Abstract
Introduction
At the University of California Irvine (UCI) in Orange County California, we published an implementation study to evaluate the use of an electronic patient-reported outcome (ePRO) tool to guide pharmacist-led chemotherapy education in newly diagnosed cancer patients. The goal of this secondary analysis was to describe the types of pharmacist-led interventions performed in the implementation study.
Methods
Using a pre-configured ePRO battery, patients’ symptoms were assessed in seven domains (cognitive impairment, physical impairment, pain, depression, anxiety, fatigue, nausea/vomiting) at each infusion visit. Other symptoms that were brought up during the visit were also documented. Based on symptom scores calculated in real-time, pharmacists implemented personalized patient care strategies and documented them in the clinical notes. Descriptive statistics were employed to evaluate the type of interventions employed for each symptom domain.
Results
Among 250 patients enrolled, the average age of participants was 60 years old (SD = 14.6), with 129 (52%) being female, and 134 (54%) of participants identified as white. The preconfigured ePRO battery identified clinically important symptoms at 47.3% of the visits, and other clinically important symptoms were identified at 23.4% of the visits. Pharmacist education was performed for 311 (55%) of 564 visits examined throughout the study. The highest proportions of pharmacological interventions were performed to manage nausea/vomiting (51 visits), followed by gastrointestinal symptoms (21 visits), and pain (16 visits). Additional provider communication was performed for managing nausea/vomiting (14 visits), pain (9 visits), and neurological symptoms (9 visits).
Conclusion
We observed a high rate of pharmacist-led education after the institution of ePRO to evaluate symptoms in patients undergoing anticancer treatment. Future studies should consider incorporating additional ePRO domains to include a wide range of patient-reported symptoms tailored to each type of anticancer treatment.
Introduction
Patients undergoing anticancer treatment experience a wide array of physical and socioemotional effects that can impact their quality of life and treatment outcomes when they are left unaddressed.1,2 There is a growing literature suggesting the benefits of incorporating patient-reported outcome (PRO) tools into routine clinical practices to facilitate patient-centered care and enhance patient-provider communication. 3 A recent global survey has shown that oncology practitioners used PRO data to facilitate communication with patients and monitor their general health status. 3 The Patient Reported Outcomes Measurement Information System (PROMIS®) is a validated National Institutes of Health (NIH)-developed electronic PRO (ePRO) tool that can be used to evaluate and monitor symptoms (physical, mental, and social) for both research and clinical purposes. 4 PROMIS® can also be integrated into the electronic health records to disseminate real-time results to all institutional healthcare professionals, maximizing its clinical use across multiple disciplines. 1 Increasing the use of ePRO in oncology settings provides an excellent opportunity to expand patient engagement beyond routinely monitoring for emerging problems. 5 This can eventually enhance the overarching goal of high-quality patient-centered cancer care. 5
At the University of California Irvine Chao Family Comprehensive Cancer Center (UCI CFCCC), we have conducted an implementation study to evaluate the role of PROMIS® to guide pharmacist-led chemotherapy education and demonstrate its feasibility to identify symptoms especially among racial and ethnic minority patients. 6 In the implementation study, we successfully developed and implemented a multilevel ePRO-driven intervention which allowed oncology pharmacists to perform interventions as deemed necessary based on patients’ symptoms. By providing oncology pharmacists access to ePRO, it allows them to objectively monitor patients’ symptoms and intervene as appropriate to optimize patients’ care. Our innovative approach aligns with the vision of the Hematology/Oncology Pharmacy Association (HOPA) regarding the role of oncology pharmacists to provide personalized supportive care. 7
In this follow up study, we described the symptoms that required pharmacists’ interventions among patients undergoing anticancer treatment, as well as the types of pharmacist-led interventions that were performed due to symptoms identified. Our study addresses the research gap regarding the lack of data showing how pharmacists can use ePRO data to better assist in caring for patients. Our study differs from other studies surrounding the use of PROMIS® by specifically focusing on the interventions provided by a pharmacist within a healthcare team. These analyses provide insights into the use of ePRO-driven pharmacist-led care to provide an improvement in health outcomes among patients undergoing anticancer treatment.
Methods
Study design
This is a secondary analysis of a published implementation study that has evaluated the feasibility of real time ePRO tools for pharmacist-led chemotherapy education and interventions. The primary study was a prospective, pragmatic, implementation study, which took place at the UCI CFCCC between July 2021 and June 2023. 6 The CFCCC infusion center is located in Orange County, California and serves a widely diverse population, making it an ideal site to conduct our study with the aim of evaluating and comparing patient-reported symptoms from their anticancer treatments between different populations. The primary study protocol received ethics approval from the UCI Institutional Review Board (#2021-6431). 6 All study participants provided written informed consent before participation.
Eligibility criteria
Eligible participants were aged 18 years or older with a new diagnosis of cancer and were planning on receiving intravenous anticancer treatment at the CFCCC. Oncology pharmacists at the CFCCC screened eligible patients by using the infusion center schedule within Epic®, the electronic health record system used by the institution. Patients who chose not to participate in the study or could not provide written consent to participate in the study due to physical or mental incapabilities were excluded.
Intervention
In our primary study, we have preconfigured an ePRO battery (PROMIS®) to assess patients’ symptoms in seven domains including cognitive impairment, physical impairment, pain, depression, anxiety, fatigue, nausea & vomiting at each infusion visit. The ePRO battery was available in English and Spanish. Medical interpreters were used for all other languages, such as Vietnamese and Korean, using video remote technology available at the CFCCC. The ePRO was completed by the patient using computer adaptive tests (CAT) and short forms through REDCap. All patients were given an iPad to complete the ePRO assessments either before or during their infusion sessions. Based on the symptom scores calculated in real-time, the pharmacists counseled patients and tailored interventions as necessary. Patients who tolerated their treatment well, denied experiencing symptoms, or declined to continue to participate in a pharmacist consultation were discharged from the study.
When symptoms were being identified during the pharmacists’ assessments, counseling and education sessions were provided by pharmacists with regard to medication and side effect management, general lifestyle advice, and emotional support. Additional symptoms that were not captured by the ePRO battery were documented and intervened as needed. Many patients may require additional interventions, including pharmacological therapies based on the pharmacist's discretion related to drug dosing changes, drug discontinuation, or initiating a new medication. Additionally, communication between the pharmacist and other healthcare providers, such as oncologists and registered nurses or nurse practitioners, may occur to further discuss patient symptoms, the need to prescribe other medications, and the impact on treatment plans.
Data collection
All pharmacist intervention data and notes detailing their patient interactions were extracted to perform data analysis. At each patient visit, pharmacists documented their interactions using two databases: (1) The PharmacyApp (an internal CFCCC infusion pharmacy database) which is a database for all UCI Health infusion pharmacists to manage drug inventory, record patient counseling notes, and maintain all pertinent pharmacy operational logs and (2) Epic® where provider communications are documented.
Outcomes: primary and secondary
The primary outcome of the study is to quantify the rate of pharmacist-led interventions across all completed study visits and to describe the distribution of patient-reported symptoms requiring pharmacist education. We classified all medications that were intervened during pharmacological interventions using the World Health Organization (WHO)'s Anatomical Therapeutic Chemical (ATC) classification system, in order to ensure a standardized approach is utilized for drug utilization research and reporting. 8 Secondary outcomes were evaluated for pharmacological interventions and provider communication performed and stratified by symptom domains.
Statistical analysis
We employed descriptive statistics to summarize participant characteristics and study outcomes categorical variables as counts and percentages, and continuous variables as means and standard deviations (SD).
For the primary outcome, the distribution of patient-reported symptoms intervened and requiring pharmacist education categorized by the specific ePRO symptom domains addressed, were summarized as percentages of the total completed study visits. Symptoms managed by pharmacists that did not belong to one of the seven ePRO symptom domains were further categorized based on body systems (i.e. gastrointestinal, dermatological, immunological, neurological, ophthalmological, reproductive, hormonal, cardiological) as shown in Table 1. For the secondary outcomes, percentages were calculated for pharmacological interventions and provider communication based on the total study visits where each specific ePRO symptom domain was addressed.
Symptoms that were not captured by the originally configured ePRO battery.

Results
Demographics
Data from 250 patients were enrolled. The average age of participants was 60 years old (standard deviation [SD] = 14.6), with 129 (51.6%) being women, and 147 (58.8%) of being married (Table 2). 134 (53.6%) of participants identified as white, 88 (35.2%) were high school graduates, and 86 (34.4%) had a full-time job at the time of participation. The average number of comorbidities each patient had was 3.4 (SD = 4.2), and the average number of medications each patient was taking at the time of the study was 8.9 (SD = 8.1). The top comorbidities among patients were hypertension (27.2%), hyperlipidemia (12.4%) and diabetes mellitus (9.2%). A total of 564 visits were captured among the 250 participants (Table 3) and the number of visits each participant completed ranged from one to five. Eighty-seven (34.8%) patients were taken off the study due to death, patient decision, or no need for further follow-up visits per pharmacists’ recommendation. The remaining participants stopped the study following pharmacists’ recommendations for no further follow-up appointments required. Clinically important symptoms that involved one of the 7 domains from our preconfigured ePRO battery were identified at 47.3% of the visits. In contrast, the other clinically important symptoms that were not part of the ePRO battery were identified at 23.4% of the visits (Table 3).
Patient demographic data.
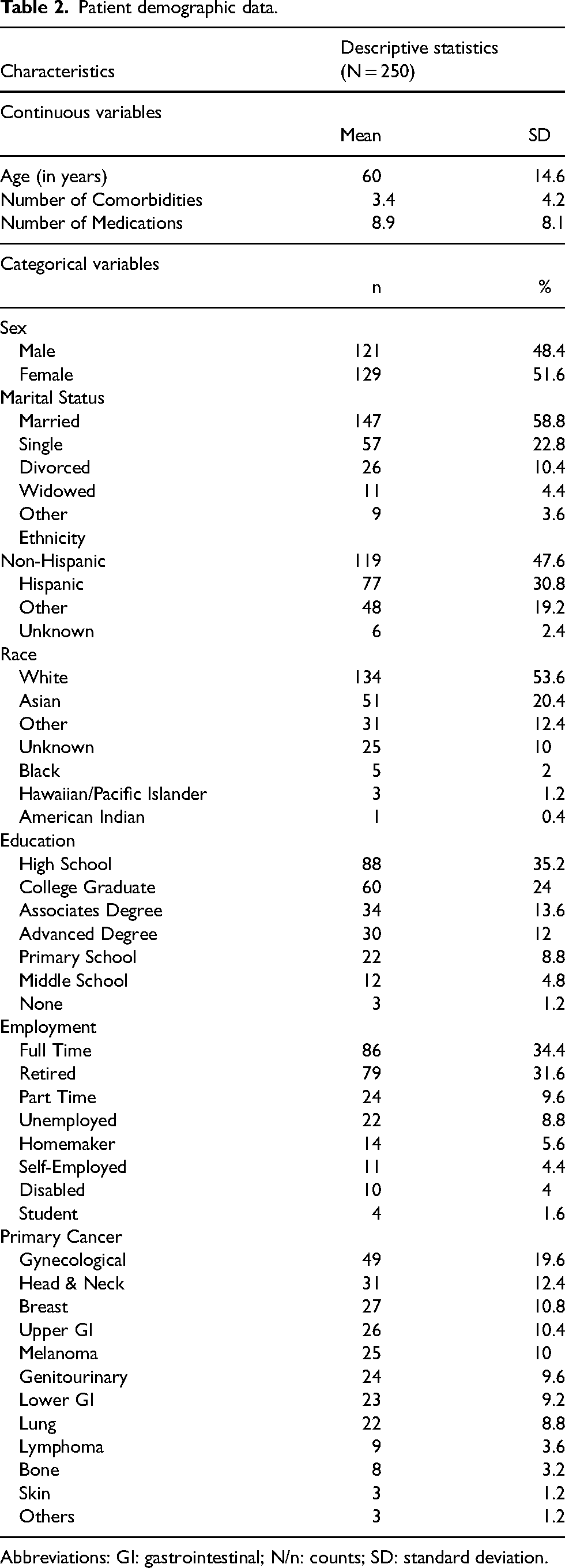
Abbreviations: GI: gastrointestinal; N/n: counts; SD: standard deviation.
Visit interventions and clinically important symptoms.

Pharmacist education
Pharmacist education was performed for 311 (55.1%) of the 564 visits examined throughout the study. Among the 564 documented pharmacist education sessions, most were related to symptoms that were not captured by our ePRO battery and nausea & vomiting, which accounted for 132 (23.4%) and 131 (23.2%) visits, respectively (Table 4). Through the ePRO battery, the least number of pharmacist education was performed for cognitive impairment (15 visits, 2.7%), and depression (24 visits, 4.3%). Among those symptoms that were not captured by the ePRO battery, 40 visits (7.1%) referred to neurological symptoms, 38 (6.7%) to gastrointestinal symptoms, 24 (4.3%) to dermatological symptoms, and 15 (2.7%) to immunological symptoms.
Symptoms requiring pharmacist interventions.
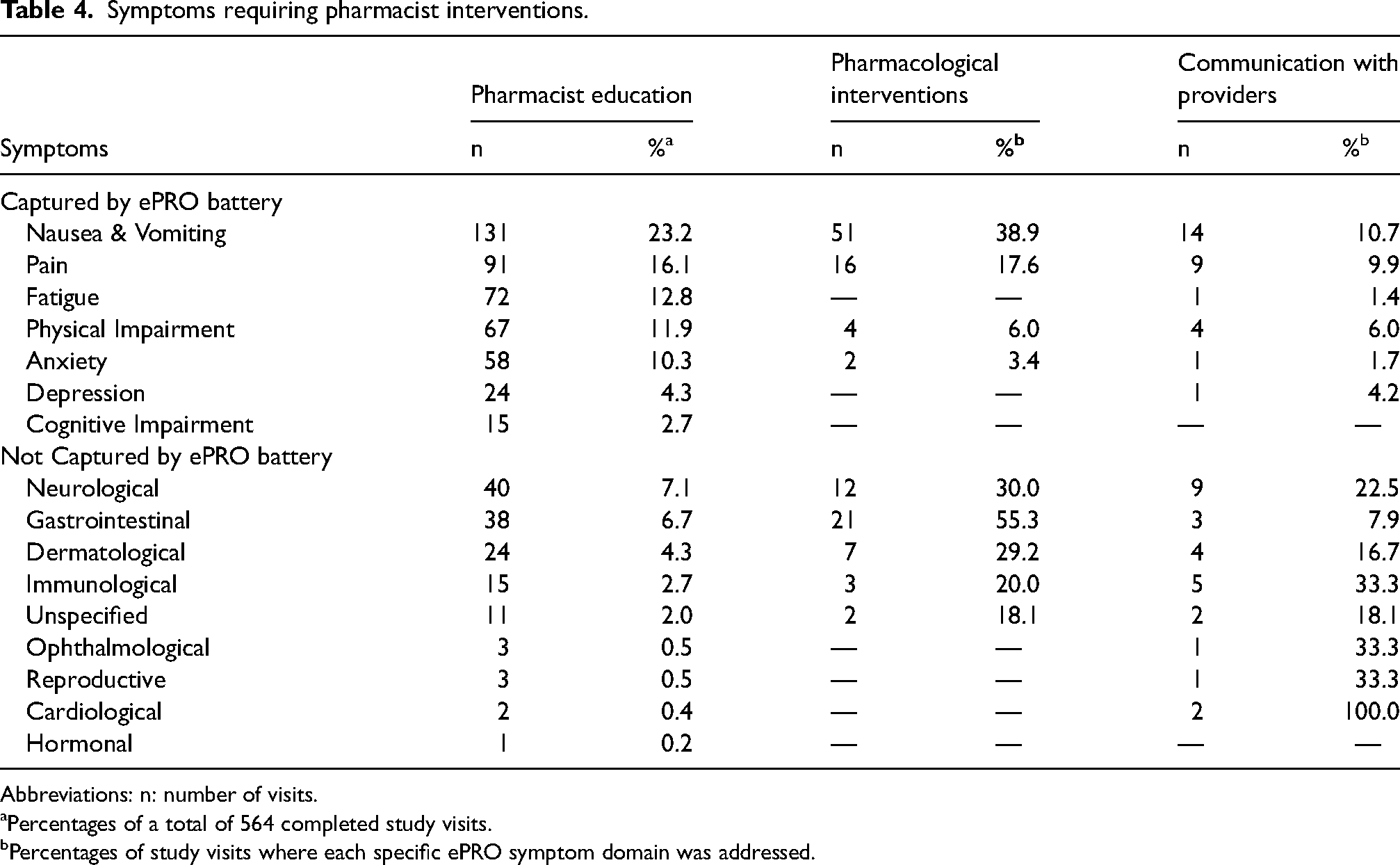
Abbreviations: n: number of visits.
aPercentages of a total of 564 completed study visits.
bPercentages of study visits where each specific ePRO symptom domain was addressed.
Pharmacological interventions
One hundred and six visits (18.8%) required pharmacological interventions. Pharmacological interventions were most likely to be required with the management of nausea & vomiting (51 visits, 38.9%), gastrointestinal symptoms (21 visits, 55.3%), and pain (16 visits, 17.6%) (Table 4). Across the visits requiring pharmacological interventions, antiemetic medications (27.4%) were the most common type of medication documented within the pharmacological interventions, followed by antipsychotic medications (20.8%) as shown in Table 5.
Classification of medications involved in pharmacological interventions using the WHO ATC system.

Abbreviations: n: number of visits.
aPercentages of a total of 106 study visits which recorded ≥1 pharmacological intervention.
Provider communication
Fifty-five visits (9.8%) required provider communication. When symptoms were being identified, nausea and vomiting (14 visits, 10.7%), neurological symptoms (9 visits, 22.5%), and pain (9 visits, 9.9%) were most likely to require communication with providers (Table 4). Thirty-six (62%) involved communication with oncologists and 14 (24%) involved communication with registered nurses or nurse practitioners.
Discussion
In this study, we observed a high rate of pharmacist-led education after the institution of ePRO to identify symptoms in patients undergoing anticancer treatment. The modest number of pharmacological interventions and provider communication performed by pharmacists may be explained by the fact that most patients’ concerns were addressed by providing additional counseling to these patients. It was surprising, however, to observe that a wide variety of symptoms were reported by patients, and they were not captured by the seven preconfigured domains of our ePRO. These findings suggest a need to configure a wider variety of symptoms tailored to specific cancer treatment types, as well as to incorporate the personalization of symptom domains within the assessment battery.
Our preconfigured ePRO greatly assisted pharmacists with patient symptom management and education. The ePRO tool allowed patients to report the symptoms that were prioritized by the investigational team. This in turn allowed pharmacists to develop and discuss tailored management plans in a timely manner that otherwise may not have been discussed if they were not made aware of the patient-reported symptoms before patient counseling. With each recurring visit, pharmacists were able to track patient symptoms and monitor for improvement or decline which then allowed them to personalize consultations for individual patients. The assessment of ePRO scores and subsequent interventions can and should also be performed by other healthcare providers, such as physicians or nurses, to provide multidisciplinary personalized care to the patients.9,10 Nevertheless, our research demonstrates the integral role of a pharmacist in a healthcare team and how beneficial the use of an ePRO tool can be in efficiently managing patient symptoms in resource-limited settings where patients may not always have access to other healthcare providers.
The open communication channel facilitated by the ePRO implementation has further created opportunities to discuss symptoms not preconfigured in the tool, suggesting improved trust in the patient-pharmacist relationship. As we analyzed the other clinically important symptoms that were raised by patients, we did not only observe a diverse range of symptoms but also learned that these symptoms accounted for the most pharmacist education interactions. This was also a concern raised by the patients and providers who were involved in this study. 11 Based on the different types of anticancer treatments that patients were admitted for, future ePRO tools could be implemented for certain anticancer treatment plans and adjusted to account for the most common ‘other’ symptoms reported by patients. Having an ePRO tool that is more specific to the type of anticancer treatment a patient is receiving would allow patients to answer the most relevant symptom-related questions and thereby reduce the questionnaire burden. 11 This would ensure that the most pertinent symptoms for each cancer treatment type are specifically addressed by the pharmacist systematically. 12
It was not surprising to observe that antiemetic medications were most involved in pharmacists’ interventions in this study, as nausea and vomiting were one of the most frequently reported patient symptoms. Among symptoms that were not captured by ePRO, we have noticed that the neurological symptoms category encompassed the largest number of symptoms not listed under the seven ePRO domains and as a result the nervous system anatomical class medications were also the second most intervened by pharmacists. However, not all the nervous system medications prescribed to our patients were given to manage neurological symptoms. One example of this which was observed in our study pertained to the use of olanzapine, which is categorized as an antipsychotic medication. Olanzapine was likely given for the prevention of chemotherapy-induced nausea and vomiting in cancer patients, specifically in those patients who were not receiving olanzapine and experienced nausea and vomiting. 13 On the other hand, medications were not intervened as frequently for symptoms such as fatigue, anxiety, or physical impairment, likely because effective pharmacological therapies are lacking for these symptoms.
Through our interventions, pharmacists were also able to communicate with healthcare providers mostly consisting of oncologists and nurses or nurse practitioners regarding symptoms that our patients were experiencing. In our study, we have observed that pharmacists communicate with providers to address symptoms such as nausea/vomiting and pain. There were fewer communications regarding other symptoms such as anxiety, depression, and fatigue. Although there was not a high prevalence of provider communication deemed necessary by pharmacists to perform in this study, the instances which required provider communication greatly helped in making sure that the patient was given the most appropriate interventions to address their symptoms.
The strengths of this study include identifying and addressing patient-reported symptoms of concern relating to anticancer treatment in patients from underserved populations and diverse backgrounds. Many of the participants in this study may not have had access to the same patient-reporting tools and high quality of treatment if we had conducted the study in a different location. This study allows patients to be active participants in their own healthcare experience and to voice their concerns and symptoms so that they can be efficiently and effectively addressed by their healthcare team. The role of a pharmacist was also highlighted in this study as a key healthcare provider who can make a great difference in patients’ anticancer treatments. The limitations of the study would be the limited number of patients enrolled and the specificity of ePRO domains assessed. Future studies should consider incorporating a larger sample size of patients as well as a greater number of relevant ePRO domains to include a wider range of patient-reported symptoms which are tailored to each type of anticancer treatment patients are receiving.
Conclusion
In conclusion, we observed a high rate of pharmacist-led education after the institution of our ePRO battery to evaluate symptoms in patients undergoing anticancer treatment. Our ePRO allowed identification of several symptoms such as nausea and vomiting and pain that required pharmacological interventions and provider communications. However, a number of other symptoms also surfaced through patient interactions. Our ePRO battery platform has also enhanced the role of a pharmacist in symptom management, allowing oncology pharmacists to provide timely interventions and to communicate with the healthcare team. Future studies should evaluate the implementation of specific ePRO tools catering to specific anticancer treatments to better address patient-reported symptoms.
Footnotes
Acknowledgements
We acknowledge Dr Kaleem Anwar, Ms Gretchen Avilar, Mr Ryan Baniel, Ms Daniella Chan, Ms Mary Dagsi, Dr Yu Ke, Ms Chelsea McKinney, Ms Reem Nasr, Ms Kayleen Ports, Dr Lari Wenzel and all the pharmacists at the CFCCC infusion center pharmacy for their assistance with this study.
Author contributions
Conception and methodology: Alexandre Chan, Ding Quan Ng; Financial support: Alexandre Chan; Administrative support: Alexandre Chan; Provision of study materials or patients: Alexandre Chan; Collection and assembly of data: All authors; Data analysis and interpretation: All authors; Manuscript writing: All authors; Final approval of manuscript: All authors; Accountable for all aspects of the work: All authors.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Hematology Oncology Pharmacy Association (HOPA) for providing research funding for this study.
Ethical approval and informed consent statements
The primary study protocol received ethics approval from the UCI Institutional Review Board (#2021-6431).
