Abstract
Introduction
Medication nonadherence is a prevalent issue in the oncology population and can be costly on the healthcare system while leading to worsened outcomes, including increased mortality. One intervention that has been shown to help promote medication adherence to oral therapies for chronic conditions is blister-packaging. However, no literature is available assessing the potential impact of blister-packaging in oncology. Therefore, we aimed to quantify the impact of blister-packaging oral oncolytics on medication adherence and healthcare costs.
Methods
An economic model was designed to assess the potential impact of blister-packaging oral oncolytics for chronic lymphocytic leukemia (CLL) patients in the U.S. The model took the perspective of the U.S. healthcare system and assessed newly diagnosed CLL cases initiating therapy, utilizing a one-year time horizon. The intervention of interest was blister-packaging oral therapies in the treatment of CLL, while the model evaluated the impact of this intervention on healthcare costs through improved patient adherence based on peer-reviewed literature.
Results
This analysis consisted of 20,700 newly diagnosed patients with CLL initiating therapy. Initiating blister-packed oral therapies saw adherence increase by 1004 patients from 12,979 patients (62.7%) to 13,983 patients (67.5%). While an increase in pharmacy costs by $61,589,670 (+$247.95 per-patient per-month (PPPM)) was seen for the population, medical costs decreased by $82,285,847 (-$331.26 PPPM). Overall, this led to a net reduction in total healthcare costs of $20,696,177 (-$83.32 PPPM).
Conclusions
Blister-packaging oral oncology drugs in CLL has the potential to reduce total healthcare expenditures by increasing medication adherence. Future studies are needed to evaluate the direct effects of blister-packaging oral oncolytics on clinical, healthcare resource utilization, and financial outcomes.
Keywords
Introduction
Medication nonadherence is a prevalent issue in the United States,1,2 leading to increased healthcare costs. 3 Nonadherence is particularly common in the oncology population, as many struggle to take their medications as prescribed and their adherence declines over time. 4 Additionally, due to the severe nature of different cancers and the high costs associated with their treatment, nonadherence to oncology treatments can both be costly on the healthcare system, and also lead to worsened outcomes, including increased mortality.5–7
Oral oncolytic utilization has been increasing in recent years.8–10 While oral agents in oncology may be more convenient,11,12 patients are required to be more responsible for their treatment and utilization. Instead of going to an infusion center with a scheduled appointment weekly or monthly to receive their therapy infusion, oral therapy requires patients to remember to take their medication regimen more frequently, sometimes multiple times a day. This has resulted in patients with oral oncolytics having lower medication adherence than those receiving infused therapies.13,14 Research has shown that oral therapy requiring daily dosing has lower odds of patients becoming adherent compared to dosing less frequently than once daily. 15
To enhance patient adherence to oral medications, various interventions have been identified.16,17 The American Society of Clinical Oncology (ASCO)/ National Community Oncology Dispensing Association (NCODA) Patient-Centered Standards for Medically Integrated Dispensing also place a high importance on recommending tools and policies to help promote patient adherence to oral oncolytics. 18 Some of these recommendations include calendars or scheduling communications to patients, as well as pill caddies. 18 One specific intervention that has consistently been shown to help promote medication adherence in several studies encompassing a variety of chronic disease states such as hypertension, cardiovascular disease, diabetes, and a composite of multiple chronic conditions is medication adherence packaging, specifically blister-packaging or calendar-packaging medications.16,17,19 Blister-packaging significantly increased the likelihood of adherence to angiotensin-converting enzyme inhibitors (ACEi) for hypertension (adjusted odds ratio (aOR: 1.13, 95% confidence interval (CI): 1.05–1.21). 19
Chronic lymphocytic leukemia (CLL) is a common blood cancer, having the highest prevalence of all leukemias in the United States with a prevalence of 197,060 in 2019 and the second highest prevalence of all blood cancers behind only non-Hodgkin lymphoma (NHL) (prevalence of 722,631). 20 There is an estimated incidence of 20,700 cases of CLL in the U.S. annually, with an estimated 4400 deaths. 21 CLL has a relatively high 5-year survival rate across all stages of 88%. 22 Given that CLL is a slow growing malignancy 20 with a long duration and newer available targeted treatments, it has started to be managed like a chronic disease. 23 The National Comprehensive Cancer Network (NCCN) Treatment Guidelines for CLL/Small Lymphocytic Lymphoma (SLL) recommend oral therapy options alone or in combination with infused or injectable therapy for every preferred and other recommended regimen in the first-line therapy setting, as well as for second or third-line therapy. 24 Oral agents are also recommended in the relapsed/refractory (R/R) setting. 24 Like most oncology settings, the rates of patients nonadherent to oral oncolytics in CLL are relatively high, ranging from 37.3% to 58.6% in the literature depending on the specific oncolytics utilized and disease stage.25–27
While studies in the literature indicate that blister-packaging medications can improve adherence, and increased adherence is linked to improved health outcomes and reduced healthcare costs, there are no studies available tying these together. Thus, this study aims to model the potential impact of blister-packaging oral CLL medications on healthcare costs in the CLL population.
Methods
Study design
An economic model was designed to assess the impact of blister-packaging oral oncolytics for CLL patients in the United States. The model took the perspective of the United States healthcare system and assessed newly diagnosed CLL cases initiating therapy, utilizing a one-year time horizon (Table 1). This time horizon was selected because the source data on the impact of adherence on healthcare costs used a one-year follow-up in their study. 25 The intervention of interest was blister-packaging oral therapies in the treatment of CLL, while the model evaluated the impact of this intervention on healthcare costs through improved patient adherence. The definition of adherence in the model was a proportion of days covered (PDC) of 80% or higher, which is consistent with validated definitions of adherence in the oncology space in the published literature.15,25,28,29 The model utilized data of the impact of adherence in CLL patients on healthcare costs from a retrospective analysis of Marketscan Commercial Claims and Medicare Supplemental databases by Dashputre et al., which also used an adherence definition of 80% or higher PDC. 25 Additionally, the source study for the impact of blister-packaging on medication adherence was a retrospective analysis of Walmart pharmacies nationwide assessing the impact of blister-packaging medications on adherence and the likelihood of patients becoming adherent based upon different medication adherence definitions. 19
Economic model input parameters.
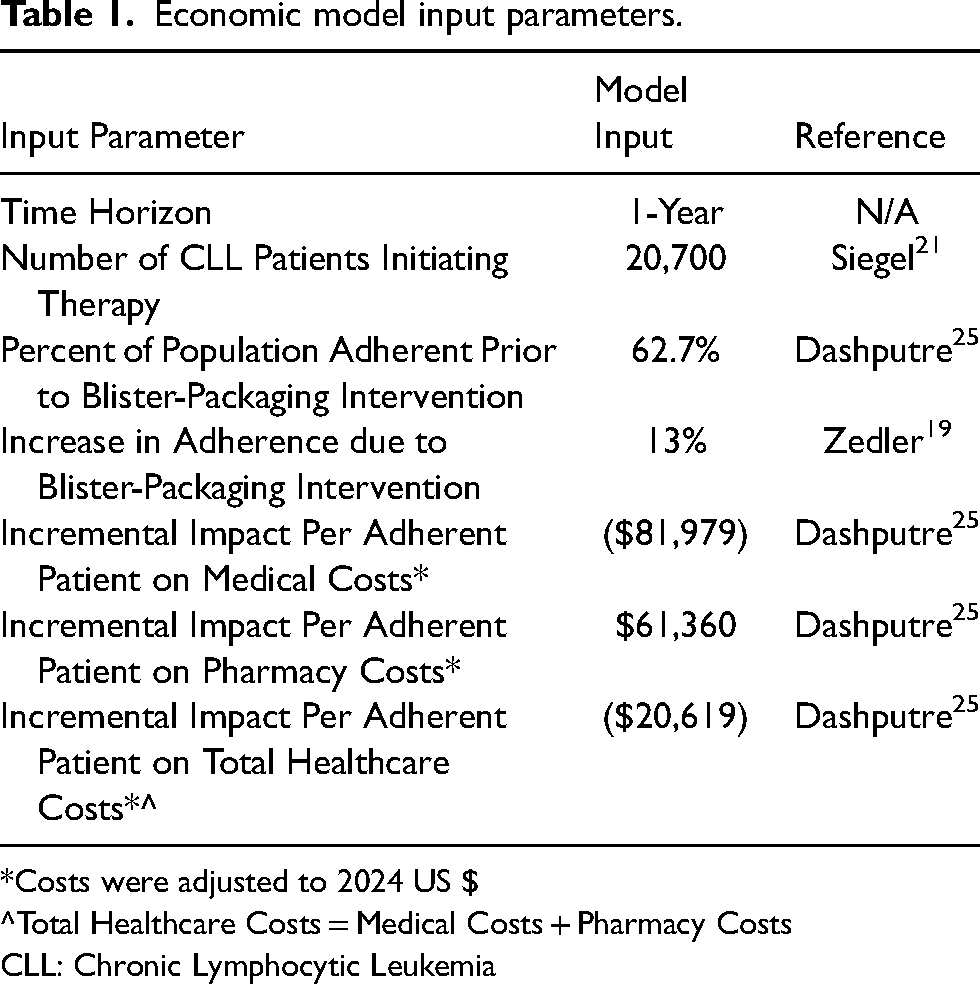
*Costs were adjusted to 2024 US $
^Total Healthcare Costs = Medical Costs + Pharmacy Costs
CLL: Chronic Lymphocytic Leukemia
Population
In this analysis, the population of interest were comprised of incident CLL patients who were initiating CLL therapy. Since this model was conducted from a U.S. healthcare perspective, the total number of incident CLL patients (20,700) in the U.S. was used in the analysis. 21 While not every patient diagnosed with CLL will initiate treatment in a given year, as many patients start in a “watch and wait” phase depending on the severity of their condition at diagnosis, 24 we used incident CLL patients as a proxy to the number of patients initiating therapy in given year. This is because while some of the incident patients will go into a “watch and wait” phase, others who were diagnosed in previous years may transition from “watch and wait” to initiating therapy. The source study by Dashputre et al. examined two specific oral agents in CLL: ibrutinib and idelalisib. 25 Current guideline recommendations for first-line treatment of CLL include oral agents as monotherapy or in combination with other infused treatments for all preferred or other recommended regimens (acalabrutinib, venetoclax, zanubrutinib, or ibrutinib). 24 While ibrutinib is still listed as a potential first-line therapy in CLL, idelalisib is now recommended only in the R/R stage. 24 This model assumes that the impact on healthcare costs will hold for any oral first-line therapy. Considering network meta-analyses (NMAs) have shown that the newer oral treatments (acalabrutinib, venetoclax, and zanubrutinib) have more favorable efficacy results in clinical trials than ibrutinib or idelalisib,30,31 it is reasonable for this is a conservative estimate. The base case of the model assumes that 62.7% of patients were adherent in the pre-period, based on the findings from Dashputre et al. 25 The Dashputre et al. study also evaluated patients on oral therapies to initially treat multiple myeloma. However, due to the agents assessed and newer first-line treatment regimens and combinations becoming available post study period, 32 along with the low baseline adherence rate found in the study (28.9%), 25 the decision was made to focus the model solely on the CLL population.
Intervention
The focus of the intervention was blister-packaging oral agents for patients with CLL. A retrospective analysis of prescription dispensing data from a national pharmacy chain, with data spanning from December 2006 through July 2009, investigated whether patients receiving blister-packed ACEi were more likely to become adherent. 19 The findings from a propensity-scored matched analysis revealed that patients initiating therapy with blister-packaged medication had 13% higher odds of becoming adherent with a PDC of at least 80% (adjusted odds ratio (aOR): 1.13, 95% CI: 1.05–1.21). 19 In the base-case analysis, it was assumed the blister-packaging intervention would lead to a 13% increase in patients becoming adherent. Even though this study did not assess the impact of blister-packaging oral oncolytics, it is the best available literature showing the impact that blister-packaging can have on medication adherence along with no studies being identified in the literature assessing the impact of blister-packaging oral oncolytics.
Outcomes
The analysis examined the overall impact of blister-packaging on total healthcare costs, including medical costs and pharmacy costs. Aggregate costs were also stratified based upon age (<65 years old, ≥ 65 years old), and payer type (commercial insurance, Medicare) based upon the proportion of patients in the Dashputre et al. study who corresponded to each category, 25 as well as geographic region (Northeast, Midwest, South, West) based upon incidence of leukemia in 2024 by state. 21 An additional outcome of interest was the per-patient per-month (PPPM) impact on healthcare costs. PPPM was calculated by dividing the aggregate cost by the total number of patients in the analysis (n = 20,700) and then dividing that by 12 months. Dashputre et al. calculated the reduction in healthcare resource utilization (HCRU) (outpatient use and inpatient/emergency department use) via adjusted incidence rate ratios (aIRR) for adherent patients versus nonadherent. 25 However, they did not provide baseline utilization for either group, which made it challenging to determine what the overall reduction would be for this population and thus was not included in this model.
Costs
The input for the impact on healthcare costs came from the Dashputre et al. analysis, which was applied to the yearly number of newly adherent patients’ post-initiation of a CLL medication in a blister-pack. 25 After adjusting costs to 2024 U.S. dollars using U.S. medical consumer price index (CPI), 33 this resulted in an annual incremental reduction in medical costs of $81,979, an increase in pharmacy costs of $61,360, and a total healthcare cost reduction of $20,619 per adherent patient. Medical costs consisted of inpatient hospitalizations, emergency department visits, and outpatient visits. 25
Sensitivity analyses
One-way sensitivity analyses were conducted for every model input parameter. The upper and lower bounds for the impact of blister-packaging on medication adherence were taken from the upper and lower bounds of the 95% confidence interval (5% to 21%) of the base case input of 13%. 19 Due to inadequate data available in the peer-reviewed literature to inform the upper and lower bounds of the remaining inputs, the remaining parameter inputs were varied by +/- 20%.
Results
This analysis consisted of 20,700 newly diagnosed patients with CLL initiating CLL therapy. Before implementing blister-packaged oral therapies, 12,979 patients (62.7%) were adherent to their CLL regimen (Figure 1). After implementation of blister-packaging oral therapies, adherence rose to 13,983 patients (67.5%), an increase of 1004 patients becoming adherent, or 4.8 percentage points more than prior.
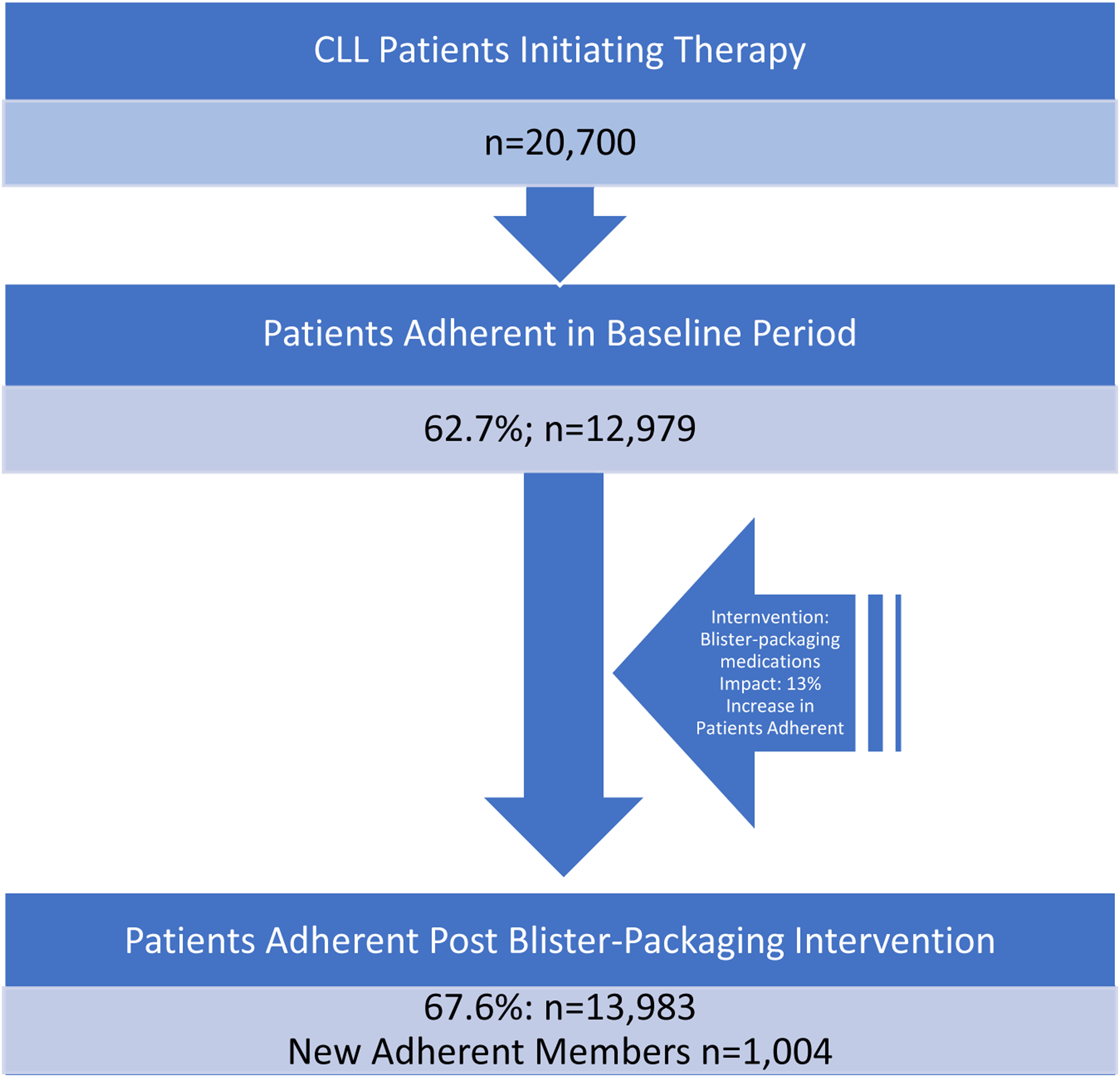
Flowchart of patients becoming adherent with blister-packaging medications.
With the increase in the number of patients becoming adherent, this analysis found that introducing blister-packaged oral oncolytics would lead to a reduction in total healthcare costs of $20,696,177 for CLL patients during the first year (Table 2). While pharmacy costs increased by $61,589,670 for the population, medical costs decreased by $82,285,847. These results equate to a PPPM pharmacy cost increase of $247.95, a PPPM medical cost decrease of $331.26, and a PPPM total healthcare cost decrease of $83.32 (Figure 2).
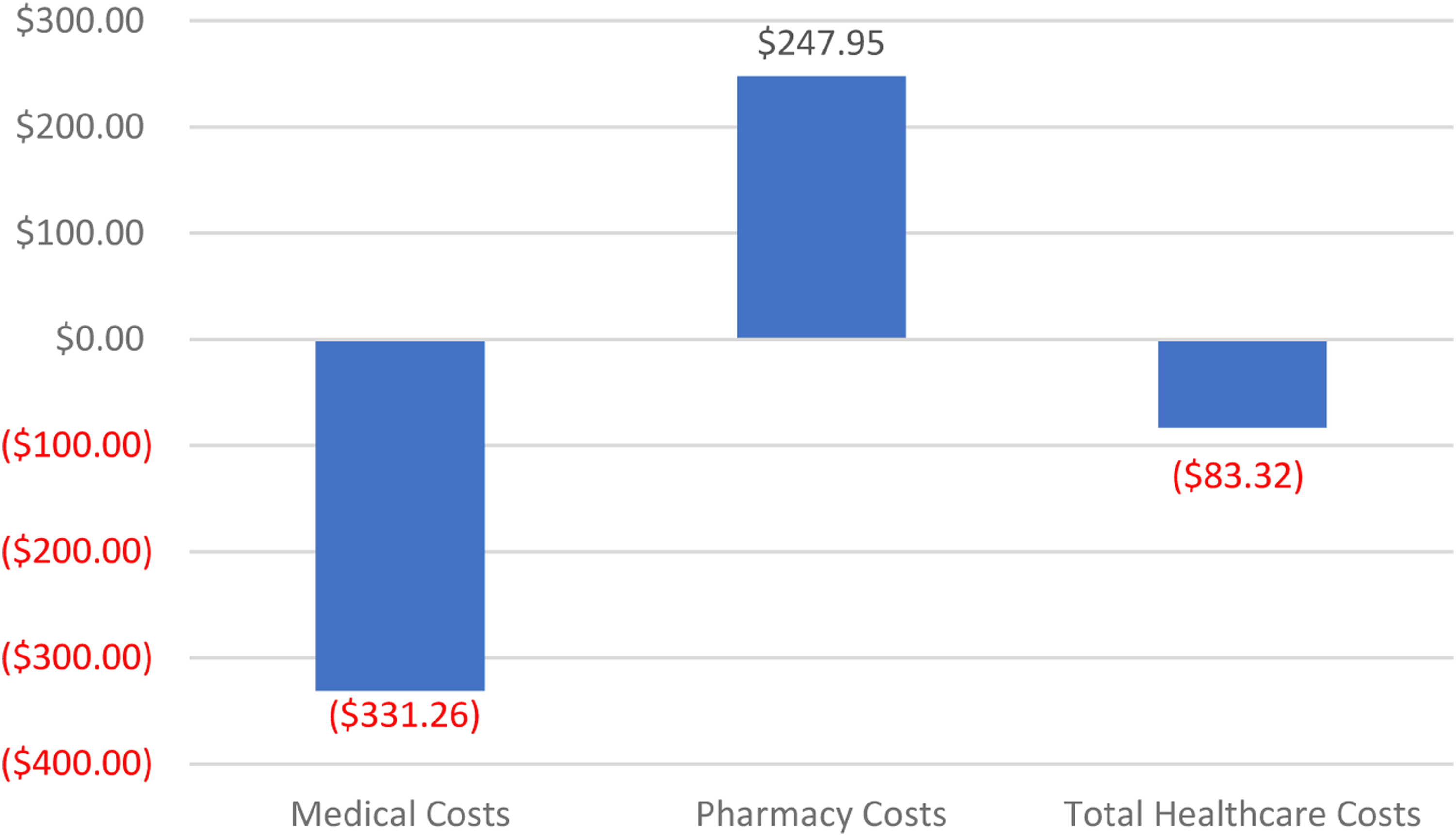
Incremental impact of increased medication adherence on per-patient per-month healthcare costs.
Impact of increased adherence on healthcare costs.
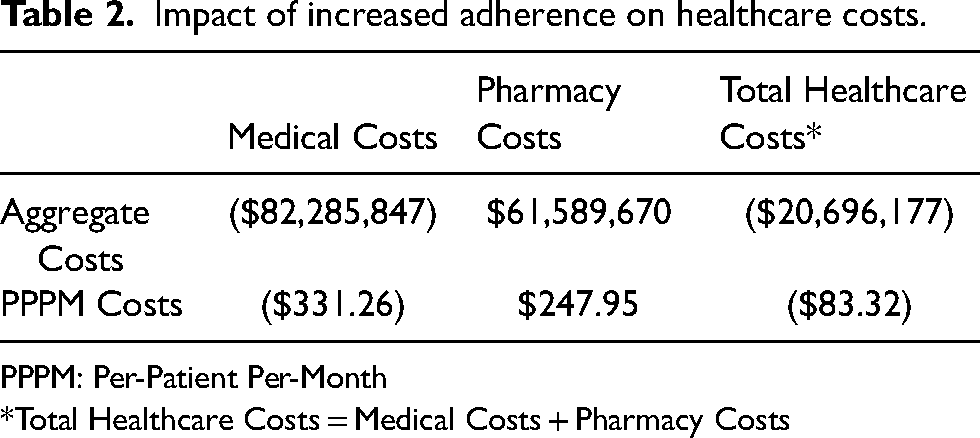
PPPM: Per-Patient Per-Month
*Total Healthcare Costs = Medical Costs + Pharmacy Costs
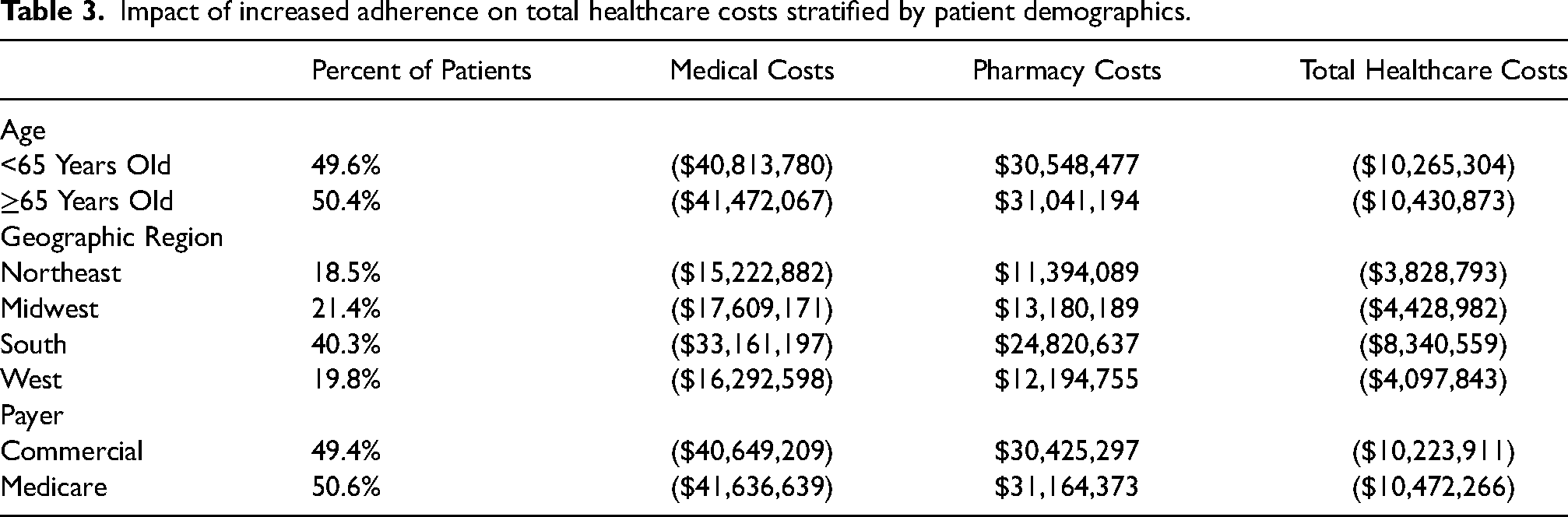
Total healthcare cost savings were almost evenly split between patients less than 65 years of age (-$10,265,304) and those 65 years or older (-$10,430,873) as well as those with commercial insurance (-$10,223,911) and those with Medicare (-$10,472,266) (Table 3). Due to differences in incidence and population in different states, the South saw the largest decrease in total healthcare costs (-$8,340,559) followed by the Midwest (-$4,428,982), the West (-$4,097,843), and the Northeast (-$3,828,793).
Impact of increased adherence on total healthcare costs stratified by patient demographics.
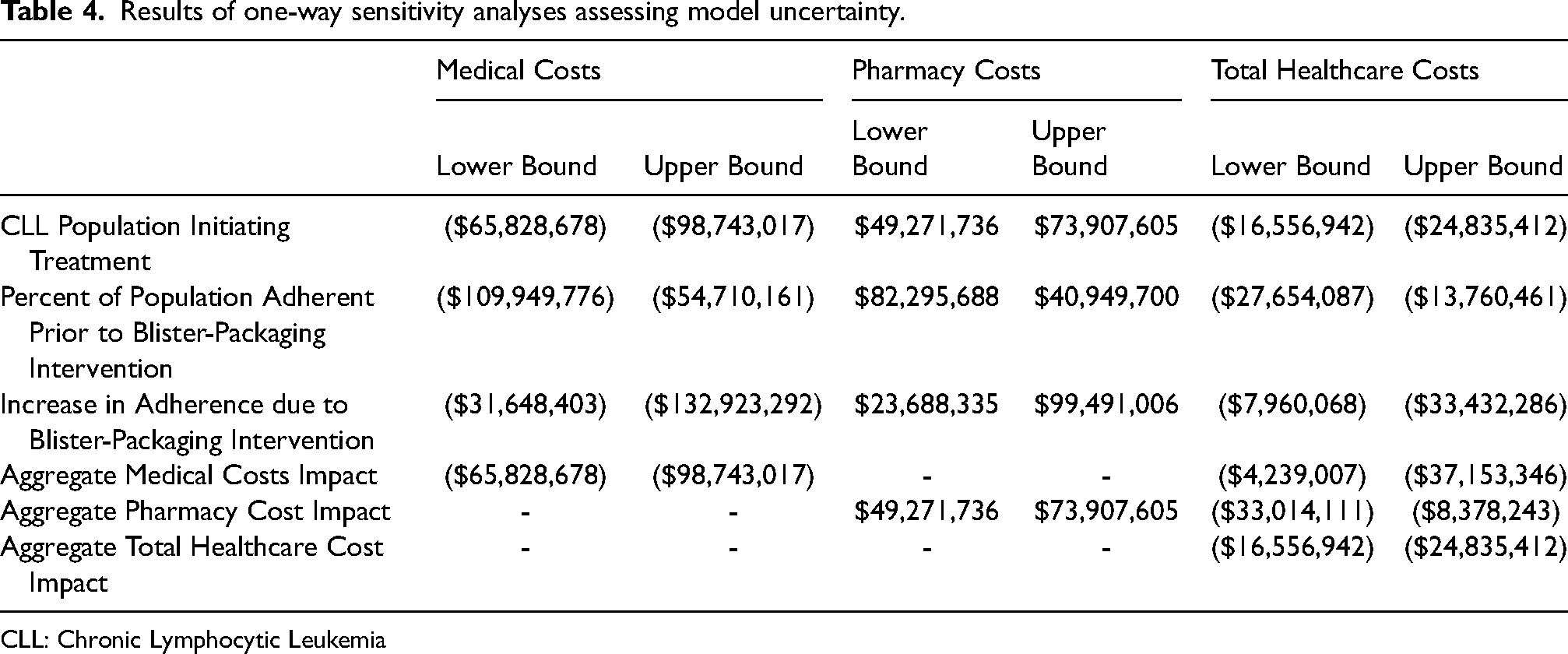
The results of the sensitivity analysis on total healthcare costs showed that the model was most sensitive to changes in the incremental impact per adherent patient on medical costs (-$4,239,007 to -$37,153,346), followed by the expected increase in adherence due to blister-packaging (-$7,970,068 to -$33,153,346), and lastly, the incremental impact on pharmacy costs (-$8,378,243 to -$33,014,111) (Table 4).
Results of one-way sensitivity analyses assessing model uncertainty.
CLL: Chronic Lymphocytic Leukemia
Discussion
This economic analysis modeled the potential impact of increased adherence from blister-packaging oral oncolytics on reduction of healthcare costs. The findings suggest that while pharmacy costs increased due to more patients becoming adherent and therefore receiving more prescriptions, overall medical costs saw a dramatic decrease due to better adherence. Specifically, a net healthcare cost savings of approximately $20.7 million was observed for patients initiating therapy for CLL.
While this case study indicated the potential impact of blister-packaging oral oncolytics for CLL specifically, this benefit could extend to other oral cancer treatments. Oral oncology drugs are listed in treatment guidelines as first line therapy in many cancer types including renal cell carcinoma (RCC), prostate cancer, colorectal cancer, non-small cell lung cancer (NSCLC), breast cancer, multiple myeloma, ovarian cancer, and chronic myeloid leukemia (CML), among many others.34–40 Blister-packaging is expected to improve adherence in these patients, thereby enhancing health outcomes and reducing healthcare costs. However, due to variations in treatment effectiveness and disease severity, it remains challenging to determine how these results from CLL would translate to other cancer types. Strategies to promote patient adherence to oral therapies, such as calendars and pill caddies, are supported by the ASCO/NCODA Patient-Centered Standards for Medically Integrated Dispensing. 18 BC Cancer, which is an governmental agency in British Columbia, Canada, also recommends blister-packaging oral oncolytics as a way to increase medication compliance and promote safe administration. 41 Due to the increased utilization of value-based contracting (VBC) in oncology,42–44 payers, pharmacies, and manufacturers have an increased incentive to promote patient adherence to oncology therapies to increase the likelihood of treatment success. Additionally, due to different accreditations available for pharmacies dispensing oncology medications,45–47 pharmacies may start to consider blister-packaging oral oncolytics as a way to help promote medication adherence. Two of the leading accreditation organizations in this space are URAC which provides accreditations for specialty pharmacies, 47 as well as NCODA which provides accreditations for medically integrated dispensing pharmacy centers of excellence. 46 As part of these accreditations, pharmacies are determined to be centers of excellence that provide quality care with initiatives to improve medication adherence and health outcomes.
The source study by Dashputre et al. found that patients adherent to CLL therapy had a reduction in HCRU with an aIRR of 0.80 (95% CI: 0.75–0.86) for outpatient use and an aIRR of 0.73 (95% CI: 0.62–0.87) for inpatient use. 25 This research also showed that adherence to multiple myeloma therapy led to a reduction in outpatient and inpatient use, with aIRRs of 0.78 (95% CI: 0.74–0.81) and 0.64 (95% CI: 0.57–0.72), respectively. 25 Adherent patients with multiple myeloma saw considerable reductions in medical costs per patient of $109,779, despite an increase in pharmacy costs of $71,673, resulting in a total healthcare cost reduction of $38,106. 25 Similar outcomes were reported in other studies, including a retrospective analysis of the Surveillance, Epidemiology, and End Results Program (SEER) Medicare database from 2007–2014 showing that hormone therapy adherence in women 65 years and older with hormone-receptor positive (HR+) breast cancer had a significant reduction in number of hospitalizations, hospitalization days, and number of outpatient office visits. 48 A retrospective analysis of Marketscan Commercial Database from January 2002 through July 2008 assessed patients taking imatinib for CML and the association of adherence to HCRU and healthcare costs using an adherence definition of greater than or equal to 85% medication possession ratio (MPR). 49 They found that adherence to imatinib had a significant reduction in mean inpatient visits (0.4 vs 4.1, p < 0.001), mean outpatient visits (30.2 vs 41.7, p < 0.001), and mean emergency room (ER) visits (0.3 vs 0.5, p < 0.001), along with significant reductions in unadjusted costs for inpatient utilization ($40,740 in 2005 US $), outpatient utilization ($22,549 in 2005 U.S. $), ER costs ($182 in 2005 U.S. $), and an increase in unadjusted costs for medications ($14,409 in 2005 U.S. $). 49 Another retrospective analysis of Marketscan Commercial Database from January 2011 through December 2015 of commercially insured patients with CML was conducted to assess the impact of adherence to tyrosine kinase inhibitors (TKIs) on HCRU and costs. 28 It was found that patients adherent to TKIs had lower aIRR for all-cause hospitalizations (aIRR: 0.32, p < 0.001) and CML-specific hospitalizations (aIRR: 0.31, p < 0.01). 28 Adherent patients also had significantly lower adjusted medical costs ($10,973 per patient) but significantly higher pharmacy costs ($26,521 per patient). 28 A retrospective cohort study using Optum's Clinformatics® Data Mart database from 2010 to 2018 showed varying results across cancer types. 29 While most results did not meet statistical significance, they found that adherence to oral cancer therapies had reductions in inpatient hospitalizations and emergency department visits for colorectal cancer and stomach cancer, while other cancers saw increases in HCRU such as liver cancer and lung cancer. 29 These mixed outcomes could be due to the study assessing HCRU for only 6 months post treatment initiation as well as only requiring one prescription dispensing compared to the other studies which included a longer follow-up and multiple prescription dispenses.
One important difference between what was seen in the source study by Dashputre et al. and the current treatment landscape in CLL is that idelalisib is no longer recommended as first-line therapy and only recommended as a potential option in R/R disease. 24 While ibrutinib is still a potential first-line therapy option, it is not listed as a “preferred regimen” but only as an “other recommended regimen”. 24 However, all therapy options first-line for both “preferred regimen” and “other recommended regimen” include an oral oncolytic either as monotherapy or in combination with a parental therapy. 24 The analysis that included ibrutinib as the only current first-line treatment may not fully reflect the effectiveness of newer agents such as acalabrutinib, venetoclax, and zanubrutinib, which were not evaluated.30,31 Consequently, it is plausible to note that utilizing ibrutinib and idelalisib data assessing first-line therapy may underestimate the true current impact across the class. Nonetheless, some of the first-line therapy regimens include providing a parental therapy (obinutuzumab or rituximab) in conjunction with the oral oncolytic. 24 While blister-packaging may impact and promote medication adherence for the oral therapy, it does not affect parental therapy, making it challenging to determine the full impact on patient outcomes and healthcare costs.
Beyond the therapy options available since the time of the source study by Dashputre et al., ibrutinib (the remaining therapy from the study that is still a potential first-line option) has seen packaging changes. Previously available in medication stock bottles during the study period, 50 it is now provided in manufacturer blister-packaging, eliminating the need for additional blister-packaging at the pharmacy. 51 With the manufacturer changing their packaging of the medication to be blister-packed, this may add further weight to the importance of blister-packaging oral oncolytics to help promote patient adherence. A search of the package inserts of all oral oncolytics on the market through 2023 shows that approximately 20% of them are available in manufacturer blister-packs. A second change to ibrutinib is that during the study period, ibrutinib was only available in 140 mg tablets and patients would have to take 3 tablets at a time to get to the required dose in CLL (420 mg). 50 However, since the study period, ibrutinib now has 420 mg tablets and patients only have to take one tablet at a time. 51 This simplification is likely to further improve adherence by reducing the complexity of the medication regimen.
Another important note from this analysis was that it modeled the number of incident CLL cases per year as the number of patients initiating therapy. This may not be fully accurate, as in CLL some patients may wait in a “watch and wait” phase after diagnosis before initiating treatment and this is a limitation of the model. 24 Without knowing the true number of patients starting initial therapy for CLL in a given year, we decided to use incident patients as a numerical average, in order to capture patients who are diagnosed and started treatment right away, as well as those who were previously in the “watch and wait” phase and then decided to initiate treatment. Due to the advancement in treatment and medication regimens in CLL over the past decade, the time to initiate treatment has been decreasing. 52 A retrospective claims analysis of Optum Clinformatics DataMart from 2013 through 2021 of patients diagnosed with CLL found that for patients first diagnosed with CLL during 2018–2021 had a shorter median time to treatment initiation (0.48 years) compared to patients first diagnosed during 2014–2017 (1.2 years). 52 However, more than half of patients in the analysis who were diagnosed with CLL did not initiate treatment during the follow-up period. 52 While some patients are being treated earlier in CLL due to the availability of targeted therapy, “watch and wait” is still recommended in treatment guidelines as a potential strategy for asymptomatic patients. 24
Out-of-pocket (OOP) costs are also a major factor in medication non-adherence for oral oncology treatments.15,53 To compound this further, oral oncolytic medication costs have been increasing year-over-year for the past decade,8–10,54 and have been among the medications with the highest annual price increase in the U.S. over the past 5 years.55–59 In 2022, ibrutinib, venetoclax, and acalabrutinib were all among the 100 medications with the largest revenue, with wholesale acquisition cost (WAC) price increases from 2.6% to 7.3%. 59 These costs can be substantial for patients with commercial insurance with high-deductible plans, as well as patients with Medicare coverage due to Medicare's Part D cost sharing structure which can see OOP cost exceed $10,000 per patient for oral oncolytics. 60 However, starting in 2025 OOP costs will be capped at $2000 per Medicare Part D beneficiary due to the Inflation Reduction Act, 61 which may help reduce the occurrence of cost-related nonadherence for Medicare beneficiaries. Additionally, the ASCO/NCODA Patient-Centered Standards for Medically Integrated Dispensing provides recommendations and resources to help assist patients in finding financial support. 18
One consideration in blister-packaging oral oncolytics is the additional precautions needed to be undertaken if said oral oncolytic is a hazardous drug. While none of the first-line recommended oral agents in CLL are hazardous drugs, several of the older oral agents that treat CLL as well as oral oncolytics that treat other malignancies are. 62 When handling and dispensing hazardous drugs, special precautions and care is needed to be taken by pharmacy personnel to prevent exposure to the worker, including wearing additional personal protective equipment.63,64 Additionally, if packaging machines are being used to fill medication vials or blister-packs, they must have required certification to handle hazardous drugs. Medically integrated dispensing pharmacies, specialty pharmacies, and other pharmacies that are considering blister-packaging oral oncolytics will have to take this into account when deciding whether to blister-pack oral oncolytics that are hazardous drugs and whether they have the equipment, processes, and/or machinery in place to do so. However, agencies such as BC Cancer recommend blister packaging oral oncolytics as a way to help promote safe administration for patients. 39
Although this analysis showed that blister-packaging may improve medication adherence rates, patient outcomes, and reduce costs, these findings need to be validated in future studies. Currently, no studies have directly assessed the impact of blister-packaging in oncology, making this analysis hypothesis-generating. Future research should explore the impact not only on adherence, but also on clinical outcomes such as treatment success, progression free survival, and overall survival, as well as patient specific outcomes such as quality of life, patient satisfaction with blister-packaging, and ease of use with blister-packaging. These studies could include prospective pragmatic clinical trials or observational studies, or retrospective claims analyses where researchers compare adherence rates between manufacturer-supplied blister packaging and standard stock vials, examining whether packaging design influences outcomes. Additionally, future studies should account for different cancer types, given the variations in medication effectiveness and disease severity across oncology.
Limitations
There were several limitations to this analysis in addition to what has already been discussed. One limitation is that the study utilized to assess the impact of blister-packaging medications on adherence was conducted in patients utilizing ACEi and not in patients taking oral oncolytics. No studies were identified that assessed the impact of blister-packaging oncology agents, so therefore we opted to utilize the results of this study as it was the best available evidence. While CLL currently is treated and managed like a chronic condition, 23 there are differences in the safety profiles of CLL agents compared to ACEi that may contribute to differences in medication adherence. ACEi have a mild adverse event profile with rates of adverse events being reported in less than 20% of patients, and treatment discontinuation rates in clinical trials ranging from 3–6%.65,66 While most adverse events for ACEi are mild, one of the more prevalent reactions is a dry cough which can lead to treatment discontinuation. Oral oncolytics on the other hand have a more severe safety profile. Of the four recommended first-line agents in the CLL space, the rates of any adverse events are greater than 90%, with adverse events with a severity of grade III or higher ranging from 50–80% in phase III trials.67–72 More severe adverse events in oral oncolytics can lead to holding treatment until the event dissipates which could result in patients appearing to be nonadherent in claims analyses, or treatment discontinuation. Additionally, treatment discontinuation in phase III trials for these agents ranged from 6% to 23%.67–72 While this leads to an important limitation in this analysis since no studies have been conducted in real-world settings assessing the impact of blister-packaging oral oncolytics, the best available evidence of blister-packaging impact was used in this hypothesis generating analysis. Given the potential uncertainty in this setting, the one-way sensitivity analyses attempted to address the variability of this input by varying the upper and lower bounds of this to assess what the potential overall impact would be of blister-packaging oral oncolytics.
Another limitation that was partially covered above is that the impact used in the model was carried out for all first-line treatments for CLL, in addition to what was assessed in the source study by Dashputre et al., and we do not know for certain what the true impact would be for the newer first-line recommended medication regimens. However, due to the improved efficacy results of newer agents,30,31 adherent patients on the newer first-line therapies may see an even larger impact on health outcomes and healthcare costs. As discussed above, this analysis used the incidence of CLL as a proxy for the number of patients with CLL who would be initiating therapy each year. While this may not be an exact number, it helps provide a best estimate. The one-way sensitivity analyses assessed the uncertainty of this input by varying the number of patients in the model initiating treatment in the U.S. by +/- 20% to account for disease incidence variability.
Conclusion
Nonadherence in oncology substantially impacts patient outcomes, HCRU, and healthcare costs. Blister-packaging oral oncology drugs in CLL has the potential to reduce total healthcare expenditures by increasing medication adherence. Future studies are needed to evaluate the direct effects of blister-packaging oral oncolytics on clinical, HCRU, and financial outcomes.
Footnotes
Author contributions
Concept and design (EPB, JDL); analysis and interpretation of data (EPB); drafting of the manuscript (EPB); critical revision of the manuscript for important intellectual content (EPB, NB, HN, PS, DD, JDL); final review of the manuscript (EPB, NB, HN, PS, DD, JDL); administrative, technical, or logistic support (EPB, DD, JDL); supervision (EPB, JDL). All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Saad, Dumitru, Nelkin, and Lucaci are employees and shareholders of Becton, Dickinson and Company. Borrelli and Barnes are employees of Becton, Dickinson and Company. Barnes is a shareholder of BioMerieux. No other conflicts of interest to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
