Abstract
Introduction
Combination therapy with BRAF and MEK inhibitors are the standard of care for treatment of BRAF V600 mutant unresectable or metastatic melanoma. Encorafenib, a BRAF inhibitor, and binimetinib, a MEK inhibitor, when taken as continued combination therapy, show long term progression-free survival benefits. 1 However, most patients (>98%) in clinical trials experienced adverse events (AEs) associated with combination therapy with 70% being serious (grade 3–4). Dose adjustments or interruptions because of AEs occurred in 55% of patients treated with encorafenib and binimetinib and 16% of patients discontinued therapy because of AEs. 2 Gastrointestinal disorders and eye disorders were the most common AEs associated with the need for dose adjustments and interruptions. AEs leading to therapy discontinuation in more than one patient were increased transaminases or blood creatinine, headache, and rash. 1
The AEs associated with combination encorafenib/binimetinib therapy are well documented and clinicians can use supportive therapies and management strategies to increase the likelihood of patients being able to persist on therapy. 3 The most common AEs observed with encorafenib/binimetinib therapy typically occur within the first 2 months after starting therapy, and AE burden tends to decrease with time on treatment. 1 Pharmacists are well-positioned to provide care for patients initiating and continuing treatment on oral anticancer medications. The Hematology/Oncology Pharmacist Association (HOPA) recommends that pharmacists, as part of a multidisciplinary oncology care team, provide patients with education and strategies for managing anticipated AEs and conduct routine follow-up monitoring while patients are on therapy. 4 With the prevalence of early onset AEs it is recommended that pharmacists contact patients for therapy assessment within 2 weeks of treatment initiation. Studies assessing the impact of pharmacist-led oral anticancer therapy monitoring programs show improved medication safety and adherence, improved adherence to monitoring standards, reduced AEs, and re-engagement of patients lost to follow up.5,6 However, there is limited data available about the impact on therapy changes of tailoring pharmacist led patient monitoring to coincide with anticipated AE onset, particularly those that occur early in therapy. The purpose of this study was to assess the impact of pharmacist led tailored monitoring of encorafenib and binimetinib on dose adjustments, interruptions, and therapy discontinuations during the first 90 days of treatment.
Methods
Study design and data
This was a single center pre-post intervention cohort study of patients initiating combination therapy with encorafenib and binimetinib for unresectable or metastatic melanoma filled through the center's specialty pharmacy or manufacturer's assistance program from July 2018 to December 2019 (pre-intervention) and April 2021 to December 2022 (post-intervention). Patients were excluded if participating in a clinical trial or receiving treatment for a non-FDA approved indication. The study was approved by the center's Institutional Review Board.
Setting
This study was conducted at a tertiary academic medical center. The center's specialty pharmacy model integrates specialty pharmacists in the outpatient clinics as part of the interdisciplinary patient care team. The oncology clinic specialty pharmacists assist patients prescribed specialty medications with access to treatment, including screening for medication safety and appropriateness, completing insurance prior authorizations and appeals if needed, providing financial assistance counseling when medications are unaffordable, and providing medication education. After initiating treatment, patients are monitored monthly for medication adherence, AEs, and healthcare utilization and yearly for response to therapy and continued therapeutic safety. Pharmacists provide interventions to optimize therapy outcomes when needed. Pharmacist intervention information (e.g., reason, type, and outcome) are documented in the specialty pharmacist patient management system.
Pre-Intervention cohort
In the pre-intervention cohort, patients received comprehensive medication education prior to initiating encorafenib and binimetinib in combination for unresectable or metastatic melanoma with BRAF V600 E or K mutation. Per clinic protocol, patients were contacted by the health system specialty pharmacy (HSSP) three times in the first 90 days of therapy to align with refill needs. Patients may have had additional contact if they reached out to the clinic or pharmacist outside of the standard touchpoints. Specialty pharmacy technicians were responsible for contacting patients regarding their medication refill by phone or through the Electronic Health Record (EHR) patient message portal approximately seven days before the patient was expected to run out of medication. Specialty pharmacists were alerted if the patient reported any problems such as missed doses of their medication, AEs, unexpected provider visits (including urgent care, emergency room, or hospitalization), medication changes, or indicated the need to speak to a pharmacist. The pharmacist would then review the alert and contact the patient if needed to provide patient education or make a recommendation to the physician for supportive therapy, medication adjustments, or additional healthcare service (e.g., laboratory monitoring, urgent care visit or clinic visit). For patients receiving manufacturer assistance, refills were managed by the manufacturer assistance program; however, any AEs or medication related issues reported to the clinic were addressed by the HSSP.
Intervention cohort
In April 2021, a tailored early treatment monitoring program was implemented for patients initiating encorafenib and binimetinib for unresectable or metastatic melanoma with BRAF V600 E or K mutation filled through the HSSP or the manufacturer's assistance program. Similar to usual care, pharmacists provided thorough counseling to patients initiating encorafenib and binimetinib. In addition, patients received a welcome kit containing over the counter items and a guide for side effect management, as well as contact information for the HSSP. The specialty pharmacist initiated patient outreach at six designated times during the 90 days following medication initiation, in addition to the standard refill calls completed by pharmacy technicians. Pharmacists performed follow-up monitoring by phone. The call schedule was designed at specific time points based on clinical trial and manufacturer data describing AE onset. Pharmacists asked patients specific questions related to anticipated AEs and provided mitigation strategies if needed during each monitoring call. Patients could report the same AE at multiple monitoring calls and multiple AEs could be reported in the same call. Interventions were also provided if patients reported other problems such as missed doses or medication changes or if they had additional questions.
Study procedures data
Data for the pre-intervention cohort was collected by retrospective chart review. Post-intervention data was captured prospectively as part of normal clinical practice and documented in the specialty pharmacy patient management system and the EHR. Research Electronic Data Capture (REDCap) hosted at Vanderbilt University was used to collect the data for analysis.7,8 The primary outcome of this study was the number of treatment interruptions, dose reductions and medication discontinuations pre- and post-intervention implementation. The type and frequency of AEs and pharmacist interventions were also described.
Statistical analysis
Descriptive statistics were used to report the data. Continuous variables were reported as median and interquartile ranges (IQRs), and categorical variables were reported as frequencies and percentages. Statistical analysis was performed using R version 4.3.1.
Results
The number of patients in the pre-intervention (n = 18) and post-intervention (n = 19) cohorts was similar. There were more males in the post-intervention cohort (pre, n = 9 [50%]; post, n = 12 [63%]) and cancer stage at time of diagnosis was more advanced (stage 4) in the post-intervention cohort (pre, n = 10 [56%]; post, n = 18 [95%]) (Table 1).
Baseline characteristics.

BRAF, B-raf proto-oncogene; ECOG, Eastern Cooperative Oncology Group; IQR, interquartile range.
In the first 90 days of treatment, 126 AEs were reported in the pre-intervention cohort and 211 AEs in the post-intervention cohort. Most patients experienced AEs within the first 2 weeks after initiation of treatment (pre 78%, post 95%). Pre-intervention, patients (56%) reported AEs most often at 14 days on therapy. Post-intervention, patients (84%) reported AEs most often at 7 days on therapy. Patients in the pre (n = 18) and post (n = 19) intervention cohorts reported fatigue (pre, n = 10 [56%]; post, n = 13 [68%]), nausea (pre, n = 8 [44%]; post, n = 14 [74%]), and diarrhea (pre, n = 3 [17%]; post n = 9 [47%]) most often (Figure 1).
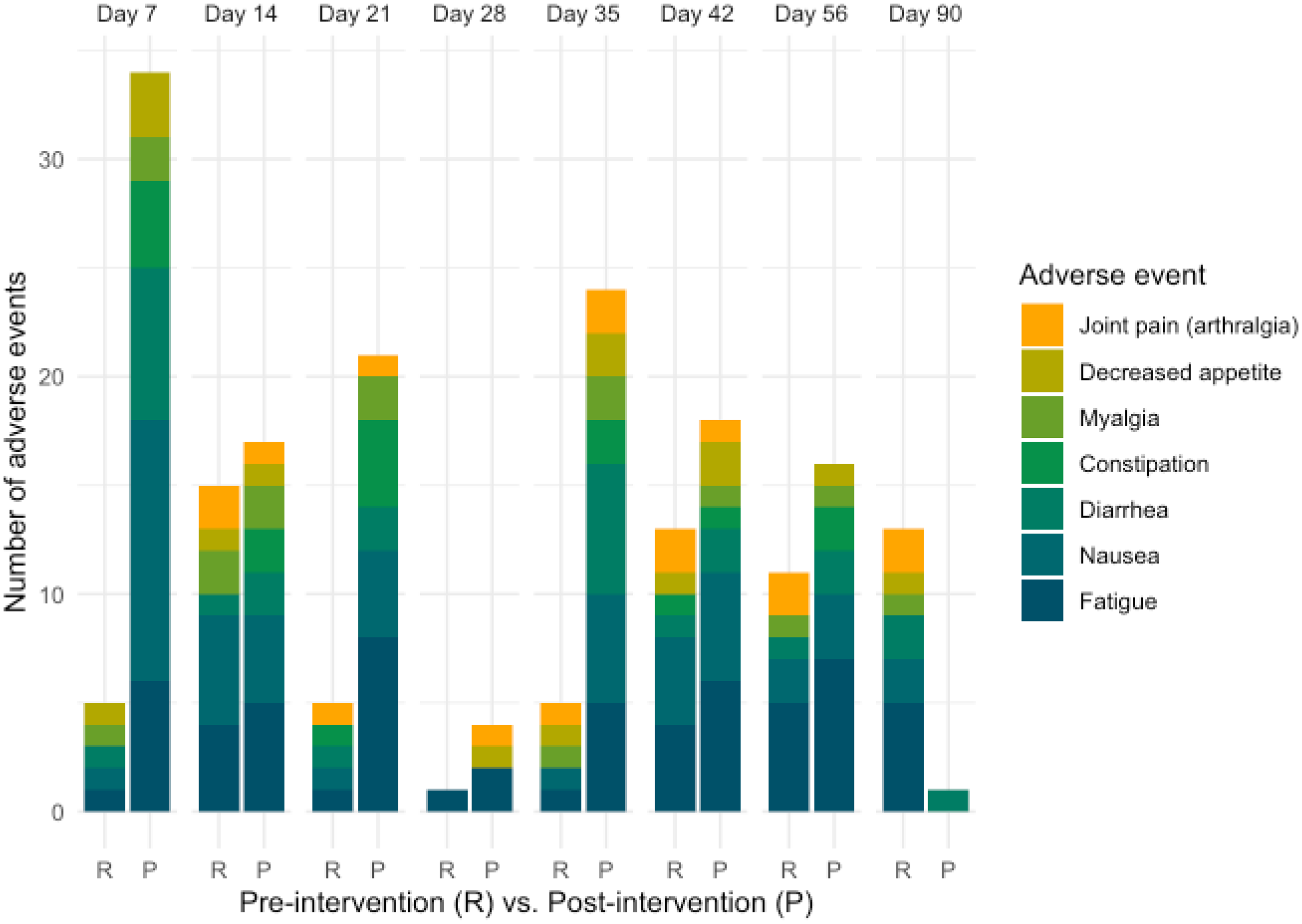
Timing and frequency of the 6 most reported AEs in both cohorts.
In the pre-intervention cohort, 39% of patients required at least one dose reduction with median time to dose reduction being 22 (IQR 18–63) days and 50 (IQR 22–67) days for encorafenib and binimetinib, respectively. In the post-intervention cohort, 47% of patients required at least one dose reduction with median time to dose reduction being 36 (IQR 23–46) days for both encorafenib and binimetinib. Dose increases, after a reduction, occurred twice in the post-intervention cohort, but not in the pre-intervention cohort. In the pre-intervention cohort, 44% of patients required one or more treatment holds with median time to first hold being 27 (IQR 12–44) days and 38 (IQR 15–45) days and median duration of hold being 11 (IQR 3–22) days and 6 (IQR 1–22) days for encorafenib and binimetinib, respectively. In the post-intervention cohort, 58% of patients required one or more treatment holds with median time to the first hold being 15 (IQR 8–30) days and median duration of holds being 10 (IQR 6–19) days for both encorafenib and binimetinib.
In the pre-intervention cohort two patients (11%) discontinued treatment within the first 90 days because of disease progression (n = 1) and clinical decline (n = 1). In the post-intervention cohort five patients (26%) discontinued treatment within the first 90 days. Reasons for discontinuation post-intervention were disease progression (n = 1), AE (generalized weakness/intolerability; n = 1), clinical decline (n = 2), and patient decision (n = 1).
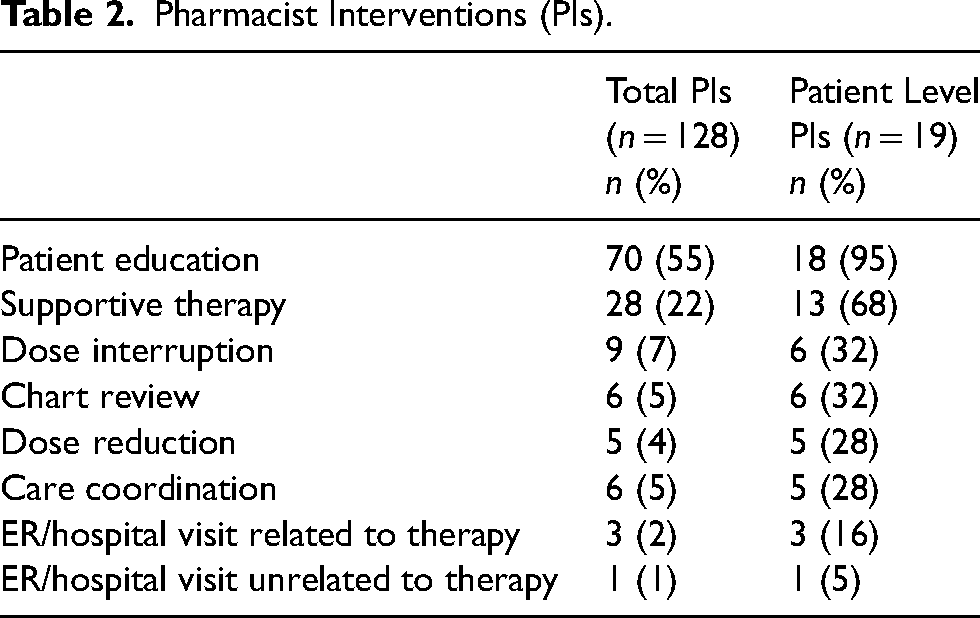
Post-intervention, 128 pharmacist interventions were performed. All patients (n = 19) received at least 1 intervention, with patient education (n = 18, [95%]) and supportive therapy (n = 13, [68%]) being the most frequent. Fewer patients had interventions related to treatment interruption (n = 6 [32%]), chart review for patient history and clinical status (n = 6 [32%]), care coordination (n = 5 [26%]), dose reduction (n = 5 [26%]), and emergency department/hospitalization (n = 4 [21%]) (Table 2).
Pharmacist Interventions (PIs).
Discussion
This relatively small pre/post cohort study found that increased pharmacist monitoring during the first 90 days of combination therapy with encorafenib and binimetinib did not impact the number of treatment interruptions, dose reductions, and medication discontinuations when comparing the post-intervention cohort to the pre-intervention cohort; however, more AEs were reported and reported earlier in the post-intervention cohort. These findings are consistent with other studies that demonstrate pharmacist-led oral chemotherapy-monitoring programs may not show significant difference in overall time on therapy, but pharmacist encounters for AE management and interventions are increased. 9 This study reinforces that clinical pharmacists are instrumental in providing patient support, with education and supportive care to mitigate AEs that commonly occur in patients initiating oral anticancer therapy.
Early and proactive management of AEs in patients on BRAF and MEK inhibitor combination therapy may result in better patient management. Monitoring patients within the first 2 weeks of initiating oral anti-cancer medications is shown to be valuable.4,10 In this study, most patients reported AEs within 7 to 14 days after the start of treatment. This supports the recommendation by HOPA that follow-up should occur during this interval when AEs are likely to present. In the post-intervention cohort, there were two instances of dose increases after an initial reduction because of AEs, indicating that early identification and management of AEs may impact resolution of AEs; therefore, allowing for the opportunity to up-titrate back to optimal dosing. 10
Tailored monitoring requires pharmacist time commitment and implementation of pharmacy systems to track and manage outcomes. A change in treatment interruptions, dose reductions, and medication discontinuations was not observed in the cohort with increased pharmacist monitoring, suggesting that increased monitoring may not be clinically impactful for all anti-cancer medications. Medication class, performance status, and advanced disease state may impact outcomes associated with tailored monitoring; therefore, further research evaluating these characteristics is needed. Additionally, benefits of increased monitoring from the patient perspective should be explored as patients with cancer have indicated pharmacy education and support are valued. 11 Maximizing productivity of the health care team is vital for oncology practices, especially with projected workforce shortages. 12 Developing and implementing efficient pharmacist monitoring programs can contribute to patient care and optimize the use of limited resources.
Limitations
This study is limited by simple size and representing a single center, oncology outpatient clinic. The implementation of the tailored early treatment monitoring program was delayed due to the COVID-19 pandemic. This could impact patient characteristics and resulting outcomes. The pre and post cohorts differed in their cancer staging, which could also have impacted results. Additionally, there may have been a shift in prescribing practices, specifically the use of nonstandard dosing protocols to manage AEs.
Conclusions
This study supports the HOPA recommendation for patient outreach within 2 weeks after initiating treatment with oral anti-cancer medications. Pharmacists play a key role in patient follow-up and results suggest that tailored monitoring may be optimized by considering factors such as medication class, patient performance status, and disease state.
Footnotes
Acknowledgement of research support
Special thanks to Carson S. Lamb, PharmD, CSP for assisting in tailored monitoring calls.
The project described was supported by CTSA awards No. UL1 TR002243 and UL1 TR000445 from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the National Institutes of Health.
Author contributions
BDL, NBS: Conceptualization, Methodology; BDL, SGW, NBS: Data Curation; BDL, JD, LC, ADZ, KWW: Data Analysis and Interpretation; SGW, JD, LC, ADZ, KWW: Data Visualization; BDL, SGW, JD, ADZ, NBS, KWW: Writing-reviewing and editing.
All authors
Final approval and accountability
Data availability statement
Due to the nature of the research, ethical, and privacy concerns supporting data is not available to be shared. Select de-identified data elements may be shared upon request to the authors.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The following authors have participated in funded research with funding going to the institution: BDL: Pfizer Inc. (2020–2023), AstraZeneca (2020–2023), ASHP Foundation (2023–2024), Takeda, Inc. (2024–Present) SGW: Pfizer Inc. (2020–2023), AstraZeneca (2020–2023), BeiGene (2022–2024), ADZ: Pfizer Inc. (2020–2023), BeiGene (2022–2024), AstraZeneca (2020–2023), ASHP Foundation (Oncology, 2023–2024), KWW: ASHP Foundation (2023–2024).
Ethical approval and informed consent
This study was approved by the institutional review board at Vanderbilt University Medical Center as a standard/expedited study (approval # 202045) on 11/12/2020.
Funding
This study was supported by a grant from Pfizer, Inc. (grant number 61420901).
