Abstract
Introduction
The demand for oncology pharmacy services is set to increase as the burden of cancer rises in sub-Saharan Africa. Oncology pharmacists may be exposed to antineoplastic drugs (ADs) and need comprehensive health and safety guidelines. The objective of the study was to assess the effectiveness of the local oncology pharmacy practice standards, by critically evaluating them against international best practice standards.
Methods
We compared the Independent Clinical Oncology Network (ICON) administration of ADs standards resource document (ICON standards) and Good Pharmacy Practice (GPP) standards with the International Society of Oncology Pharmacy Practitioners (ISOPP) Standards for the safe handling of cytotoxics, and the Quality standard for the oncology pharmacy service (QuaPos), using 10 domains: transport of ADs, working arrangements, education and training, engineering controls, use of personal protective equipment, risk management, medical monitoring of personnel, cleaning procedures, accident management and documentation, labelling and checking procedures.
Results
The ICON standards align closely with international best practice standards, but the GPP standards focus only briefly on the compounding of ADs.The GPP standards are outdated and some of the stipulations are erroneous. Oncology pharmacists would do better to adhere to the more comprehensive ICON standards, although these standards also need to be updated in line with best practice.
Conclusion
Revising and improving both these local standards in consultation with key role players in the oncology pharmacy industry will go a long way in protecting the health and safety of oncology pharmacists in South Africa.
Keywords
Introduction
With the incidence of cancer expected to increase by 70% in sub-Saharan Africa by 2030, 1 the role of the oncology pharmacist is more important than ever, in correlation with the high demand for oncology pharmacy services in the region. In South Africa, there are no formal training programmes for oncology pharmacists or official guidelines. Special registration as an oncology pharmacist with the South African Pharmacy Council (SAPC) is not required. 2 With a 2024 market revenue of US$ 0.57 billion (projected to increase to US$ 0.78 billion by 2029), the South African oncology drug market is rapidly expanding. 3 This will lead to higher volumes of anti-neoplastic drugs (ADs) passing through the health care system and greater numbers of individuals involved in the mixing of the drugs being exposed.
In 2022, von Grunigen, Geissbuhler 4 reported that there were major gaps in the safe handling of AD drugs in low- and middle-income countries (LMICs) although upper-middle-income countries (like South Africa) performed appreciably better than lower-income countries. They used a free online tool (Cyto-SAT) to assess 134 items grouped into 10 domains and 28 sub-domains in oncology pharmacy. Considerable variation in the content and scope of safe handling guidelines and pharmacy practices was also observed in a study in which 24, mainly developed countries, including the United States, Canada, United Kingdom, continental Europe, Japan and Australia, participated. 5 Authors of a study from Ethiopia (another LMIC) reported that, in their setting, only 37.6% of health professionals who handled ADs had good cytotoxic handling practices. 6 Fazel et al. (2022) reviewed studies of barriers to the safe handling of cytotoxic drugs in Canada, which included poor handling (46% of reviewed papers), poor safety culture (41%) and inconsistent policies (36%), while also showing that implementing certain facilitators such as adequate safety training (41%), leadership support (23%), and consistent policies (21%) can have a positive impact on oncology pharmacy practice. 7
In 2018, the Independent Clinical Oncology Network (ICON) “Chemotherapy standards and guidelines” were published, with a focus on safety, competency, and error reduction. The document specifically mentioned the lack of nationally recognised guidelines for AD administration in South Africa. 8
All pharmacies (community and institutional) in the private sector in South Africa, in which ADs are prepared, are affiliated with one of two managed care organisations, viz. the Independent Clinical Oncology Network (ICON) or the South African Oncology Consortium (SAOC). Both organisations have guidelines to which the respective oncology practices must adhere. While the ICON standard on the compounding of ADs is available publicly, the SAOC standard is not.
In addition to the ICON or SAOC standards, all pharmacies that are registered with the SAPC are legally obliged to adhere to the Good Pharmacy Practice (GPP) standards in terms of the South African Pharmacy Act No. 53 of 1974. 9 However, not all ICON- and SAOC-affiliated pharmacies are registered with the SAPC. Those that are not registered are not legally required to apply the GPP standards.
Although oncology pharmacy is not a recognised speciality in South Africa, some individuals who have experience in mixing ADs belong to the South African Society of Oncology Pharmacists (SASOPh). Membership is voluntary and only around 60
The objective of this study was to assess the effectiveness of the ICON and GPP standards in protecting the health and safety of those handling ADs in private pharmacies in South Africa, by critically reviewing and evaluating the two local standards against the international best practice ISOPP standards and the QuaPos standard.
Methods
The GPP, ICON, ISOPP and QuaPos standards were reviewed, using 10 oncology pharmacy-related domains derived from two international best practice standards recommended by the SASOPh, to identify gaps and contradictions in the local guidelines. The standards as well as the determination of the comparisons were reviewed by the first author (RB). The domains used were the transport of ADs, working arrangements, education and training, requirements for facilities and engineering controls, use of personal protective equipment (PPE), risk management, environmental monitoring and medical monitoring of personnel, cleaning procedures, accident management and documentation, labelling and checking procedures.
Results
The GPP standards
The GPP standards are aimed at pharmacy practice in general and are not specific to oncology pharmacy. One relatively short section of the GPP standards (5 of 222 pages) is dedicated to the practice of the mixing of ADs and was last updated in 2010. Nine of the 10 domains are covered; no mention is made of risk management. Some of the statements made in the GPP are inaccurate. These include: “no adequate means of monitoring and measuring inadvertent adsorption [of cytostatics] have been devised”, and “protective clothing must include … a single-use surgical mask” (which does not provide adequate protection against exposure to ADs). In addition, the recommendations are not written in a logical sequence.
The ICON standards
The ICON standards, last updated in 2018, are more comprehensive than the GPP standards, regarding the compounding of ADs. While the functions and roles of the oncology nurse and the oncologist are comprehensively described in separate sections, no specific details are provided about the role of the oncology pharmacist; the compounding of ADs is assigned to the oncology nurse. All 10 oncology pharmacy-related domains are covered.
The ISOPP standards
The ISOPP standards for the safe handling of cytotoxics, which are specific to oncology pharmacy, were first published in 2007. Refinement of safe handling practices led to the latest 2022 edition of the standards. They provide a comprehensive, structured approach to the practice of oncology pharmacy and all 10 oncology pharmacy-related domains are discussed in detail.
The QuaPos standard
The globally recognised QuaPos standard was developed by the European Society of Oncology Pharmacy. The most recent edition was published in 2021. The standard is specifically aimed at the oncology pharmacist and the practice of oncology pharmacy, and cover all 10 domains.
Comparison of the GPP, ICON, ISOPP and QuaPos standards
The GPP standards, with their 32 items, are outdated and inadequate regarding oncology pharmacy practice. They mention oncology pharmacy practice only briefly in one of 49 sections, with limited focus on the compounding of anti-neoplastic drugs. They do not provide the detailed and in-depth guidance that the ICON, ISOPP and QuaPos standards do. These three standards are structured, using appropriate and relevant headings, and address the various aspects of oncology pharmacy practice systematically.
Even though recommendations for good oncology pharmacy practice are mentioned in the section in which the function of the oncology is described in the ICON standards, rather than in a section about the oncology pharmacist, the recommendations themselves compare favourably with the ISOPP and QuaPos standards. However, the ICON standards do not discuss the practice of oncology pharmacy as extensively as the other two standards, nor do they provide the same level of detail.
Oncology pharmacy-specific domains: performance of the South African standards
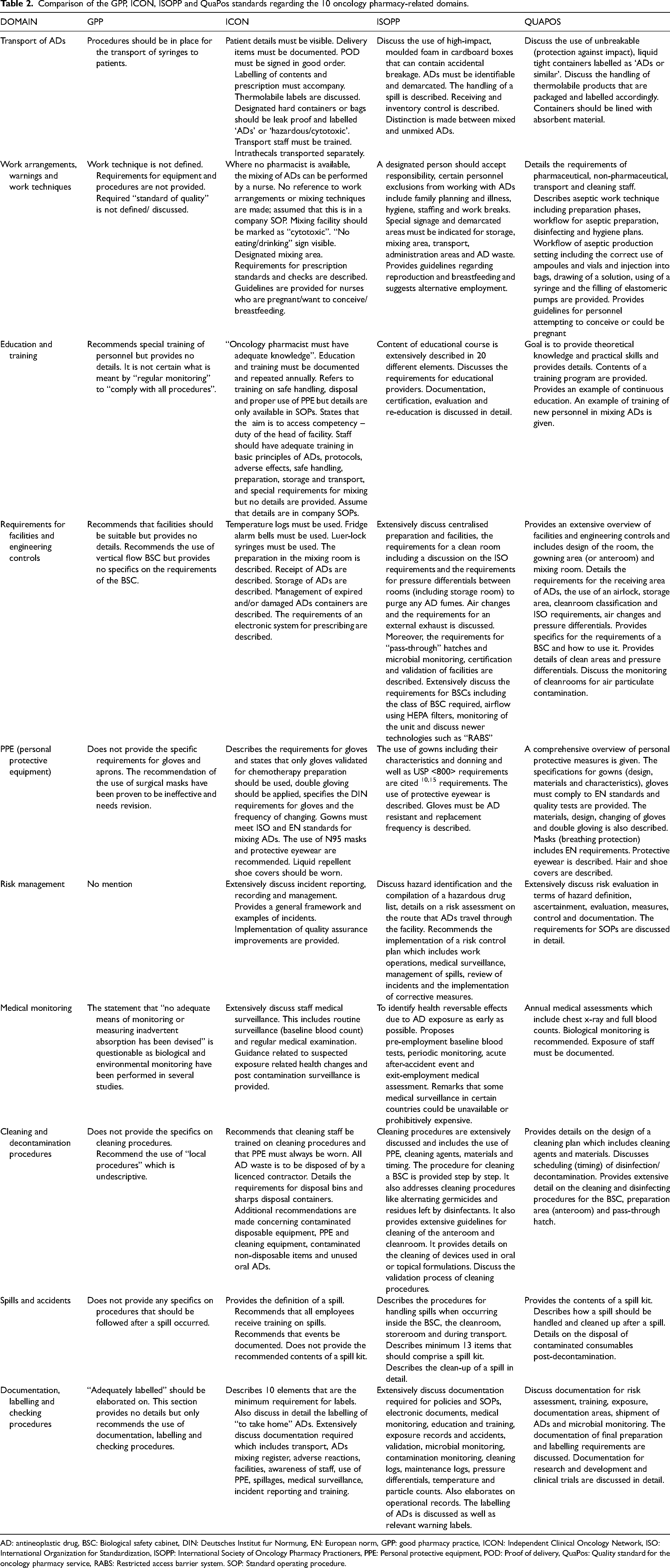
The differences between the four standards regarding the 10 oncology pharmacy practice domains, are provided in Table 1.
List of abbreviations.

Transport of anti-neoplastic drugs
Although the GPP standards mention the safe transport of ADs, additional details that are provided in the ISOPP and QuaPos standards are missing. The GPP standards state that ADs must be transported in protected syringes (to prevent accidental spillage) that are individually labelled with essential information such as the name and strength of the medication, and the patient details. The ICON standards are more comprehensive than the GPP standards and provide more details regarding the transport and receipt of ADs. They compare well with the ISOPP and QuaPos standards as they include recommendations regarding the use of unbreakable, liquid-tight containers, staff training, and documentation and procedures in the event of an accident or ‘spill’. The ICON standards specifically distinguish between mixed and unmixed ADs, advocating the use of high-impact moulded foam during transport for added protection of AD containers. The ISOPP and QuaPos standards both recommend that the admixtures be delivered directly to the ward/infusion lounge and that all staff involved in the transport be appropriately trained on procedures relating to accidental spillage. The ISOPP and QuaPos standards also provide more details than the ICON standards about the labelling of containers to identify that they contain ADs.
Work arrangements, warnings and work techniques
Work arrangements, warnings and work techniques are only briefly mentioned in the GPP standards. The ISOPP and QuaPos standards include the effectiveness of work arrangements (administrative controls) in the hierarchy of exposure control. The ICON standards state that, in the absence of an oncology pharmacist/pharmacy, compounding can be done by a registered nurse, but omit detailed mixing techniques. The ISOPP and QuaPos standards emphasize the importance of administrative controls, including clear signage to warn staff about exposure to ADs, designated areas, and detailed aseptic work techniques, reconstitution and compounding of ADs, cytotoxic waste, and spills. The ISOPP and QuaPos standards also comprehensively describe safe work techniques, including compounding ADs in a biological safety cabinet (BSC), sterile mixing, and pharmaceutical microbiology. All four standards recommend the use of luer-lock syringes for compounding ADs. Reference is made to personnel who wish to conceive or who are pregnant in all except the GPP standards.
Education and training
The GPP standards state, in several places, that staff should receive “adequate training”, but do not provide any details about the content or other issues related to the training. The ICON standards mention adequate training in basic principles of ADs, protocols, adverse effects, safe handling, preparation storage, and transport, but does not elaborate on any of these aspects. The ISOPP and QuaPos standards, however, do provide comprehensive guidance about course content, education providers, certification and recertification requirements, and documentation of training. The QuaPos standard also provides a practical example of continuous education.
Requirements for facilities and engineering controls
The GPP standards require that facilities are suitable for the preparation of ADs but provide no additional details regarding engineering controls. The ICON standards specify the requirements for the preparation of ADs in the mixing room, receipt and storage, management of expired ADs, and electronic prescribing of chemotherapy. In contrast, the ICON and ISOPP standards offer comprehensive requirements, including cleanroom specifications, air changes, pressure differentials, and the use of vertical flow BSCs. The QuaPos standard further details the monitoring, validation, and microbial safety of cleanrooms, underscoring the importance of meticulous engineering controls in maintaining a safe environment.
Personal protective equipment
Personal protective equipment includes masks, gowns, gloves, head and shoe coverings. There are variations regarding the use of other PPE in all the standards. The only types of PPE mentioned in the GPP standards are surgical masks, eye protection, single-use plastic aprons, and “appropriate” gloves (undefined). The ICON standards comprehensively describe the use of PPE. For example, they emphasize the importance of using EN 374 gloves, gowns that meet international standards, N95 masks, and head and shoe coverings. All relevant PPE is comprehensively discussed in the ISOPP and QuaPos standards. Both standards recommend the use of FFP3 type masks. The ISOPP standards make extensive reference to the United States Pharmacopoeia Chapter 800 for the requirements of the use of PPE, 10 including AD-resistant gloves. The QuaPos standard describes the use of EN standards/requirements related to the use of PPE, as well as the design of gloves and the frequency with which they should be changed.
Risk management
The GPP standards do not specifically address risk management, but the ICON standards provide a comprehensive framework for incident reporting, and management and implementation of quality assurance improvements. Risk management receives special attention in both the ISOPP and QuaPos standards. The ISOPP standards emphasize hazard identification, risk assessment, and the implementation of a risk control plan, including medical surveillance and incident management. The QuaPos standard provides an extensive evaluation of risks, with detailed requirements for standard operating procedures (SOPs), including a template of an SOP that can be used for risk management. In addition, incident reviews are discussed and procedures for documenting accidents are provided.
Medical monitoring
The GPP standards provide no details about environmental and medical monitoring. However, it erroneously states that “no adequate means of monitoring or measuring inadvertent absorption has been devised”. The ICON standards, in comparison, discuss medical monitoring in detail – in line with the ISOPP and QuaPos standards. All three state that blood tests (including full blood counts and liver function tests) and medical examinations should be part of routine examinations. The QuaPos standard recommends annual examinations. Included is the recommendation to record ‘baseline’ measurements for new employees and ‘exit’ medical examinations when employees leave the company.
Cleaning and decontamination procedures
The GPP standards state that surfaces should be “thoroughly cleaned immediately after use according to local procedures” but do not provide details about the cleaning methods. The ICON standards state that N95 masks and protective eye wear should be used during the cleaning process but do not go into detail about that process. They do, however, refer to an internal SOP about cleaning, which is not included in the standards themselves. Cleaning and decontamination procedures are, however, extensively described in both the ISOPP and QuaPos standards, which specify the use of PPE during decontamination, and the cleaning agents and materials (such as gauze) that should be used. Cleaning procedures for specific areas in the pharmacy are provided, including the anteroom and storage areas, the BSC, and devices used in oral and/or topical formulations. The ISOPP and QuaPos standards also require cleaning procedures to be documented.
Spills and accidents
The GPP standards state that “correct action is taken following any accidental spillage or contamination of the person”, but do not stipulate what those “correct actions” are. They also state that “accidental spillage or contamination of the operator must be dealt with immediately and procedures must be established for the treatment of the skin, eyes and other surfaces”. Again, the specific procedures are not described. The importance of training and documentation regarding spills is included in the ICON standards, without the provision of practical steps for handling spills. Although the ICON standards do not describe the required contents of a spill kit, they do require that such a kit is available in the workplace. They also recommend that all staff receive training on procedures following a spill and that all such events be documented. The ISOPP and QuaPos standards offer comprehensive instructions, including the contents of spill kits and detailed procedures for managing spills in various environments, such as the BSC, cleanroom, storeroom, and during transport. The contents of a spill kit, and procedures that need to be followed after an accidental spill in the BSC, cleanroom, storeroom, or during transport are comprehensively discussed.
Documentation, labelling and checking procedures
The GPP standards briefly mention the necessity for ADs to be “adequately labelled” but lack comprehensive guidance. In contrast, the ICON standards extensively detail the labelling requirements, particularly for take-home medications, and outline the necessary documentation for various processes, including transport of ADs, an AD mixing register, adverse reactions register, spills register, history of medical monitoring of staff, incident reporting, and training. Both the ISOPP and QuaPos standards provide detailed documentation protocols for the maintenance of operational records, labelling requirements, policies, SOPs, electronic documents, medical monitoring, education and training, exposure records, accidents, microbial monitoring, environmental monitoring, cleaning records, maintenance records, and BSC servicing records.
Discussion
Of the thousands of pharmacies registered with the SAPC, only five have the word ‘oncology’ in their names (as per the SAPC website, August 2024). All five are private pharmacies and are registered as either institutional (hospital) or community (retail) pharmacies as there is no option to register with the Council as an oncology pharmacy.
All registered pharmacies are obliged to adhere to the GPP standards. Only a few of the approximately 160 private oncology pharmacies in South Africa are registered with the SAPC; the others are affiliated with one of the two managed care organisations (the ICON or the SAOC), and adhere to their specific practice standards.
The objective of this study was to assess the effectiveness of the locally-used GPP and ICON standards against international oncology pharmacy best practice standards in protecting the health and safety of those handling ADs in private pharmacies in South Africa. The ICON standards proved to be more comprehensive than the GPP standards and compared well with the two international best practice standards recommended by the SASOPh.
While the GPP standards provide a basic framework for the transport of ADs, they lack the details included in the ICON, ISOPP and QuaPos standards. The GPP standard's lack of specificity, especially regarding labelling of containers and training of staff about spills during transport, could compromise the safety of employees.
There are also major gaps in the GPP standards regarding work arrangements, warnings, and work techniques when handling ADs, although these are adequately covered in the ICON standards. While both the GPP and ICON standards emphasise the necessity for adequate education and training, neither of these locally used standards define ‘adequate’, nor do they provide sufficient detail about the content.
Regarding the use of PPE, the GPP standards’ stipulation for the use of surgical masks when mixing ADs is incorrect. Surgical masks have been proven to provide ineffective protection during the compounding of ADs. 11 The appropriate masks to use are FFP3 masks (recommended in the ISOPP and QuaPos standards). The latter provide superior protection from AD exposure than do surgical masks.
Risk management, which incorporates risk identification, risk measurement and assessment, risk mitigation, and risk reporting and monitoring, is an important aspect in protecting health and safety, in all industries. 12 Therefore, it is concerning that it is not mentioned in the GPP standards with respect to oncology pharmacy practice.
The GPP standards inaccurately state that no adequate monitoring methods for AD exposure exist. This may be because the standards were published more than a decade ago. Many developments have been made in this area since 2010, and there are now well-established methods that can be used to measure and monitor biological and environmental contamination of ADs in oncology pharmacies.13,14 The medical monitoring stipulated in the international standards should be included in the GPP standard, including blood tests to measure baseline liver function tests, annual medical examinations, etc.
The GPP standards provide minimal guidance for cleaning and decontamination, While the ICON standards underscore the importance of staff training and the use of PPE, they fall short in stipulating specific cleaning methods. Both standards need to be updated to incorporate the guidance provided by the ISOPP and QuaPos standards, i.e., PPE use, cleaning agents, specific area procedures, and documentation requirements.
The GPP and ICON standards provide general recommendations regarding what to do in the event of an accidental AD spill, but neither provides details about what procedures to follow if a spill occurs or the required contents of spill kits. The contents of a spill kit are provided in the ISOPP standards, viz. written instructions, warning signs and PPE, a spill mat for small spills, a pillow for large spills, use of alkaline detergent solutions, etc., all of which should be added to the GPP and ICON standards.
There are significant differences in the standards, regarding documentation, labelling, and checking procedures for ADs. Again, it is the GPP standards that need to be expanded to include issues such as labelling requirements – particularly for take-home medications – and relevant documentation related to the transport of ADs, reporting of adverse reactions, maintenance of operational records, and medical and environmental monitoring.
Recommendations
It is crucial to update and revise local standards such as the GPP to align with international practices like ISOPP and QuaPos. Implementing comprehensive training programs, mandating advanced engineering controls in pharmacy facilities, and promoting the use of personal protective equipment (PPE) is essential steps to safeguard healthcare professionals. All pharmacies should be registered with the SAPC. The benefits of registration include, amongst others, professional recognition, which enhances credibility and trustworthiness among patients and healthcare providers; access to resources, guidelines, and updates provided by the SAPC that help them to stay informed about best practices and regulatory changes; access to Continuous Professional Development programmes, which ensure that the pharmacy and staff remain up-to-date with the latest developments in the field; and opportunities to network with other professionals and organizations within the SAPC. The GPP standards state only that facilities should be suitable for AD preparation, without specifying details about engineering controls Chemotherapy is transported long distances across the country on a daily basis, so it is essential that appropriate protocols are followed to ensure the safe transport of ADs and the of protection of those involved.
Conclusion
The adverse health effects that are related to occupational exposure to ADs are potentially serious, and revision of the standards related to the compounding of ADs in all pharmacies – whether registered with the SAPC or affiliated with the managed care organisation – is long overdue. The GPP standards (published in 2010) are very outdated. Additionally, some of the stipulations in the standards are erroneous, which could compromise the health and safety of oncology pharmacists (i.e., government employees) if followed. Until a concerted effort is made to revise the GPP standards, they remain redundant with regard to oncology pharmacy practice. All oncology pharmacists would do better to adhere to the more comprehensive ICON standards, although these standards also need to be updated in line with best practice, having last been revised in 2018. Revising and improving both these local standards in consultation with key role players in the oncology pharmacy industry will go a long way in protecting the health and safety of oncology pharmacists in South Africa Table 2.
Comparison of the GPP, ICON, ISOPP and QuaPos standards regarding the 10 oncology pharmacy-related domains.
AD: antineoplastic drug, BSC: Biological safety cabinet, DIN: Deutsches Institut fur Normung, EN: European norm, GPP: good pharmacy practice, ICON: Independent Clinical Oncology Network, ISO: International Organization for Standardization, ISOPP: International Society of Oncology Pharmacy Practioners, PPE: Personal protective equipment, POD: Proof of delivery, QuaPos: Quality standard for the oncology pharmacy service, RABS: Restricted access barrier system. SOP: Standard operating procedure.
Footnotes
Author contributions
RB conceptualised the study, designed the methodology, collected the data, contributed to the interpretation of the results, wrote the first draft of the manuscript. DB supervised the study, contributed to the design, critically reviewed the final draft of the manuscript. GN supervised the study, contributed to the design of the study, methodology and interpretation of results, critically reviewed and edited the manuscript. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
