Abstract
Introduction
Cisplatin has ubiquitous use throughout many cancer types and is incorporated in various regimens. Nephrotoxicity is a well-known side effect of cisplatin, occurring in up to one third of patients. Early clinical data showed elevated peak concentrations are correlated with increased incidence of nephrotoxicity, suggesting prolonging infusion duration may abate this known toxicity. A recent study showed there was no difference in rates of acute kidney injury (AKI) between rapid (1-h) and standard (3-h) infusions of cisplatin, however, cisplatin was administered inpatient with prolonged and aggressive hydration methods. Therefore, we aimed to compare outcomes in patients who received rapid-infusion vs. standard-infusion cisplatin in the outpatient setting at our institution.
Methods
This single center, pre-post intervention study evaluated outpatient visits between January 2022 and March 2024. Cisplatin was administered over 3-h at Mays Cancer Center, UT Health San Antonio, prior to an institutional practice change to 1-h (rapid infusion) that took place February 2023. The co-primary outcomes were the maximum decrease in eGFR from baseline and the incidence of stage 1 or higher AKI. Secondary outcomes included rates of treatment modification and rates of healthcare resource utilization.
Results
A total of 53 patients with a chemotherapy regimen containing cisplatin met study criteria. Baseline characteristics were similar for the standard (n = 28) and rapid infusion (n = 25) groups. The maximum decrease in eGFR in the standard infusion vs. rapid infusion group did not differ (15.2 vs 18.0 mL/min/1.73 m2, respectively; p = 0.89). The incidence of stage 1 or higher AKI was not significantly different between the standard and rapid infusion cohorts (p = 0.13). No differences were identified for the secondary outcomes including rates of regimen modifications or hospitalizations due to AKI or time to AKI in 120 days after the first cisplatin dose.
Conclusion
In this pre-post intervention analysis neither occurrence nor severity of nephrotoxicity appeared to be affected by the infusion rate of cisplatin in the outpatient setting. Rapid infusion of cisplatin over 1-h appears to be a feasible option without increased safety risk for patients.
Objective
The purpose of this pre-post intervention study was to compare nephrotoxicity outcomes in patients who received rapid or standard infusion of cisplatin in the outpatient setting.
Introduction
Cisplatin is a chemotherapeutic agent used in the treatment of multiple types of cancer. Nephrotoxicity induced by cisplatin is a common adverse event that limits its utility. 1 Cisplatin has many mechanisms of causing acute kidney injury (AKI) including oxidative stress, inflammation, and proximal tubular injury. 2 Numerous strategies to circumvent cisplatin-induced nephrotoxicity have been studied. Hydration and forced diuresis reduce cisplatin half-life and urinary cisplatin concentration to prevent AKI. A systematic review found that providing low-volume hydration with potassium and magnesium supplementation over a short-duration is safe and feasible for limiting cisplatin-induced AKI. 3
There is no consensus on cisplatin infusion duration. The manufacturers’ package inserts recommend a 4–8 h infusion. 4 However, study-specific protocols detail rapid-infusion rates as short as 30 min and continuous infusion rates as well. 5 Pharmacokinetic studies indicate a correlation between peak plasma concentrations of cisplatin and nephrotoxicity.6,7 Therefore, rapid infusion of cisplatin could result in an increased peak concentration and ultimately lead to increased incidence of nephrotoxicity. Shorter infusion duration could potentially reduce time spent at outpatient visits and optimize chemotherapy administration sequencing, patient care and ultimately improve quality of life.
To date, only one clinical study has been published evaluating rapid cisplatin infusion of 1-h compared to 3-h infusions. 8 This study investigated renal function changes in patients with lung cancer receiving cisplatin. This study evaluated patients in the inpatient setting along with prolonged aggressive hydration with normal saline. Results from this study support the use of rapid-infusion cisplatin as the standard of care in this patient population. 8 However, cisplatin was administered inpatient and patients received continuous hydration for 24 h, which may not appropriately represent an outpatient population receiving cisplatin.
It is unclear whether the infusion rate of cisplatin affects the incidence of cisplatin-induced nephrotoxicity in patients with other types of cancer or who receive different hydration and diuresis regimens. Moreover, because cisplatin is most frequently administered in the ambulatory setting, it remains unknown if a rapid-infusion rate of cisplatin is clinically sound. Therefore, the purpose of this study was to compare nephrotoxicity outcomes in patients who received rapid- or standard-infusion of cisplatin for different cancer diagnoses in the outpatient setting.
Methods
Study design
This study was a retrospective pre-post intervention cohort study of patients at UT Health San Antonio - Mays Cancer Center (MCC), an NCI-Designated Cancer Center, who received cisplatin during an outpatient visit between January 2022 and March 2024. Patient consent for inclusion in the study was waived. Cisplatin was administered over 3-h (standard infusion) at MCC prior to an institutional practice change to 1-h (rapid infusion) in February 2023. All patients received cisplatin in the outpatient setting with pre- and post-hydration according to institution protocol. Patients were given 1 liter of normal saline intravenously administered over 60 min both before and after the cisplatin infusion. The infusion time of normal saline was in addition to the cisplatin infusion. Potassium and/or magnesium supplementation was added if patients had abnormal electrolytes resulted on the lab draw just prior to infusion. Fluid administration time was extended if electrolyte supplementation was added to ensure that no more than 10 milliequivalents of potassium were infused per hour. Patients also received intravenous dexamethasone 12 mg, fosaprepitant 150 mg, and palonosetron 0.25 mg for antiemesis prior to cisplatin.
Patients were included if they had a diagnosis of lung, bladder, or head and neck cancer and were receiving a cisplatin-containing chemotherapy regimen at the start of treatment. The patient's initial dose of cisplatin must have been at least 45 mg/m2. Patients must have had two serum creatinine (SCr) values available prior to the first dose of cisplatin.
Patients were excluded if they did not have a SCr value available within 21 days after a cisplatin dose. Patients who had an estimated glomerular filtration rate (eGFR) of <30 mL/min/1.73 m2 or classified as having chronic kidney disease (CKD) stage 4 prior to receiving cisplatin were also excluded. Patients who received more than six cycles of cisplatin or who previously received cisplatin in the past year of inclusion date were excluded from the study. Patients on clinical trial or receiving investigational therapy were excluded as well.
Baseline characteristics were collected for all patients included. Demographic characteristics included age, sex, race/ethnicity, weight, height, BMI, and baseline SCr. Baseline SCr was defined as the average of the patient's two most recent SCr values recorded prior to starting cisplatin therapy. This baseline SCr value was used to calculate the patient's baseline eGFR using the Modification of Diet in Renal Disease (MDRD) equation. The baseline eGFR value was used to determine the patient's baseline CKD stage using the Kidney Disease Improving Global Outcomes (KDIGO) 2012 CKD guidelines.
Treatment characteristics collected included cancer diagnosis, cisplatin dose, adjunctive therapies such as mannitol and/or electrolyte supplementation, concurrent chemotherapy given as part of the treatment regimen, number of treatment cycles, cumulative cisplatin dose, and dose frequency.
Outcomes
The primary outcomes were the maximum decrease in eGFR from baseline and the incidence of AKI stage 1 or higher for rapid infusion cisplatin (1 h) compared to standard infusion cisplatin (3 h). Acute kidney injury stages were defined in accordance with KDIGO 2012 guideline on AKI (Table 1). Renal function measures were collected after each cisplatin dose, and the maximum difference in eGFR for each patient after completion of their cisplatin treatment plan was calculated.
KDIGO 2012 staging of AKI.

The secondary outcomes included interventions to modify the cisplatin treatment regimen due to toxicity such as discontinuation of cisplatin, additional hydration appointments, and dose reductions. If dose reduction occurred, the percent of dose decrease was recorded. If treatment was discontinued due to toxicity, this was recorded. For every cisplatin treatment, the earliest SCr value after the dose (within 21 days) was collected. Other secondary outcomes included any hospital and/or emergency department visit due to toxicity, the maximum increase in SCr from baseline, and time to AKI within 120 days after the first cisplatin dose. If the patient had an emergency department (ED) or hospitalization record during treatment, the physician note was reviewed to determine the reason for admission. Primary problems for admission including volume overload, decreased urine output, or flank pain were recorded as ‘hospital/ED visit for nephrotoxicity.’ To account for potential confounding factors that may contribute or prevent AKI, a multivariate analysis was performed.
Statistical analysis
Student's t-tests were used for normally distributed continuous data, and Wilcoxon-rank sum tests were used for non-normally distributed continuous data. A chi-square or Fisher's exact test was used to compare categorical data, including demographic characteristics and outcomes. A multivariable linear regression analysis for the primary outcome of maximum decrease in eGFR from baseline was conducted. Age, baseline eGFR, cumulative cisplatin dose, receipt of mannitol, along with the infusion group were entered into the model. Risk for AKI by infusion group in 120 days was evaluated using Kaplan Meier analysis with log-rank tests. To assess association of infusion groups, hazard ratios (HRs) were calculated with 95% confidence intervals (CIs) using Cox-proportional hazards model and adjusted for age, baseline eGFR, receipt of mannitol and cumulative cisplatin doses up to after 6 cycles. SPSS 29.0 (IBM Corp, Armonk, NY, USA) was used for all statistical analyses. Statistical significance was calculated using a p-value of < 0.05.
Results
A total of 59 patients with lung, bladder, or head and neck cancer received their first dose of a cisplatin-containing regimen between January 1, 2022 and March 31, 2024. Four patients were excluded due to being concurrently enrolled in a clinical trial and receiving an investigational therapy, and two patients were excluded due to baseline eGFR classifying them as CKD stage 4.
A total of 53 patients were included in the final analysis. The demographic characteristics (Table 2) did not differ between the rapid and standard infusion cohorts. Baseline renal function, as well as treatment characteristics were also similar among the two groups. There were 5 unique chemotherapy regimens in the 1-h infusion group with the most common being gemcitabine (32%), etoposide (20%), and docetaxel with 5-fluorouracil (20%). Nine different chemotherapy regimens were used in the 3-h infusion group including docetaxel, methotrexate, and vinblastine (29%), etoposide (21%), and gemcitabine (18%).
Baseline characteristics.
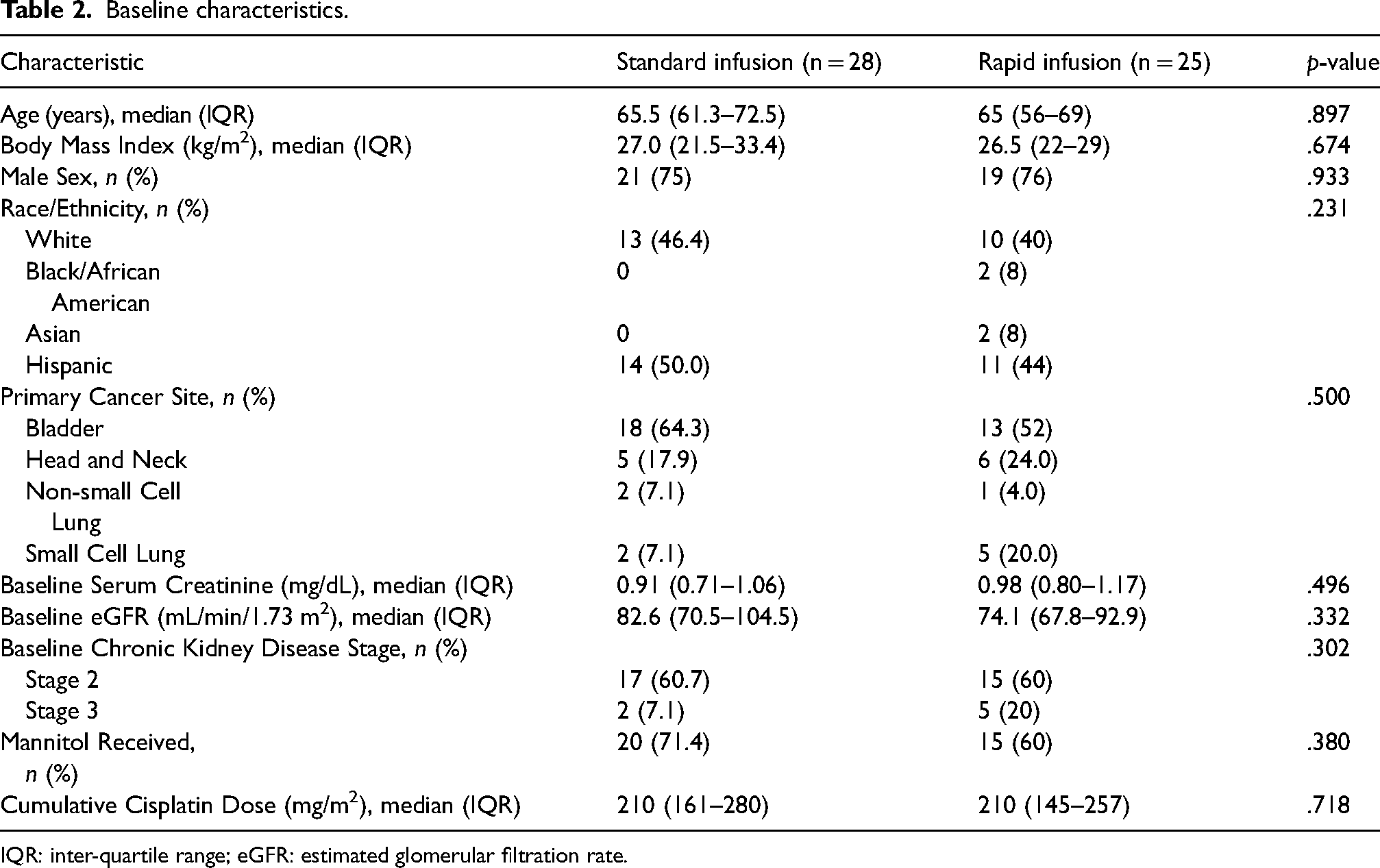
IQR: inter-quartile range; eGFR: estimated glomerular filtration rate.
For the primary outcome, the mean maximum decrease in eGFR was not significantly different between the rapid infusion and standard infusion group (−18.0 vs. −15.2 mL/min/1.73 m2, p = .89). A student's t-test found no significant difference between the rapid and standard infusion cohorts in the incidence of stage 1 or higher AKI (32% vs. 22%, p = .13) (Figure 1).
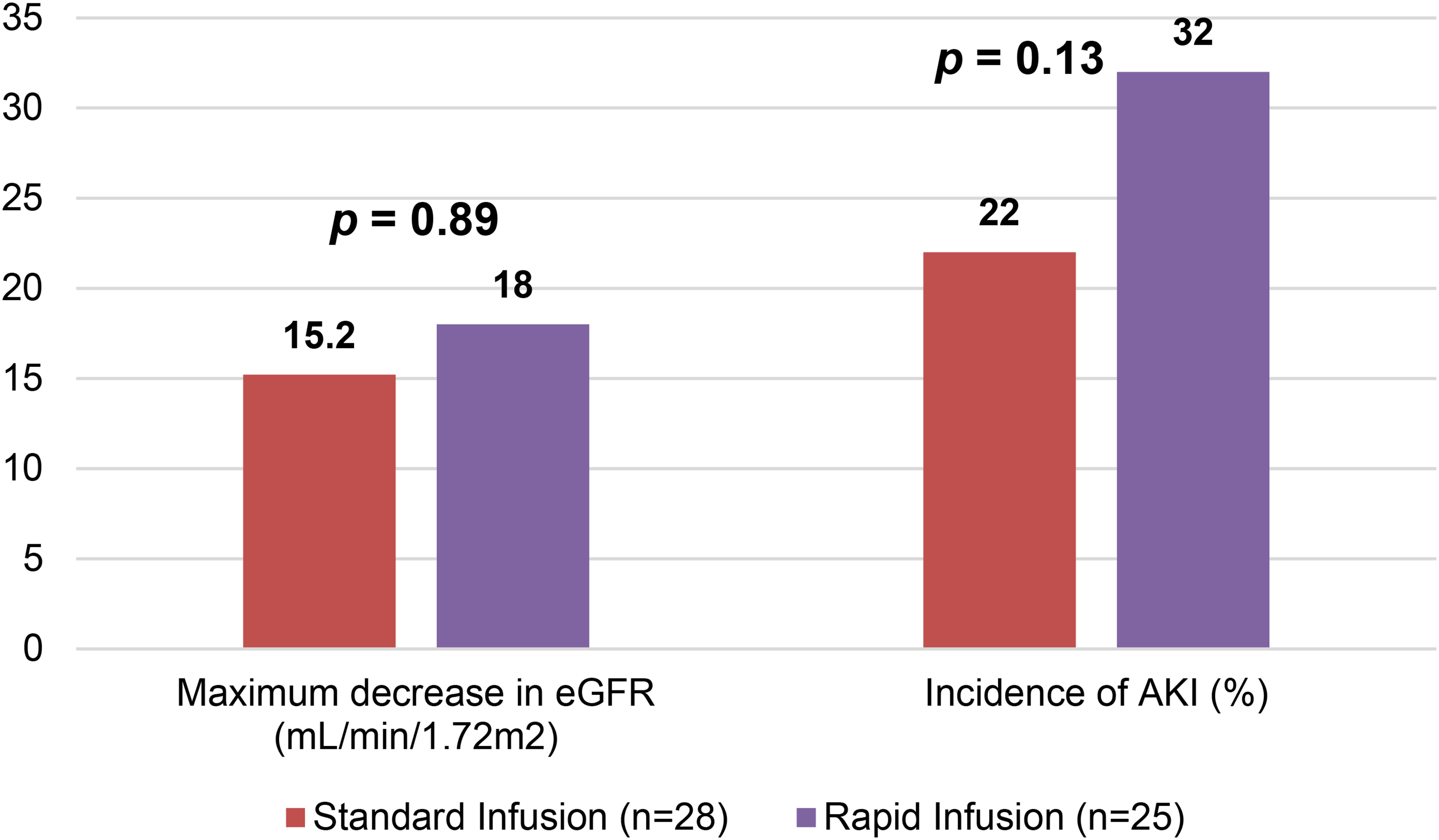
Primary outcomes.
There was no significant difference in cisplatin dose reduction or discontinuation between the two groups. In patients that did undergo a dose reduction, the median cisplatin dose reduction was 25% (4–30) in the rapid infusion group and 47% (17.5–57.5) in the standard infusion group (p = .56). Patients in the rapid and standard infusion groups had no significant difference in additional hydration appointments required (12% vs. 17.9%, p = .55) or hospital/ED visits due to toxicity (8.0% vs. 7.1%, p = .91). The maximum increase in baseline SCr was also not significantly different between the two cohorts (1.24 vs. 1.20, p = .49) (Table 3).
Secondary outcomes.
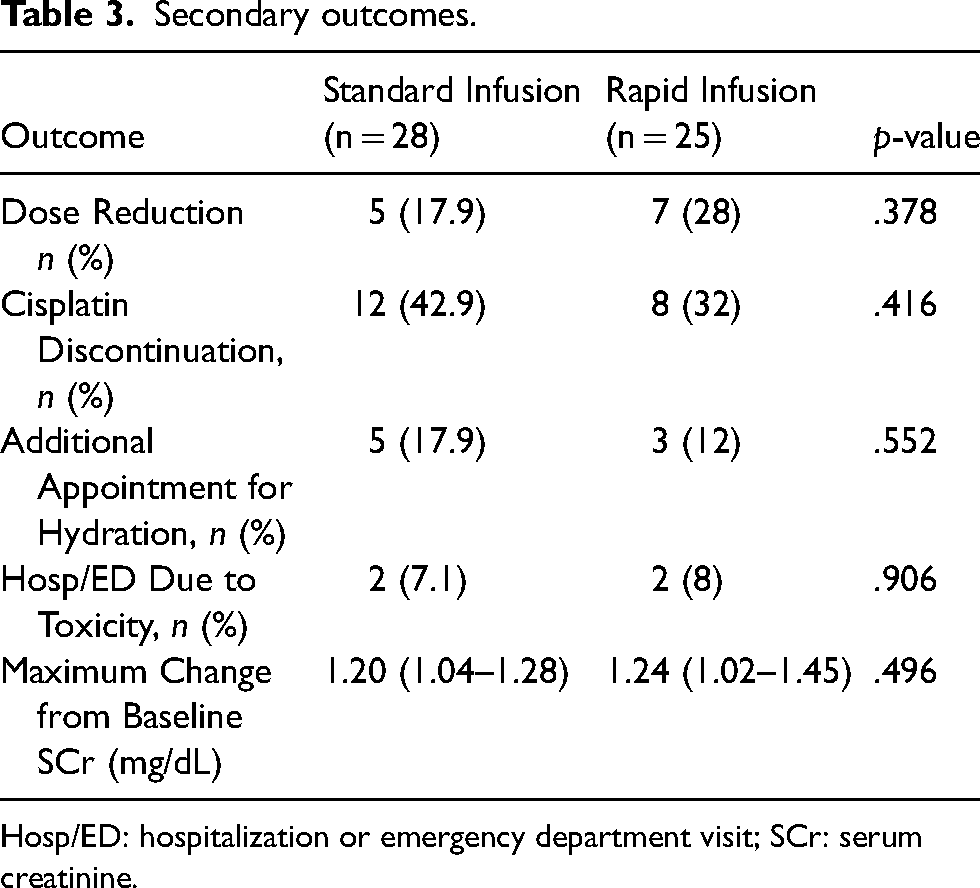
Hosp/ED: hospitalization or emergency department visit; SCr: serum creatinine.
The multivariate analysis showed that each year of increase in age was positively associated with a decrease in eGFR from baseline (OR = .344, 95% CI = .036–.652, p = .029). Lower baseline eGFR was positively associated with a decrease in eGFR (OR = .237, 95% CI = .087–.387, p = .002). Cumulative cisplatin dose was also positively associated with a decrease in eGFR from baseline (OR = .047, 95% CI = .005–.088, p = .028). Infusion time, race, and electrolyte supplementation were not associated with risk of AKI (Table 4).
Multivariate analysis of confounding variable.

eGFR: estimated glomerular filtration rate.
The time to AKI at 120 days after the first cisplatin dose was not significantly different between the 1-h and 3-h infusion groups (Figure 2). The median time to AKI at 120 days was 63 days in the 1-h infusion group and 52.5 days in the 3-h infusion group.
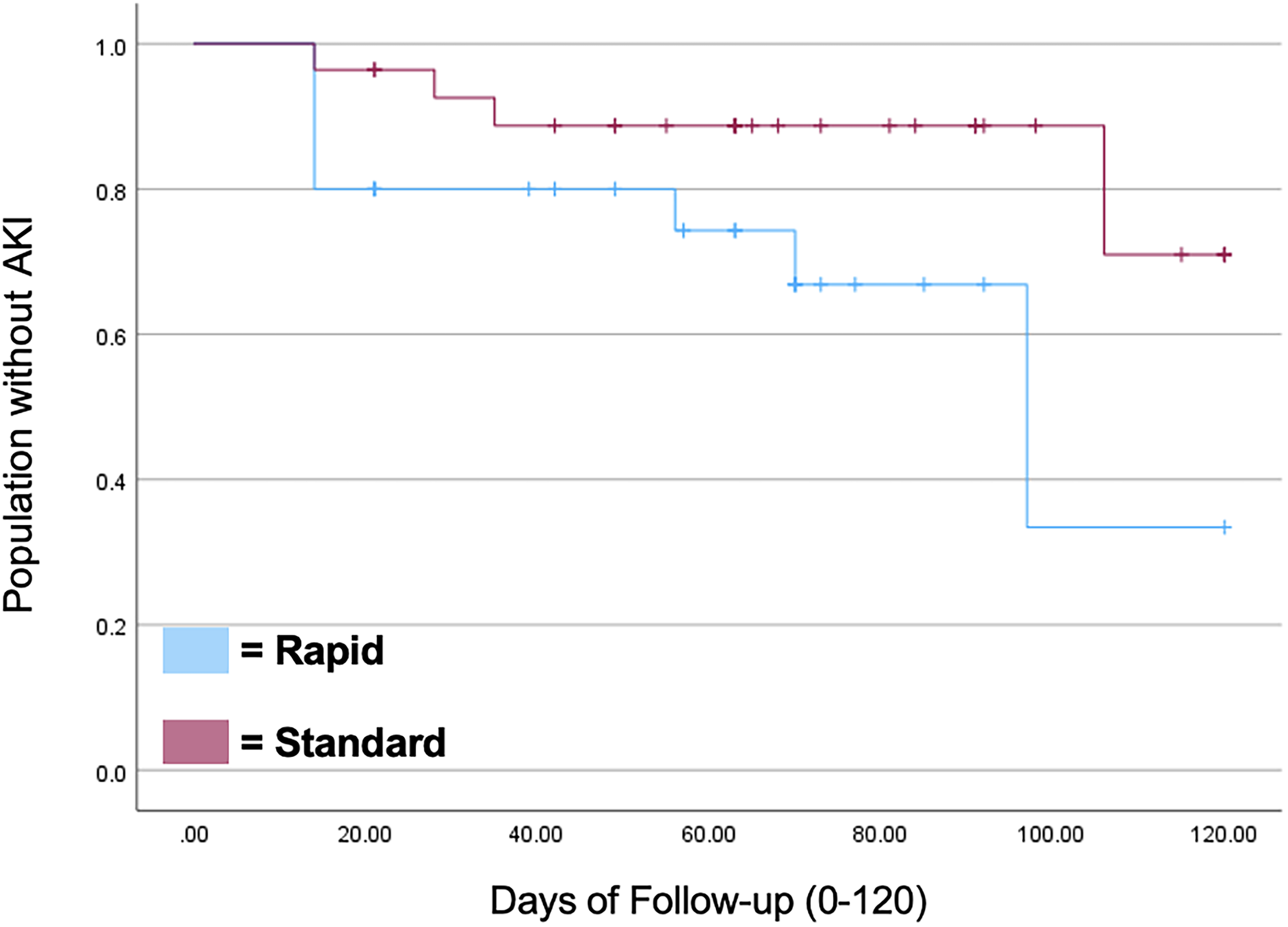
Kaplan-Meier curve of acute kidney injury occurrence within 120 days of first cisplatin dose.
The Cox proportional hazards model showed that age, baseline eGFR, and receiving mannitol were not significantly associated with risk of AKI development in 120 days after first cisplatin dose (Table 5).
Cox proportional hazards model for AKI occurrence within 120 days.
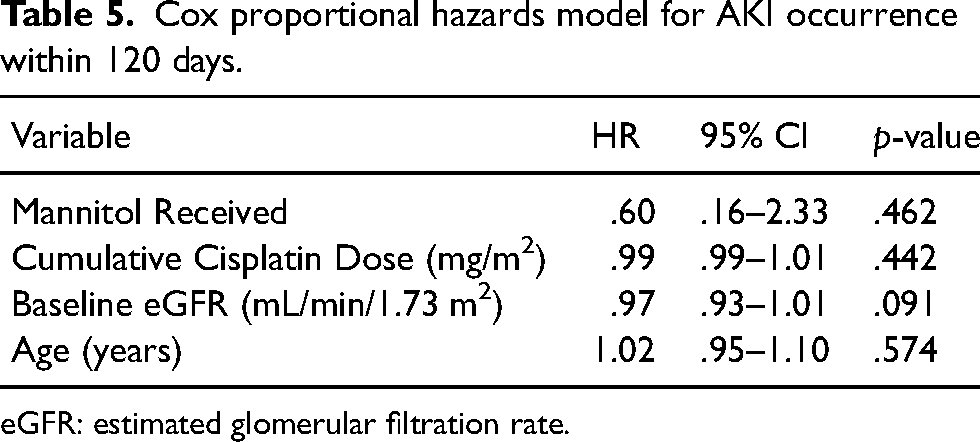
eGFR: estimated glomerular filtration rate.
Discussion
To the authors’ knowledge, this is the first study to compare rapid and standard infusion rates of cisplatin in the outpatient setting. These results align with the only other study to investigate the relationship between infusion duration and cisplatin-induced nephrotoxicity. 8 The similarities and differences between the two studies are worth noting. The results of both studies demonstrate that renal function is not affected by the infusion rate of cisplatin. However, the study by Mense and colleagues only included patients with lung carcinoma. Our study differs by including patients with bladder, lung, or head and neck cancer to better represent the ubiquitous use of cisplatin. Furthermore, our study included patients who received at least 1 dose of cisplatin during the study period and evaluated their renal function after each dose. The previous study's primary outcome of eGFR after 3 cisplatin doses may not account for renal function changes or recovery after the first two doses. Lastly, our study performed additional analyses to determine time to AKI and potential patient risk factors for AKI development.
Mense and colleagues included patients that received aggressive and standardized hydration regimens before and after each cisplatin infusion. 8 They found the incidence of nephrotoxicity in both rapid and standard infusion groups similar to those of other studies, ranging between 20–30%. 9 In our study, although the incidence of AKI in the rapid infusion group (32%) did not significantly differ from the standard infusion group (22%), the higher incidence in the rapid infusion group could be explained by a number of different reasons. There was a variety of hydration schedules used for our patients with different electrolytes supplemented, addition of mannitol, and additional hydration appointments documented. Additionally, the rapid infusion group had a higher number of patients with baseline CKD stage 3 (20% vs. 7.1%, p = .302) which puts those patients at higher risk for acute renal decline. 10 Our study also defined stage 1 AKI as an increase of 0.3 or greater in SCr from baseline which is a lower threshold for AKI development than the previous study (defined as 1.5–1.9 times greater than baseline). A study by Kang and colleagues found that stage 1 AKI development was an independent predictor of all-cause mortality in patients with cancer. 11 Their study utilized the same definition of a 0.3 or greater increase from baseline SCr when determining the association with all-cause mortality. Therefore, using the lower threshold to identify AKI allows for a more inclusive understanding of this complex patient population.
In this group of patients, age, baseline eGFR, and cumulative cisplatin dose were found to be positively associated with a decrease in eGFR from baseline. Multiple studies have explored risk factors associated with cisplatin-induced AKI, however, many of them have conflicting results. One study found that age was not associated with AKI development from cisplatin in their population. 12 Another study reports age as a significant risk factor but did not find a relationship between baseline eGFR and AKI development. 13 In addition, the study found cisplatin dose to be a risk factor for cisplatin-induced AKI but did not explore the relationship between cumulative cisplatin dose with AKI incidence.
This study is the first to analyze the difference in time to AKI between the two infusion groups. As seen in Figure 2, patients in both the rapid and standard infusion group developed an AKI within 20 days after their first cisplatin dose. The timing of AKI aligns with the study results from Oda and colleagues who found that the majority of patients developed an AKI in 7–9 days after cisplatin treatment. 12 Although the results show that there was no significant difference in time to AKI in 120 days, the p-value of 0.059, indicates a potential trend towards significance. Furthermore, we performed a Cox proportional hazards model to identify patient risk factors for earlier AKI development. Age and baseline eGFR were not associated with time to AKI. Further analyses will be needed to determine if other patient characteristics are risk factors for faster time to AKI in this population. Mannitol did not negatively affect the time to onset of AKI when administered with cisplatin. This reinforces the data that mannitol is protective against cisplatin-induced AKI in certain populations. 14
While our study explored many variables to determine the safety of rapid infusion cisplatin, there are many limitations that should be considered. First, due to the sample size, the study may not have enough power to detect differences between the regimens, a limitation that was not addressed in the previous study. The inclusion of multiple carcinoma types made it difficult to control for all adjunct chemotherapy regimens used with cisplatin without having too many categories. Furthermore, patient comorbidities were not collected from the electronic health record. Both diabetes and hypertension have been associated with cisplatin-induced AKI in other studies. 15 The authors acknowledge the limitation of not including these variables. Additionally, by screening for emergency department or hospitalizations during treatment using the primary problem of volume overload could result in visits that are not specific to cisplatin-induced AKI. Conditions such as heart failure, cirrhosis, and baseline CKD can predispose patients to a volume overload state that may not be cisplatin-induced. Lastly, although this publication intends to give a better understanding of outcomes in patients receiving rapid-infusion cisplatin across a broad range of patients, this does introduce heterogeneity in patient population, therapies, and interventions.
Future studies with larger sample sizes are needed to confirm the safety of rapid-infusion cisplatin in the outpatient setting. Addressing the limitations in the present study by accounting for comorbidities and adjunct chemotherapy agents will be helpful in providing a holistic picture of which population will benefit from shorter infusion duration without additional risks.
Conclusions
Our study further demonstrates that neither the severity nor incidence of nephrotoxicity is affected when shortening cisplatin infusion duration from 3 h to 1 h. By including a heterogeneous population of patients with different cancers, the results support the introduction of rapid infusion cisplatin even in the absence of aggressive hydration and inpatient monitoring to save patients time and healthcare utilization costs. The potential to decrease chair time by 2 h with each rapid infusion of cisplatin can help patient and institution buy-in. Additional research is recommended to ensure external validity and provide understanding of the long-term effects of rapid infusion cisplatin.
Footnotes
Author contributions
EA: Conceptualization, Methodology, Investigation, Writing – Original Draft. JGH: Conceptualization, Methodology, Validation, Writing – Review and Editing, Supervision. GCL: Software, Formal Analysis.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and informed consent statement
The Ethics Committee of the Mays Cancer Center, UT Health San Antonio, waived the need for ethics approval and patient consent for the collection, analysis and publication of the retrospectively obtained and anonymized data for this non-interventional study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
