Abstract
Introduction
Malignant hematology (MH) patients are susceptible to invasive fungal infections due to prolonged neutropenia and immunosuppressive therapies, which may require voriconazole therapy. Although voriconazole therapeutic drug monitoring (TDM) is common, evidence describing this practice is limited. The primary objective of this study was to describe the current practice of voriconazole TDM in MH patients at the Princess Margaret Cancer Centre (PM).
Methods
A retrospective chart review was conducted for MH inpatients initiated on voriconazole at PM between November 1st, 2019 and November 13th, 2020. Data regarding voriconazole doses, levels, dose changes, and adverse effects were collected. The primary endpoint was the proportion of patients with initial voriconazole levels within therapeutic range (1–5 mg/L).
Results
Fifty-six patients were included in the study. The most common reason for starting voriconazole was possible invasive fungal infection (44 patients, 78.6%). Fifty-one patients (91.1%) received a loading dose of voriconazole, averaging 386.5 ± 78.5 mg. The average maintenance dose was 242.1 ± 45.7 mg. An average of 2.6 ± 2.9 levels were drawn per patient with an average level of 3.2 ± 2.4 mg/L. Forty-one patients (73.2%) had an initial voriconazole level within therapeutic range and 90 out of 145 total levels (62.1%) were within therapeutic range. There were 52 dose modifications made; 31 (60.8%) doses adjusted, 12 (23.5%) doses held, and 9 (17.6%) doses discontinued. For the 31 dose adjustments, 26 (83.9%) had a level redrawn and 17 (65.4%) of those levels were within therapeutic range. Twenty-three (41.1%) patients developed adverse effects, 8 (34.8%) of which were associated with supratherapeutic levels. Of these 23 patients, 19 (33.9%) experienced transaminitis, 3 (5.4%) experienced both transaminitis and neurotoxicity, and 1 (1.8%) experienced photopsia.
Conclusion
Overall, 41 (73.2%) patients achieved an initial voriconazole level within therapeutic range. Of these 41 patients, 30 (73.2%) remained within therapeutic range for the duration of their inpatient voriconazole therapy. These findings suggest that the current practice of voriconazole TDM at our institution is yielding largely positive results, but still has room for improvement.
Introduction
Malignant hematology (MH) patients are susceptible to invasive fungal infections (IFIs) due to prolonged neutropenia.1,2 The European Organization for Research and Treatment of Cancer/Mycoses Study Group (EORTC/MSG) classifies IFIs as “proven”, “probable”, or “possible”. 3 Triazole antifungals are the preferred agents for the treatment and prevention of IFIs in most patients. 1 The Infectious Diseases Society of America (IDSA) recommends primary treatment with voriconazole for invasive pulmonary aspergillosis (IPA). 1 This drug exhibits wide intra- and inter-patient variability, owing to a range of factors including a non-linear relationship between dose and serum level, patient age, drug-drug interactions (as it is both a substrate and inhibitor of CYP2C9, CYP2C19, and CYP3A4), and genetic polymorphisms of CYP2C19.4–7 Studies have shown that voriconazole trough levels are correlated with the occurrence of adverse events and/or treatment response.8–10 The British Society for Medical Mycology recommends a target voriconazole trough concentration of greater than 1 mg/L for the treatment of IFIs and a voriconazole trough of less than 4–6 mg/L to minimize drug-related toxicities. 11 Our local Sinai Health System (SHS)/University Health Network (UHN) Antimicrobial Stewardship Program (ASP) Guidance Document on Voriconazole Monitoring and Adjustment recommends targeting voriconazole trough levels between 1–5 mg/L for improved outcomes and reduced toxicity. 12
There is some literature available describing voriconazole TDM in MH patients.13–17 Yi and colleagues conducted a retrospective cohort study (n = 151) evaluating adult inpatients who received posaconazole or voriconazole TDM, in which 54% of the total voriconazole levels were within therapeutic range, defined as 1–5.5 mg/L. 13 Sebaaly and colleagues conducted a retrospective chart review (n = 88) evaluating voriconazole TDM in MH patients, solid organ transplant recipients, or patients with pulmonary disease, in which 53% of patients had initial voriconazole levels within the target range, defined as 1–5.5 mg/L. 14 Guinea and colleagues conducted a retrospective study (n = 107) evaluating voriconazole serum concentrations collected from patients at 6 different hospitals in Madrid, with 42.1% of those patients having a diagnosis of a hematological malignancy. They found that 71.3% of all voriconazole levels were within therapeutic range, defined as 1–5.5 mg/L. 15
Neofytos and colleagues conducted a multicenter trial at 4 academic institutions in North America. 16 Patients were randomly assigned to receive either standard dosing of voriconazole without TDM or TDM-driven voriconazole dosing. However, this trial was discontinued prematurely due to an inability to enroll patients in the non-TDM-arm. The authors attributed this to the non-blinded nature of this study, resulting in a reluctance of clinicians to enroll patients into the non-TDM-arm. 16 Saini and colleagues prospectively investigated voriconazole levels in MH patients from June 2007 to September 2010 receiving oral voriconazole. 17 The study team found considerable variability in the voriconazole levels, ranging from 0 mg/L to 16.6 mg/L. 17
With the increase of published literature supporting the practice of voriconazole TDM, it has become standard of practice at the Princess Margaret Cancer Centre (PM), with pharmacists assuming more responsibility for this. Pharmacists have extensive training in medication management as well as TDM, and at our institution, already have medical directives in place for the management of other medications such as vancomycin. At the time of this study, voriconazole levels were being ordered by prescribers with recommendations for dose adjustments and subsequent levels made by pharmacists, when appropriate. TDM is performed for all patients receiving voriconazole, using our local guidance document to aid clinicians in decision-making. The document includes guidance on when to order levels and recommendations on managing subtherapeutic and supratherapeutic levels. 12 A proposal for a voriconazole medical directive to order and monitor serum voriconazole trough levels, as well as make the necessary dose adjustments, was put forth by PM pharmacists. Our study aims to evaluate recent practice in voriconazole TDM in MH patients at PM.
Objectives
Primary objective
The primary objective of our study was to describe the current practice of voriconazole TDM in MH inpatients at PM.
Secondary objectives
The secondary objectives were to: a) evaluate the safety of voriconazole by assessing the proportion of patients with adverse effects while on voriconazole who also had supratherapeutic drug levels; and b) measure the proportion of therapeutic drug levels achieved after dose modification of sub- or supratherapeutic levels during inpatient admission.
Materials and methods
Design
This retrospective chart review was conducted at PM. The study protocol was approved with a waiver for the need of informed consent by the University Health Network Research Ethics Board (REB), approval number 20–6220.1.
Individuals who were diagnosed with a hematological malignancy, admitted on a malignant hematology unit and started on voriconazole as an inpatient between November 1st, 2019 and November 13th, 2020, and had at least one trough voriconazole level drawn were included in this study. Patients were excluded if they were started on voriconazole in the outpatient setting, or if they were initiated on voriconazole at a different institution. Potential study participants were identified using our pharmacy medication management system to determine patients who were prescribed voriconazole. All data were collected from this system and our institution's electronic patient record.
Upon initiating voriconazole, data regarding the reason for voriconazole use, previous antifungal use, baseline renal, and baseline hepatic function were collected. All voriconazole trough levels drawn while patients were admitted were included in our study. Data regarding the timing of the voriconazole levels were collected from the electronic patient record. Trough levels were further analyzed to evaluate whether they were drawn according to the SHS/UHN ASP Voriconazole Monitoring and Adjustment guidance (before the 6th dose if a loading dose was given or on the 6th day of therapy if a loading dose was not given). 12
The adverse events evaluated in this study were elevated liver enzymes (aspartate transaminase (AST) and alanine transaminase (ALT)), elevated bilirubin, visual disturbances (photopsia), and neurotoxicity (visual and/or auditory hallucinations, altered mental status, agitation, and involuntary myotonic movements). The Common Terminology Criteria for Adverse Events (CTCAE) v.5 was used to define elevated liver enzymes and bilirubin (Figure 1). 18 At our institution, the upper limit of normal (ULN) for AST is 34 U/L and for ALT is 40 U/L. The ULN for bilirubin is 22 μmol/L.

Transaminitis and elevated bilirubin as per Common Terminology Criteria for Adverse Events (CTCAE) v.5. ULN = Upper Limit of Normal.
The study team created a list of drugs with the potential to affect voriconazole trough levels due to pharmacokinetic interactions (see Appendix A). This list was used to evaluate whether patients receiving interacting medications had voriconazole trough levels within therapeutic range.
Outcomes
The primary outcome was defined as the proportion of patients with an initial voriconazole trough level within therapeutic range (1–5 mg/L). The secondary outcomes were defined as the proportion of patients who experienced adverse events while on voriconazole who also had supratherapeutic drug levels and the proportion of therapeutic drug levels achieved after dose modification of sub- or supratherapeutic levels during inpatient admission.
Data analysis
Descriptive statistics were performed to report the patient demographics and study outcomes. Data analysis was conducted using Microsoft Excel software.
Results
Patient demographics
There were 123 inpatients prescribed voriconazole during our data collection period (Figure 2). 56 (45.5%) of these patients met eligibility criteria and were included in the study. The remaining 67 (54.5%) patients were excluded for the following reasons: 31 patients (46.3%) were started on voriconazole as an inpatient before the study period, 15 patients (22.4%) were started on voriconazole as an outpatient or on a previous admission, 10 patients (14.9%) did not have a trough level reported in the electronic patient record, 5 patients (7.5%) were admitted before the study period, 3 patients (4.5%) were started on voriconazole at a different institution, 2 patients (3%) had their first voriconazole level drawn as an outpatient, and 1 patient (1.5%) did not have a diagnosis of a hematological malignancy.

Flow diagram for study participants.
An overview of study patient demographics can be found in Table 1. There were 34 males (60.7%), the mean age was 60 ± 14.5 years, and the mean patient weight was 73.9 ± 17 kg. Most patients (73.2%) had a primary diagnosis of acute myeloid leukemia (AML).
Patient demographics (n = 56).
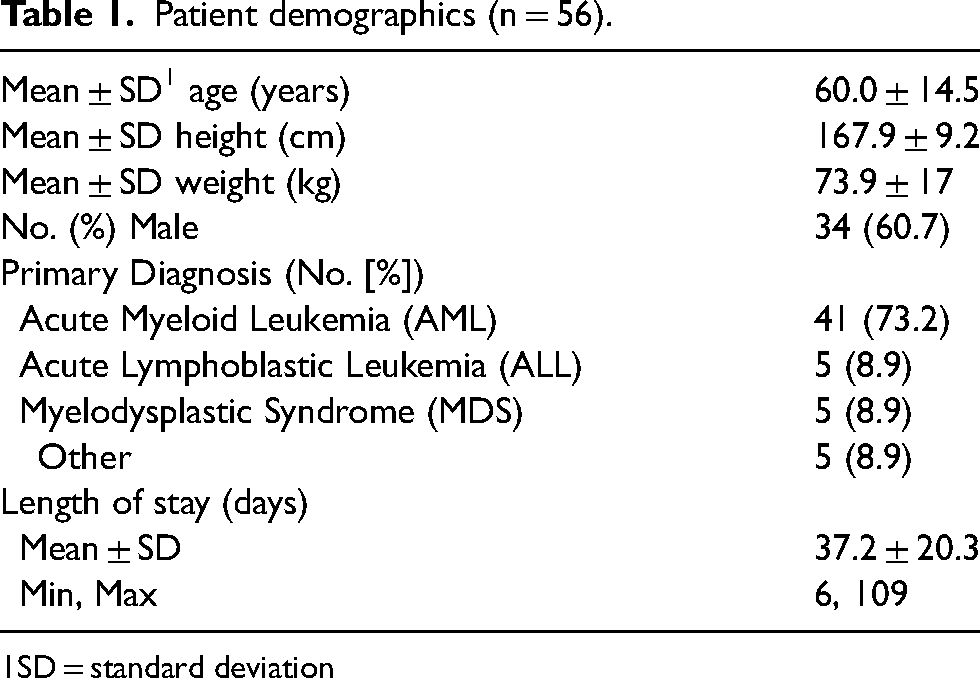
SD = standard deviation
Initiation of voriconazole
Forty-four (78.6%) patients were started on voriconazole due to possible IFI, as defined by EORTC/MSG. Nine (16.0%) patients were prescribed voriconazole for probable IFI. Two patients (3.6%) were prescribed voriconazole for proven IFI, one with positive blood cultures for Candida kefyr and the other with positive blood cultures for Trichosporon asahii. One patient (1.8%) was prescribed voriconazole as antifungal prophylaxis.
Forty-four (78.6%) patients received previous antifungals during their admission prior to initiation of voriconazole, with caspofungin (45.5%) and fluconazole (45.5%) being the most common antifungal agents used in these patients. Fifty-one (91.1%) patients received a loading dose of voriconazole. The average loading dose was 386.5 ± 78.5 mg (5.4 ± 0.7 mg/kg) and the average maintenance dose was 242.1 ± 45.7 mg (3.4 ± 0.6 mg/kg). The majority of patients had normal baseline renal function, with 83.9% of patients having a creatinine clearance (CrCl) greater than or equal to 60 mL/min prior to initiating voriconazole therapy. The majority of patients also had normal hepatic function prior to starting voriconazole with forty-four (78.6%), forty-seven (83.9%), and thirty-nine (69.6%) patients having bilirubin, AST, and ALT within normal limits, respectively. Table 2 summarizes additional data related to the initiation of voriconazole therapy.
Initiation of voriconazole (n = 56).
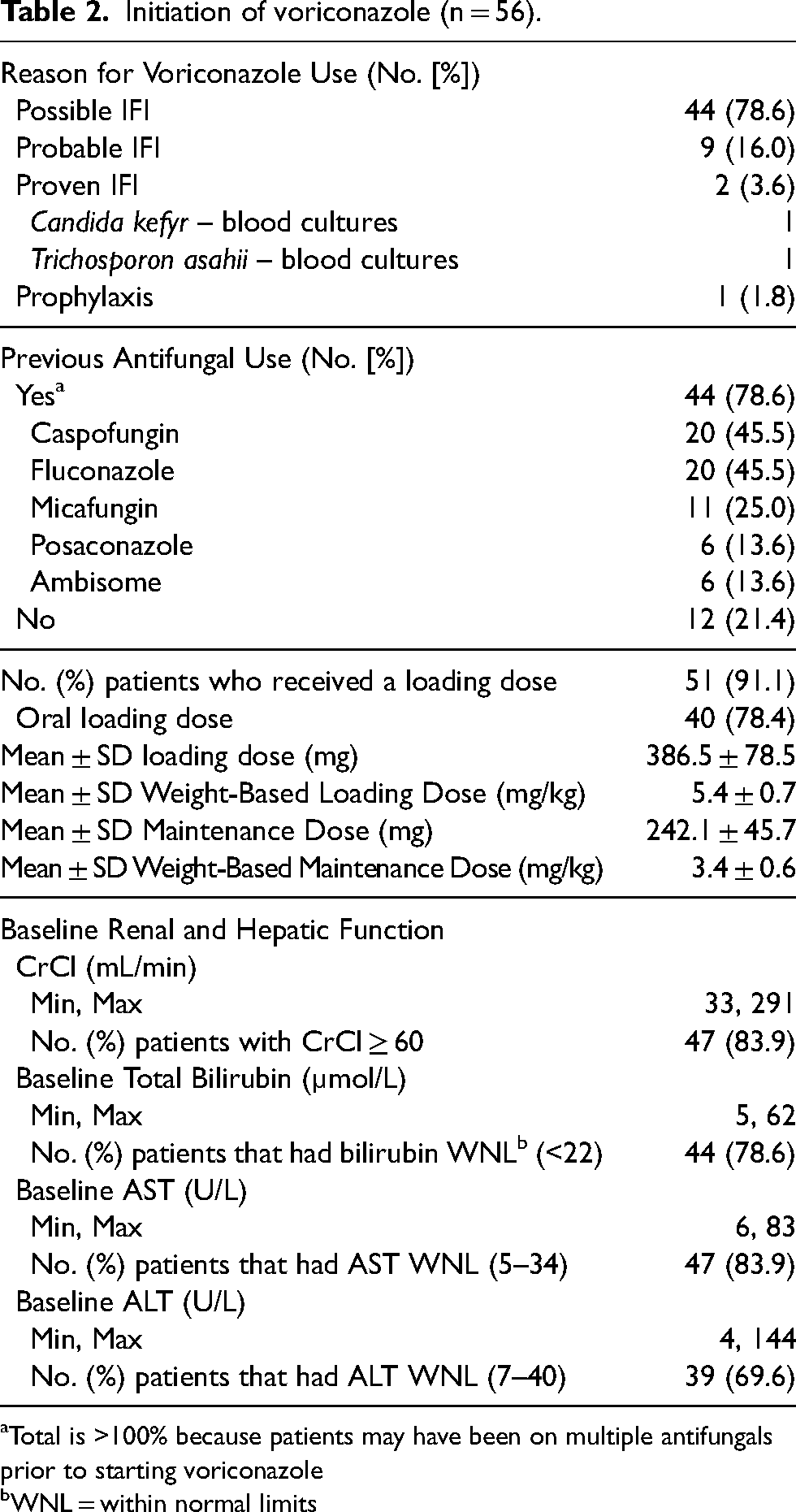
Total is >100% because patients may have been on multiple antifungals prior to starting voriconazole
WNL = within normal limits
Voriconazole therapeutic drug monitoring
An overview of voriconazole therapeutic drug monitoring at our institution can be found in Figure 3. There were 41 (73.2%) patients who had an initial voriconazole level within therapeutic range. Of these, 30 (73.2%) patients remained within therapeutic range for the duration of their inpatient voriconazole therapy. For the other 11 (26.8%) patients, 10 (24.4%) had levels outside therapeutic range after the second level, and 1 (2.4%) patient had levels outside therapeutic range after the third level. For the 10 patients who had the second voriconazole level outside of therapeutic range, 7 (70.0%) were supratherapeutic and 3 (30.0%) were subtherapeutic. Of these 10 patients, 3 (30.0%) patients needed one dose adjustment to reach the therapeutic range, 2 (20.0%) patients needed two dose adjustments, and 5 (50.0%) patients discontinued voriconazole following their second level due to adverse effects (n = 2) or other reasons (n = 3). The 1 patient whose levels were outside therapeutic range after the third level had subtherapeutic levels and needed 2 dose adjustments to reach the therapeutic range.
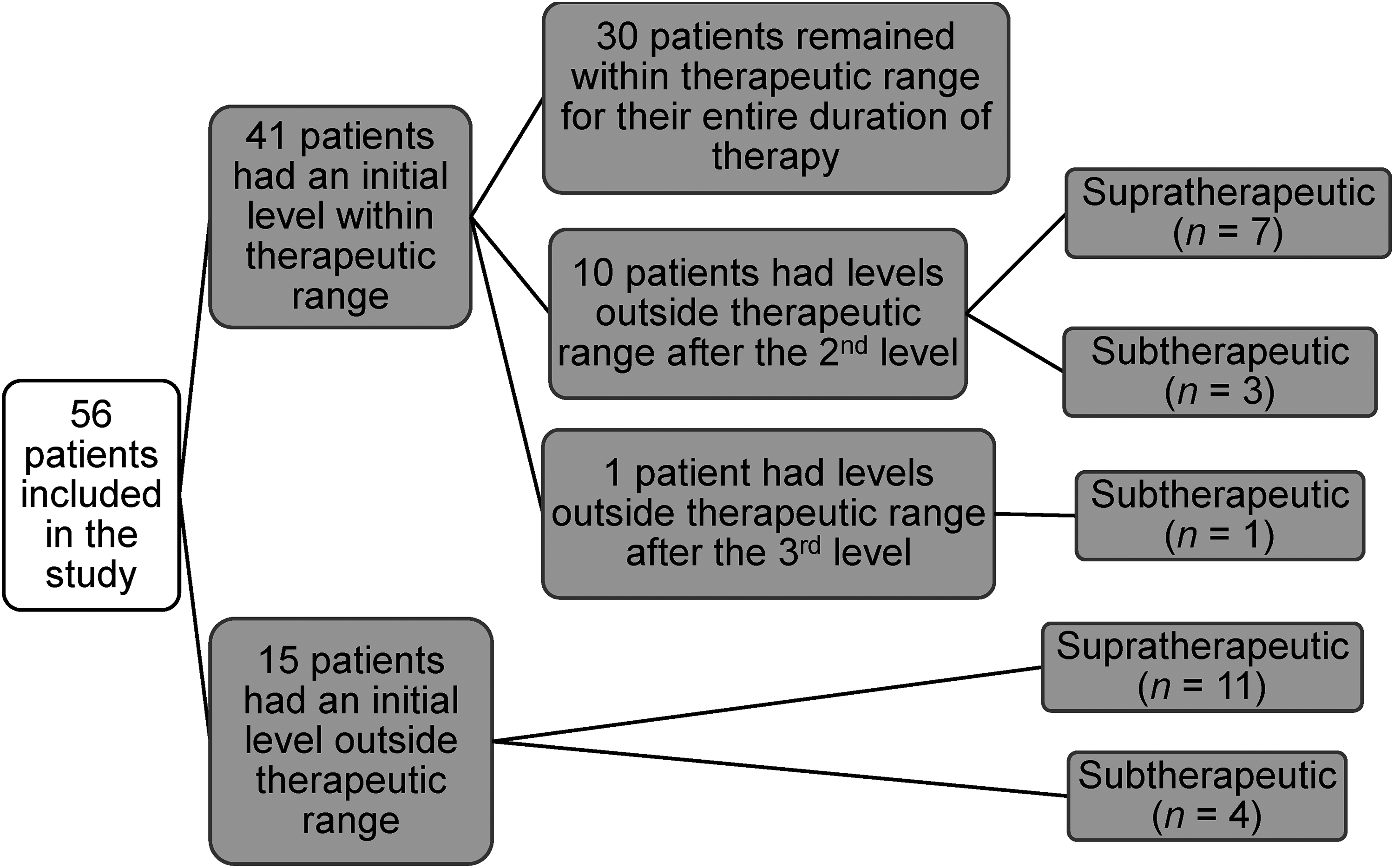
Flow diagram for voriconazole therapeutic drug monitoring.
For the 15 patients who had an initial voriconazole level outside of therapeutic range, 11 (73.3%) were supratherapeutic and 4 (26.7%) were subtherapeutic. Additionally, for those same 15 (26.8%) patients, 2 (13.3%) patients needed one dose adjustment to achieve therapeutic range, 4 (26.7%) patients needed two dose adjustments, 2 (13.3%) patients needed three dose adjustments, and 7 (46.7%) patients discontinued voriconazole following their initial level due to adverse effects (n = 5) or other reasons (n = 2).
In total, there were 145 voriconazole levels drawn, with each patient having an average of 2.6 ± 2.9 levels. Ninety (62.1%) of these levels were within therapeutic range, 24 (16.5%) were subtherapeutic, and 31 (21.4%) were supratherapeutic. In total, there were 52 dose modifications made, including 31 (60.8%) dose adjustments, 12 (23.5%) doses held, and 9 (17.6%) doses discontinued. For the 31 dose adjustments, 26 (83.9%) had a level redrawn in the inpatient setting and 17 (65.4%) of those levels were within therapeutic range. The remaining dose adjustments that did not have a level redrawn as an inpatient were due to discharge from hospital and follow-up continuing in the outpatient setting. Outpatient voriconazole levels are outside the scope of this study. Further information on inpatient voriconazole TDM can be found in Table 3.
Voriconazole therapeutic drug monitoring.

SD = standard deviation
Timing of level corresponds to the first trough level drawn after initiation of voriconazole. For example, a value of 7 means that the trough level was drawn prior to the 7th dose of voriconazole
As per the SHS/UHN ASP Voriconazole Monitoring and Adjustment Guidance Document (before the 6th dose if a loading dose was given or on the 6th day of therapy if a loading dose was not given). 12
Adverse effects
There were 23 (41.1%) patients who experienced adverse effects while on voriconazole, 8 (34.8%) of which had supratherapeutic voriconazole levels. Table 4 provides further details on the 8 patients who experienced adverse effects while voriconazole levels were supratherapeutic. Nineteen (33.9%) patients experienced transaminitis, 3 (5.4%) experienced both transaminitis and neurotoxicity, and 1 (1.8%) experienced photopsia. For transaminitis specifically, there were 28 distinct episodes in total with 12 (42.9%) instances of Grade 1 transaminitis; 12 (42.9%) of Grade 2; 3 (10.7%) of Grade 3; and 1 (3.5%) of Grade 4. Of the 23 patients who experienced adverse effects, 5 (21.7%) discontinued voriconazole therapy.
Patients who experienced adverse effects with supratherapeutic levels (n = 8).

MDS = myelodysplastic syndrome. AML = acute myeloid leukemia. DLBCL = diffuse large B-cell lymphoma
Drug interactions
Thirty-nine patients (69.6%) were receiving at least one of the interacting drugs screened (Appendix A). Thirty-four patients (87.2%) received a proton-pump inhibitor (pantoprazole), which can theoretically increase voriconazole levels via CYP2C19 inhibition, and one patient (2.6%) received letermovir, which can decrease voriconazole levels due to CYP2C19 induction. There were two patients (5.1%) who received pantoprazole and letermovir in combination, and one patient (2.6%) who received pantoprazole and fluoxetine, with fluoxetine potentially increasing voriconazole concentrations due to CYP2C19 inhibition. For the 34 patients receiving pantoprazole, 25 (73.5%) had an initial level within therapeutic range, 6 (17.7%) had a supratherapeutic initial level, and 3 (8.8%) had a subtherapeutic initial level. Eighteen (52.9%) of these 34 patients experienced an adverse effect during their hospital admission and 4 (22.2%) of these adverse effects were associated with supratherapeutic levels. For the one patient receiving letermovir, their initial voriconazole level was within therapeutic range and they did not experience an adverse effect during their admission. For the two patients who received both pantoprazole and letermovir, their initial voriconazole levels were within therapeutic range and they did not experience any adverse effects screened for. Finally, for the patient who received both pantoprazole and fluoxetine, their initial voriconazole level was supratherapeutic, but they did not experience an adverse effect. Of note, this patient's voriconazole was discontinued and the reason for this was unclear.
Discussion
The purpose of this study was to understand the current practice of voriconazole TDM in MH patients at PM and determine the proportion of patients with an initial voriconazole trough level within therapeutic range during inpatient admission. Overall, 73.2% of patients had an initial voriconazole level within therapeutic range. The retrospective chart review conducted by Sebaaly and colleagues found that 53.0% of patients had an initial voriconazole level within therapeutic range. 14 For all levels drawn, 62.1% of total levels were within therapeutic range in our study. This is slightly higher than the retrospective cohort study conducted by Yi and colleagues, in which 54% of the total voriconazole levels were within therapeutic range. 13 For the voriconazole levels outside therapeutic range, our study had more supratherapeutic than subtherapeutic levels (21.4% versus 16.5%) whereas the study by Yi and colleagues had more subtherapeutic than supratherapeutic levels (26% versus 20.4%). 13 The authors attributed their higher proportion of subtherapeutic levels to a large number of levels being drawn early, prior to attaining steady state concentrations (37%). 13 Of note, our study defined a serum trough concentration of 1–5 mg/L as the therapeutic range, as opposed to 1–5.5 mg/L in both the aforementioned studies.13,14 These differences can make it difficult to directly compare results between studies. Our local guidance of trough concentrations between 1–5 mg/L is based on the recommendations from the British Society for Medical Mycology that recommends a target voriconazole trough concentration of greater than 1 mg/L for the treatment of IFIs and a voriconazole trough of less than 4–6 mg/L to minimize drug-related toxicities. 11 Although many studies use the upper range of 5.5 mg/L, trough concentrations associated with a greater probability of toxicity varied between studies and included greater than 4 mg/L, greater than 5 mg/L, and greater than 6 mg/L. 11 Our local guidance is also based on Canadian recommendations from Laverdiere and colleagues, which recommend a voriconazole trough concentration with an upper limit of 5 mg/L for efficacy and an upper limit of 5.5 mg/L for safety. 19 After taking both the British and Canadian recommendations into consideration, our institution adopted 5 mg/L as our upper limit.
Timing of trough levels is another important consideration when assessing the appropriateness of TDM. According to the SHS/UHN ASP Voriconazole Monitoring and Adjustment guidance, trough levels should be drawn before the 6th dose if a loading dose is given and on the 6th day if no loading dose is given. 12 In our study, the average timing of a level being drawn was prior to the 7.1 ± 1.8th dose. Since almost all patients (91.1%) in our study received a loading dose, our results indicate that most trough levels (93.1%) were drawn at the appropriate time. Five (8.9%) patients did not receive a loading dose and due to our retrospective study design, it was difficult to determine the reason for this. However, one patient did receive voriconazole for prophylaxis which could explain why no loading dose was given. Reasons for the other four patients could be prescriber preference or prescriber reluctance due to underlying renal or hepatic dysfunction. Another important consideration for timing of voriconazole levels is that at the time of data collection, voriconazole levels were not being processed on Saturdays, Sundays, and statutory holidays at our institution. This may contribute to the variability in scheduling the first voriconazole level. On average, voriconazole loading doses were 5.4 ± 0.7 mg/kg while the manufacturer's recommendation is 6 mg/kg. Additionally, maintenance doses were 3.4 ± 0.6 mg/kg while the manufacturer's recommendation is 4 mg/kg. 4 Voriconazole was administered orally in 78.4% of patients in this study. The slight discrepancies between the mean voriconazole loading and maintenance doses found in this study compared to the manufacturer's recommendation could be attributed to clinician rounding. Voriconazole tablets are only supplied as 50 mg and 200 mg tablets, and at our institution, IV doses are rounded to the nearest 50 mg increment.
For the 41 patients whose initial voriconazole trough levels were within therapeutic range, 20 of them had a trough level redrawn in the inpatient setting, half of which were within therapeutic range. This further highlights the challenges with voriconazole TDM and maintaining patients within therapeutic range, due to its wide intra- and inter-patient variability. Due to our retrospective study design, it was difficult to determine why the remaining 21 patients did not have a trough level redrawn as an inpatient. However, the most likely explanation is these patients had their trough levels redrawn in the outpatient setting. For the 15 patients whose initial voriconazole trough levels were outside of therapeutic range, the majority either required multiple dose adjustments to reach therapeutic range (6, 40.0%) or did not achieve therapeutic range during their admission (7, 46.7%). This highlights the high intra-and inter-patient variability associated with voriconazole and the potential impact that this variability can have when performing voriconazole TDM in clinical practice. Seventeen of the 26 (65.4%) patients whose initial voriconazole level resulted in a dose adjustment had subsequent voriconazole trough levels within therapeutic range. This is encouraging since for these patients, clinician intervention resulted in a positive impact on voriconazole TDM.
There were 23 (41.1%) patients who experienced potential voriconazole-related adverse effects during their hospital admission. This was very similar to the findings by Park and colleagues, where 42% of patients experienced adverse effects. 20 However, only 8 of the 23 patients (34.8%) in our study had supratherapeutic levels. The most common adverse effect that the patients in our study experienced was transaminitis (39.3%), the majority of which were mild (42.9% Grade 1 and 42.9% Grade 2), whereas 27% of patients experienced transaminitis in the study by Park and colleagues. 20 Of note, their study did not report the frequency based on grade of the transaminitis, although they did define transaminitis based on CTCAE v.4. 20 The etiology of transaminitis can be multifactorial with possible contributors being chemotherapy, other hepatotoxic medications, infection, and the hematological malignancy itself. Based on this, it is difficult to attribute the transaminitis solely to voriconazole use. It is also important to note that voriconazole can cause transaminitis even when trough levels are within therapeutic range. 4
All three patients who presented with signs or symptoms of neurotoxicity had supratherapeutic drug levels (5.4, 8.4, and 10.8 mg/L). These findings are consistent with that of Jin and colleagues, who found that higher voriconazole trough levels (greater than or equal to 5.5 mg/L) were correlated with neurotoxicity. 10 There were no patients in our study who developed neurotoxicity with voriconazole levels within therapeutic range. However, neurotoxicity can still occur in these patients, although less likely. Similar to transaminitis, the etiology of neurotoxicity can be multifactorial and since we were limited to retrospectively collecting data from patient charts, it can be difficult to attribute the neurotoxicity solely to voriconazole use.
The main challenge associated with this study is the retrospective nature. The data extracted was obtained from patient charts, which may be incomplete, and made it difficult to obtain information on adverse effects, particularly photopsia and neurotoxicity.
Another important consideration is that our study collected data on inpatient voriconazole levels alone. Voriconazole levels are also followed in the outpatient setting, and we excluded patients that only had outpatient levels drawn. There were also patients in our study that had their first level drawn as an inpatient, but had subsequent levels drawn as an outpatient. This limits our ability to fully capture voriconazole TDM for the entire duration of voriconazole therapy. We excluded outpatient levels in our study because patients are generally not monitored as closely and it introduces other confounding factors, such as adherence to therapy.
An additional limitation of this study is the lack of inferential statistics performed, particularly due to a smaller sample size. Therefore, it is difficult to make inferences regarding the relationships observed in certain subgroups of patients. For example, while all three patients who experienced neurotoxicity had supratherapeutic levels, we cannot conclude that patients who have supratherapeutic levels are more likely to experience neurotoxicity from the results of this study. Our study was a descriptive study, so our goal was to describe the practice of voriconazole TDM at our institution, and not to establish correlations between variables of interest.
There are many opportunities for further research in voriconazole TDM. Future directions include research in voriconazole TDM in the outpatient setting, which would improve the continuity of care from the inpatient to the outpatient setting. Additionally, further investigations in factors that impact voriconazole trough levels and their impact on clinical outcomes would be of interest, such as the routine use of CYP2C19 genotyping to identify poor and rapid metabolizers of voriconazole. Although CYP2C19 polymorphisms have been shown to be a predictor of voriconazole concentrations, its clinical implications have not been well established. 21 If larger-scale studies find a positive impact on clinical outcomes, CYP2C19 genotyping could potentially be used to guide the initial dosing of voriconazole at our institution. Since the completion of this study, a pharmacist medical directive to complete voriconazole TDM and adjust doses has been approved at our institution. With this TDM service, we hope to justify our pursuit of the pharmacist TDM medical directive for voriconazole. Future pharmacy-related research may involve evaluation of the pharmacist-led voriconazole TDM post-implementation of the medical directive.
Conclusion
Overall, 41 (73.2%) patients achieved an initial voriconazole level within therapeutic range and 90 (62.1%) of the total levels drawn were within therapeutic range. For the 41 patients with an initial voriconazole level within therapeutic range, 30 (73.2%) of them remained within therapeutic range for the duration of their inpatient voriconazole therapy. For the 15 patients with an initial voriconazole level outside therapeutic range who remained on voriconazole, 1 to 3 dose adjustments were needed to achieve therapeutic range. Therefore, these findings suggest that the current practice of voriconazole TDM at our institution is yielding largely positive results, but still has room for improvement.
Footnotes
Author contributions
JFlo contributed to literature review, data acquisition and analysis, drafting of the article and revisions, approval for publication, and taking responsibility for the manuscript. JFla contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. SP contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. PD contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. IP contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. LH contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. KY contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Appendix A: Drugs and Potential Effect(s) on Voriconazole Levels Reviewed

| Drug | Effect on voriconazole level? (Increase or Decrease) |
|---|---|
| Rifamycins (rifampin and rifabutin) | Decrease |
| Carbamazepine | Decrease |
| Long-acting barbiturates (phenobarbital) | Decrease |
| Phenytoin | Decrease |
| Letermovir | Decrease |
| Oral contraceptives containing ethynyl estradiol and norethindrone | Increase |
| Proton pump inhibitors
omeprazole (most potent inhibitor out of the PPIs) dexlansoprazole esomeprazole lansoprazole pantoprazole rabeprazole |
Increase |
| Cimetidine | Increase |
| Fluoxetine/fluvoxamine (moderate 2C19 inhibition) | Increase |
| HIV protease inhibitors
ritonavir atazanavir darunavir nelfinavir |
Increase/decrease depending on the agent (ritonavir would decrease – most significant drug interaction out of protease inhibitors) |
| Non-Nucleoside Reverse Transcriptase Inhibitor (i.e., efavirenz)
efavirenz etravirine |
Increase for etravirine Decrease for efavirenz |
| Cobicistat | Increase |
