 17th, 2023. Institutional review board (IRB) approval was obtained through each respective institution prior to study initiation. VTE prophylaxis was administered according to the clinical practice of each participating hospital; only one center had an institution guideline for dosing enoxaparin in patients with BMI > 40 kg/m2. Enoxaparin dose, route, and frequency were recorded. Patients were compared in two separate groups: those that received standard enoxaparin dosing versus those that received an adjusted dosing regimen. Standard dose VTE prophylaxis was defined as enoxaparin 40 mg SC daily. Adjusted dose enoxaparin was defined as any total daily dose greater than 40 mg SC once daily.
Patients
Patients included were 18 years of age or older, had a BMI greater than 30 kg/m2, received VTE prophylaxis with enoxaparin for greater than two days, and received antineoplastic therapy within six months prior to the initial enoxaparin administration during the study period. Patients were excluded if they received therapeutic anticoagulation, had a history of hematopoietic stem cell transplantation (HSCT) or prior administration of chimeric antigen receptor T-cell (CAR-T) therapy, received a bispecific antibody (BiTE) during the index admission, received less than two days of enoxaparin, changed enoxaparin regimens ≥ 48 h after admission, or were pregnant. Admissions beyond the first patient encounter during the study period were censored from data collection.
A total of 200 patients, or all available patients if fewer than 200, were screened at each institution. Each institution's data analytics team pulled patient data, and a random number generator was used to select patients for screening if the initial list provided by the analytics team consisted of more than 200 patients to limit selection bias. Each institution conducted independent reviews of included patients through electronic medical records. To ensure standardization, the research team utilized an data abstraction manual and met monthly to review any questions that arose. Study data were collected and managed using REDCap electronic data capture tools hosted at the University of California, San Francisco.7,8
Outcomes
The primary outcome was the cumulative incidence of HA-VTE (defined as a VTE event diagnosed during admission or within 90 days after discharge). This was confirmed through imaging, including doppler ultrasounds of the extremities or computed tomographies of the chest available in each respective institution's electronic medical record. Secondary outcomes included type of HA-VTE, incidence of bleeding, bleed type, and mortality during hospitalization. In accordance with the International Society on Thrombosis and Haemostasis (ISTH) criteria, bleeding types were classified as major bleeding, clinically relevant non-major bleeding (CRNMB), or minor bleeding. Major bleeding was defined as fatal bleeding and/or symptomatic bleeding at a critical site (such as intracranial, intraspinal, intraocular, retroperitoneal, intra-articular, pericardial, intramuscular with compartment syndrome), bleeding that results in a decrease in hemoglobin of 2 g/dL or more, or bleeding requiring a transfusion of 2 units or more of blood. 9 CRNMB was defined as any sign or symptom of hemorrhage (e.g., more bleeding than would be expected for a clinical circumstance, including bleeding found by imaging alone) that does not fit the criteria for definition of major bleeding but does meet at least one of the following criteria: requiring medical intervention by a healthcare professional, leading to increased level of care within the hospital, and/or prompting a face to face (i.e., not just a telephone or electronic communication) evaluation. Minor bleeding was defined as overt bleeding not meeting the criteria for major bleeding or CRNMB. 9
Statistical analysis
Descriptive statistics were used to describe the enrolled patients. Continuous variables were reported as median and interquartile range (IQR). Categorical variables were reported as frequency and proportion of observation. Differences in baseline characteristics, efficacy, and safety outcomes between groups were analyzed using Pearson's Chi-squared test or Fisher's exact test and Mann-Whitney U test. Data was analyzed using STATA© (StataCorp. 2023, release 18 College Station, TX, USA: StataCorp LP).
Results
After 800 patients were screened for eligibility, 330 patients were included in this study (Figure 1). Common reasons for exclusion included being on therapeutic anticoagulation, receiving HSCT, and having a change of enoxaparin regimen 48 h or more after admission. Three hundred patients received standard enoxaparin dosing, and 30 patients received an adjusted enoxaparin regimen for VTE prophylaxis.
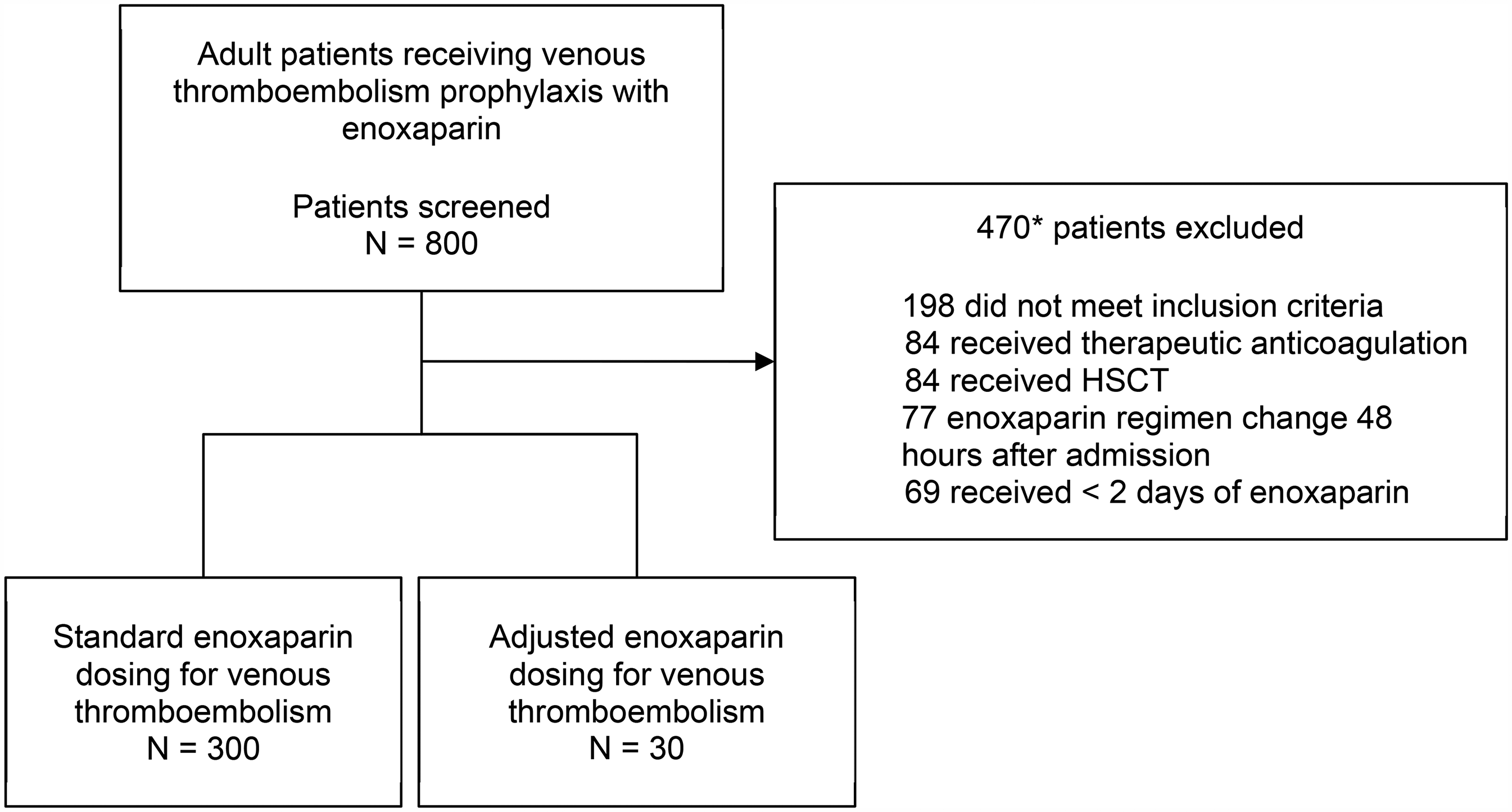
Patient flow diagram (*the number of patients excluded does not add up to 470 due to individual patients meeting multiple exclusion criteria).
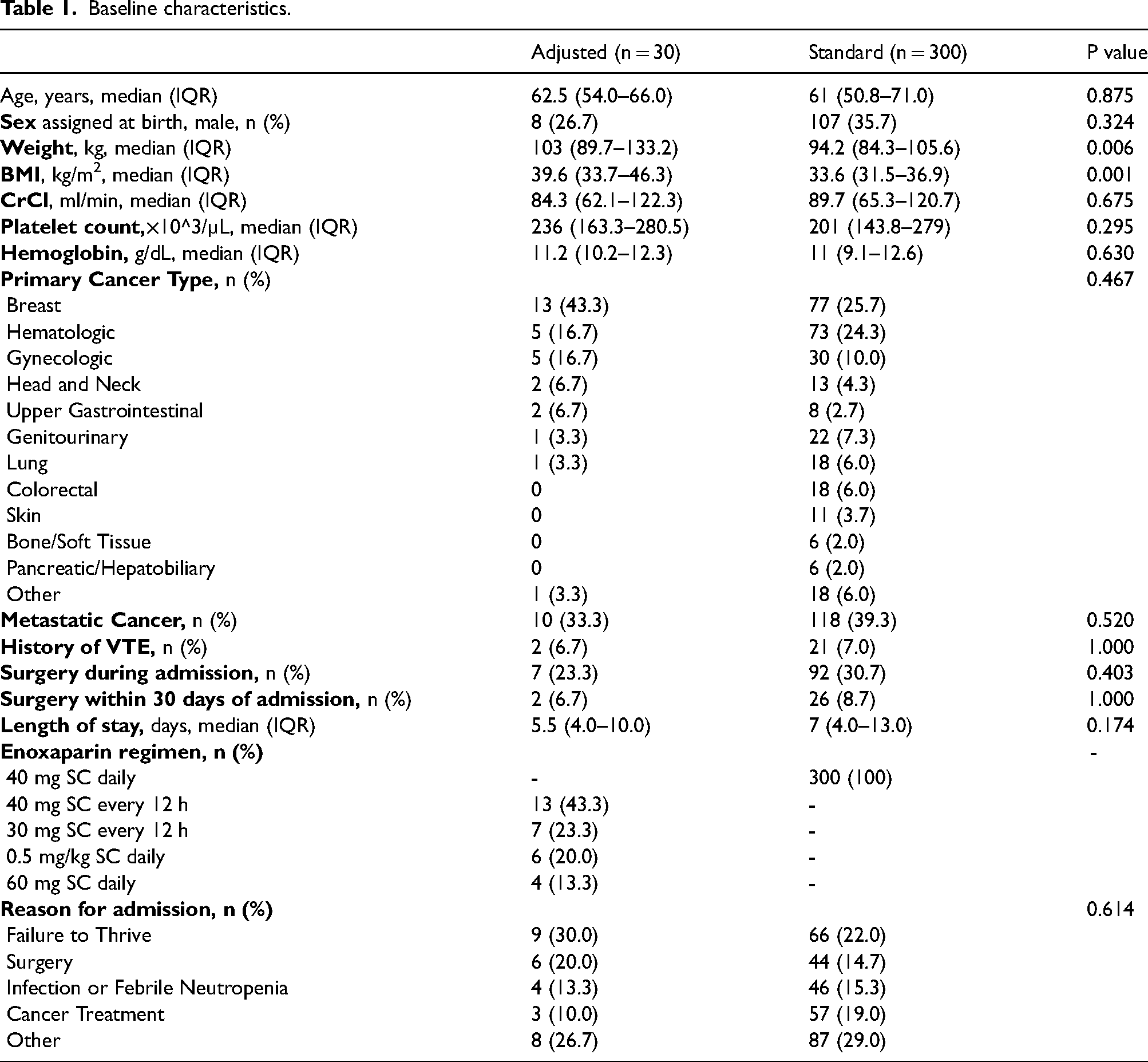
The most common adjusted enoxaparin regimen was 40 mg SC every 12 h, followed by 30 mg SC every 12 h, 0.5 mg/kg SC daily, and 60 mg SC daily (Table 1). Baseline characteristics were similar between groups except for weight and BMI, which were higher in the adjusted group (median weight 103 kg [89.7–133.2] vs 94.2 kg [84.3–105.6], p = 0.006; BMI 39.6 kg/m2 [33.7–46.3] vs 33.6 kg/m2 [31.5–36.9], p = 0.001).
Baseline characteristics.
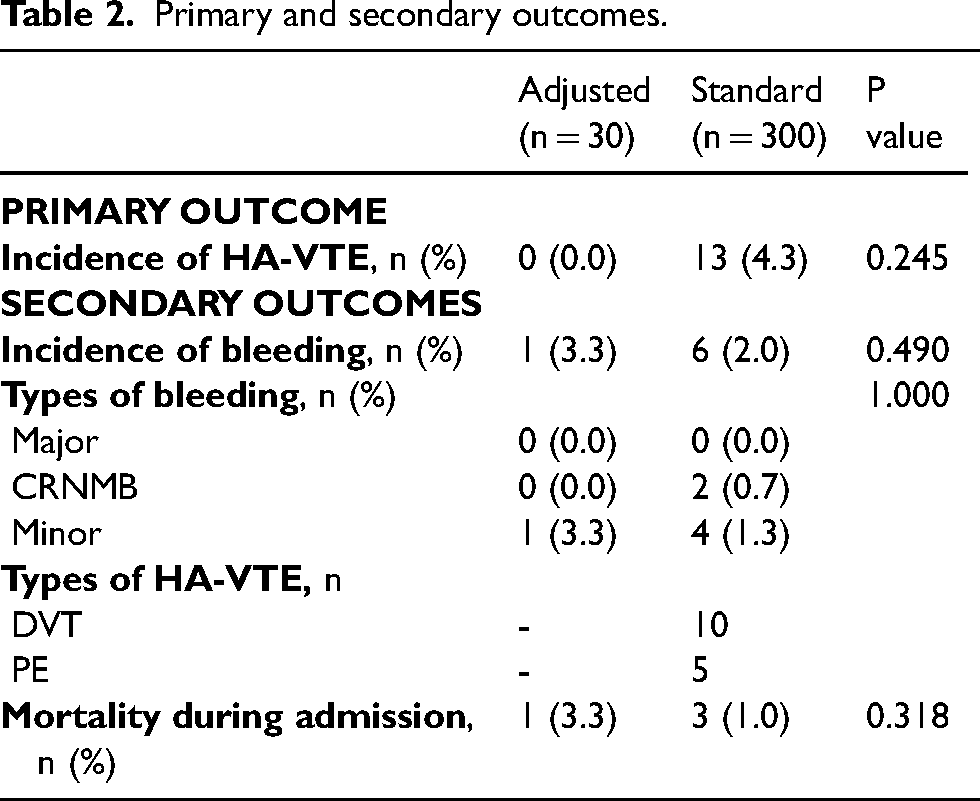
No HA-VTE events occurred in the adjusted group while 13 HA-VTE events occurred in the standard group (0% vs 4.3%, p = 0.245; Table 2), which included 10 DVTs and 5 PEs. Both DVT and PE occurred in two patients, and two out of ten DVTs were catheter-related. Patients who experienced HA-VTE had a median weight of 93.3 kg (IQR 84.5–102 kg) with a median BMI of 32.2 kg/m2 (IQR 31.8–35.2 kg/m2). The incidence of bleeding was similar between the adjusted group and the standard group (3.3% vs 2.0%, P = 0.490). Four patients expired during admission (one in the adjusted group and three in the standard dose group, P = 0.318; Table 2). The causes of death were due to the progression of cancer and not related to VTE or bleeding.
Primary and secondary outcomes.
Discussion
We observed more HA-VTE events in the standard enoxaparin dosing group, though this study was underpowered to detect statistical significance. The incidence of HA-VTE in the standard dose group was higher than what has previously been reported in medical patients. Current literature suggests the rate of HA-VTE in general medical patients occurs in up to 1.2% of patients and increases with longer length of hospitalization, with many events occurring after discharge. 1 In our study, 4.3% of total patients experienced HA-VTE (all contributed by the standard group), and 11 of 13 VTE events occurred post-discharge. It is established that many HA-VTE events occur post-discharge, and the event rate we observed was in line with current literature. However, there are no large studies designed to capture the rate of HA-VTE as it applies to our study population.
This higher rate of HA-VTE may signal a gap in guideline recommendations and clinical practice. NCCN only recommends dose adjustment for this patient population for BMI greater than 40 kg/m2; however, it is established that enoxaparin drug levels inversely correlate to a patient's total body weight, which is seen in patients whose body weight differs only slightly from average. This suggests standard dose enoxaparin may be inadequate for above-average weight patient and particularly in patients with BMI between 30–40 kg/m2. 10 The higher median BMI in our adjusted dosing group may have been because patients with BMI > 40 kg/m2 were more likely to receive adjusted dosing in accordance with NCCN dosing guidelines. In our study, however, patients who experienced HA-VTE had BMIs ranging from 30.5 to 43.3 kg/m2.
While identifying the right patient to adjust doses in, selecting the appropriate adjusted dose is also nebulous. In our study, less than half of those patients who received dose adjustments actually received the NCCN recommendation. We noted seven patients received enoxaparin 30 mg SC every 12 h, which is a regimen widely used for VTE prophylaxis in trauma patients and those undergoing major surgery regardless of weight. 11 Because these patients were surgical patients it was less clear which dosing strategy should be applied when patients meet multiple criteria (e.g., obesity and trauma surgery) for dose adjustment. There is emerging discussion that twice-daily dosing of VTE prophylaxis may be a superior approach versus standard once-daily dosing.6,11,12 This is hypothesized to relate to the pharmacokinetics of enoxaparin and the half-life of roughly 4.5 h. By dosing enoxaparin twice-daily, in the same fashion as therapeutic anticoagulation, a patient may achieve a more favorable kinetic profile that optimizes peak and trough plasma anti-Xa activity. 12 Though studies vary on the recommended dosing, and what constitutes a target trough anti-Xa goal for prophylaxis, our findings point to the broad lack of standardization in what constitutes standard versus adjusted enoxaparin dosing in practice.
This study had several limitations. Fewer patients with a BMI between 30 and 40 kg/m2 received adjusted dosing. This is unsurprising, as current NCCN guidelines suggest consideration of adjusting prophylactic enoxaparin doses for patients with a BMI of > 40 kg/m2. This may be due to the overall lack of available literature for patients who meet defined criteria of obesity (BMI >30 kg/m2), but do not have guidance for HA-VTE prophylaxis. Given the retrospective, multi-center study design, there was inherent variability in institutional practice and policy which limited the sample size we were able to capture. Between University of California Health Systems, there are no consensus guidelines that all centers share, which leaves dosing decisions up to the discretion of the treating clinician and current literature. Additionally, concomitant medications which could influence the risk of thrombosis were not collected or assessed. Finally, our patients had limited lengths of stay, and the inability to capture post-hospitalization events at outside institutions that do not utilize the same electronic medical record system may have resulted in lower HA-VTE rates.
Further research with a greater representation of each cancer type and a larger sample size, particularly in the adjusted dose cohort, is needed to generalize this study's outcomes and provide external validity to the findings. It could be suggested to create a standardized platform in which HA-VTE events are reported to improve accuracy in capturing the outcome's true prevalence, as well as associated patient characteristics. Institutions could additionally move towards a standardized guideline for VTE prophylaxis, which may strengthen the number of patients who receive prophylaxis and at the appropriate guideline-directed dose.
Conclusion
There was no statistical difference in HA-VTE occurrence between standard and adjusted enoxaparin dosing amongst patients with cancer and a BMI greater than 30 kg/m2. However, the lack of any HA-VTE event in the adjusted group implores further evaluation of these dosing regimens given the potential for improved efficacy. Additionally, patients who received adjusted doses did not appear to experience increased rates of bleeding. The clinical significance of these findings is unclear due to the limitations of a small sample size and the overall lack of power to detect a difference in outcomes. Moreover, low HA-VTE event rates regardless of the thromboprophylaxis strategy used limit the ability to comprehensively assess the efficacy of various enoxaparin doses in this population. Given the current gap in primary literature and the scarcity of real-world evidence, these results suggest further studies be conducted to determine proper thromboprophylaxis strategies in patients with cancer and a BMI greater than 30 kg/m2.
Footnotes
Authors’ contribution
TP researched literature and conceived the study. All authors were involved in protocol development and gaining ethical approval. CA, JA, KG, BC, and CT conducted chart reviews, performed data analysis, and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final manuscript version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent statements:
Institutional review board (IRB) approval was obtained through each participating institution prior to study initiation. Informed consent was not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
