Abstract
Background
Although syringe preparation for large-volume (>3 mL) subcutaneous (SC) drugs represents a significant workflow burden for pharmacists, their preferences for alternatives such as on-body delivery systems (OBDSs) are unexplored.
Objective
To evaluate pharmacists’ preferences for preparing OBDSs vs. syringes.
Methods
In this cross-sectional study, pharmacists in US academic or community settings with experience preparing SC daratumumab/hyaluronidase (HYAL), rituximab/HYAL, pertuzumab/trastuzumab/HYAL, and/or efgartigimod/HYAL completed a double-blinded, 21-item, online survey that included questions about preferences regarding the preparation of prefilled syringes versus an OBDS.
Results
Thirty pharmacists completed the survey. 100% responded that the OBDS appeared to be easy to prepare and easy to learn how to prepare and preferred it to syringe preparation. In response to a preparation scenario without reduced warming time that included preparation specifics, 86.67% preferred OBDS preparation to the syringe used to administer daratumumab/HYAL due to (1) time required to prepare the drug, (2) effort required to prepare the drug, and (3) optionality in drug preparation location. 29 pharmacists (96.67%) felt that the OBDS would reduce burden, 30 (100%) felt that it would improve efficiency, and 27 (90%) felt that it would reduce preparation errors. 22 pharmacists (73.33%) felt that the OBDS could provide optionality since it can be prepared outside of the pharmacy, and 100% felt that OBDS preparation would eliminate needlestick injuries.
Conclusion
Pharmacists reported that an OBDS would be easy to prepare and to learn how to prepare and would improve pharmacy efficiency and safety compared with syringes used for large-volume SC drug administration.
Introduction
Although pharmacists and pharmacy technicians are instrumental in ensuring accurate medication preparation and safe administration, little research attention has been paid to their preferences regarding subcutaneous (SC) drug preparation. Drugs for which this insight would be useful, among others, include daratumumab/ hyaluronidase (HYAL) (Darzalex Faspro) for the treatment of multiple myeloma; rituximab/ HYAL (Rituxan Hycela) for the treatment of follicular lymphoma, previously untreated diffuse large B-cell lymphoma, or chronic lymphocytic leukemia; pertuzumab/ trastuzumab/ HYAL (Phesgo) and trastuzumab/ HYAL (Herceptin Hylecta) for the treatment of human epidermal growth factor receptor 2-positive breast cancer; and efgartigimod/ HYAL (Vyvgart Hytrulo) for the treatment of anti-acetylcholine receptor antibody-positive generalized myasthenia gravis. Biologics such as these are among a rapidly growing class of large, structurally complex therapeutic proteins used to treat a range of serious conditions, most commonly cancer and autoimmune disorders.1,2
While intravenous (IV) administration is appropriate for many biologics, SC administration is often preferable for longer-term and chronic conditions and doses are currently most commonly prepared in clinics with manually pushed syringes and direct oversight. 1 Due in part to the difficulties associated with increasing the concentration of biologics, these drugs are often administered as large-volume formulations, and a vial with a syringe for extraction of the drug from the vial is usually the first commercially available administration format for these large-volume formulations. 1
While not necessary, coformulation with HYAL is sometimes used in large-volume SC formulations to enhance permeation and increase injection speed. 1 Unfortunately, HYAL coformulation is associated with a range of preparation and administration challenges due to its high viscosity, which also leads to the use of larger-bore needles in compounding than are necessary when large-volume SC drugs are not coformulated with HYAL. 1 When using a large-volume SC delivery system like an on-body delivery system (OBDS), coformulation with HYAL is not needed.
Depending on the steps involved in their preparation, the time required for manual preparation of SC biologics varies (e.g., 8.7 min for trastuzumab, 3 10.8 min for rituximab, 4 28.1 min for daratumumab due to its warming time). 5 SC biologics are prepared in clinic and hospital pharmacies with a time- and labor-intensive multistep process involving vials, transfer needles, and syringes; for example, the current preparation protocol for daratumumab/ HYAL syringes is summarized in Table 1. This preparation process represents a significant burden for pharmacists and pharmacy technicians and may also increase the risk of needlestick injuries and musculoskeletal issues associated with repetitive, forceful movements. Research into the prevalence and prevention of repetitive strain injuries in pharmacists and pharmacy technicians is unfortunately limited. Significant increases in the average number of compounded sterile preparations per month, such as following the integration or expansion of health service networks, can result in hand-related repetitive strain injuries for pharmacists and pharmacy technicians. 6 A few of the most relevant strategies for the prevention or reduction of these repetitive strain injuries have included replacement of higher-gauge (20 or 19) and longer (1.5-inch) needles with lower-gauge (18 or 16) and shorter (1-inch) alternatives to reduce the manual pressure required to transfer the same amount of fluid and the introduction of automated compounding systems. 6 However, these strategies would not be needed with the utilization of an OBDS that would allow for nurse preparation at the patient's chairside.
Preparation protocol for daratumumab / hyaluronidase.
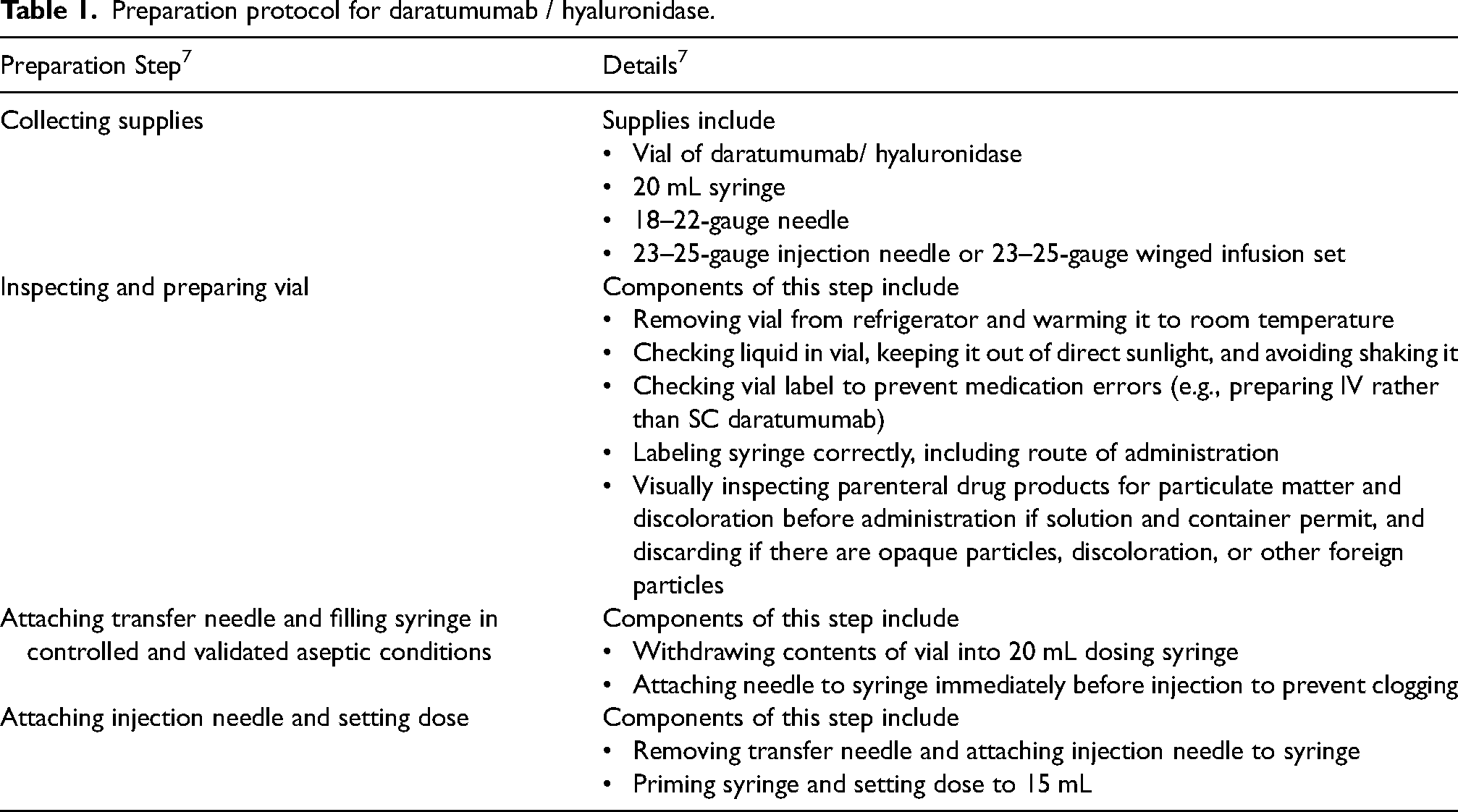
OBDSs can replace the use of syringes for the administration of large-volume SC formulations and eliminate the need for coformulation with HYAL. 1 OBDSs may mitigate physical strain and errors for pharmacists and pharmacy technicians due to a streamlined, single-step, hands-free filling process and needleless preparation. The manual preparation step for an OBDS can be more efficient than traditional syringes because they are designed to automatically fill once preparation is initiated, which would allow preparers to attend to other responsibilities. OBDSs may potentially reduce or eliminate the time-consuming step of warming drugs to ambient temperatures, although this benefit requires additional research. In the situation that the drug vial is refrigerated and the OBDS is left at room temperature, the mechanism for potential drug warming via the OBDS revolves around transfer of heat during the preparation process and while the OBDS is worn. Warming time is currently a burden for pharmacists and pharmacy technicians, and reducing the total warming time required (e.g., from 15 to 5 min) or eliminating the need for it would allow for faster turn-around time for providing treatment to the patient and could reduce drug waste. A prospective, cross-sectional, time and motion survey study with 26 participants comparing IV to SC daratumumab found that preparation of daratumumab/ HYAL took a median of 28.1 min, of which half would be spent waiting for the drug to reach room temperature. 5 Similarly, efgartigimod/ HYAL must be removed from the refrigerator at least 15 min before injection to allow for it to warm to room temperature. 8 While a 15-min warming period might not seem overly burdensome at first, when applied across numerous patients—for example, accumulating to 10 h of warming time per month for 40 patients—it represents a substantial net delay in facilitating treatment delivery to the patient.
OBDSs provide optionality in the preparation site, notably by enabling nurses to prepare medications right at the patient's chairside, thus alleviating some of the sterile compounding workload placed on pharmacies. This shift is particularly significant given the challenges associated with training, standardization, monitoring, and regulating sterile compounding processes, which have historically been fraught with issues. Such problems include compounding errors—ranging from incorrect doses or concentrations to contamination or improper labeling—that can lead to severe complications, or in the worst cases, fatal outcomes for patients.9–16 These compounding errors may also be expensive, as many oncology drugs are paired with on-patent regimens. Additionally, the variability in practices, such as the selection of the appropriate needle diameter for SC drugs, exacerbates the risk of adverse events, including syringe blockages, unnecessary backflow, and increased patient discomfort. 16 Despite these concerns, the exact prevalence of such errors and their consequent adverse effects remains largely unquantified. 14
Preparation and administration with an OBDS can significantly minimize occupational exposure by securely containing the medication and preventing any accidental spillage or aerosolization, thereby protecting healthcare workers and patients from potentially harmful drug exposure. Chairside preparation or at-home patient self-administration (when appropriate) could alleviate workflow burdens and the risk of preparation errors for pharmacies by reducing or eliminating the necessity for SC drug preparation by pharmacists and pharmacy technicians, including sterile hood compounding, without compromising the integrity of medication review. Alternatively, pre-mixing medication can also increase the risk of drug waste due to patient no-shows, cancelled appointments, therapy changes, or discharge against medical advice. 17
Due to industry interest and investment and because most large-volume SC biologics are used to treat cancers, 2 which are increasing in prevalence, 18 the challenges faced by pharmacists when preparing SC syringes are likely to increase in frequency and severity. To address the shortcomings of syringes and syringe pumps, therefore, an OBDS (enFuse, henceforth referred to as Product X) was developed for the delivery of large-volume SC drugs. In the US, product X is a drug-device combination product that goes through clinical trials then gains approval for use with each specific drug. To date, it has been FDA-approved for the self-administration of pegcetocoplan (EMPAVELI) for the treatment of paroxysmal nocturnal hemoglobinuria. 19
In the context of the challenges faced by pharmacists and pharmacy technicians when preparing SC drugs, the gaps in the literature reviewed here, and the potential benefits of an OBDS, the objective of this study was to evaluate pharmacists’ preferences for preparing a manually pushed, prefilled syringe for viscous drugs coformulated with HYAL versus an OBDS (Product X, shown in Figure 1) that does not require coformulation of drugs with HYAL and can allow for chairside preparation by nurses.

Product X on-body delivery system.
Methods
Study design and participants
This was a double-blinded, online survey study run by a third-party vendor with a database of more than 1.5 million respondents, including pharmacists. Pharmacists were sent an online survey to complete between 4 October and 11 October 2023. The responses collected and presented here are from academic or private practice pharmacists in the US. These individuals reported, in a set of screening questions, their experience with preparing medications such as daratumumab/ HYAL, rituximab/ HYAL, pertuzumab/trastuzumab/ HYAL, trastuzumab/ HYAL, and/or efgartigimod/ HYAL.
Study survey and procedures
The full list of survey questions can be found in Supplementary Information. The survey included 15 screening questions collecting information about professional experience. Following completion of the screening questions, pharmacists were given a written introduction to the survey and shown a 30-second, unbranded demonstration video of an OBDS (Product X, shown in Figure 1) followed by a 4-minute presentation of how Product X works. Once the introduction video and presentation were viewed, the pharmacists completed the survey. The 21-item survey included questions about the apparent ease of preparing and learning how to prepare Product X and thoughts and preferences regarding the preparation of Product X or a syringe for large-volume SC biologics.
Statistical analysis
Responses to survey questions were tabulated and descriptive statistics were calculated from the survey data. Results for questions asked with Likert scale response options are presented as mean (standard deviation [SD]) or as number and percentage of participants (n [%]) choosing each category, and results for questions with binary responses are presented as number and percentage of participants (n [%]). No formal hypothesis testing was performed.
Ethics
Because this hypothesis-generating, non-interventional study was comprised entirely of an anonymous, online survey, institutional review board approval was not required. For informed consent, at the outset of the survey, participants were required to agree to the survey vendor's compliance statement. This compliance statement included stipulations that participants would be compensated for their time, that they were legally allowed to disclose information asked about in the survey, and that they had read the vendor's privacy policy. Respondents who chose not to agree to the compliance statement were unable to complete the survey and were excluded from the sample.
Results
Participant characteristics
A total of 30 pharmacists completed the survey; details about their professional experience are presented in Table 2. Participants lived in New York (n = 5), Indiana and Pennsylvania (n = 4 each), North Carolina (n = 3), New Jersey and Ohio (n = 2 each), and Alabama, Arizona, Arkansas, California, Georgia, Massachusetts, Missouri, Montana, Nevada, and South Carolina (n = 1 each). Most participants (16 [53.33%]) worked in university-affiliated academic medical centers, 9 (30.00%) worked in non-university-affiliated community hospitals, 4 (13.33%) worked in private practice, and 1 (3.33%) worked in long-term care. Most participants were directors of pharmacy (n = 13, 43.33%) or clinical pharmacy specialists (n = 11, 36.67%), 4 (13.33%) were infusion floor pharmacists, and there was 1 (3.33%) home infusion pharmacist and 1 (3.33%) long-term care pharmacist. Participants had been practicing in a clinical setting for a mean of 17.77 years (SD 9.93).
Participant professional experience.
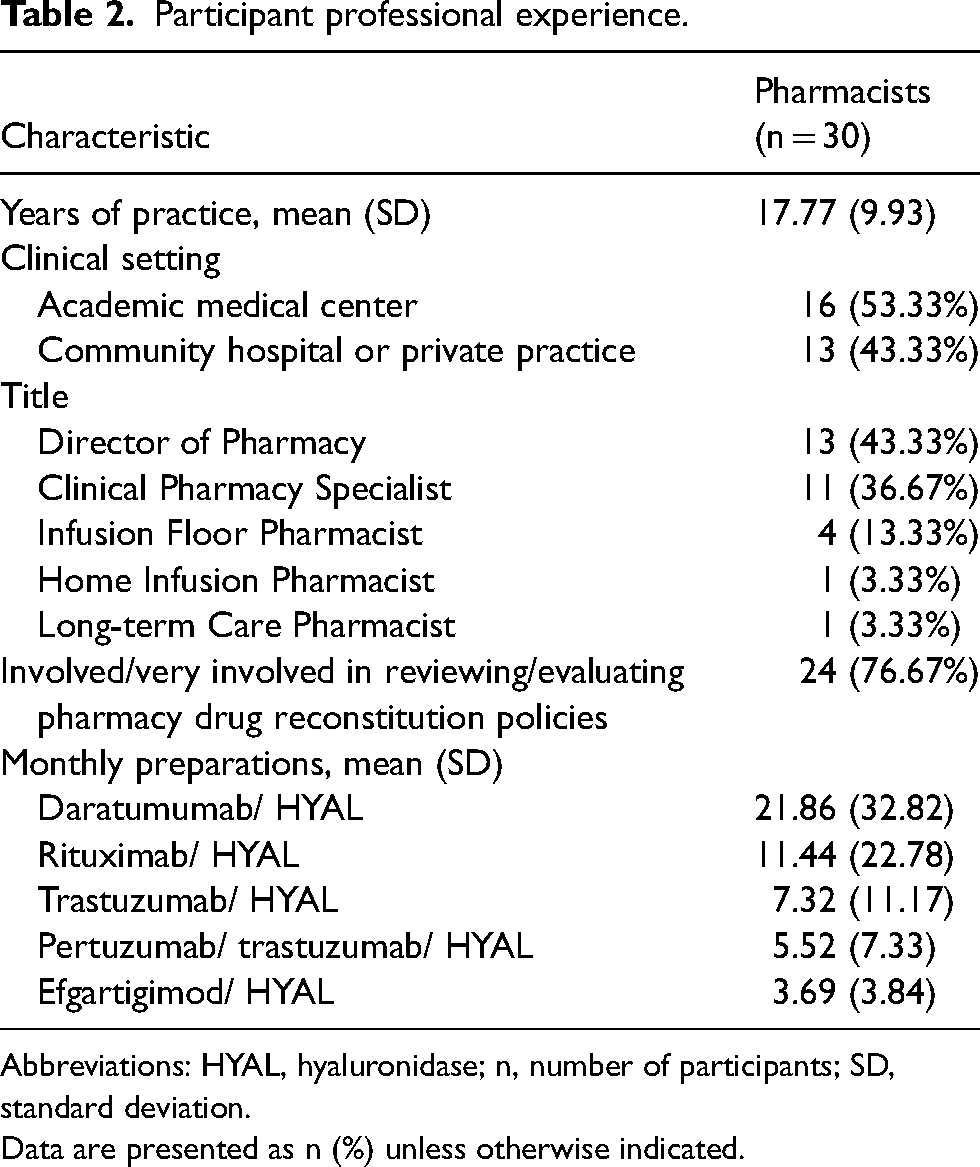
Abbreviations: HYAL, hyaluronidase; n, number of participants; SD, standard deviation.
Data are presented as n (%) unless otherwise indicated.
With respect to familiarity with the pharmacy preparation of SC drugs, on a scale of 1–5 (1 = not familiar, 5 = very familiar), pharmacists rated themselves as having a mean familiarity of 4.60 (SD 0.76) for daratumumab/ HYAL, 4.23 (SD 0.99) for rituximab/ HYAL, 4.23 (SD 0.96) for trastuzumab/ HYAL, 3.93 (SD 1.15) for pertuzumab/ trastuzumab/ HYAL, and 2.83 (SD 1.69) for efgartigimod/ HYAL. Mean number of preparations per month were 21.86 (SD 32.82) for daratumumab/ HYAL, 11.44 (SD 22.78) for rituximab/ HYAL, 7.32 (SD 11.17) for trastuzumab/ HYAL, 5.52 (SD 7.33) for pertuzumab/ trastuzumab/ HYAL, and 3.69 (SD 3.84) for efgartigimod/ HYAL. On a 1–5 scale (1 = not involved, 5 = very involved), participants rated themselves as having a mean 4.20 (SD 1.11) level of involvement with reviewing and evaluating policies governing drug reconstitution in the pharmacy.
Survey results
Ease of learning and completing preparation
All pharmacists (100%) reported that preparation of the OBDS, which involves inserting a vial into the transfer base, appeared to be easy to learn and easy to complete for pharmacists and pharmacy technicians.
Preference for preparation of OBDS vs. syringe
In response to images of Product X showing its needleless preparation and of syringes showing their manual preparation, 100% of the pharmacists reported that they would prefer preparing the OBDS over the preparation of the syringes currently used to administer large-volume SC biologics such as daratumumab/ HYAL and rituximab/ HYAL.
In a detailed scenario that provided specifics about the preparation of SC daratumumab/ HYAL without a reduction in warming time (Table 3), 26 pharmacists (86.67%) preferred preparation of the OBDS over that of the syringe currently used to administer daratumumab/ HYAL for the treatment of multiple myeloma, assuming safety and efficacy were comparable. Of these 26 pharmacists, 13 pharmacists (50%) rated their preference as strong, 11 pharmacists (42.31%) rated their preference as moderately strong, and 2 pharmacists (7.69%) rated their preference as moderate, with a mean overall preference on a scale of 1–5 (1 = weak preference, 5 = strong preference) of 4.42 (SD 0.63). The top 3 most important factors affecting preference in this scenario were (1) the amount of time required to prepare the drug, (2) the amount of effort required to prepare the drug, and (3) the optionality provided by Product X in drug preparation location.
Preparation considerations included in detailed scenario regarding daratumumab/ hyaluronidase preparation.

In a detailed scenario that provided specifics about the preparation of SC daratumumab/ HYAL with a reduction in warming time, all of the pharmacists (100%) preferred preparation of the OBDS to the syringe currently used to administer SC daratumumab/ HYAL. This preference was strong among 12 pharmacists (40%), moderately strong among 16 pharmacists (53.33%), and moderate among 2 pharmacists (6.67%), with a mean overall strength of preference on a scale of 1–5 (1 = weak preference, 5 = strong preference) of 4.33 (SD 0.60). The top 3 most important factors affecting preference in this drug-specific scenario were (1) the amount of time required to prepare the drug, (2) elimination of the warming step, and, tied for (3), storage time of the prepared drug and amount of effort required to prepare the drug; these results are shown in Figure 2. In an open-ended question about reasons for preference and strength of preference, responses included “less prep time and lack of needles,” “ease of preparation, less staff time, reduced waste for no-shows,” “less consumable and labor costs,” “less risk of manual injury from repetitive motion, ability to reconstitute at the chairside,” “patient convenience and moving patients out of the infusion center to a home care setting,” and “simplicity, safety, and increased flexibility of prep and admin.” In comparison, in a drug-agnostic scenario, the 3 most important features of preparation were (1) hands-free filling, (2) elimination of the warming step, and (3) optionality in the site of preparation.
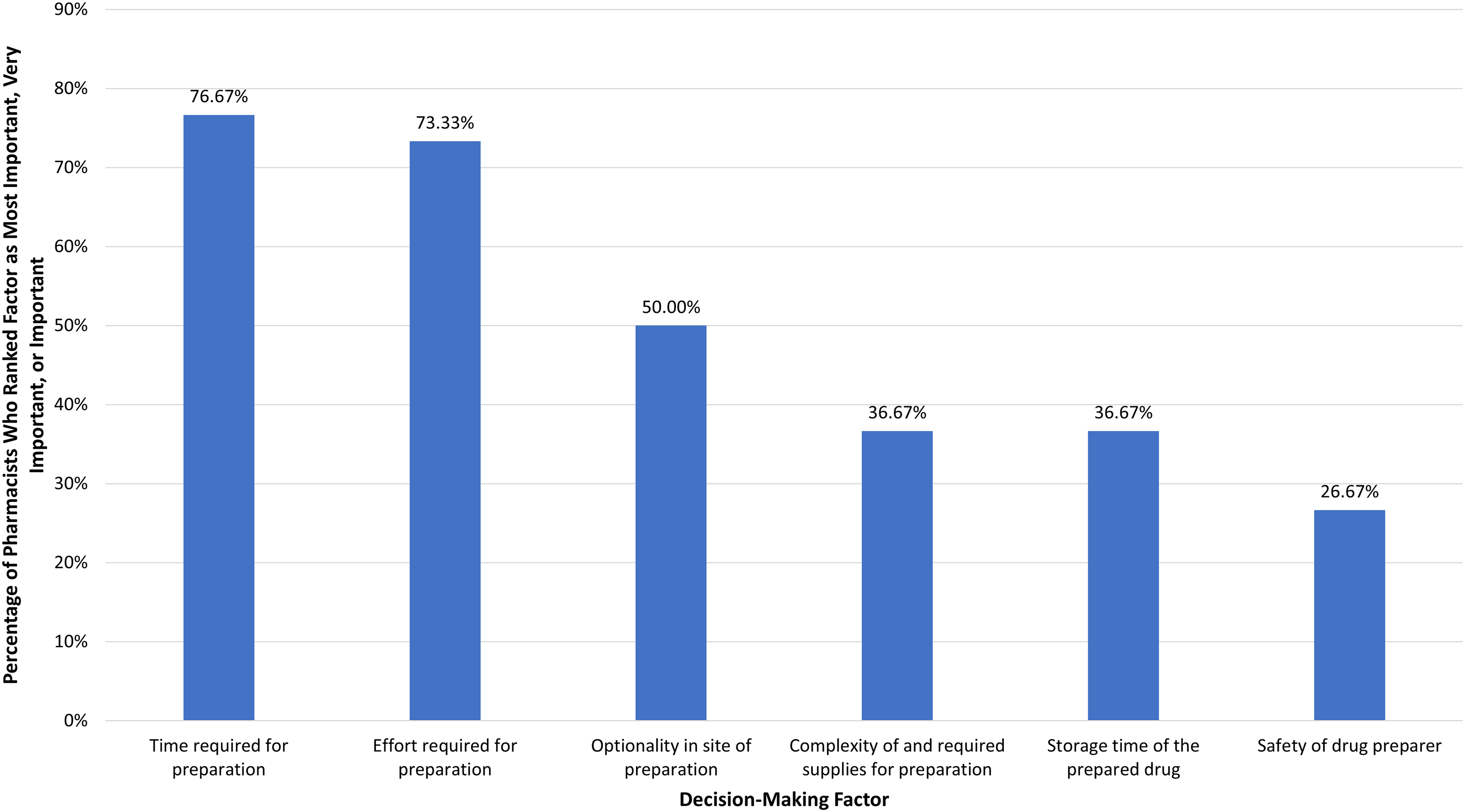
Percentage of pharmacists rating factors as most important, very important, or important in influencing decision-making regarding preparation of daratumumab/ hyaluronidase (survey question 6 in Supplementary Information).
Overall, regardless of the drug being prepared or the details of preparation, the 4 most valuable attributes of an SC administration method in decision-making about drug preparation were (1) hands-free, single-step preparation; (2) elimination of warming time; (3) flexibility in preparation site; and (4) needleless preparation. These results are further illustrated in Figure 3.
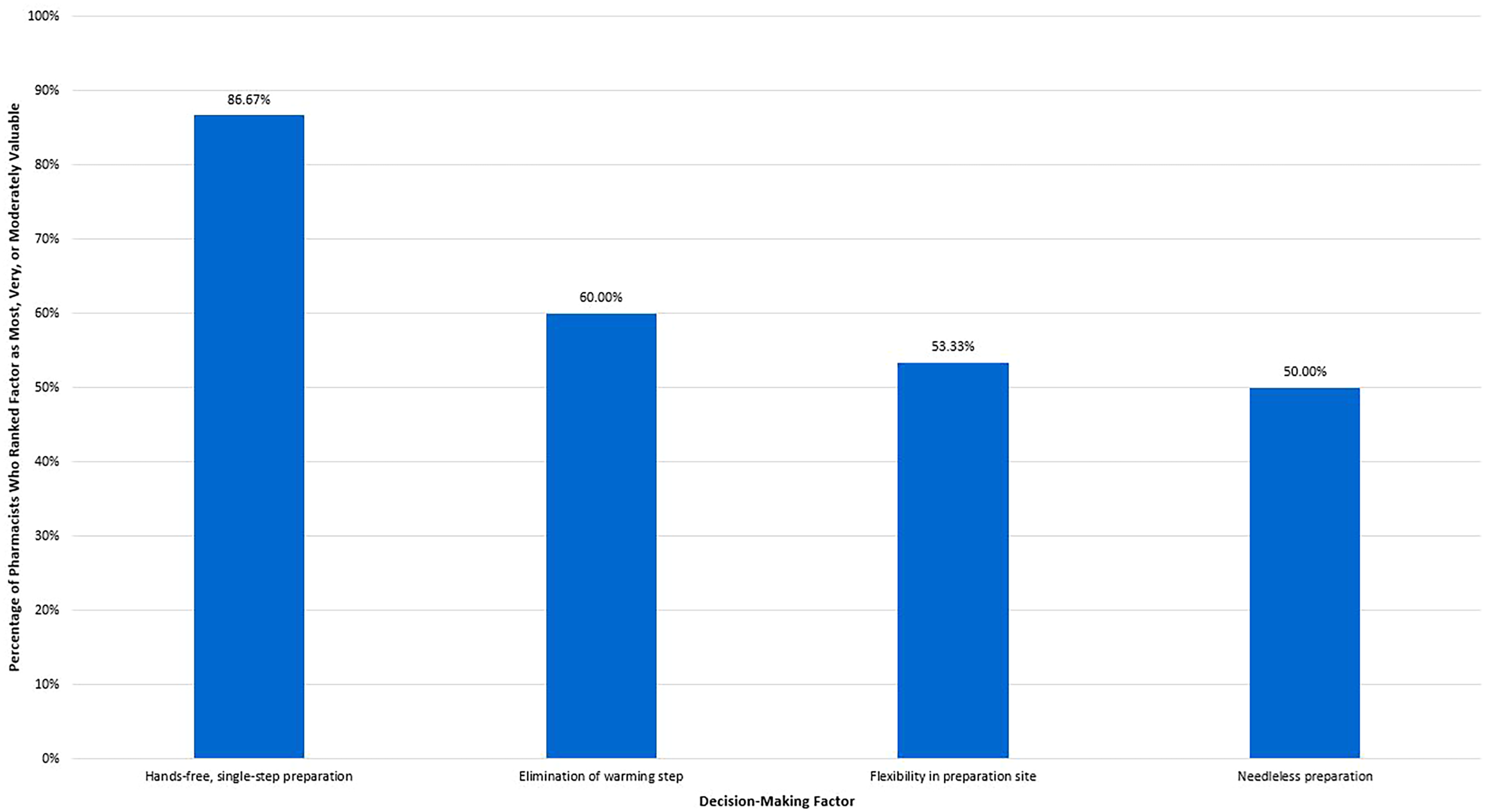
Percentage of pharmacists rating factors as most important, very important, or moderately important in decision-making regarding preparation of subcutaneous administration methods (survey question 11 in Supplementary Information).
This exploratory study was not powered for statistical comparison by participant characteristics. However, breaking down the sample by clinical setting, 100% of pharmacists from each setting (n = 16 for academic medical center, n = 9 for community hospital, n = 4 for community practice, n = 1 for other) preferred preparation of Product X to syringes for large-volume SC drugs and for the treatment of multiple myeloma compared with daratumumab syringes if Product X could eliminate warming time, with the average strength of this preference (1: weak, 5: strong) being 4.38, 4.11, 4.5, and 5, respectively. The preference for Product X vs. the daratumumab syringe mildly weakened without the elimination of warming time, with 81.25% of academic medical center, 100% of community hospital, 75.00% of community practice, and 100% of other setting pharmacists preferring preparation of Product X to that of daratumumab syringes, with the average strength of this preference being 4.46, 4.22, 4.67, and 5, respectively.
Similarly, breaking down the sample by years of experience, 100% of pharmacists from each bracket of experience (n = 8 for 0–10, n = 15 for 11–12, n = 2 for 21–30, n = 5 for >30 years of experience) preferred preparation of Product X to syringes for large-volume SC drugs and for the treatment of multiple myeloma compared with daratumumab syringes if Product X could eliminate warming time, with the average strength of this preference (1: weak, 5: strong) being 4.5, 4.2, 4, and 4.43 respectively. The preference for preparation of Product X vs. the daratumumab syringes mildly weakened without the elimination of warming time, with 87.50% of pharmacists with up to 10 years of experience, 86.67% of pharmacists with 11–20 years of experience, and 100.00% each for pharmacists with 21–30 or >30 years of experience, with the average strength of this preference being 4.31, 4.20, 4.75, 4.43, respectively.
Benefits of an OBDS if used for all large-volume SC drugs in the clinic setting
Results regarding the benefits of the replacement of syringes with OBDSs for the delivery of all large-volume SC drugs in the clinic setting are depicted in Figure 4. With respect to physical burden, 29 pharmacists (96.67%) felt that the OBDS would reduce burden on pharmacists and pharmacy technicians due to its less complex and labor-intensive drug preparation process. All of the pharmacists (100%) felt that the OBDS would improve efficiency (faster preparation of drugs) due to its single-step, hands-free filling process even without the elimination of warming time. A total of 27 pharmacists (90%) felt that the OBDS would reduce errors associated with the preparation process due to its single-step, hands-free filling process, and 22 pharmacists (73.33%) felt that the OBDS could provide optionality in adjusting hospital protocols regarding preparation location.
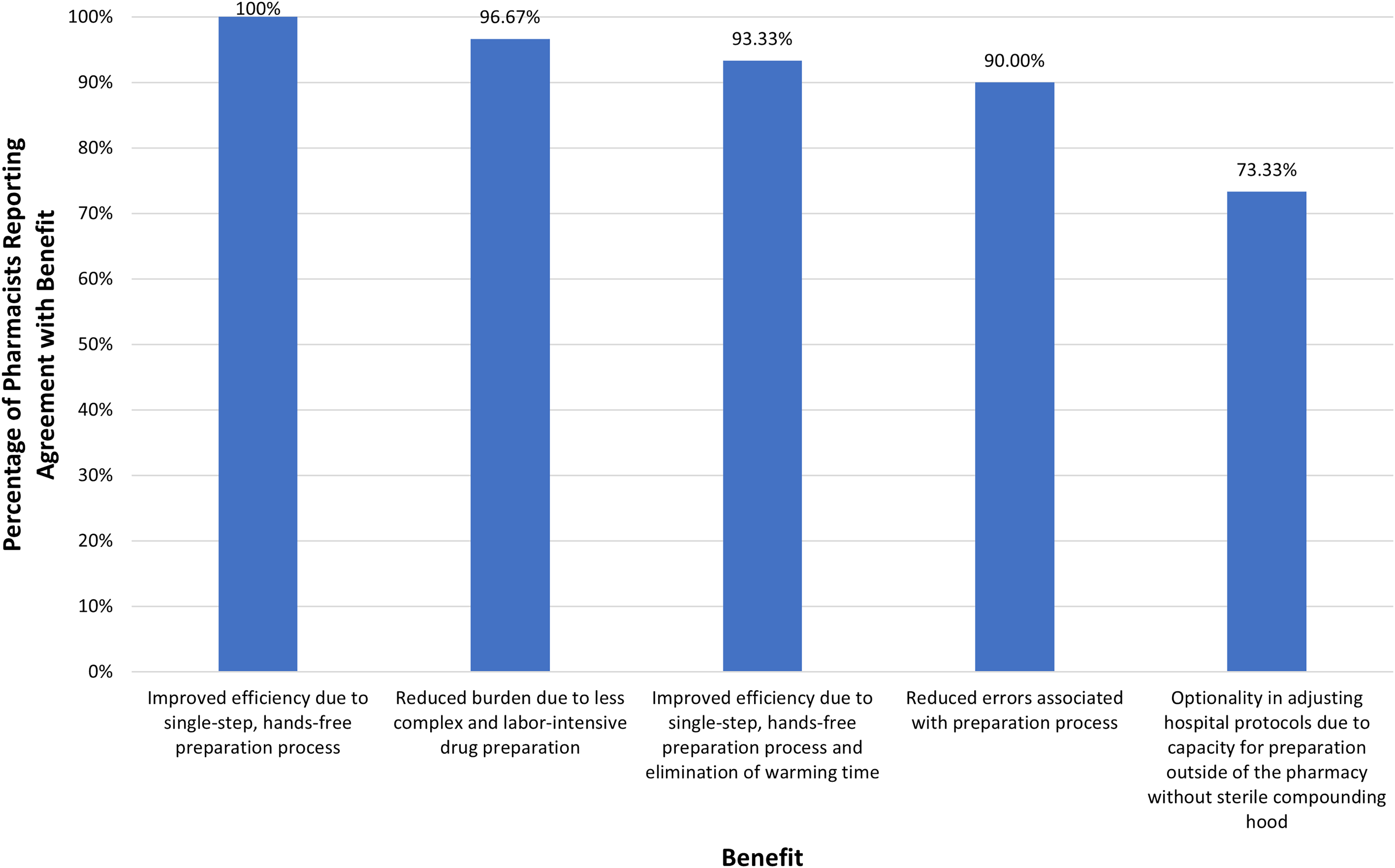
Percentage of pharmacists reporting agreement with benefits of using Product X for delivery of all large-volume subcutaneous drugs in the clinic (survey questions 12–14 in Supplementary Information).
Needlestick injuries
A total of 25 pharmacists (83.33%) reported that they or a pharmacist or pharmacy technician they knew had received a needlestick injury from a syringe during sterile compounding. The most commonly selected acceptable rate of needlestick injuries over their careers (mean 17.77 years) in pharmacists and pharmacy technicians was <1% (16 [53.33%]), followed by 1–2% (10 [33.33%]), with a weighted average acceptable rate of 1.5% (range 1.1%–1.9%). All (100%) of the pharmacists felt that preparation of the OBDS would eliminate the risk of needlestick injuries for pharmacists and pharmacy technicians.
Knowledge of needle gauge used for SC administration
In a series of 3 questions about daratumumab/ HYAL (administered with a 23–25-gauge needle), rituximab/ HYAL (administered with a 25–30-gauge needle), and pertuzumab/ trastuzumab/ HYAL (administered with a 25–27-gauge needle), pharmacists were asked to indicate what gauge(s) of needle(s) is used to administer these drugs or whether they did not know the correct needle size used. For daratumumab/ HYAL, there were no incorrect responses regarding needle gauge, but 6 pharmacists (20%) indicated that they did not know the correct needle size. For rituximab/ HYAL, 4 pharmacists (13.33%) answered incorrectly and 7 (23.33%) indicated that they did not know the correct needle size, for a total of 36.67% incorrectly informed or uninformed pharmacists. Finally, for pertuzumab/ trastuzumab/ HYAL, 2 pharmacists (6.67%) answered incorrectly and 7 (23.33%) indicated that they did not know the correct needle size for a total of 30.00% incorrectly informed or uninformed pharmacists.
Discussion
In this double-blinded, online survey study of 30 pharmacists in the United States, participants reported that an OBDS, Product X, appeared to be easy to prepare and easy to learn how to prepare and expressed an overwhelming preference for the hands-free filling and needleless preparation afforded by an OBDS like Product X over the preparation process currently required for traditional syringe administration of large-volume SC formulations such as daratumumab/ HYAL or rituximab/ HYAL. Although the specific reasons and ranking of reasons varied by preparation scenario details, factors commonly affecting this preference included hands-free filling and needleless preparation, potential elimination of the warming step, and optionality in the site of preparation.
As briefly reviewed in the introduction, OBDSs could have several benefits over traditional, manually pushed syringes for pharmacists and pharmacy technicians. The factors affecting decision-making about the preparation of SC administration methods considered most valuable in this sample of pharmacists were hands-free, single-step filling, optionality in the site of preparation, and potential elimination of the warming step. Most pharmacists further agreed that the benefits of replacing syringes with an OBDS such as Product X for all SC administrations in the clinic setting would include improved efficiency, reduced burden on pharmacists and pharmacy technicians, reduced errors associated with the preparation process, and optionality in adjusting hospital protocols due to capacity for preparation outside of the pharmacy without a sterile compounding hood. Because an OBDS is a closed system and therefore carries no aseptic or exposure concerns, it can be prepared at the patient's chairside. This optionality in the site of preparation is further supported by the fact that OBDSs can be stored in automated medication dispensing systems (e.g., the BD Pyxis ES platform) that allow for secure medication storage in patient care units for use by nurses as needed. Chairside preparation by nurses when patients are ready for injections would reduce wasted drug due to patient no-shows or self-discharges against medical advice. Patient no-shows could themselves be reduced due to the alleviation of needle phobia possible with a hidden needle mechanism. 20 Storage in patient care units could further alleviate challenges with limited pharmacy storage capacity by eliminating the need to prepare and store drugs in advance of administration.
Drug wastage represents a significant cost to healthcare systems and unnecessarily escalates the cost of care, particularly when the drugs in question are higher in cost. 21 Unfortunately, less is known about wasted drug product amount and cost due to patients being unwilling or unable to attend clinic appointments, resulting in patient no-shows. 22 However, while drugs such as daratumumab/ HYAL must be used within 4 h of preparation, the unpunctured vial for use in an OBDS such as Product X can be safely used for up to 24 h after preparation (see Table 3). This substantial prolongation of drug stability should significantly reduce drug wastage due to no-shows.
While most pharmacists in this study had received or knew a pharmacist or pharmacy technician who had received a needlestick injury, the weighted average acceptable rate of needlestick injury was 1.5% and all reported that Product X would eliminate the risk of needlestick injuries for pharmacists and pharmacy technicians. Needlestick injuries are often underreported; a low reporting rate therefore does not necessarily correspond to a low rate of injuries. 23 Needlestick injuries may cause physical and emotional distress, incur direct and indirect costs such as follow-up care and missed workdays, and carry a risk of transmission of bloodborne pathogens such as HIV and hepatitis B or C. 23 Unfortunately, research into needlestick injuries in pharmacists and pharmacy technicians is extremely limited, in part because pharmacists and pharmacy technicians are often not included in studies evaluating needlestick injuries in healthcare workers. In one cross-sectional study of 524 government pharmacists in Perak, Malaysia, the overall prevalence of needlestick injury was 23.1%, of which 10.3% did and 12.8% may have involved contamination. 7 Failing to report needlestick injury was common among these pharmacists, with reasons for not reporting needlestick injuries including not knowing whose duty it was to report (45.5%) and being too busy (44.7%). 7 Additional research into needlestick injury prevalence in pharmacists and pharmacy technicians and potential prevention methods is clearly needed.
The lack of a systematic approach to important considerations such as the selection of the appropriate needle diameters for SC injections can result in increased difficulty with administration and near-misses and adverse events associated with using injection devices, including syringe blockage, backflow, and patient pain. 24 Preventable adverse events such as these are associated with significant and unnecessary costs for hospitals and patients and can require the needless use of additional equipment and staff time.25,26 The findings that 20%–37% of the pharmacists surveyed were unfamiliar with the correct needle gauge used for the administration of daratumumab/ HYAL, rituximab/ HYAL, or pertuzumab/trastuzumab/ HYAL are therefore a cause for concern.
The United Kingdom provides a useful case study for how capacity challenges can lead to less optimal conditions for drug preparation. Although SC injection of mAbs can reduce treatment time by up to 97% compared with IV infusions,4,5,27 their regimens often involve months or years of multiple treatment cycles. These lengthy treatment regimens, in combination with the expanding availability of these treatments and the rapid increase in the number of patients requiring them, 28 can cause significant clinic backlog issues in chemotherapy units. 29 The combination of these factors has resulted in a crisis in the NHS's ability to deliver systemic anti-cancer therapies (SACT) to patients. 30 There are multiple potential solutions for these capacity challenges, such as expanding opening hours, improving scheduling procedures, staggering appointments, or facilitating preparation of mAbs by nurses. 29 Although increasing clinic capacity can result in untenable pressure on pharmacies to prepare sufficient drug product, one recent pilot study found that nurse-led mAb preparation reduce this pressure. 29
This study has limitations, including a small sample size, the potential for self-report bias, and the use of a survey that was not comprehensively validated (i.e., principal components analysis was not conducted and internal consistency was not evaluated). As the survey was only completed by a small number of pharmacists in the United States, the results may not generalize to other pharmacists in the United States or in other countries. Different countries have varying regulations regarding sterile compounding, which are influenced by organizations (e.g., United States Pharmacopeia) and institutional policies. In countries where pharmacy technicians or pharmacists are primarily responsible for daily medication preparation, the results described here are generally applicable. However, in regions where nurses are also preparing medications, the findings may be less generalizable due to differences in roles and responsibilities. To address these variations, a follow-up study is currently underway to explore these questions among pharmacy staff in the United Kingdom. Finally, although it was not comprehensively validated, the survey was evaluated for content and face validity by the authors and was then pretested with 3 pharmacists before its use in this study.
These hypothesis-generating findings will hopefully encourage additional research into potential improvements in the large-volume SC formulation preparation experiences of pharmacists and pharmacy technicians and in hospital pharmacy efficiency that may be possible with the replacement of current SC syringes with OBDSs.
Conclusion
This study provides valuable insights into the preferences of pharmacists regarding the preparation of administration methods for large-volume SC formulations such as daratumumab/ HYAL. Overall, the pharmacists surveyed here reported that they would prefer preparing an OBDS to the syringes currently used to administer large-volume SC drugs and felt that the use of such an OBDS for the administration of all large-volume SC formulations in the clinic setting could provide several key benefits, including improved pharmacy efficiency, reduced preparation time and errors, and preparation setting optionality. Optionality in preparation site would allow the SC administration method to be prepared at the patient's chairside by nurses or by patients in the home setting when appropriate, further reducing the burden on pharmacists.
Supplemental Material
sj-docx-1-opp-10.1177_10781552251326574 - Supplemental material for Evaluating pharmacist preferences: Preparation of a novel on-body delivery system vs. high-resistance, manual syringes for large-volume subcutaneous drugs
Supplemental material, sj-docx-1-opp-10.1177_10781552251326574 for Evaluating pharmacist preferences: Preparation of a novel on-body delivery system vs. high-resistance, manual syringes for large-volume subcutaneous drugs by Mehul Desai, Mitchell Blewett, Angela Yaniv, Adam Smith, Prit Patel, Catherine Loughran and Omar Rahman in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
Writing and editorial support for this manuscript were provided by Terri Levine, MSc, PhD, CMPP. The authors would like to thank the pharmacists who participated in this study.
Author contributions
Conceptualization: MD; Methodology: MD, OR; Formal Analysis: MD, OR; Investigation: MD; Resources: MD; Data Curation: MD, MB, AY, AS, PP, CL, OR; Writing – Original Draft: MD, MB, AY, AS, PP, CL, OR; Writing – Review and Editing: MD, MB, AY, AS, PP, CL, OR; Visualization: MD, MB, AY, AS, PP, CL, OR; Supervision: MD; Project Administration: MD; Funding acquisition: MD. All authors reviewed and approved the final version of the manuscript.
Consent to participate
For informed consent, at the outset of the survey, participants were required to agree to the survey vendor's compliance statement. This compliance statement included stipulations that participants would be compensated for their time, that they were legally allowed to disclose information asked about in the survey, and that they had read the vendor's privacy policy. Respondents who chose not to agree to the compliance statement were unable to complete the survey and were excluded from the sample.
Consent for publication
Not applicable.
Data availability
Data collected for the preparation of this manuscript are available from the corresponding author.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mehul Desai and Omar Rahman are employees of Enable Injections, Inc. Mitchell Blewett, Angela Yaniv, Adam Smith, Prit Patel, and Catherine Loughran have no relevant disclosures to report.
Ethical considerations
Because this non-interventional study was comprised entirely of an anonymous, online survey, institutional review board approval was not required.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Enable Injections, Inc.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
