Abstract
Background
The practice of paediatric oncology pharmacy (POP) has been in existence in Africa for several decades. However, most POP practitioners lack formal education and training. Training and credentialing of pharmacists requires the availability of an appropriate curriculum developed to meet local needs. Hence, the African POP technical working group (TWG) of the International Society of Paediatric Oncology (SIOP) embarked on a process to develop a curriculum in POP.
Method
The curriculum development followed a modified Delphi approach: the formalisation of the POP TWG, the initial development of the curriculum, community and stakeholder engagement via a modified Delphi scoring, and the finalisation of the curriculum at the pharmacy workshop of the SIOP Africa Congress in South Africa.
Results
A POP TWG of 23 members with diverse experiences was formed. The initial curriculum, focusing on 36 theoretical and 5 clinical components, was developed. The modified Delphi process restructured the 36 theoretical contents to 27, the 5 clinical contents to 9, and introduced 6 research modules. The theoretical and clinical components were assigned 86 and 240 credit hours respectively, with a research component culminating in a published manuscript. Twenty-eight individuals from 13 countries attended the workshop, resulting in unanimous approval of the curriculum.
Conclusions
The SIOP Africa Pharmacy TWG has developed a POP curriculum specific to Africa. The implementation of the curriculum through African universities and specialist colleges is imperative to reduce the education and training gaps in POP.
Background
Africa faces unique healthcare challenges that make it impractical and inappropriate to adopt a paediatric oncology pharmacy (POP) curriculum directly from high-income countries (HICs). A significant burden of childhood cancer exists, with many cases in Africa diagnosed at advanced stages due to limited awareness, diagnostic delays, and inadequate healthcare infrastructure.1–3 Additionally, the number of trained pharmacists, particularly those specialising in paediatric oncology, is extremely low, and in many countries, they do not exist. 4 The majority of African nations are classified as low-income or lower-middle-income countries,5,6 meaning healthcare resources, including access to essential medicines and diagnostic tools, are often scarce. Furthermore, African healthcare systems are heavily burdened by communicable and non-communicable diseases which compete for already limited resources. 7 A curriculum designed for well-resourced settings fails to account for these challenges, making it essential to develop a context-specific curriculum that prepares pharmacists to navigate the realities of paediatric oncology care in Africa.
The African continental branch of the International Society of Paediatric Oncology (SIOP) known as SIOP Africa comprises physicians, nurses, pharmacists, nutritionists, members of civil society and other professionals in paediatric oncology. It aims to improve and disseminate knowledge of childhood malignant diseases and their management in African countries. The SIOP Africa paediatric oncology pharmacy group was formed at the 14th SIOP Africa conference in March 2022 in Kampala, Uganda to improve the standards of POP in Africa. 8
The management of children with cancer in many African countries is led by adult oncologists, paediatricians or non-specialist physicians. 9 Chemotherapy admixture and administration are often left to nurses, physicians or untrained pharmacists. 9 Although these pharmacists may have extensive experience in POP, they have not benefited from formal education, training and certification in the field. Available POP textbooks and specialist courses are tailored for high-income country audiences and hardly address challenges faced by pharmacists attempting to provide specialised care in highly resource-constrained settings. 9
Curriculum development is a key strategy for overcoming the challenges of education, training, and certification faced by healthcare professionals in Africa, including pharmacists specialising in paediatric oncology.10,11 Current practices in POP and expectations from pharmacists themselves and the healthcare community should be key considerations in developing a standard curriculum that responds to the needs of Africa.12,13
Several techniques have been utilised in curriculum development with the ultimate goal of producing well-rounded professionals capable of delivering on their professional responsibilities.13,14 To produce dynamic professionals, various curricula have adopted general, career-specific and technical contents.15–17 Traditional curriculum development approaches have included the search for existing relevant curricula, identifying curriculum gaps and taking steps to develop enhanced curricula that address these gaps. 13 These curriculum development approaches have been successful in producing curricula that augment professional growth and development.18–20 Curriculum development essentially encompasses needs assessment, goal setting, syllabus design, methodological/materials considerations, testing/teaching and evaluation.21,22 Nevertheless, the dynamic nature of curriculum development permits scientifically justifiable modifications of recommended approaches. However, goal-setting remains an integral element in curriculum development. 13
In addition, community and stakeholder acceptance of curricula remains a critical aspect in ensuring the successful implementation, sustainability and relevance of paediatric oncology pharmacy training in Africa, fostering greater integration into healthcare systems and improving patient outcomes.23,24 Conducting a needs assessment before curriculum development stimulates community and stakeholder interest and commitment.25,26 SIOP Africa highlighted the relevance of POP education and training in achieving the World Health Organisation (WHO) Global Initiative for Childhood Cancer (GICC) goals. 9 The African Community of SIOP, through a review of literature and conference proceedings, affirmed the limited exposure and experience to paediatrics and by extension, paediatric oncology to the clinical pharmaceutical workforce. 9 The need for an African POP curriculum was identified in collaboration with community and stakeholder engagement to further strengthen the call for specialised paediatric oncology pharmaceutical service delivery.
Based on the established needs 9 and the need to set an explicit goal in the development and standardisation of a curriculum,21,22 the SIOP Africa POP technical working group (TWG) embarked on the development of an African POP curriculum to enhance the delivery of safe and advanced POP services across Africa. This manuscript details the processes and rationale of the creation of a POP curriculum specific to the African setting.
Methodology
The methodology of the African POP curriculum development has been presented: In Figure 1.
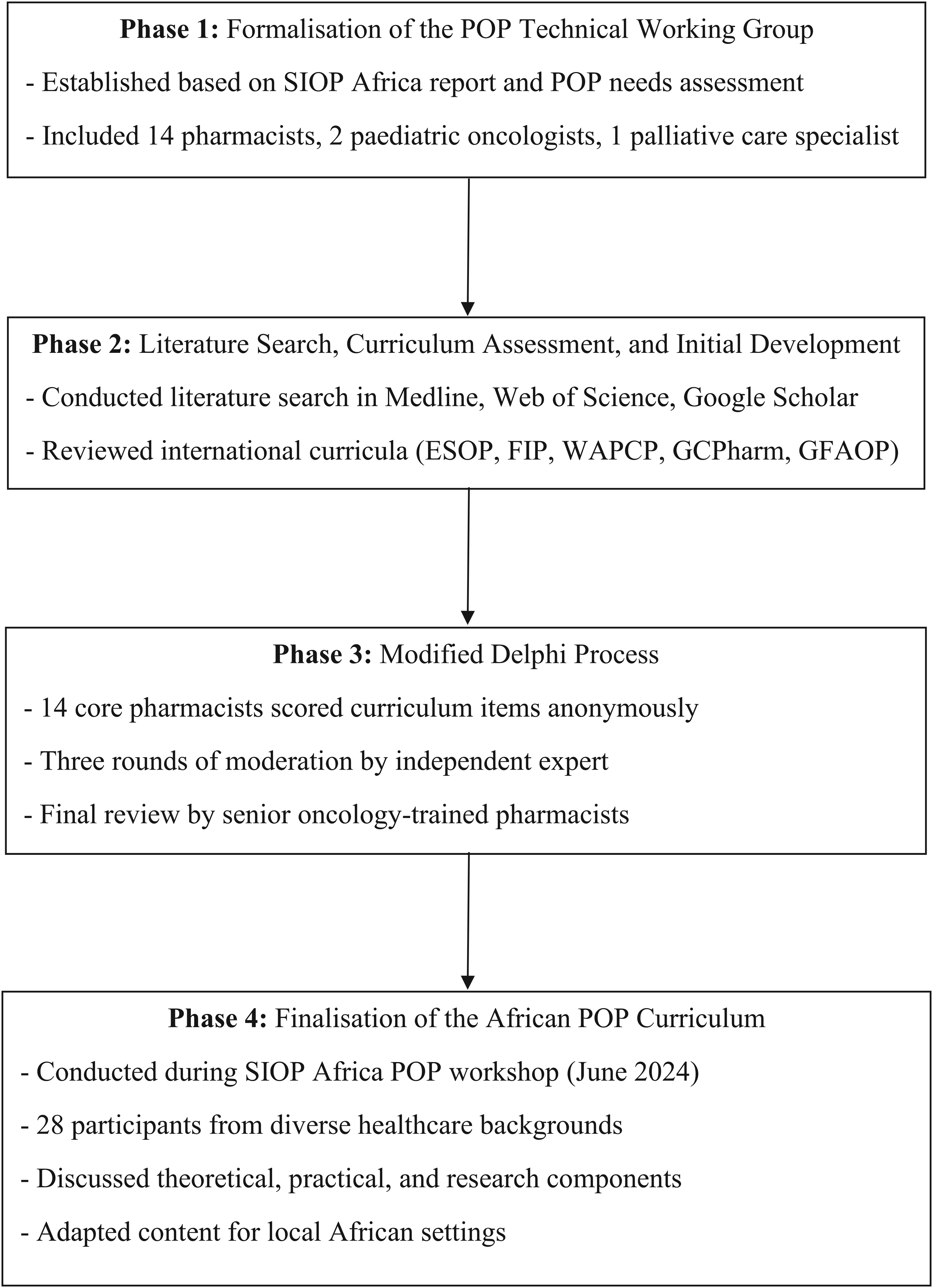
Methodological Flow Diagram for the African POP Curriculum Development.
Formalisation of the paediatric oncology pharmacy (POP) technical working group (TWG)
Based on the SIOP Africa community-wide report 9 and the POP needs assessment findings, 27 the POP TWG was established. The TWG consisted of 14 core African pharmacists who are trained in or work in POP, two African paediatric oncologists and an African palliative care specialist.
Literature search, curriculum assessment and initial development
We conducted an English language literature search on Medline via EBSCOhost, Web of Science and Google Scholar using MeSH terms including “curriculum’’, “paediatric’’, “oncology’’ and “pharmacy’’. We extended the search to grey literature via citation screening to ensure completeness.
Readily available and accessible curricula including those from the European Specialisation in Oncology Pharmacy (ESOP), 28 the International Pharmaceutical Federation (FIP), 29 the West African Postgraduate College of Pharmacists (WAPCP), 30 the Ghana College of Pharmacists (GCPharm) Paediatric Oncology Training Programme, 31 the FIP knowledge and skills reference guide for professional development in cancer care 32 and the Groupe Franco-Africain d’Oncologie Pédiatrique (GFAOP) pharmacy training syllabus 33 were considered for inclusion.
Modified Delphi process
The 14 core members of the African POP curriculum TWG were invited to participate in a modified Delphi process of the initial curriculum content. A senior paediatric oncologist served as the independent moderator. The initial curriculum content was sent to the pharmacists via email with participant blinding to ensure anonymity. The core members scored each theoretical and clinical skill item as “interesting to learn”, “important to learn” or “essential to learn” with a score of 1, 3 and 5 respectively. The assigned scores were converted to percentages. The items that scored ≥ 80% were included in the curriculum. The 14 core members were then requested by the independent moderator to suggest topics relevant to local African settings. After three rounds of moderation according to the modified Delphi process, topics that met widespread approval were collated into one document. The remaining topics that were not included were screened by two senior paediatric oncology-trained pharmacists, from Tanzania and France, for potential inclusion.
Finalisation of the African POP curriculum
The final development phase took place during the SIOP Africa POP workshop in June 2024 in Johannesburg, South Africa. The 28 participants included pharmacists, oncologists, nurses, social workers and other allied health professionals. Based on their specialities and practice areas, the workshop participants were divided into four groups facilitated by TWG pharmacists. To ensure that participants were fully briefed on the relevant concepts, presentations delivered by members of the core group included an analysis of SIOP Global Mapping Programme (GMP) results relevant to pharmacy,34,35 and feedback from a survey on education and training needs conducted before the workshop. This model was chosen to incorporate prior GMP findings, representing a baseline of paediatric oncology capacity on the continent, as well as more recent feedback by POP practitioners. The team aimed to optimise interaction by contextualising participants’ responses to the GMP survey and the subsequent, pharmacy-specific education and training survey.
Table 1 details the workshop programme outline. The theoretical, practical and research components, with associated topics, were discussed to develop sub-topics and improve quality. The workshop tasks focused on identifying the essential content for Africa and how standard topics could be adapted for local settings.
SIOP Africa 2024 paediatric oncology pharmacy pre-conference workshop programme outline.

Results
In Phase 1, completed in November 2023, the POP TWG comprised 17 core members: 14 core African pharmacists, 2 senior paediatric oncologists and a palliative care specialist. By June 2024, the team was expanded to increase multidisciplinarity with 23 active members representing 10 Anglophone and Francophone African countries, and 2 European countries. The 14 core African pharmacists included a French pharmacist with extensive experience in teaching in Francophone Africa, curriculum development and GFAOP activities. The inclusion of a palliative care specialist ensured that this vital component was represented. The senior paediatric oncologists, acted as facilitators of a blend of senior and junior professionals to make maximal use of various experiences and build capacity. Between the formation of the TWG and the commencement of the SIOP Africa 2024 conference, the group held 26 virtual meetings.
The literature search, curriculum assessment and initial development of the African POP curriculum focused on theoretical (36 items) and clinical skills (5 items). Independent from the core POP topics, considered universal to all settings and countries, the TWG included basic paediatric pharmacy concepts supporting POP principles and considered crucial to paediatric oncology practice in African settings.
The modified Delphi process structured the 36 theoretical items into 27 and renamed them as theoretical modules with various facets. The 5 clinical skills items were expanded to 9 clinical skills modules. The 14 core African pharmacists suggested the addition of 6 research modules which increased the content categorisation from 2 to 3: theoretical, clinical skills and research content. Each of the theoretical modules was assigned credit hours totalling 86 h. The clinical skills modules were allocated a total of 720 practical hours at a rate of 3 practical hours per credit hour (240 credit hours). The research modules will require candidates to complete a research project with a minimum of one publishable manuscript. The allocation of the credit hours was completed by 2 core African pharmacists based on their clinical and academic pharmacy practice experience.
The finalisation of the African POP curriculum at the SIOP Africa POP workshop was preceded by 8 presentations from the core members detailing the delivery of POP services at different hospitals in Africa, the findings of the African POP curriculum needs assessment survey, pathophysiology and therapeutics of paediatric cancers, a paediatric cancer case and tumour board simulation, the role of POP practitioners in addressing medication shortages, available training programmes in Ghana and Tanzania, and the product of the modified Delphi process. The 28 workshop participants took part in the curriculum discussions, affirming the necessity to include research components to the theoretical and practical competencies in the African POP curriculum. The African POP curriculum was then discussed and finalised. The course curriculum will be supplemented by a freely accessible textbook covering all the agreed-upon topics.
Despite the success of the development process, several challenges were encountered. The diversity of practices and resource availability across different African countries posed a challenge in standardising certain aspects of the curriculum. Additionally, ensuring sustained engagement and participation from all stakeholders required continuous effort and coordination. Addressing these challenges necessitated a flexible approach, allowing for modifications based on feedback and the evolving needs of the healthcare landscape, and advocating for hybrid delivery of course contents. We were not able to identify POPs from Lusophone (Portuguese-speaking) countries but efforts are being made to include them in future.
Discussion
This innovative project showcases collaboration between pharmacists and other healthcare professionals, emphasising the creation of content designed by Africans, for Africans, and tailored to the African healthcare context. It highlights the involvement of pharmacists in collaborative healthcare teams and the importance of prioritising both what pharmacists consider essential and what paediatric oncologists and other healthcare professionals expect from pharmacists. The development of the African POP curriculum marks a significant step towards addressing the educational and training needs of pharmacists in paediatric oncology across Africa. This initiative responds to the pressing need for specialised education in paediatric oncology pharmacy, as highlighted by SIOP Africa leaders. 9 The comprehensive curriculum development approach, which combined literature reviews, curriculum searches, a modified Delphi process, and extensive stakeholder engagement, ensures that the curriculum is both contextually relevant and academically rigorous. 36
The incorporation of POP curricula from the GFAOP, the West African Postgraduate College of Pharmacists, and the Ghana College of Pharmacists provided a foundational framework for developing the harmonised African curriculum, ensuring continuity with previously established standards. The curriculum incorporates several uniquely African elements to address local challenges and healthcare realities. It includes specialised modules on managing medication shortages given the frequent disruptions in drug availability across Africa.37,38 Recognising the high burden of late-stage cancer diagnoses, it places a strong emphasis on palliative care, with a dedicated specialist included in the curriculum development team. Research components require pharmacists to produce a publishable manuscript, encouraging the generation of Africa-specific evidence in POP. The curriculum also integrates local case studies and tumour board simulations to ensure practical application in African healthcare settings. Additionally, it is designed for both Anglophone and Francophone Africa, with bilingual adaptation facilitated by a French pharmacist. Community and stakeholder engagement was integral to its development, ensuring that the curriculum aligns with the needs of African healthcare professionals and institutions.
Although contextually subjective and open to various modifications, a Delphi process comprising a minimum of two rounds is appropriate and various rounds have successfully been utilised in curriculum development.39,40 In this modified Delphi process, three Delphi rounds were performed and various modifications were incorporated before and after the Delphi rounds to reflect the unique contextual needs of the study. The review of literature and existing curricula sources, in addition to assisting in the initial drafting of the African POP curriculum, significantly guided the allocation of credit hours and research output load. This strategy was adopted to ensure that the assigned credit hours meet the quality standards of implementation collaborators, higher academic institutions and educational accreditation boards and councils. 41 The flexibility highlighted by the TWG permits further modifications to meet the particular requirements of various collaborative authorities across Africa and motivates the awarding of degrees and professional certifications as paediatric oncology pharmacists of various grades.
The curriculum development process, representing a collaboration between pharmacists, paediatric oncologists and a palliative care specialist from diverse backgrounds and regions, fostered inclusivity and regional acceptance This diversity ensured a holistic view of the needs and practices in POP across Africa, leading to a curriculum that is adaptable to various local settings. The incorporation of theoretical training, clinical skills and research contents within the African POP curriculum underscores the commitment to producing well-rounded paediatric oncology pharmacists. By addressing both foundational paediatric pharmacy concepts and advanced oncology-specific knowledge, the curriculum will potentially prepare pharmacists to deliver high-quality care in paediatric oncology in largely resource-constrained settings. The emphasis on research skills is crucial in fostering a culture of continuous improvement and innovation within the field. 42
A major advantage of the proposed curriculum is the delivery of course content on a hybrid platform permitting candidates to have job flexibility. This approach will ensure that the limited and widely dispersed pharmacists who practice paediatric oncology will not leave their duty posts for further knowledge and deprive patients of scarce pharmaceutical services. Future efforts should focus on the implementation and evaluation of the African POP curriculum. The experiences of candidates who enrol can be harnessed to assess the effectiveness of the curriculum and identify areas for improvement. Furthermore, continuous professional development opportunities for pharmacists, including refresher courses and advanced training, will be essential in maintaining the relevance and impact of the curriculum.
Further recommendations for the curriculum implementation
The following recommendations, in line with the submissions of Brown (1998) and Richards (2001),21,22 have been proposed;
Methodological/materials considerations: The development of teaching resources, practical guides and textbooks to support the syllabus. This recommendation is envisioned as an accreditation and credentialing technique. Testing/teaching: The implementation of the African POP curriculum with the generation of a report illustrating the implementation methodology and outcomes. Evaluation: The development and implementation of monitoring and evaluation checklists to conduct an impact assessment of the curriculum and reporting findings for quality improvement.
Conclusion
The African POP technical working group has developed a robust, locally relevant, African-sensitive training curriculum which represents a pivotal advance in the education and training of POPs in Africa. With theoretical, clinical and research competencies, the curriculum can be adapted according to the level of training needs applicable to member countries. The time adaptability and proposed flexible implementation format ensure sustainability and effectiveness in improving paediatric oncology pharmacy practice across Africa. The curriculum will further be developed for implementation and enhance the award of dual qualifications in response to the particular needs of potential candidates and collaborators.
Footnotes
Abbreviations
Acknowledgements
On behalf of SIOP Africa, the authors recognise the workshop participants for their contributions.
Authors’ contributions
EW and KBZ: Study conception, study design, data collection, data analysis and drafting of the original manuscript. SNM, SGOA, TB, ACC, VDM, AMF, AFA, SA, LCI: Study conception, study design, data collection and review of the manuscript. BP, SK, JvH and JAG: Study design, data analysis, review of the manuscript and supervision. All authors reviewed and approved the final version of the manuscript.
Availability of data and materials
All relevant data are within the manuscript and its Supporting Information files. However, the final curriculum is available on an explicit request to the African POP curriculum development group.
Consent for publication
This article is published on behalf of SIOP Africa.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical clearance
Ethical approval for the study was granted by the Antwerp University Hospital Ethics Committee (Project ID: 6880).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The 2024 SIOP Africa paediatric oncology pharmacy workshop received sponsorship from Bristol Myers Squibb. Participants and team members received individual sponsorships from institutions such as World Child Cancer.
