Abstract
Introduction
Lorlatinib is a potent third-generation anaplastic lymphoma kinase/c-ros oncogene 1 (ALK)/ROS1 oral tyrosine kinase inhibitor that has broad coverage of acquired resistance mutations and is currently indicated for the treatment of adult patients with metastatic non-small cell lung cancer (NSCLC) whose tumors are ALK-positive.
Case report
In this case, we aimed to present the safety and effectiveness of lorlatinib use in a patient diagnosed with ALK-positive metastatic NSCLC who underwent hemodialysis 3 days a week.
Management & Outcome
A 76-year-old female patient has been undergoing regular hemodialysis for about 2 years. A brain magnetic resonance imaging (MRI) was taken due to headache and a mass was detected. She was diagnosed with lung adenocarcinoma as a result of excisional biopsy. Positron emission tomography/ computed tomography (PET/CT) showed a mass in the hilar region of the left lung and multiple lymphadenopathy in the mediastinum. In February 2023, 100 mg lorlatinib was started daily. There was no significant regression in PET-CT and no brain MRI residue during follow-up. The patient has been continuing lorlatinib for approximately 1 year with almost complete response, with no side effects other than hypercholesterolemia.
Discussion
We presented our experience using lorlatinib in a patient with metastatic ALK + NSCLC undergoing hemodialysis. Although the dosage of lorlatinib in hemodialysis patients is still controversial, our case report indicates that 100 mg lorlatinib was safe in this patient.
Introduction
ALK rearrangements are present in ∼3% of non-small cell lung cancer (NSCLC) patients, respectively, providing opportunities for targeted therapeutic intervention. 1 Lorlatinib is a potent third-generation anaplastic lymphoma kinase/c-ros oncogene 1 (ALK)/ROS1 oral tyrosine kinase inhibitor (TKI) that has broad coverage of acquired resistance mutations and is currently indicated for the treatment of adult patients with metastatic NSCLC whose tumors are ALK + as detected by an FDA-approved test. 2 It was recently reported from an interim analysis of the CROWN phase III study that in patients with previously untreated, advanced ALK + NSCLC, progression-free survival (PFS) based on blinded independent central review was signifcantly longer in those who received first-line lorlatinib compared with crizotinib (hazard ratio 0.28; 95% CI 0.19–0.41). 3
The main purpose of presenting this case is to evaluate the safety and efficacy of lorlatinib as first line treatment in ALK + metastatic NSCLC patients on hemodialysis.
Case report
In January 2023, a 76-year-old female non-smoker was admitted to the medical oncology with complaints of headache. The patient has a 15-year history of type 2 diabetes mellitus (DM) and hypertension. Since 2015, she has been followed up in the nephrology clinic with a diagnosis of chronic kidney disease secondary to type 2 DM and has been undergoing hemodialysis three days a week for four hours per session since October 2021. The patient underwent brain magnetic resonance imaging (MRI) which revealed a 4 × 3 centimeter (cm) mass in the left occipital lobe and a 2 × 3 cm mass in the frontal lobe (Figure 1). Additionally, thorax and abdomen computer tomography (CT) imaging showed a 3 × 3.5 cm mass in the left lung hilar region and multiple pathological lymphadenopathies in the mediastinum. The patient underwent brain mass excision. The pathology report indicated mucinous adenocarcinoma metastasis that was positive for TTF-1 and Napsin-A, and compatible with lung metastasis as the primary focus. During the postoperative period, there were no residual lesions found on the brain MRI (Figure 2). The patient received SBRT treatment of 30 Gy to the left parietal and left occipital regions in 5 fractions.
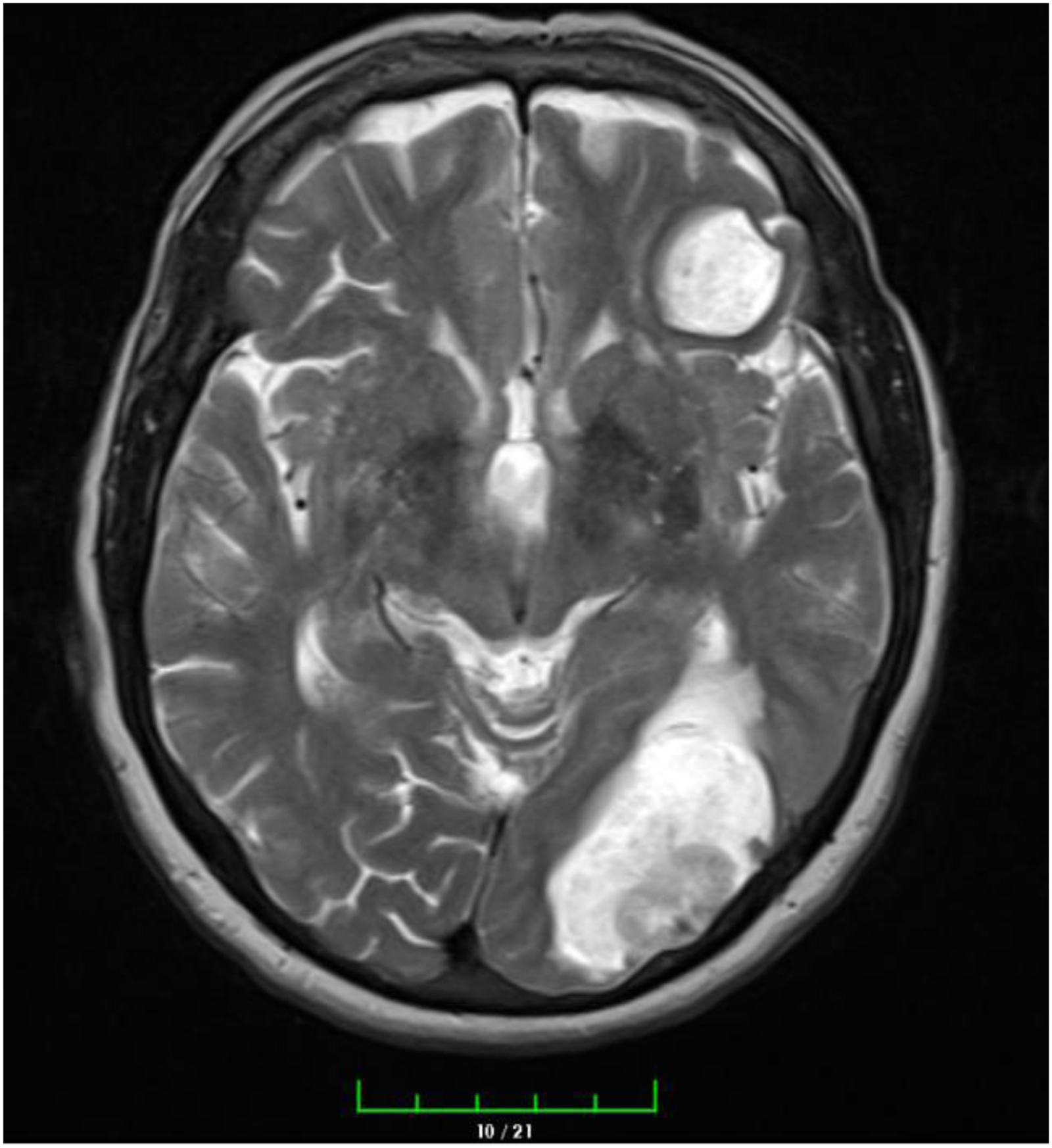
Preop brain MRI.
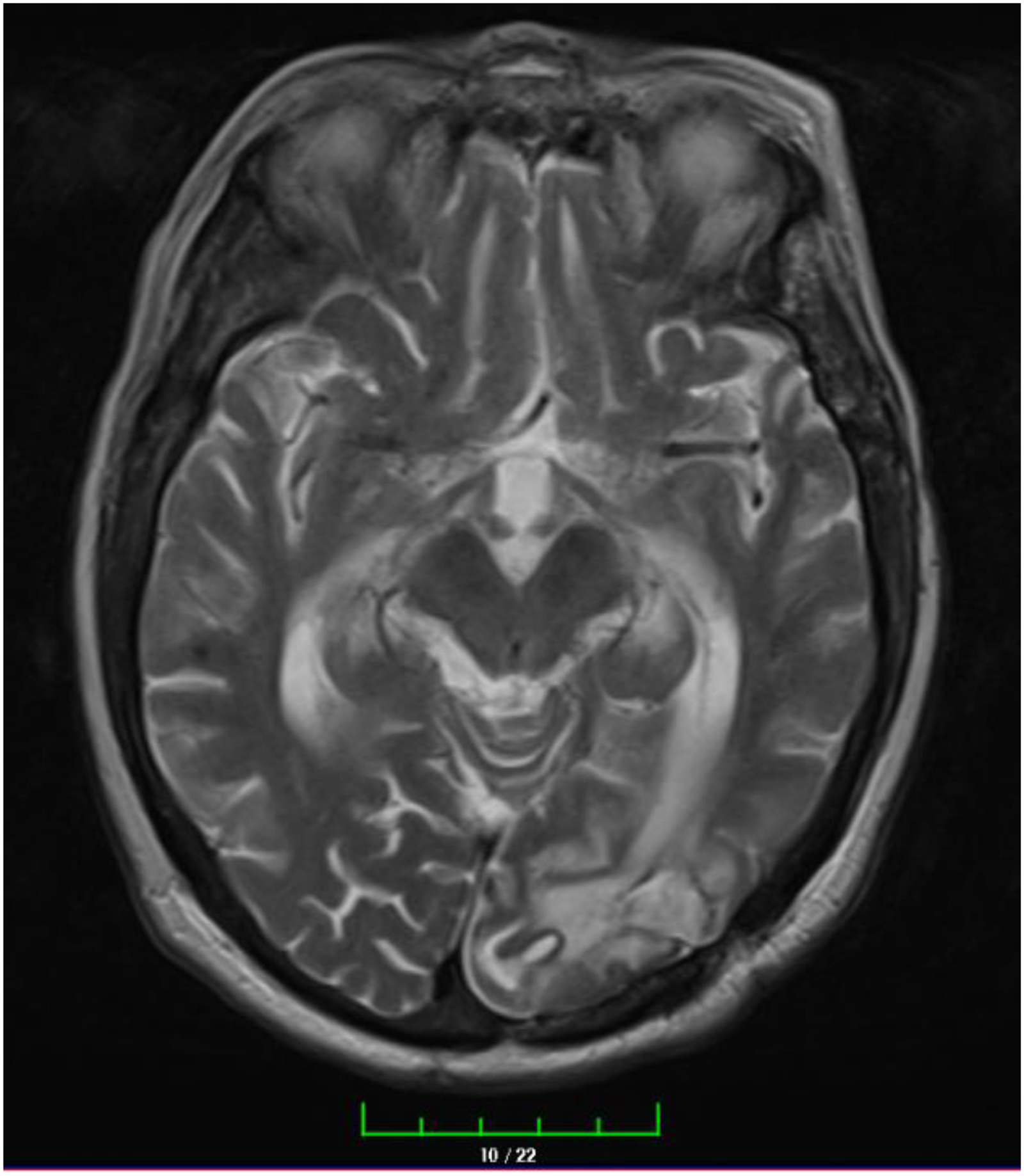
Postop brain MRI.
Positron emission tomography/ computed tomography (PET/CT) with fluorine-18-2- fluoro-2-deoxy-D-glucose (FDG) revealed a 3 × 4 cm mass (SUVmax:14) in the left lung hilar region, multiple metastatic lymphadenopathies, the largest of which was an 18 × 25 mm mass (SUVmax:13) in the mediastinum, and nodular lesions measuring 18 × 10 mm (SUVmax:3.4) in the right adrenal gland and 19 × 13 mm (SUVmax:5.3) in the left adrenal gland. Echocardiography showed an left ventricular ejection fraction of 60%, minimal mitral insufficiency, and an electrocardiogram in sinus rhythm. The ALK rearrangement rate was determined to be 80% using the Floresan in Situ Hibridizasyon (FISH) method (ZytoLight SPEC ALK Dual Colour Break Apart Probe). Additionally, the EGFR and BRAF mutations were negative, and ROS-1 rearrangement was not present. The immunohistochemical PD-L1 Tumour Proportion Score (TPS) was negative. The patient's Eastern Cooperative Oncology Group (ECOG)-performance score (PS) was 2. In February 2023, lorlatinib was initiated at a standard dose of 100 mg orally once daily (QD). The patient underwent physical evaluation every four weeks, including monitoring of electrocardiography (EKG), neurological symptoms, weight, edema, and laboratory values such as lipid profiles, low-density lipoproteins (LDL), high-density lipoproteins (HDL), and triglycerides (TG). After a month, the patient's total cholesterol level was found to be 270 mg/dL, TG level was 244 mg/dL, which were higher than the normal range (total cholesteral 0–200 mg/dL, TG 0–150 mg/dL). As a result, the endocrinology clinic was consulted, and rosuvastatin 10 mg, a statin group antihyperlipidaemic agent, was started orally QD and continued with lorlatinib. At the third-month control, PET-CT showed significant regression. No metastatic lesions on brain MRI and total cholesterol levels were normal. No drug-related side effects were observed during follow-up, including neurologic symptoms, bradycardia, and edema and was regularly on haemodialysis. PET-CT performed in January 2024 was almost compatible with a complete response, brain MRI showed no pathological contrast enhancement and ECOG-PS rose to 1. The patient has been continuing lorlatinib for approximately 1 year with almost complete response, with no side effects other than hypercholesterolemia.
Discussion
Lorlatinib is a novel, highly potent, third-generation macrocyclic ALK/ROS1 TKI that competitively binds to the adenosine triphosphate binding pocket and blocks ALK-dependent oncogenic signaling. Lorlatinib has also been designed to penetrate the blood-brain barrier, in part by minimizing p-glycoprotein-1-mediated efflux, which can lead to poor blood-brain barrier penetration. 4 Lorlatinib is administered orally, with or without food. The recommended dose is 100 mg once daily unless there is disease progression or intolerable toxicity. It is absorbed immediately and the highest plasma concentration is reached in 1–2 h. Pharmacokinetic studies have shown that variables such as age, gender and race do not affect plasma exposure. 5
Advers reactions reported in the phase I/II study of lorlatinib, included hypercholesterolemia, hypertriglyceridemia, central nervous system (CNS) effects (mood disorder, cognitive disorder, and speech disorder), weight gain, edema, peripheral neuropathy, diarrhea, constipation, fatigue, arthralgia, and vision disorder. Of these, the most common side effects associated with lorlatinib are hypercholesterolemia, hypertriglyceridemia, edema, weight gain, and peripheral neuropathy. 6 Hypercholesterolemia and hypertriglyceridemia usually occurred within the first few weeks of treatment (median time to onset, 15 days) and were not common reasons for dose delay (3.4% and 4.7%, respectively) or dose reduction (0.7% and 1.7%, respectively) and did not result in any permanent discontinuations across the study. 7 Our patient developed mild hyperlipidemia in the first month and lipid levels were controlled with antihyperlipidemic treatment. We did not reduce or delay the dose of lorlatinib.
Lorlatinib is metabolized primarily by CYP3A and UGT1A4. Renal excretion of lorlatinib is limited. 8 Based on findings from nonclinical in vitro studies and in vivo metabolic profiling, lorlatinib was found to be extensively metabolized via oxidation and conjugation. In two human absorption, distribution, metabolism and excretion studies, also known as mass balance studies, studies, unchanged lorlatinib accounted for <2% of the dose in the urine, indicating minimal urinary excretion of the parent drug. 9 Therefore, renal impairment would not be expected to have a major impact on the pharmacokinetics or safety of lorlatinib. However, results from a population pharmacokinetic analysis showed that baseline creatinine clearance (CLcr) was a statistically significant clearance (CLcr) was a statistically significant predictor of lorlatinib of the variability in lorlatinib plasma clearance. The median estimated single-dose lorlatinib clearance was 18% and 26% lower in NSCLC patients with mild and moderate renal impairment, respectively, in this analysis. 10
No dose adjustment of lorlatinib is necessary in patients with mild to moderate renal dysfunction. A recently published phase 1 study in adults with mild, moderate, and severe renal ımpairment, the single 100 mg lorlatinib dose administered to all participants was well tolerated. Participants with mild and moderate renal impairment did not experience clinically meaningful increases in lorlatinib exposure; therefore, no lorlatinib dose adjustment is recommended in these populations. Currently, safety data following multiple-dose administration of lorlatinib in participants with severe renal impairment is limited. Thus, there could be a theoretical increased risk of those toxicities typically reported with continuous lorlatinib dosing. Patients with severe renal impairment had 41% higher lorlatinib concentrations due to CLcr decrases, and instead of the 100 mg QD lorlatinib starting dose, a lower starting dose of 75 mg QD is recommended in this population. 11 However, the manufacturer's labeling does not recommend dose adjustment in patients with end-stage renal disease requiring hemodialysis. Because there are no clinical studies. Our patient, who regularly undergoes hemodialysis, has been using 100 mg lorlatinib for approximately 12 months and has not experienced any side effects other than manageable hyperlipidemia.
Conclusion
We presented our experience using lorlatinib in a patient with metastatic ALK + NSCLC undergoing hemodialysis. Our case is the first to describe the experience of lorlatinib in a patient receiving hemodialysis treatment. Although the dosage of lorlatinib in hemodialysis patients is still controversial, our case report indicates that 100 mg lorlatinib was safe in this patient. During lorlatinib treatment, the patient's periodic serum lipid levels, neurological and psychiatric consultation evaluation, edema and weight gain should be closely monitored. Especially in hemodialysis patients, edema may be more serious and increase the need for hemodialysis. Therefore, special attention should be paid to edema.
Footnotes
Author contributions
A.F.G.: conception, data collection, writing, editing and approval of the final draft; M.K.E: conception, data collection, editing and approval of the final draft; O.Y.: study design, writing, editing and approval of the final draft; B.E.K.: study design, data collection; M.Araz.: editing and approval of the final draft; M.Artaç: editing and approval of the final draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
Written informed consents were obtained from patient.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
