Abstract
Purpose
The time to complete oncology pharmacist tasks is needed to determine workload and productivity. The Hematology/Oncology Pharmacy Association (HOPA) and the Hematology/Oncology Practice and Research Network (PRN) of the American College of Clinical Pharmacy (ACCP) partnered with the aim of establishing consensus on the time required to complete oncology pharmacy tasks.
Methods
Fifteen patient care tasks and 9 non-patient care tasks, commonly completed by oncology pharmacists were each assigned an average amount of time to be completed. This list was then converted into 24 statements and the Delphi survey method was utilized with an expert panel to arrive at consensus between December 2023 and February 2024. Consensus was defined as at least 75% agreement. The complete manuscript was endorsed by HOPA and ACCP Hematology/Oncology PRN.
Results
Thirty-three pharmacist-experts agreed to participate in this survey with all participating in round 1, and 29 (87.9%) participating in round 2. In round 1, 9 tasks achieved consensus, with 7 of these being classified as patient care associated. Seven statements reaching 65% but less than 75% agreement were deemed to reach borderline consensus. Eight statements failed to achieve at least 65% agreement and were modified based on respondent feedback. In round 2, 15 statements were included with all achieving consensus. At the completion of round 2, all 24 statements reached consensus, and the survey was deemed complete.
Conclusion
This project produced the first comprehensive consensus statements for the average time for a US-based oncology pharmacist to complete common patient and non-patient care-related tasks.
Introduction
The growing incidence of cancer as well as the expanding complexity of cancer treatment has increased the demand for oncology services in the United States (US). 1 Oncology pharmacists have swiftly filled a gap by providing various pharmacotherapy-related clinical services, such as medication reconciliation, transitions of care coordination, patient/caregiver education, medication access, medication dosing and monitoring, cancer treatment selection, and ordering and/or reviewing labs and other pertinent tests. 2 Pharmacy leaders expect the demand for oncology pharmacists and creation of new oncology pharmacist positions to increase in a variety of settings, including academic and community cancer centers.3,4
Despite this, there is a paucity of published studies quantifying oncology pharmacist scope, workload, and productivity. This deficiency limits both the evaluation of current oncology pharmacist positions and justification of new positions. The few research studies that have attempted to define oncology pharmacy workload are either single-center, observational time-motion studies, or primarily focused on categorizing the type of interventions and quantification of pharmacist tasks.5–8 These studies have limited generalizability; while oncology pharmacists are performing similar job-related tasks, the distribution of effort tasks for a particular position varies greatly between institutions. 2 This contrasts with other oncology providers whose productivity is often defined by relative value units derived from patient complexity, length of visit, and specific procedures performed. 9
To aid oncology pharmacists and pharmacy leaders, the amount of time necessary to complete each type of oncology pharmacist task is needed to determine workload and productivity. HOPA and ACCP Hematology/Oncology PRN partnered to survey hematology/oncology pharmacists with the aim of establishing a consensus on the time required to complete specific oncology pharmacy tasks.
Methods
Design
This study was deemed to be exempt from review. The Practice Outcomes and Professional Benchmarking Committee (POPBC) within HOPA utilized the Further Defining the Scope of Hematology/Oncology Pharmacy Practice publication to compile a list of the 24 oncology pharmacist tasks. 10 These tasks were deemed to be common regardless of practice setting (inpatient, ambulatory, etc.) or specialty (medical oncology, surgical oncology, hematopoietic stem cell transplantation, or a mixed population). These tasks were classified as either patient care (15 out of 24) or non-patient care tasks (9 out of 24).
POPBC committee members assigned an average amount of time to complete each of the 24 common tasks based on their expert experience. Each task's average time estimation included a range and described the amount of time a pharmacist needs to complete the associated task greater than 80% of the time it is performed. These time averages were then combined to create 24 consensus statements. Five basic demographic questions and an open-ended comment field were added to complete the Delphi survey. For each consensus statement, six responses were available: unable to answer, strongly disagree, disagree, neither agree or disagree, agree, and strongly agree. To limit the risk of bias, all statements were worded similarly starting with the phrase “The task will take an average of…to complete” and included a denominator (per drug, per patient, etc.). The survey was uploaded and administered utilizing the online platform SurveyMonkey under an Enterprise Plan which included enhanced security and compliance features (https://www.surveymonkey.com/).
To validate the time assigned by POPBC to each pharmacist job task, a Delphi survey was conducted between December 2023 and February 2024 using previously established survey techniques.11–14 The list of experts who would receive the Delphi Survey was derived from the membership of ACCP Hematology/Oncology PRN. In October 2023, there were 247 US-based, full-pharmacist members. An email was sent to these individuals requesting participation as an expert panel member from those currently practicing in direct patient care roles, supervising those in direct patient care roles, or having practiced in a direct patient care role within the previous 3 years. Thirty-three members agreed to participate as the expert panel in the Delphi Survey.
The 33 experts were sent an email asking them to complete the electronic, online survey. Reminder emails were sent at regular intervals for 3 total weeks to non-respondents. A round was considered complete 1 week after the final reminder email. Once complete, all information collected was analyzed to identify the degree of consensus. Based on previous published studies utilizing this method, an a priori decision was made to consider greater than or equal to 75% agreement (agree or strongly agree) as consensus.12–14 Statements that reached this consensus threshold were removed from future rounds of the survey. Statements that fell between 65% and 75% agreement were included unmodified in the next round to confirm borderline consensus. Statements that fell below 65% agreement were modified by the POPBC based on open-ended respondent feedback and the modified versions were included in the next round of the survey. In subsequent rounds, results from the previous round as well as any modifications were noted for each included statement. This process was planned to be repeated for up to 3 rounds or until consensus was reached for all 24 statements. Only respondents who completed the previous round were invited to participate in the subsequent round. The complete manuscript was endorsed by the Research Council and Board of Directors of HOPA satisfying the requirements to be considered a statement. It was also endorsed by the membership of ACCP Hematology/Oncology PRN satisfying the requirements to be considered an official opinion of the PRN. It does not necessarily represent an official ACCP commentary, guideline, or statement of policy or position.
Data analysis
Responses were managed to maintain responders’ anonymity. The percentage of agreement was calculated as the number of respondents selecting either agree or strongly agree divided by the total number of experts providing a level of agreement on that particular statement (excluding those who selected unable to answer), multiplied by 100%, with greater than or equal to 75% agreement being considered consensus reached, 65–75% being considered borderline consensus reached and less than 65% being considered non-consensus reached. All the data were analyzed using SurveyMonkey's intrinsic data analysis tool and Windows Excel, version 2402 (Microsoft, Corp, Redmond, WA).
Professional organizations
HOPA is a national organization supporting hematology/oncology pharmacy professionals with nearly 4000 members. A variety of committees within HOPA undertake more targeted work. The POPBC is a HOPA committee focused on supporting research initiatives that demonstrate the value of hematology/oncology pharmacy professionals. The POPBC committee is comprised of thirteen US-based oncology pharmacists and one ex-officio student pharmacist. Members come from a variety of institutions and care settings including inpatient, infusion center, ambulatory, informatics, specialty, and pharmacy management.
ACCP is a professional and scientific society serving more than 15,000 clinical pharmacists by providing leadership, education, advocacy, and resources to enable clinical pharmacists to achieve excellence in practice, research, and education. Full-pharmacist membership is restricted to those who have met specific criteria related to practice, education, research, and leadership. The Hematology/Oncology PRN was established to connect oncology clinical pharmacists to enhance professional support and collaboration based on common practice and research interests.
Results
Expert demographics
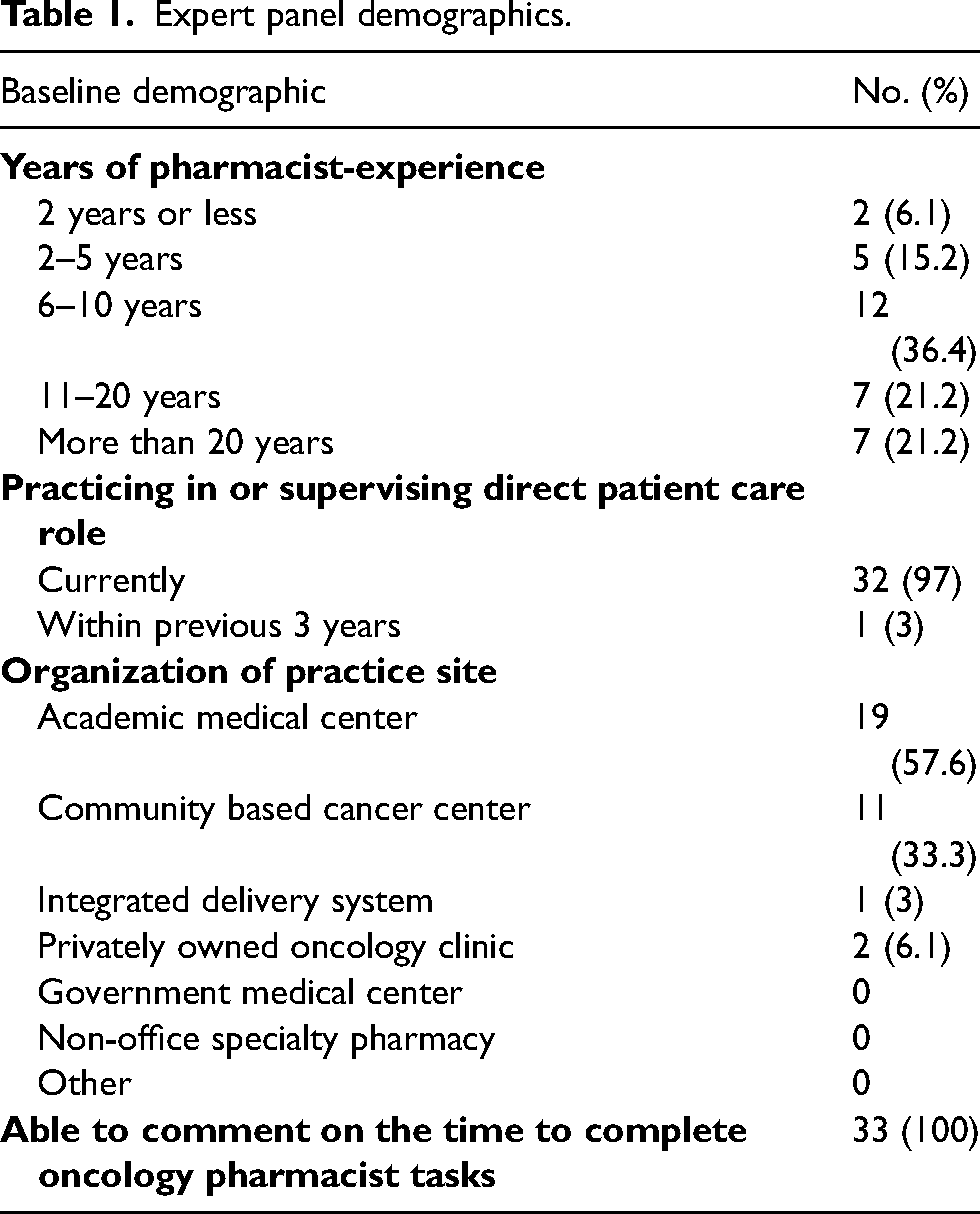
Of the 33-oncology pharmacist-experts who agreed to participate in this survey, all participated in round 1, and 29 (87.9%) participated in round 2. Table 1 summarizes the basic demographics related to participants’ practice experience and setting. All experts indicated they were able to speak to the time it takes to complete oncology pharmacist tasks and completed the entire survey.
Expert panel demographics.
Round 1 results
Twenty-four statements were included for assessment of agreement in round 1. The number of respondents providing an answer other than “unable to answer” for individual statements ranged from a low of 26 (78.8%) to high of 33 (100%) with 20 of the statements having at least 30 responses. Nine tasks (37.5%) achieved greater than or equal to 75% agreement, with 7 of these being classified as patient care associated. These were deemed to have met consensus in round 1 and removed from the future rounds of the survey. Seven statements reaching 65% but less than 75% agreement were deemed borderline consensus and were included in round 2 unmodified. Eight statements failed to achieve at least 65% agreement and were modified. All these statements were modified to increase the average time necessary to complete the task based on the open-ended feedback of round 1 respondents. One of the modified statements was edited for clarity by adding the term “metabolism specific” to specify which type of pharmacogenetic data would be used for dosing modifications based on the open-ended feedback provided by round 1 respondents. The level of agreement for each statement meeting consensus in round 1 is provided in Table 2 for patient care tasks and Table 3 for non-patient care tasks.
Consensus for patient care related tasks reached in round 1.

Respondents who chose “unable to answer” were excluded from the total number of responses for that statement
Consensus for non-patient care related tasks reached in round 1.
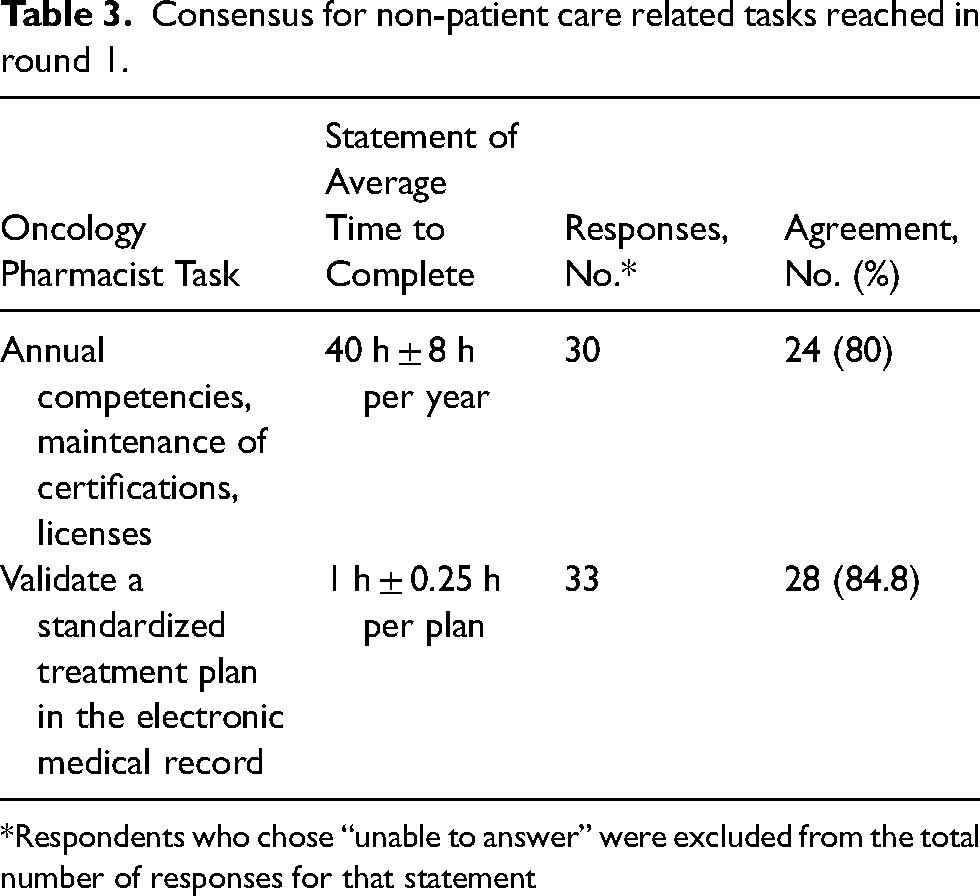
Respondents who chose “unable to answer” were excluded from the total number of responses for that statement
Round 2 results
Fifteen statements were included for assessment of agreement in round 2. The number of respondents providing a level of agreement for individual statements ranged from a low of 26 (89.7%) to high of 29 (100%) with 12 of the statements having at least 28 responses. Of the15 statements, all achieved at least 75% agreement. These were deemed to have met consensus in round 2 and removed from the future rounds of the survey. The level of agreement for each statement is provided in Table 4 for patient care tasks and Table 5 for non-patient care tasks. At the completion of round 2, all 24 statements reached the pre-specified level of consensus, and the survey was deemed complete. The complete consensus statements are summarized in Table 6.
Consensus for patient care related tasks reached in round 2.

Updated from average time in round 1 if < 65% agreement was reached.
Respondents who chose “unable to answer” were excluded from the total number of responses for that statement.
Consensus for non-patient care related tasks reached in round 2.
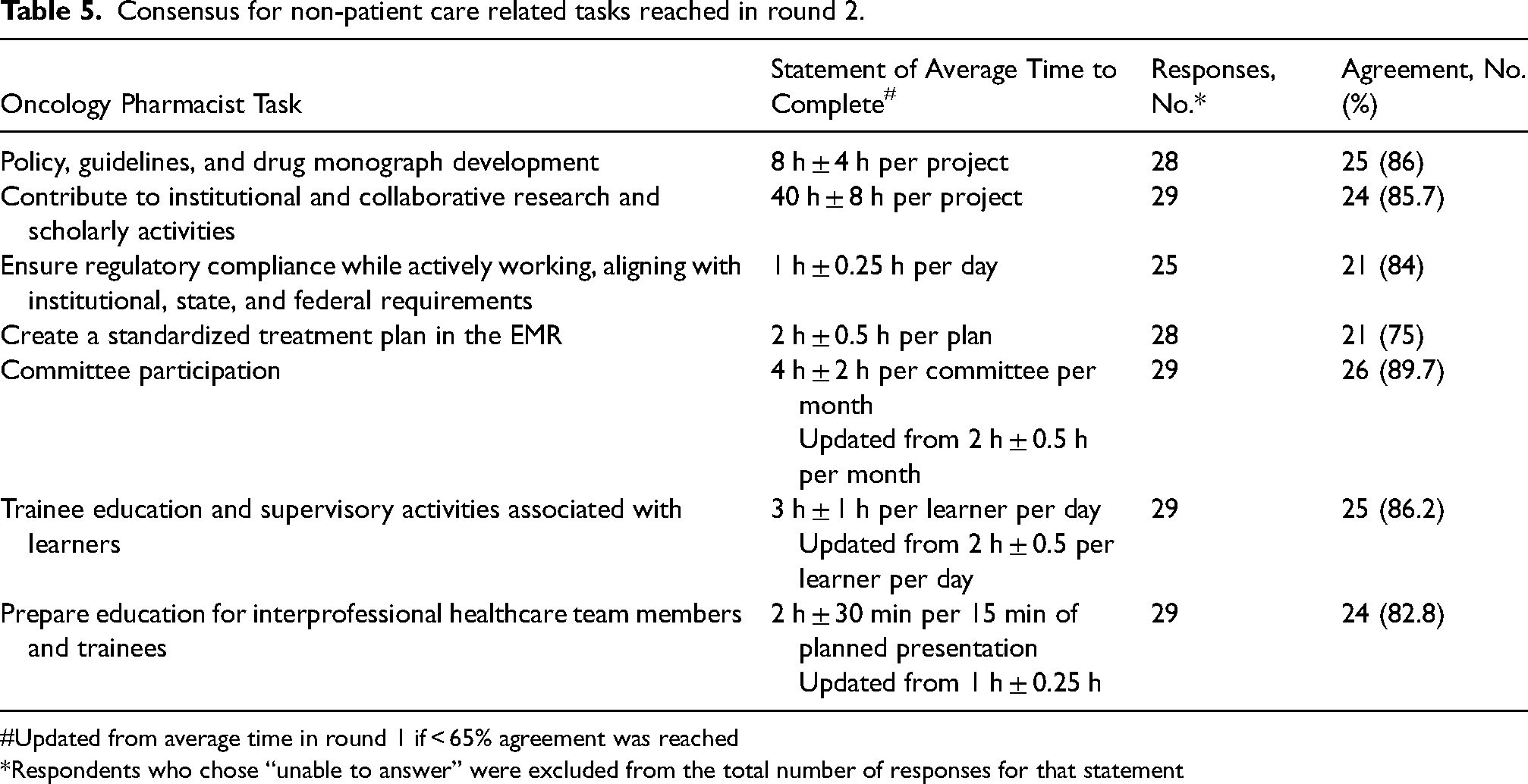
Updated from average time in round 1 if < 65% agreement was reached
Respondents who chose “unable to answer” were excluded from the total number of responses for that statement
Summary of consensus statements.

Discussion
This study met its objective and reached consensus regarding the amount of time required for oncology pharmacists to complete common tasks associated with their roles. These findings have been reviewed and endorsed by HOPA and ACCP Hematology/Oncology PRN—two of the largest national organizations representing oncology pharmacists in the US. There are no other published guidelines that can be generalized across practice settings and patient populations to help measure the workload of oncology pharmacists. Utilizing the data herein, practicing oncology pharmacists can evaluate how their expected job tasks may fit within their work hours. This will empower frontline employees and help pharmacy leaders facilitate more informed, data-driven conversations. The use of an expert panel of practicing oncology pharmacists from a variety of settings provides confidence that the results are applicable to different practices. This data may help mitigate the contribution of unrealistic workloads to oncology pharmacist burnout. A large US-based survey of oncology pharmacists found that those with high levels of burnout reported working more hours per week and reported a greater intent to leave their current job as compared to those without burnout. 15
The consensus statements provided in this study will also help pharmacy leaders. It will provide the tools necessary to determine workload distribution, benchmarking, and provide useful data that can be used for the creation of new positions. For example, if there are a certain number of tasks that need to be completed in a practice setting, these results can be readily utilized to help determine the number of oncology pharmacist FTEs required to meet those needs. The findings presented herein can also be relevant in a real-world setting where tasks are unevenly distributed among various oncology pharmacists. Furthermore, this ties position justification directly to job-specific tasks as opposed to the traditional pharmacist justification method of cost-savings. 16 The results, which outline the time and resources required for task completion, can also be easily understood by the non-oncology hiring managers and non-pharmacist leadership, further enhancing their practical value.
The consensus statements presented are consistent with results from previous studies that included a component of time in their evaluations of oncology pharmacists. One study evaluating an oncology pharmacist in a newly established medical oncology clinic determined that most patient education sessions lasted less than 30 min. 5 A second study, evaluating oncology pharmacists at a single institution, found that most medication therapy management visits (of which greater than 60% focused on education of chemotherapy) took an average 20 min of face-to-face time and 18 min of documentation time. 7 Both are consistent with the consensus statement that a patient education session takes on average 30 ± 6 min. An additional study that detailed the productivity of a single oncology pharmacist over a 14-day period found that approximately half of pharmacist interventions took more than 10 min to complete. 6 This again is in line with the consensus statements where nearly all of the patient care tasks take, on average, more than 10 min to complete.
It is difficult to place too much weight on the percentage of agreement in those tasks that reached consensus in round 1 versus round 2 given the biased nature of the controlled feedback incorporated into round 2 as part of the Delphi method. Nonetheless, it seemed that a larger proportion of patient care related tasks reached consensus in round 1 as compared to non-patient care tasks. This speaks to some of the heterogeneity that exists in non-patient care tasks. Pharmacists often receive less formal training in these non-patient care areas, may have less dedicated time to complete non-patient care related tasks, and may be more likely to prioritize patient care tasks. There were a few tasks, based on open-ended feedback from round 1, that required drastic modifications. The statement associated with “facilitating access to medications via working with insurance companies and financial counselors” was revised to increase the time requirement by 3-fold (from 15 to 45 min) between the 2 rounds. Time requirements for “policy, guideline, and drug monograph development” were revised by a 4-fold increase, and the “trainee education and supervisory activities associated with learners” task was revised to 3 h per learner per day from an original time of 2 h. This highlights the need for prospective studies that directly measure the amount of time it takes pharmacists to complete the tasks to validate these consensus statements.
Limitations of this study include the relatively small number of experts who completed the Delphi Survey. These experts also tended to be experienced clinical pharmacists with 78% with 6 or more years of experience working primarily in academic centers and community centers. New oncology pharmacists or those completing tasks outside of normal job functions may not complete tasks in the same amount of time. While the composition of the expert panel was solicited from a large pool, it is possible that those with strong opinions or those who feel overworked were more likely to volunteer—introducing bias. This may be supported, at least in part, by the fact that to reach consensus many of the statements were revised to increase the amount of time necessary to complete the associated task.
Conclusion
This project produced the first comprehensive consensus statements for the average time necessary for an oncology pharmacist to complete 24 common patient and non-patient care-related tasks for pharmacists practicing in academic and community centers in the US. The statements are endorsed by HOPA and ACCP Hematology/Oncology PRN and may be used to justify, measure, and evaluate oncology pharmacists across the US. However, future studies are required to confirm these consensus statements.
Footnotes
Acknowledgements
We would like to acknowledge Dr Kevin White, MD, PhD for his insight and advice as it related to the Delphi survey method and expertise in presenting the survey results in an effective manner.
We would also like to acknowledge student pharmacist, Sara Ali, for her contribution as an active member of the POPBC during the duration of this project.
Author contributions
Conception and design: SG, JS, KA, GR
Collection and assembly of data: SG, FR, KA, BG, LL, NP, DQ
Data analysis and interpretation: All authors
Manuscript writing: All authors
Final approval of manuscript: All authors
Accountable for all aspects of the work: All authors
Consent to participate
This study was deemed to be exempt from review and the requirement to consent by the IRB of the University of California, Irvine.
Data availability
All data collected as a part of this study is presented in this manuscript. Any questions can be directed to the corresponding author.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study was deemed to be exempt from review by the IRB of the University of California, Irvine.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hematology/Oncology Pharmacy Association.
