Abstract
Introduction
Ado-trastuzumab emtansine (T-DM1) is employed in the treatment of patients with HER2-positive breast cancer. The most common side effects are fatigue, diarrhoea, anaemia, transaminase elevation and drug-induced thrombocytopenia. This report describes a patient with metastatic breast cancer who developed drug-induced lupus due to T-DM1.
Case Report
A 54-year-old woman was diagnosed with breast cancer in March 2018. She underwent modified radical mastectomy and axillary lymph node dissection (pT2N1aM0). Following supraclavicular lymph node metastasis in May 2018, she received 8 cycles of docetaxel, trastuzumab, and pertuzumab. In December 2020, the patient presented with axillary and intra-abdominal lymph node metastases, along with bone metastases observed on PET/CT scan. Treatment with T-DM1 and zoledronic acid was initiated. After 18 months on T-DM1, she developed drug-induced lupus. Her symptoms resolved with hydroxychloroquine treatment and discontinuation of T-DM1.
Discussion
Drug-induced lupus is a clinical syndrome that shares similar features with systemic lupus erythematosus (SLE). The majority of patients present with symptoms such as arthralgia and myalgia. Hydralazine and procainamide are high-risk drugs for drug-induced lupus. Symptoms usually develop after months or years of use, but may also develop suddenly. Our patient also received TDM-1 treatment for 18 months. We present a case of TDM-1-associated drug-induced lupus in a patient with metastatic breast cancer. This is the first case of TDM-1-related drug-induced lupus reported in the literature.
Introduction
Ado-trastuzumab emtansine (T-DM1) is an antibody-drug conjugate consisting of the monoclonal antibody trastuzumab linked to the maytansinoid DM1. It has potential antineoplastic activity. The trastuzumab fragment in this conjugate binds to human epidermal growth factor receptor 2 (HER2) on the tumour cell surface and is then internalised into the cell. DM1 containing catabolites are released during lysosomal catabolism and bind to tubulin, disrupting mitotic dynamics and leading to cell death. 1 TDM-1 is approved for adjuvant treatment of HER2-positive metastatic breast cancer patients who have been previously treated, as well as HER2-positive breast cancer patients who have an incomplete pathological response to neoadjuvant trastuzumab and taxane therapy. Treatment with TDM-1 resulted in improvements in both progression-free survival and overall survival. 2 It is important to be aware of the side effects of TDM-1 for its tolerability and safety. The most frequently reported side effects include fatigue, diarrhoea, anaemia, transaminase elevation, and mild to moderate bleeding, which is known to be related to drug-induced thrombocytopenia. Additionally, TDM-1 may cause rheumatological system symptoms, such as arthralgia and myalgia.3,4
This report presents a patient with metastatic breast cancer who developed drug-induced lupus due to TDM-1.
Case report
In March 2018, a 54-year-old woman with hypertension presented with a palpable mass in her left breast and a Figures lymphadenopathy in her left axilla. Tru-Cut biopsy was performed on the palpable mass in the left breast, and the pathology report indicated invasive carcinoma(oestrogen receptor(ER) 80%, progesterone receptor(PR) -, HER2 +++, KI-67 30%) (Figures 1 and 2) The patient underwent modified radical mastectomy and axillary lymph node dissection operations (pT2N1aM0).
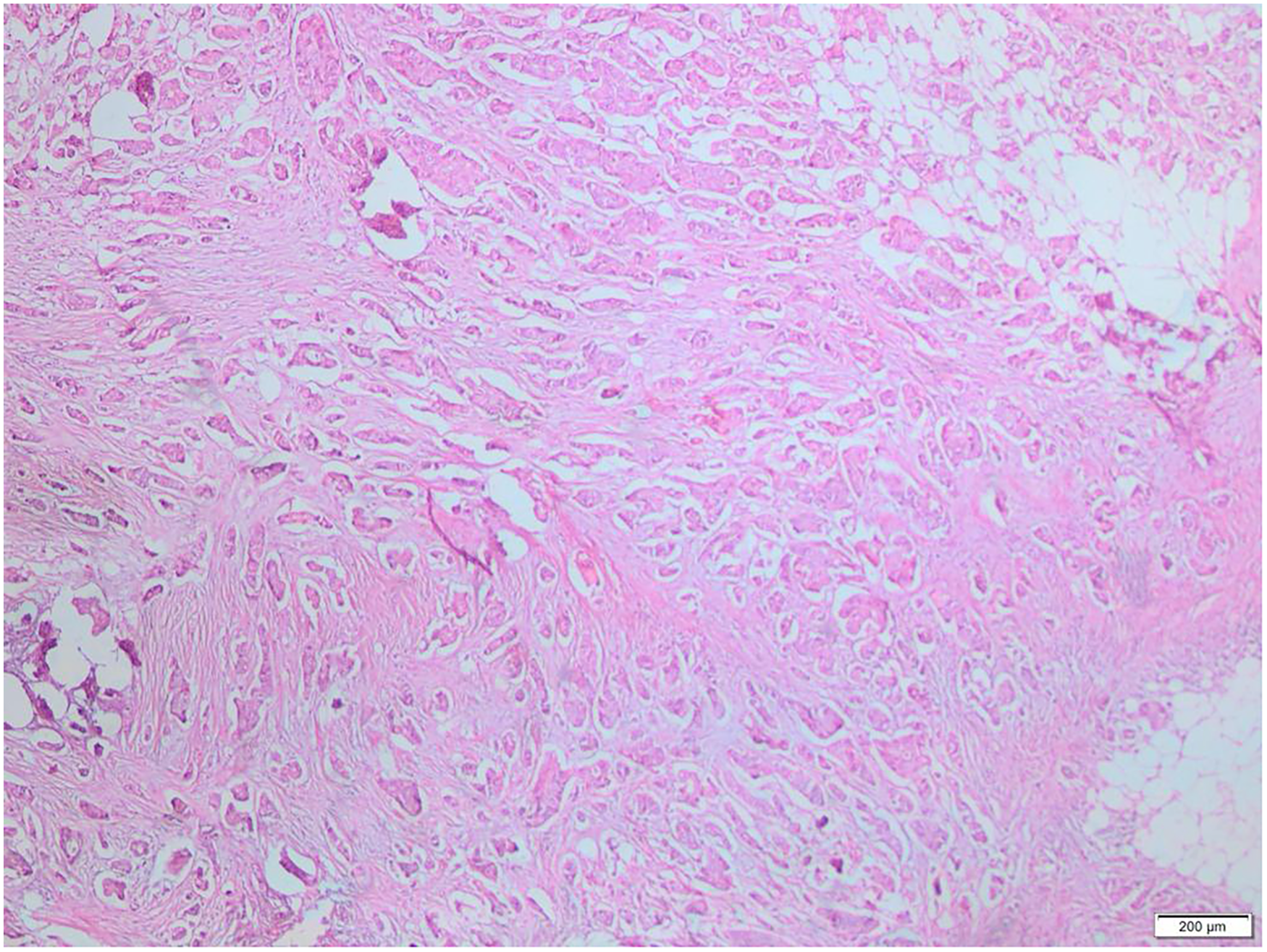
H&E stained sections show tumour infiltrating fibrolipomatous tissues.
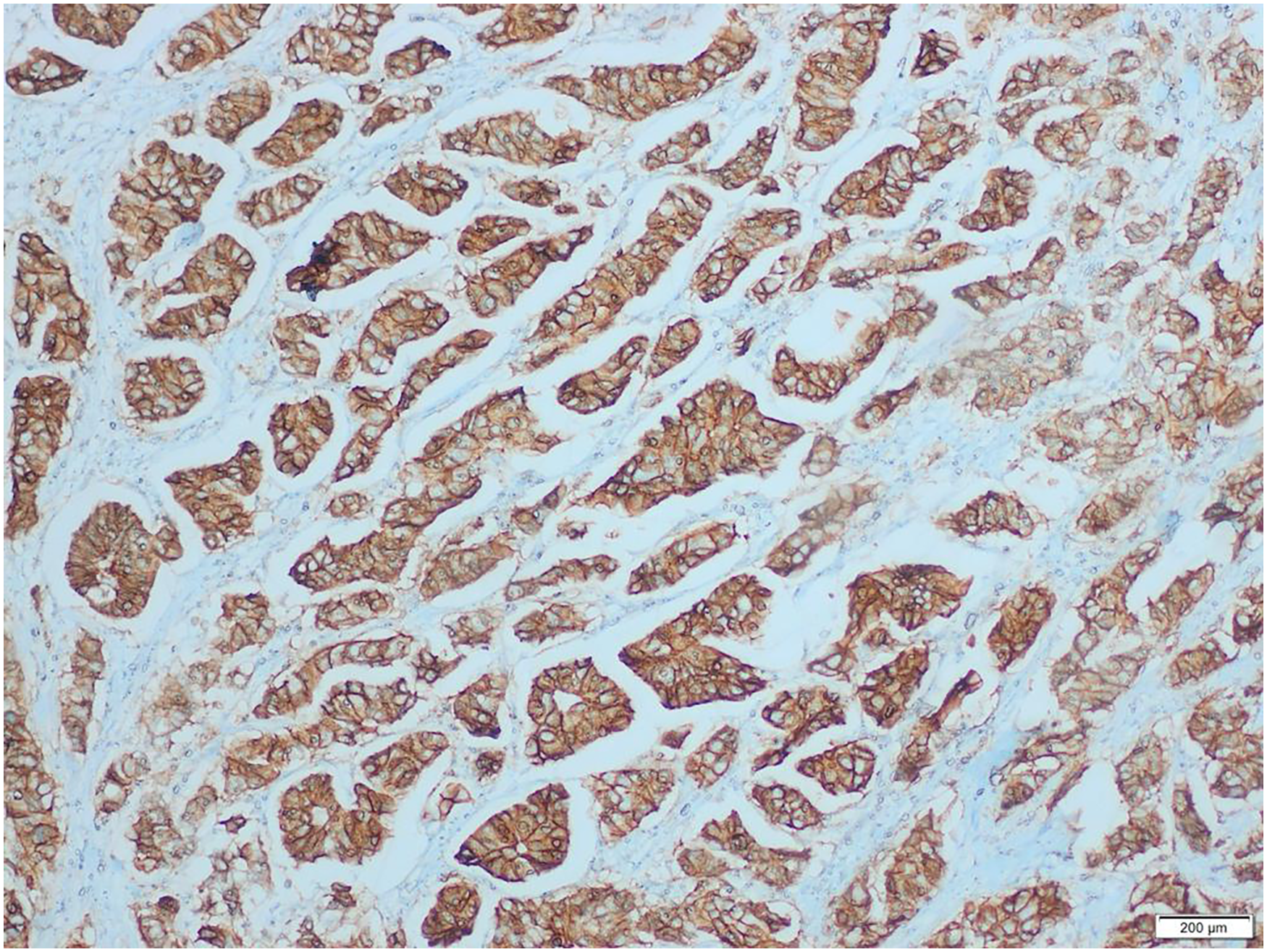
H&E stained sections show tumour infiltrating fibrolipomatous tissues.(her-2 3+).
Postoperative examination revealed left supraclavicular lymphadenopathy. A lymph node biopsy was performed (ER 50%, PR-, HER2+++) in May 2018. Pathology was invasive carcinoma metastasis. 8 cycles of docetaxel, trastuzumab and pertuzumab treatment were given.
Following chemotherapy, maintenance treatment with trastuzumab, pertuzumab, and letrozole was initiated. Adjuvant radiotherapy was performed in March 2019. The patient has axillary and intra-abdominal lymph node metastases as well as bone metastases on PET/CT scan in December 2020. TDM-1 and Zoledronic acid treatment was then initiated. Three months later, PET/CT scan showed marked regression of the lesions and treatment was continued (Figures 3 and 4).
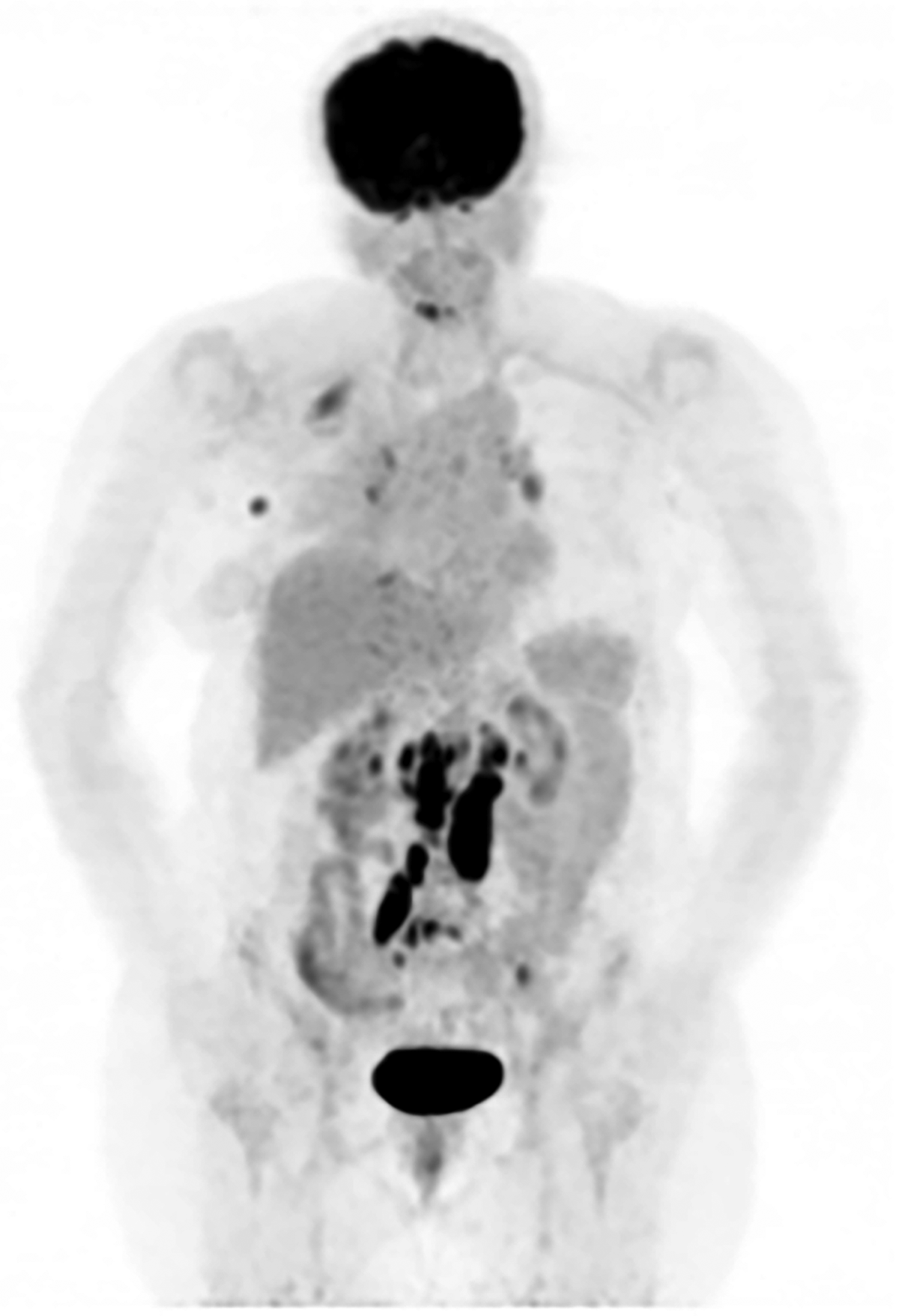
PET/BT before TDM-1.
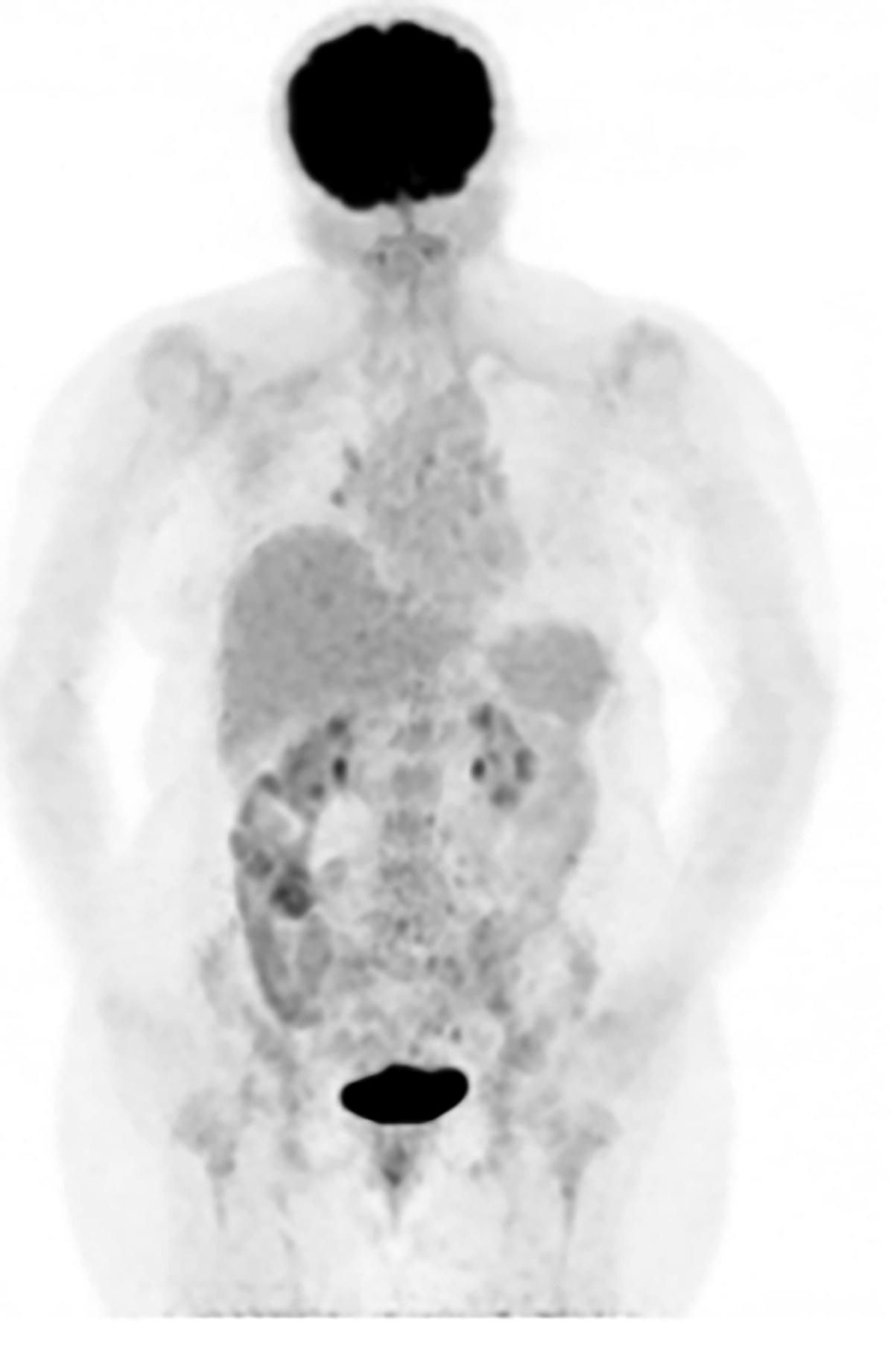
PET/CT after 3 months of TDM-1.
In December 2022, the patient complains of redness of the legs, arthralgia and fatigue and is referred to rheumatology. In July 2023, the patient, who was ANA (antinuclear antibodies) and anti dsDNA positive, was diagnosed with drug-induced lupus and hydroxychloroquine 200 mg twice daily was started (Figure 5). T-DM1 treatment was discontinued after disease progression. The patient's symptoms resolved with hydroxychloroquine treatment and discontinuation of T-DM1 treatment. The patient was diagnosed with lupus due to TDM-1.
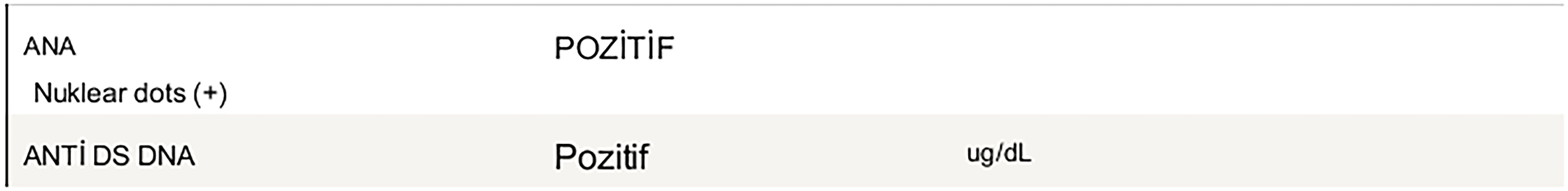
ANA – Anti dsDNA.
Methods
We searched the PubMed and Google Scholar databases from their earliest records until November 28, 2023.
Discussion
Certain drugs can interfere with the autoimmune response by inducing autoantibodies. However, not all patients who develop autoantibodies show signs of autoantibody-associated disease. Drug-induced lupus is a clinical syndrome that shares similar features with systemic lupus erythematosus. (SLE). Elevated serum levels of ANA are often caused by this, but only a small number of patients with these antibodies develop clinical symptoms that suggest drug-induced disease, such as rash, serositis, or arthritis. 5 Drug-induced lupus was first described in 1953 when lupus-like symptoms were observed in patients treated with hydralazine. Procainamide and anticonvulsants have also been found to cause drug-induced lupus. Nowadays, drug-induced lupus is becoming more common with the increasing use of biologic agents. 6 ANA is initially induced by administration of the drug to a susceptible individual. Clinical symptoms typically appear later, with 80% of patients presenting with musculoskeletal symptoms such as arthralgia and myalgia. 7 Symptoms usually develop after months or years of use, but can also develop suddenly. Hydralazine and procainamide are high-risk drugs for drug-induced lupus. Most patients develop drug-induced lupus 3 years or more after starting drug therapy. 8 Our patient also received TDM-1 treatment for 18 months.
We scored our case using the Naranjo Adverse Drug Reactions Probability Scale. The adverse drug reactions are assigned to a probability category from the total score as follows: definite if the total score is 9 or greater, probable for a score of 5–8, possible for a score of 1–4, and finally unlikely for a score of 0. In our case, we calculated a score of 8.
The clinical and serological manifestations of drug-induced lupus are specific to the drug and may differ from the symptoms associated with hydralazine and procainamide. The patient treated with TDM-1 developed a rash, arthralgia, and fatigue. As this is the first reported case of lupus caused by TDM-1, it is unclear whether there is a drug-specific clinical condition.9–11
As TDM-1 becomes more widely used, we will gain greater insight into its effectiveness and potential complications. Clinicians should remain vigilant for rare adverse events and respond accordingly, although it is generally well-tolerated.
Conclusion
We present a case of drug-induced lupus associated with TDM-1 in a patient with metastatic breast cancer. This is the first reported case of TDM-1-related drug-induced lupus in the literature.
Supplemental Material
sj-docx-1-opp-10.1177_10781552241276191 - Supplemental material for Drug induced lupus associated with Trastuzumab emtansine in a patient with metastatic breast cancer
Supplemental material, sj-docx-1-opp-10.1177_10781552241276191 for Drug induced lupus associated with Trastuzumab emtansine in a patient with metastatic breast cancer by Oğuzhan Yıldız, Ali Fuat Gürbüz, Melek Karakurt Eryılmaz, Murat Araz, Talat Aykut, Özlem Şahin, Naciye Hilal Büyükboyacı, Zeliha Çelik and Mehmet Artaç in Journal of Oncology Pharmacy Practice
Footnotes
Author Contribution
Concept — OY and MA. Design — OY and MA. Pathology figüre ZÇ collection: MKE, TA. Supervision — all authors. Data collection and/or processing — all authors. Analysis and/or interpretation — AFG, MA. Literature search — OY, AFG, MKE, TA. Writing — OY, MA. Radiological Imaging provision ÖÇ. Critical reviews — all authors. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
