Abstract
Introduction
The utilization of CDK4/6 inhibitors has led to compromised survival rates for breast cancer patients. Consequently, certain treatment aspects, involving adherence and drug-to-drug interactions, are gaining prominence. To develop chemotherapy regimens that are both effective and efficient, our main objective was to thoroughly characterize the drug-drug interactions that occur between cyclin-dependent kinase inhibitors and concurrently prescribed medications in hospitalized breast cancer patients.
Methods
In the current retrospective analysis, (January 2017 to January 2023), the baseline characteristics of patients under CDK4/6 chemotherapy were collected by reviewing the patient's medical records. Utilizing drug interactions checker softwares including Micromedex® online database system, Drugs.com® interaction checker, and UpToDate Lexicomp®, the potential for drug-drug interactions was further assessed.
Results
In this retrospective analysis, total of 75 co-medications were prescribed along with palbociclib and ribociclib. Upon analysing all co-prescribed classes of drugs, the potential drug interactions of palbociclib and ribociclib with analgesics, acid-reducing agents, and statins occurred frequently in cancer patients. In the 21-patient cohort, 17 patients (80.95%), were found to be having prevalence of potential drug-drug interactions out of which 41.26% had major pharmacokinetic interactions, 42.85% were moderate ones, while 15.87% were pharmacodynamic interactions.
Conclusion
The retrospective analysis identified the potential risks associated with drug-drug interactions of cyclin-dependent kinase 4/6 inhibitors. Potentially, the application of drug interaction detectors could facilitate additional implementation of research specially designed for interventions aimed at enhancing patient care.
Keywords
Introduction
Drug-drug interactions (DDIs) are frequently observed in cancer patients due to the administration of multiple medications, including hormonal therapy, anticancer drugs, and adjuvant treatments to manage comorbidities. Comprehending DDIs is an essential aspect of drug development as a significant alteration in exposure due to co-administered drugs can result in a loss of effectiveness or an adverse drug reaction (ADR), depending on the therapeutic window of the affected drug.1,2 DDIs are currently of significant concern in the fields of clinical oncology and clinical trial eligibility. The observed trend is attributed to the significant and swift rise in the number of treatments for oral chemotherapy, specifically those that are molecularly targeted therapies, with a considerable proportion of which are subject to metabolic and transport processes facilitated by cytochrome P450 (CYP) isozymes and P-glycoprotein (P-gp) respectively.3,4 The likelihood of hospitalized patients being impacted by DDIs is higher due to the prevalence of comorbidities, polypharmacy, and frequent modifications to therapy in this population. The occurrence of pharmacokinetic and pharmacodynamic DDIs has also been associated with an elevated likelihood of treatment failure, drug-related morbidity, and mortality. 5
DDIs have been identified as the cause of 20–30% of all ADRs in the general population. Additionally, it has been estimated that DDIs are responsible for approximately 4% of mortality in patients receiving chemotherapy. 6 According to Nigel (2014), the prevalence of CYP3A-mediated interaction potential with tyrosine kinase inhibitors and concurrently administered medications was 47%, 22%, and 11% for substrates, inhibitors, and inducers, respectively. 7 In multi-centric retrospective research conducted by Santiago Osorio et al. (2018), 105 patients with chronic myeloid leukaemia were given 134 tyrosine kinase inhibitors along with other medications. And a total of 159 DDIs were found, with at least one possible DDI containing 55 different medications and a mean of 4.8 concurrent medications. 8
In 2015, the USFDA granted accelerated approval, and in 2017, regular approval for the utilization of letrozole-based first-line therapy along with cyclin dependent kinase (CDK) 4/6 inhibitors palbociclib and ribociclib in postmenopausal women with advanced breast cancer which are human epidermal growth receptor factor-2 (HER-2) and hormone receptor (HR) positive. In India, both palbociclib and ribociclib are being prescribed in clinical set-up since 2017 and 2019 as a standard of care in the treatment of hormone positive (HR+)/ HER-2 negative metastatic breast cancer patients. In the intervention arm of palbociclib the incidence of all-grade gastrointestinal (GI) toxicities ranged from, nausea 24.5–51.5%, vomiting 14– 29.3%, diarrhoea 19.1–35%, and decreased appetite 12.8–18.6%. 9 For optimal management of these distressing symptoms of oral chemotherapy, National comprehensive cancer network (NCCN) has provided clinical practice guidelines for oncology palliative care and supportive care therapy.10,11 However, this may result in potential DDIs with respect to CYP450/transporter-mediated pharmacokinetic or pharmacodynamic interactions. Healthcare professionals are tasked with the responsibility of preventing harmful DDIs through various strategies. These strategies include avoiding interacting combinations, spacing co-administered medications, monitoring for early detection, implementing computerized screening or decision support systems, and providing patient education on prescription and non-prescription medications. Numerous studies have documented the dependency of clinicians on established computerized databases, both licensed and free, such as Lexi-Interact®, Micromedex® Drug Interactions, Drugs.com®, Medscape®, and Epocrates, for identifying potential DDIs.12,13 The occurrence of pharmacokinetic DDIs is most commonly associated with various iso-enzymes of the hepatic CYP450 and drug transporters, such as P-gp. 6
Since palbociclib and ribociclib have overlapping substrate specificity with CYP3A4 and P-gp, it makes them suitable targets for DDIs in chemotherapy regimens.14–17 There is a limited amount of systematic data available regarding the occurrence and mechanisms of interactions mediated by CYP3A4 and P-gp in real-world hospitalized patients receiving CDK4/6 inhibitors chemotherapy in India. Therefore, aim of this retrospective study is to assess the prevalence of potential DDIs associated with the use of CDK4/6 inhibitors in chemotherapy. The objective of this study is to categorize these interactions according to their severity and risk rating, by employing the Drugs.com®, IBM Micromedex® and UpToDate Lexicomp® drug interaction checking databases. It is imperative for pharmacists and clinicians to possess knowledge regarding the characteristics, operational capabilities, benefits, and drawbacks of diverse databases pertaining to DDIs. Additionally, the study also seeks to identify the potential DDIs, comorbidities, and the number of concurrently prescribed medications.
Methods
Study subjects and design
A five-year retrospective search was performed on the inpatient medical records of breast cancer patients at tertiary care Kasturba Hospital, Manipal, India from the period January 1st, 2017, to January 30th, 2023. The study protocol was approved by the Institutional Ethical Committee, with reference number IEC1:99/2023. A complete enumeration of the 1036 patients diagnosed with breast cancer were included in the current retrospective study. The investigation included 21 patients who were treated with CDK 4/6 inhibitor therapy, either premenopausal or postmenopausal, aged between 18 to 85 years as per the inclusion criteria. The inclusion criteria for this study involved selecting patient data with a minimum one-day hospital stay and CDK4/6 inhibitors chemotherapy initiation. The final analysis comprised 21 patients diagnosed undergoing CDK 4/6 chemotherapy. Drug interaction checkers such as Drugs.com®, IBM Micromedex® and UpToDate Lexicomp® were utilized to examine the medication regimen of each cancer patient on CDK4/6 inhibitors chemotherapy during their hospitalization, to determine the possibility of potential DDIs. To ascertain DDIs between concurrently administered medications and CDK4/6 inhibitors employed in breast cancer chemotherapy, the generic names of all drugs were entered into the drug interaction checker tools provided by IBM Micromedex®, Drugs.com®, and UpToDate Lexicomp®. All interactions tools were used till 31st August 2023. The electronic medical record system was used to gather data on each patient, including their age, sex, concurrent medication administration with palbociclib/ribociclib, and the number of comorbidities. The DDIs were categorized as either pharmacokinetic or pharmacodynamic based on their mechanism of action.
Statistical data analysis
Descriptive statistics were used to describe the demographic characteristic of patients, treatment, comorbidities, number of drugs prescribed per patient, and classification of drug interactions. Categorical and continuous data were analysed using descriptive statistics.
Results
Demographic information
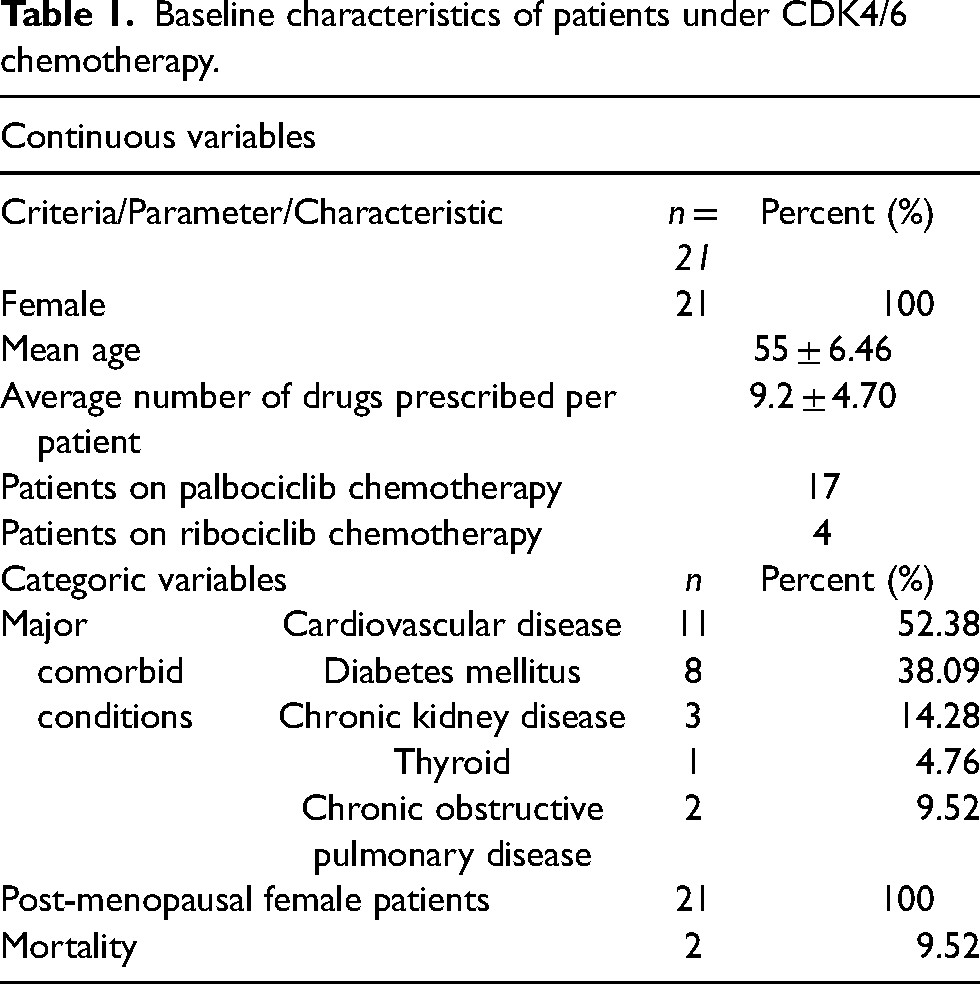
A comprehensive data analysis was conducted on twenty-one patients who were admitted for chemotherapy with CDK4/6 inhibitors (i.e., palbociclib and ribociclib) and met the specified inclusion criteria in order to identify pDDIs. The average age of the participants was 54.16 years, in range of 42 to 75 years. Out of the entire population, all female patients with breast cancer were identified in the postmenopausal stage. A large proportion of the patients had a hospital stay duration of more than five days. The patient characteristics are presented in Table 1. The most prevalent comorbidities observed in cancer patients were cardiovascular disease (52.38%), diabetes mellitus (38.09%), and chronic kidney disease (14.28%).
Baseline characteristics of patients under CDK4/6 chemotherapy.
Prevalence of potential DDIs
A comprehensive assessment of the most frequently prescribed medications that were found to have at least one DDI among the participants of the retrospective study is presented in Table 2. After conducting an analysis of the entire collection of co-prescribed drugs, it was ascertained that 7.93% of them belonged to the cardiovascular drug category, 12.38% to the antidiabetic drug category, 21.42% to the treatment of nausea/vomiting-related side effects, 4.76% to psychiatric therapies, 19.48% to analgesics, 6.19% to antimicrobials, and 24.60% to co-medications of hormonal therapy (Figure 1). A cumulative count of seventy-five co-medications have been given to be administered with palbociclib and ribociclib chemotherapy. Figure 2 illustrates the prevalence of certain pharmacokinetic and pharmacodynamic pDDIs as determined by the software Micromedex® and UpToDate®. The findings derived from Drugs.com® indicated that the predominant classification of DDIs was as follows: pharmacokinetic interactions were classified into two categories: major (12.11%) and moderate (77.77%), which are primarily mediated by inhibition of CYP450 enzymes or efflux transporters. Contraindication-related DDIs were not detected during the investigation conducted with Micromedex®. Significantly, out of the 22 most frequently prescribed medications that have been linked to potential DDIs, Drugs.com® identified two interactions and IBM Micromedex® identified eight interactions, all of which were categorized as major pharmacokinetic interactions. Drugs.com® has documented a cumulative count of 31 potential interactions, of which 12 are classified as major or moderate pharmacodynamic interactions. Of the total, 58.33% were categorized as moderate, while 41.66% were categorized as major (Table 3).
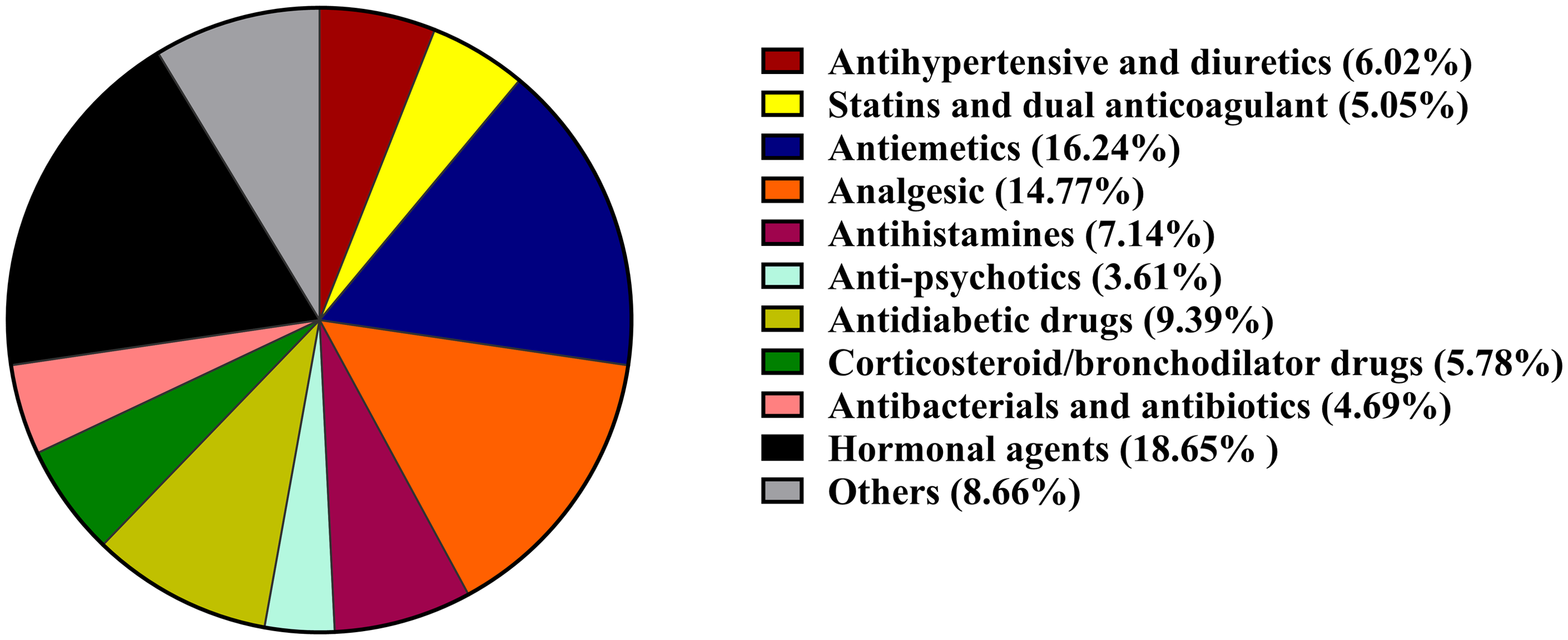
Pie chart representing the frequency of concomitant medications prescribed in CDK4/6 inhibitors chemotherapy of palbociclib and ribociclib associated with risk of DDIs. (*DDI -drug–drug interaction).
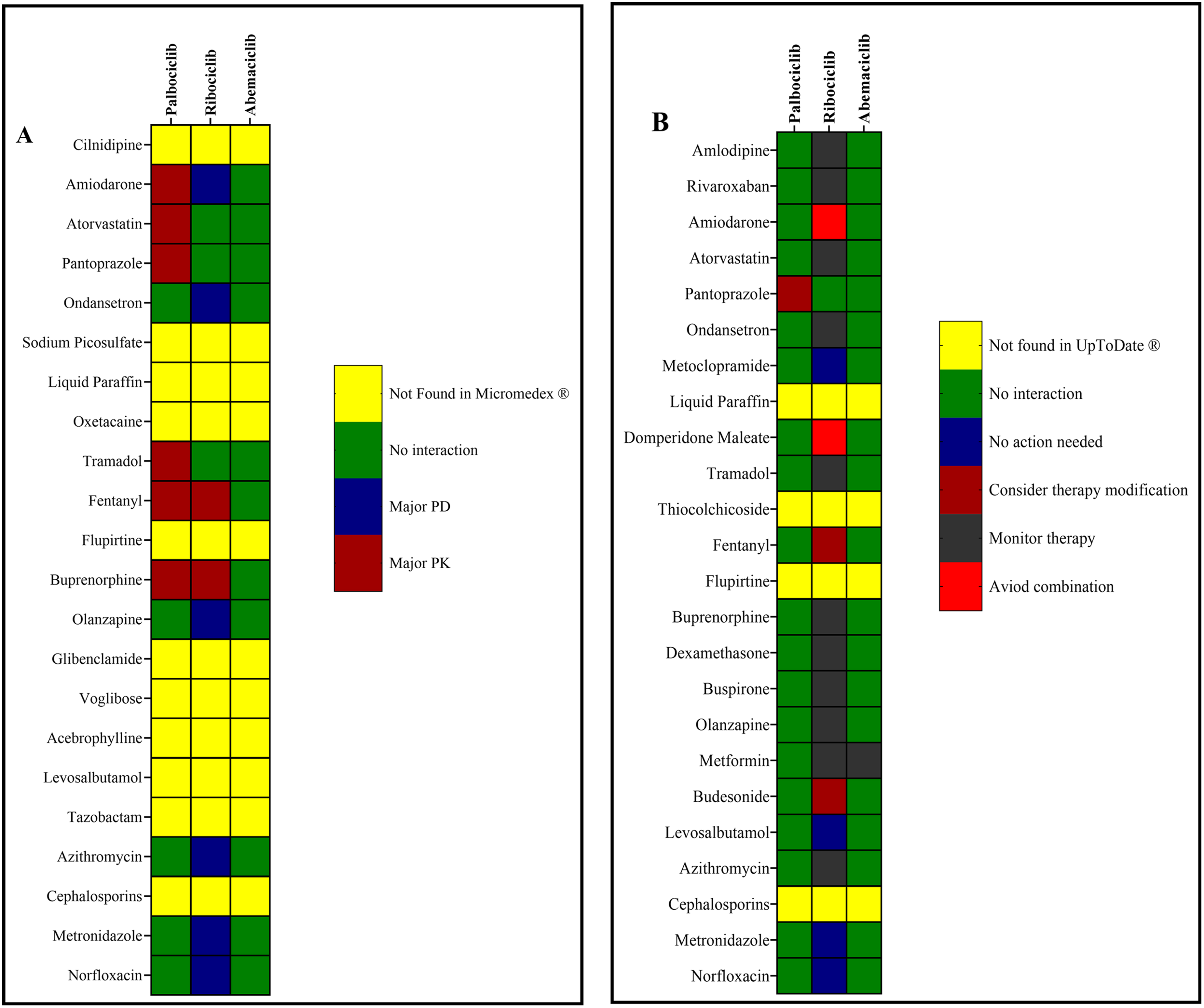
Potential DDIs (pDDIs) categorized based on risk rating. A) Severity of risk rating of pDDIs (minor, moderate, major, and contraindicated) by Micromedex®, B) pDDIs risk rating by UpToDate Lexicomp®.
Top identified prescribed medications with the potential of DDIs.
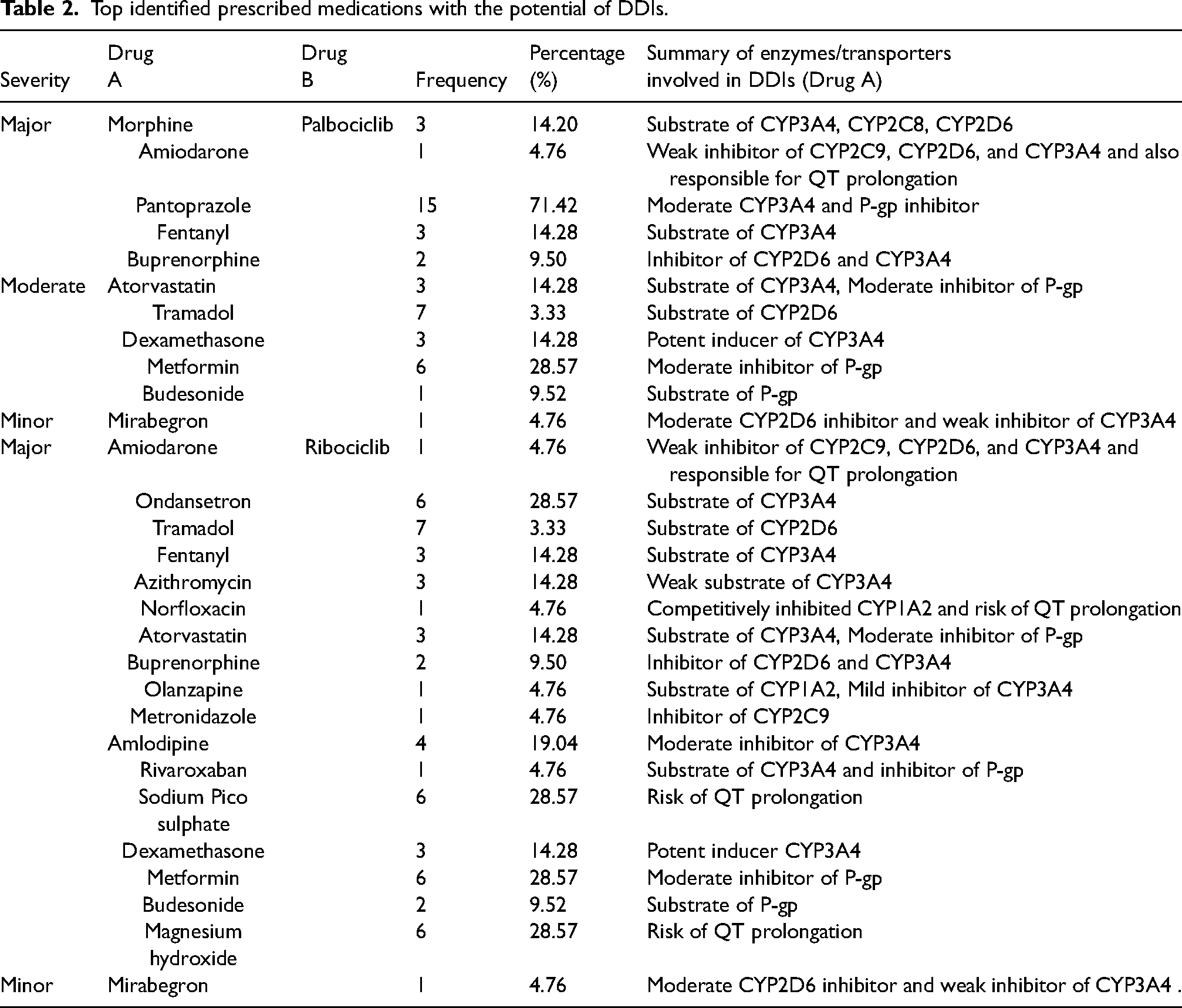
Potential DDIs identified using IBM Micromedex® and drugs.com®.
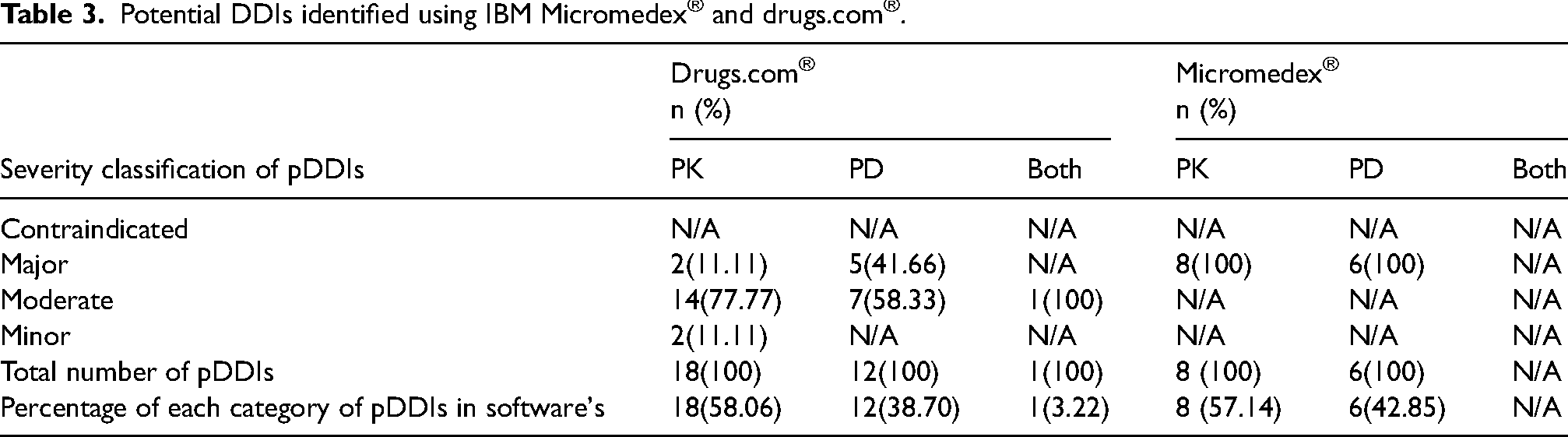
Micromedex® classified the severity categories of the identified interactions as major pharmacokinetic (57.14%) and major pharmacodynamic (42.85%). Eight main pharmacokinetic and pharmacodynamic DDIs between palbociclib/ribociclib and co-administered drugs in a cohort of twenty-one hospitalized patients were analyzed in detail in Figures 2A and 2B. Out of the patients who underwent chemotherapy with palbociclib and ribociclib, it was identified that 17 patients (representing 80.95% of the sample) had a high prevalence of potential DDIs. In addition, a total of fifteen patients (71.42% of the sample) demonstrated moderate pharmacokinetic interactions. Three patients out of the cohort were found to have encountered pharmacodynamic interactions however it is worth mentioning that 9.5% of these interactions were rated as major, whereas 4.7% were rated as moderate. Based on the analysis performed by UpToDate Lexicomp®, it was determined that 11.76% of the prescribed medications were contraindicated for co-administration with palbociclib/ribociclib, and 70.58% necessitated therapy monitoring. Furthermore, it is worth noting that 17.64% of the combinations under consideration possessed the capacity to alter the therapeutic approaches. It has been proposed that the concurrent use of amiodarone and domperidone with CDK4/6 inhibitors may be contraindicated, as illustrated in Figure 2B. In the presence of cardiovascular drugs, the incidence of potential DDIs was found to be greater, according to the study. However, there were no interactions observed with loop diuretics, ACE-II inhibitors, beta blockers and antihistamines denoting that their prescription would be safe with CDK inhibitor chemotherapy (Supplementary file 1 : Figure S1, Table S1).
Discussion
This retrospective study conducted at Kasturba Hospital in Manipal, Karnataka, India, discovered that a significant proportion of patients on CDK4/6 inhibitor therapy experienced DDIs. Each patient received a minimum of one co-medication, with 18 out of 21 patients receiving more than five prescription medications. A comprehensive examination of patients taking CDK4/6 inhibitors revealed 63 pharmacokinetic and pharmacodynamic interactions. Alongside 42.85% of the 21 patients in the cohort, 41.26% experienced significant pharmacokinetic interactions. Whereas,15.87% of the identified interactions were pharmacodynamic in nature, of which 14.8% were significant. The data from IBM Micromedex® revealed that interactions were most commonly caused by co-administration of analgesics, accounting for 35.71% of cases. Statins and antibiotics followed closely behind, contributing to 21.42% of interactions. Out of all the interactions that were recorded, a significant 57.14% were related to pharmacokinetics, while the remaining interactions were focused on pharmacodynamics. The IBM Micromedex® data indicated that pharmacokinetic interactions were mainly mediated by the inhibition of CYP450 enzymes and efflux transporters, including P-glycoprotein, by co-prescribed drugs such as pantoprazole, tramadol, and amiodarone. These interactions could lead to alterations in the plasma concentrations of CDK 4/6 inhibitors, thereby contributing to pharmacokinetic drug-drug interactions. Additionally, buprenorphine and fentanyl have also been documented to exhibit significant pharmacokinetic interactions, as per IBM Micromedex®.
Whereas, based on the latest UpToDate data, there are significant interactions between analgesics, statins, antiemetics, antipsychotics, and antibiotics. A small percentage of DDIs, 11.76%, had contraindications, indicating that the treatment was not appropriate for them. Therapy adjustments were required in 17.64% of cases, indicating optimal effectiveness. Among the three databases/software utilized for predicting drug interactions, Drugs.com identified a total of 31 drug-drug interactions (DDIs), while IBM Micromedex® predicted 14 interactions, as shown in Table 3. UpToDate identified 18 drug combinations with potential for drug-drug interactions. It was observed that IBM Micromedex® identified only those interactions classified as major pharmacokinetic/pharmacodynamic interactions, while Drugs.com also recognized interactions categorized as moderate and minor. In UpToDate, the majority of reported interactions were categorized as “monitor therapy.” The drug-interaction checkers collectively enhanced the understanding of potential drug combinations that may lead to drug-drug interactions, thereby assisting in the development of improved therapeutic regimens.
The study provided clear evidence of the drug interactions between CDK 4/6 inhibitors, palbociclib and ribociclib, with analgesics and statins. Particularly, aspirin (14.28%), fentanyl (14.28%), and tramadol (33.33%) that are known to be substrates of CYP450 enzymes and P-gp transporters. Additionally, pantoprazole (71.42%) has been identified as a moderate substrate of the CYP3A4 isoenzyme. A considerable proportion of DDIs associated with palbociclib were found to be major interactions, accounting for 45% and amongst the total interactions, 90%, were identified to be of pharmacokinetic. In the case of ribociclib, among the total interactions observed, 77.77% were classified as pharmacokinetic interactions, while the remaining 22.22% were categorized as pharmacodynamic interactions. The drug combination of atorvastatin and dexamethasone has been identified as the most frequently encountered DDI with clinical significance. Statins, including atorvastatin, lovastatin, and simvastatin, have demonstrated dual functionality as inhibitors and substrates of P-gp. Furthermore, elevating experimental data indicates that atorvastatin inhibits CYP450 enzymes and efflux transporters such as P-gp, multi-drug resistance-associated protein 2 (ABCC2), and breast cancer resistance protein (ABCG2).18–20 The DDIs that were commonly observed involved the combination of amiodarone and palbociclib, affecting the CYP3A4 enzyme, as well as atorvastatin and palbociclib/ribociclib, affecting the activity of P-gp transporter. Spironolactone ranks as the second most frequently prescribed medication in the present study. Multiple studies have substantiated the inhibitory effect of spironolactone on P-gp. 6 The concurrent administration of digoxin and spironolactone has resulted in an elevation of digoxin's plasma concentration. 21 It is important to acknowledge that a majority of these drugs are concurrently implicated in both CYP3A4 and P-gp pathways, thereby further increasing the unpredictability of the final outcome. Palbociclib and ribociclib, have demonstrated distinct cardiotoxic effects of uncertain etiology. Ribociclib, in particular, is also linked to the prolonging of the QT interval. 22 Regarding this matter, it has been observed that among the co-prescribed drugs, antibiotics, antimycotics, and antibacterials exhibit the greatest potential for pharmacodynamic interactions through the mechanism of QT prolongation.
The limitation of this study was that it depicted challenges with respect to less sample size to conclude. Despite this shortcoming, the findings of this the current investigation successfully ascertained the frequency, array, and variables associated with probable DDIs facilitated by P-gp and CYP3A4 in the context of CDK4/6 inhibitors chemotherapy. The occurrence of drug interactions can potentially affect quality of life of the patient, and the failure to detect these interactions in a clinical setting can result in a higher incidence of morbidity. Hence, it is advisable to conduct a thorough assessment of potential drug interactions when administering medications for commonly occurring comorbidities, such as diabetes and hypertension.
Conclusion
The current investigation successfully determined the frequency, and variables associated with probable DDIs mediated by P-gp and CYP3A4 in the context of CDK4/6 inhibitors chemotherapy. Further investigation is necessary to comprehensively examine the impact of DDIs on adverse drug events and clinical outcomes, such as the comparison of hospitalization rates. In addition, therapeutic drug monitoring is a potential approach for examining actual clinical significance of DDIs within the field of oncology. This area of investigation will hold significant importance in future studies.
Supplemental Material
sj-docx-1-opp-10.1177_10781552251314811 - Supplemental material for A single-centre retrospective evaluation of potential drug-drug interactions in breast cancer patients undergoing CDK 4/6 inhibitors chemotherapy
Supplemental material, sj-docx-1-opp-10.1177_10781552251314811 for A single-centre retrospective evaluation of potential drug-drug interactions in breast cancer patients undergoing CDK 4/6 inhibitors chemotherapy by Prajakta Harish Patil, Mrunal Pradeep Desai, Gayathri Baburaj, Mahadev Rao, Vijayanarayana Kunhikatta, Karthik Udupa, Ananth Pai and P. C. Jagadish in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
The authors wish to express their gratitude to Manipal College of Pharmaceutical Sciences, Manipal Academy of Higher Education and Kasturba Hospital Manipal for providing the requisite resources and facilities for conducting this study. Prajakta Patil is thankful to ICMR, New Delhi, India for providing Junior research fellowship. Mrunal Desai would like to acknowledge Lady Tata Memorial Trust, Mumbai, India for providing Junior research fellowship. Gayathri Baburaj would like to acknowledge DST-INSPIRE Fellowship, Department of Science and Technology, New Delhi, India.
Author contribution
J.P.C, M.R. and P.P. conceptualized this study. M.R., K.U., and A.P. allowed provision of study material or patients, P.P., and M.D. acquired collection and/or assembly of clinical data. P.P., M.D., G.B. and V.K. were responsible for the data analysis and interpretation of the data. J.P.C, M.R. and V.K., were involved in data curation. P.P. and M.D. drafted the manuscript. J.P.C., M.R., V.K., K.U., and A.P. final approval of the manuscript. All authors have read and approved the current version of the manuscript.
Availability of data and material
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Consent for publication and participation
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Study approval was obtained from the Institutional Ethics Committee of Kasturba Medical College and Kasturba Hospital, Manipal, Institutional Ethics Committee Review Boards before data acquisition (KMC and KH IEC Reference No-IEC1:99/2023). This retrospective study involving breast cancer patients was conducted in accordance with the ethical standards of Institutional Ethics Committee of Kasturba Medical College and Kasturba Hospital, Manipal. No animals were used in this research.
Funding
The project is funded by the Indian Council of Medical Research, Government of India (grant number 2020–4462) and an Intramural fund from Manipal Academy of Higher Education, Manipal.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
