Abstract
Objective
This longitudinal study aimed to evaluate the overall efficacy of mouthwashes in oral mucositis pain and mucositis xerostomia in advanced nasopharyngeal carcinoma (NPC) patients undergoing concurrent chemoradiotherapy (CCRT) at different phases throughout treatment.
Methods
A longitudinal study enrolled 79 advanced NPC subjects receiving CCRT. The subjects were interviewed prospectively three times over 7 weeks for pain and xerostomia scores based on the various types of mouthwash used. The median pain score difference and median xerostomia score difference were utilised to determine mouthwash superiority.
Results
Participants completed three interviews, during which 480 instances of mouthwash use were observed throughout different phases of the treatment period. The results showed that the median pain scores between mouthwashes differed significantly, H-Stat(3) = 30.0, 25.7 and 26.0, respectively, with p < 0.001 for all three interviews. The pain score reductions of lidocaine mouthwash (median = 2, interquartile range (IQR) = 3, 2 and 2.75 over the three interviews, respectively) were significantly higher than those of benzydamine and sodium bicarbonate mouthwashes. There were no significant differences between the studied mouthwashes in their xerostomia score reductions.
Conclusions
Lidocaine mouthwash was superior in managing oral mucositis pain at all phases throughout the entire chemoradiotherapy treatment for advanced NPC patients. There was insufficient evidence to determine the preferred mouthwash for treating oral mucositis xerostomia.
Keywords
Introduction
Oral mucositis is characterised as inflammation, erythema and ulceration of the surrounding oral mucosal tissues. 1 Complications, such as mucosal pain, xerostomia, dysphagia, malnutrition and immunocompromised infections, may arise as a consequence of oral mucositis, occasionally leading to the interruption or discontinuation of ongoing cancer treatment. Notably, oral mucositis remains as the most common complication arising from concurrent chemoradiation therapy (CCRT) in patients with nasopharyngeal carcinoma (NPC).
Patients with advanced NPC who have received CCRT typically in the range of 66–70 Gy in doses with a weekly platinum agent over 7 weeks are at a heightened risk of developing oral mucositis. 2 The high incidence of oral mucositis in these patients ranges up to 85%, causing patients severe discomfort and inevitably impairing their daily quality of life.
CCRT triggers pain by eliciting sensitised pro-inflammatory mediators released from injured oral cavity tissues. Additionally, it induces xerostomia by damaging the salivary glands, leading to reduced saliva production and dryness in the mouth. 3 It is believed that xerostomia and pain are correlated. 4 The intricate dynamic complexity underlying the mechanisms leading to the initiation and progression of mucositis is ascribed to many factors, including patient-specific variables, disease characteristics, chemotherapy, radiotherapy and other general factors.5,6
Multiagent combination treatment options to prevent or treat ongoing mucositis, such as cryotherapy, laser therapy, mucosal growth factors and others, are not feasible for most healthcare settings.1,7 Therefore, oral mouthwashes are still the preferred treatment because of their availability and affordability. 8
The Multinational Association of Supportive Care in Cancer (MASCC) and the International Society of Oral Oncology (ISOO) provide the following recommendations: (a) benzydamine mouthwash to prevent oral mucositis pain in patients receiving moderate dose RT (<50 gy); (b) 0.2% morphine mouthwash to treat mucositis pain in patients receiving CCRT; and (3) bland mouth rinses for oral hygiene care and comfort. There is still a lack of evidence to conclusively address the numerous questions about managing mucositis pain in practice. 1 There is a critical need to assess the ongoing effectiveness of other majorly used mouthwash, such as lidocaine mouthwash and sodium bicarbonate, in addressing oral mucositis among patients with advanced NPC undergoing CCRT. Moreover, the potential of these mouthwashes to halt the progression of mucositis severity remains unexplored. Many tertiary centres are employing these mouthwashes as part of routine treatment protocols.6,9
The MASCC suggests using topical mucosal lubricants, salivary substitutes, sugar-free lozenges and chewing gums for oral mucositis xerostomia. 10 Currently, no evidence suggests using any specific mouthwash for mucositis related xerostomia. Although bland rinses are known to provide moist comfort and maintain oral hygiene, the rationale for their specific use in xerostomia treatment needs to be investigated. 11 To date, no study has suggested the most preferred or effective universal mouthwash for mucositis pain, mucositis xerostomia or both.8,12,13
This complexity can be further attributed to the multitude of specific factors involved, such as the type and dose of chemoradiotherapy, interruptions in treatment, extent and grade of mucositis, immune function status, side effects of chemo-radiation, dysphagia, mucosal healing rate, trismus, taste and sensory changes and more. These factors can significantly impact a patient's experience with mouthwashes in managing pain and xerostomia during treatment.14–16
The effectiveness of mouthwashes remains subjective to the patient's perspective as the mucositis level of pain and xerostomia are relative to the individual, which may fluctuate during treatment. The major mouthwashes primarily utilised for oral mucositis include benzydamine, lidocaine and sodium bicarbonate mouthwash.6,9 There is a need to suggest the preferred mouthwash from a patient-centred approach that encompasses symptom reduction at any point in the overall treatment process.
This study aims to evaluate mouthwashes used in treating oral mucositis pain and mucositis xerostomia by determining the overall difference in pain reduction and xerostomia scores before and after each mouthwash at different phases throughout treatment. The goal is to provide healthcare providers with evidence-based insights to select the overall most suitable oral mouthwash for effectively managing oral mucositis.
Methods
Study design
A longitudinal study was done to collect data from a total of 79 subjects who were diagnosed with advanced NPC and underwent chemoradiotherapy in a tertiary oncology hospital from April 2019 to April 2023. Subjects were recruited by clinical pharmacists specialised in oncology through convenience sampling.
Inclusion criteria:
Patients with advanced NPC that have undergone CCRT for a total of 66–70 Gy, administered in 30–35 fractions over 7 weeks, with a weekly platinum agent when clinically fit Patients using at least one or more of the three types of mouthwash (i.e. lidocaine mouthwash, benzydamine mouthwash and sodium bicarbonate mouthwash) in any sequence throughout the treatment of oral mucositis More than 18 years old Patients with incomplete data, non-compliance, or those who passed away. Patients who were prescribed other forms of mouthwash beyond the major three types of mouthwash
Exclusion criteria:
Data collection process and instrument
Subjects with NPC scheduled for CCRT 66–70 Gy/30–35#/7 weeks with a weekly platinum radiosensitiser were subject to routine head and neck preparation protocols, including dental clearance and oral hygiene care. The initiation of each mouthwash (Supplemental Table S1) was based on doctors’ assessments. Each time a mouthwash was prescribed, the subject was instructed by doctors and nurses to use the mouthwash by standard practice (Supplemental Table S2).
Clinical pharmacists in oncology were internally trained to interview subjects to assess their pain and xerostomia score using the validated International Association for the Study of Pain (IASP) Faces Pain Scale-Revised version (FPS-R) 17 and a simplified Modified Xerostomia Score Scale 18 (Supplemental Table S3), respectively. A data collection form was employed, with each mouthwash assessed separately for a pre and post-score. Subjects who were not compliant with the prescribed mouthwash(es) were excluded.
Patients were interviewed during the 14th to 16th fraction (week 3), 24th to 26th fraction (week 5), and 33rd to 35th fraction (week 7) of CCRT via a standardised data collection instrument. Interviews conducted at weeks 3, 5, and 7 were considered separate events. The study data collected at multiple time points throughout the treatment process enabled comprehensive assessments of mouthwash superiority based on the subject's experience. Clinical pharmacists were randomly allocated for each interview to minimise biases of treatment, complications, and interviewers.
Clinical pharmacists ensured the subjects understood that the data collected were exclusively for mucositis symptoms relevant to the local effects on the internal oropharynx area resulting from chemoradiotherapy. Any symptoms not relevant to the area of effect from oral mouthwashes were filtered out. Nystatin oral suspension, prescribed for candidiasis prevention or treatment, was utilised as the placebo arm in this study, with clinical pharmacists integrating it into pain score and xerostomia score evaluations. Nystatin (placebo) was compared directly to other forms of oral mouthwashes
Outcome measures
The median difference scores for mucositis pain and mucositis xerostomia from all three interviews were utilised to evaluate the superiority of each mouthwash for its ability to reduce pain and xerostomia throughout the entire treatment course. The mouthwash with the highest score was deemed more effective.
Statistical analyses
We treated the findings of each observation of mouthwash use from the same subject in each interview as a new finding and applied Kruskal–Wallis and post hoc Dunn's tests to evaluate significant pain reduction difference and xerostomia reduction difference with a significance level of p = 0.05.
Ethical considerations
The study protocol was approved by the Medical Research and Ethics Committee, KKM/NIHSEC/P19-245(7) with study registration number: NMRR-18-3457-45095. The study aim was explained to each subject, and written informed consent was obtained from all the participants.
Results
Initially, 79 subjects were recruited after formal consent was obtained. Over the treatment period, the subject count decreased from 79 to 68 at the first interview, 61 at the second interview and 55 at the third interview. This decline in subject count was due to an incomplete data collection process influenced by various factors, such as failure to meet inclusion criteria, loss of follow-up, detection of non-compliance, COVID-19 infection and patient mortality (Supplemental Figure S1). Data analysis was performed thrice on 68 subjects after the first interview, 61 after the second interview, and 55 after the third interview (Table 1). Among the participants, the majority were males comprising 82.35% at interview 1, 81.97% at interview 2 and 83.64% at interview 3, with average ages of 51.42 years, 51.05 years and 52.15 years, respectively. At the time of cancer diagnosis, all subjects were at least in Stage III or above, with 96.6% of them receiving a radiation dose of 70 Gy. Most subjects underwent intensity-modulated radiotherapy (IMRT), accounting for 52.94% at interview 1, 52.46% at interview 2 and 52.73% at interview 3. None of the subjects experienced interruptions in any form of radiotherapy treatment. Concurrent IV cisplatin chemotherapy was administered weekly to most subjects, with proportions of 88.24% at interview 1, 88.52% at interview 2 and 85.45% at interview 3. However, many experienced interruptions in chemotherapy treatment, resulting in varying cumulative chemotherapy doses. Mucositis among subjects shifted towards higher grades as treatment progressed from the first interview to the third. The average weight loss experienced by subjects steadily increased from 2.56 kg at the first interview to 4.4 kg at the second interview and 5.4 kg at the third interview. Most subjects did not require any form of analgesics at interview 1 (69.11%) and interview 2 (57.38%), but inevitably required systemic analgesics by interview 3 (60%).
Characteristics of subjects by interview.
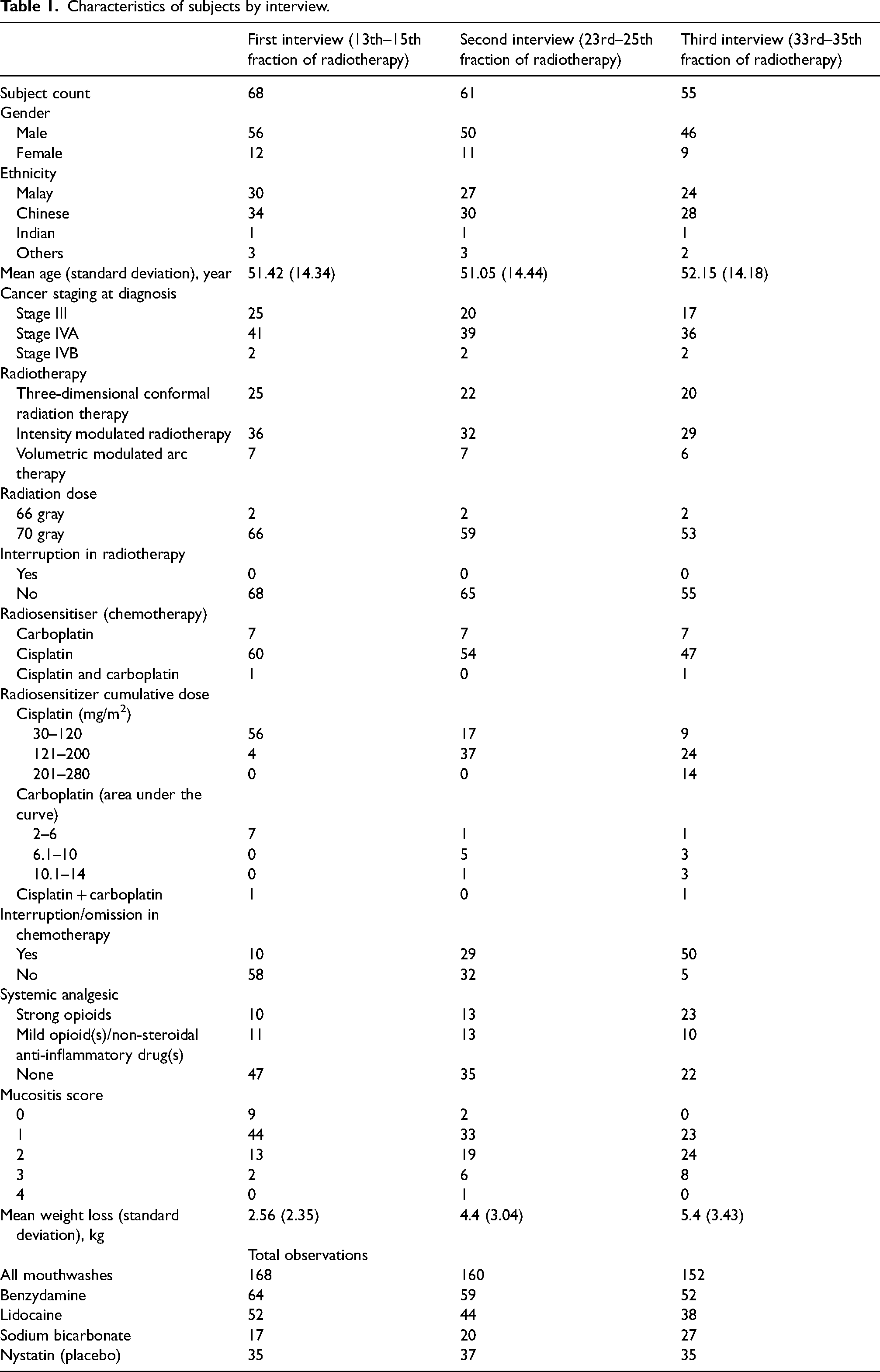
Throughout the course of the three interviews conducted over 7 weeks, a total of 480 instances of mouthwash usage were observed among the subjects. This comprised 168 instances during the first interview, 150 during the second interview and 152 during the third interview, with each mouthwash within the same subject treated as a separate observation.
Effect on pain score reduction
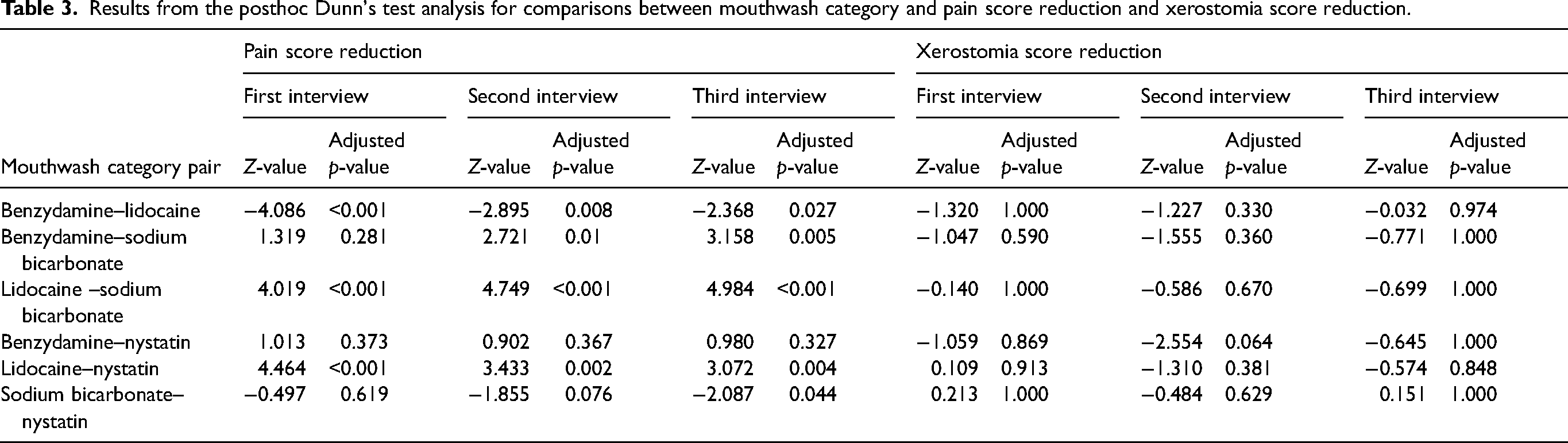
A total of 480 observations (Table 2) were documented to evaluate the efficacy of mouthwash in reducing pain scores across three interview phases. Lidocaine mouthwash demonstrated the greatest median pain score reduction (observation = 134; median = 2, 2 and 2; IQR = 3, 2 and 2.75, respectively, over the three interviews) among all mouthwash categories, as outlined in Table 2. Meanwhile, the median pain score reduction for benzydamine, nystatin and sodium bicarbonate was 0 for all interviews, except for a pain score reduction of 1 for benzydamine during the third interview (Table 2). There existed statistically significant differences in pain score reduction among the various mouthwash categories, H-Stat(3) = 30.0, 25.7 and 26.0, respectively, with p < 0.001 for all three interviews and the post-hoc Dunn test indicating that the median pain score reduction was significantly differing between lidocaine–benzydamine, lidocaine–sodium bicarbonate and lidocaine–nystatin (placebo) across all three interviews, as outlined in Table 3. This illustrates that lidocaine mouthwash was significantly superior to other mouthwash categories in reducing oral mucositis pain. Benzydamine (observation = 175; median = 0, 0 and 1; IQR = 2, 2 and 2, respectively, over the three interviews) demonstrated statistically better pain score reduction than sodium bicarbonate only in interviews 2 and 3 (Table 3). Overall, sodium bicarbonate–nystatin (placebo) [except in the third interview] and benzydamine–nystatin (placebo) were statistically insignificant in pain score reduction.
Mouthwash and their pain and xerostomia score reduction.
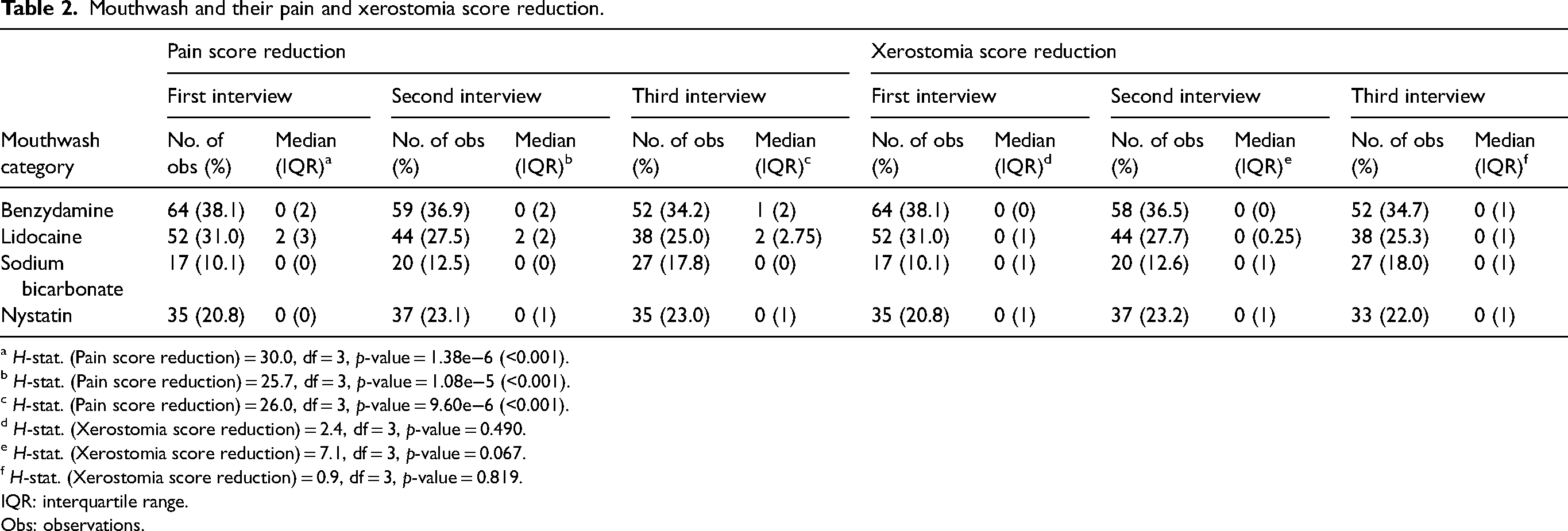
a H-stat. (Pain score reduction) = 30.0, df = 3, p-value = 1.38e−6 (<0.001).
b H-stat. (Pain score reduction) = 25.7, df = 3, p-value = 1.08e−5 (<0.001).
c H-stat. (Pain score reduction) = 26.0, df = 3, p-value = 9.60e−6 (<0.001).
d H-stat. (Xerostomia score reduction) = 2.4, df = 3, p-value = 0.490.
e H-stat. (Xerostomia score reduction) = 7.1, df = 3, p-value = 0.067.
f H-stat. (Xerostomia score reduction) = 0.9, df = 3, p-value = 0.819.
IQR: interquartile range.
Obs: observations.
Results from the posthoc Dunn's test analysis for comparisons between mouthwash category and pain score reduction and xerostomia score reduction.
Interestingly, our findings revealed instances of negative pain score reductions within the population, indicating a worsening of pain following mouthwash use. Remarkably, benzydamine mouthwash exhibited the highest number of observations, with pain worsening, totalling 18 observations. The observed pain score reductions ranged from −1 to −5 (Figure 1).
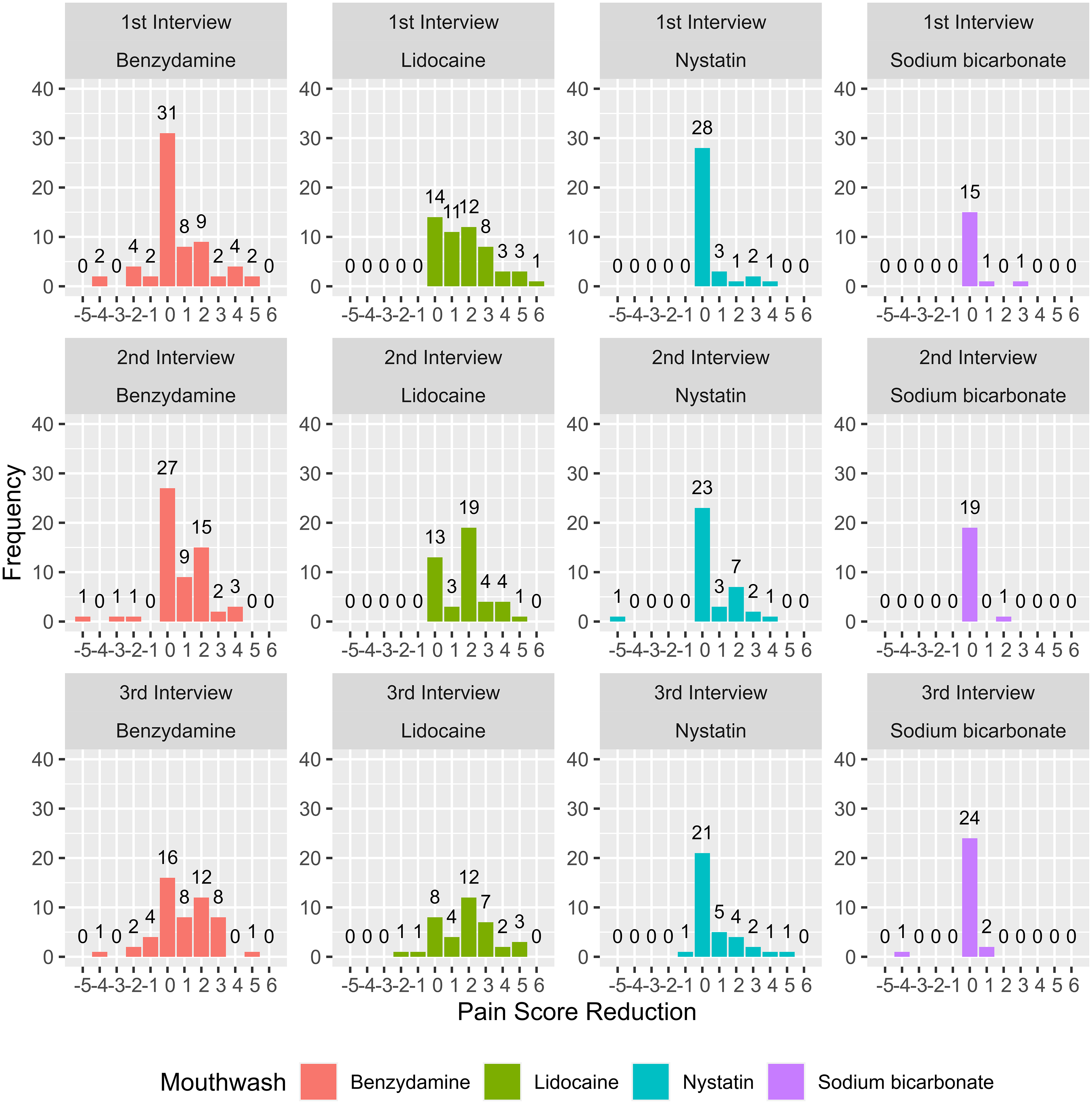
Histogram of pain score reduction by mouthwash. A negative pain score reduction indicates an increase in pain after using mouthwashes.
Effect on xerostomia score reduction
A total of 477 observations (Table 2) were collected to evaluate the effectiveness of mouthwash usage on xerostomia score reduction over three interview periods after excluding three incomplete data points from the analysis. Across all mouthwash categories, the median xerostomia score reduction remained at zero, with IQR ranging from 0 to 1. We found no significant difference in xerostomia score reduction between the various mouthwash categories (p = 0.067, as indicated in Table 3). None of the evaluated mouthwashes showed superior efficacy in treating xerostomia, as depicted in Figure 2.
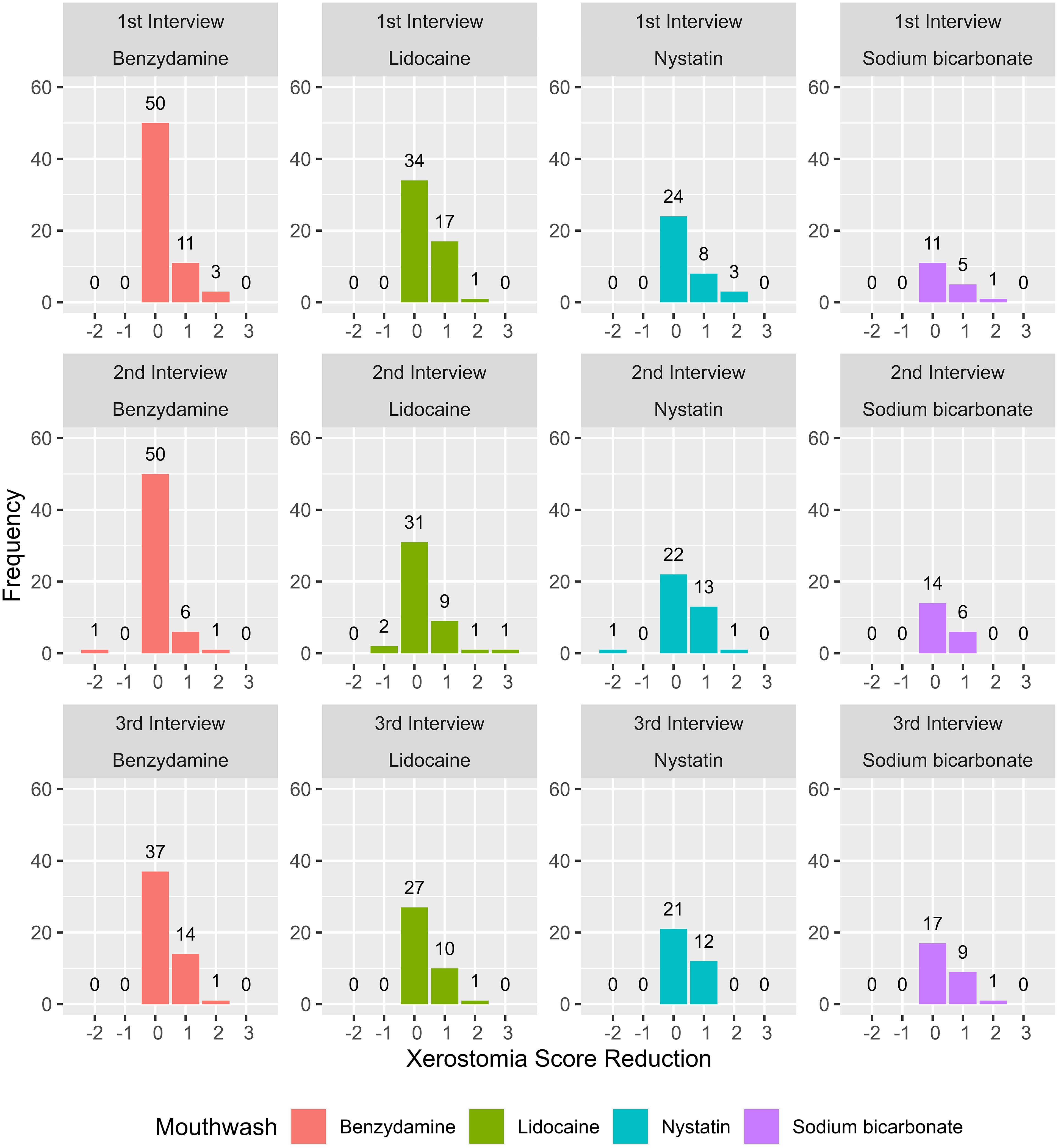
Histogram of xerostomia score reduction by mouthwash. A negative xerostomia score reduction indicates worsening of xerostomia after using mouthwashes.
Duration, dose and frequency of mouthwash usage
The study population revealed that the median dose and frequency of mouthwash used correspond with routine practice from the study site. By contrast, the median duration of mouthwash use was approximately 10 s compared to the routine practice duration of 30 s (Table 4).
Observed median dose, frequency and duration during interviews.

Discussion
We observed several key findings that have important implications for clinical practice and future research.
Effectiveness of mouthwashes
Our results on the pain score difference in Table 3 note that lidocaine mouthwash is suggestive of being the overall most effective in treating oral mucositis pain throughout treatment. Lidocaine 2% had a higher median pain score reduction and proved statistically significant at the first, second and third interviews.
Lidocaine in our study was the clear overall first choice mouthwash for the treatment of mucositis pain at different treatment phases that correlated well to the magnitude of changing confounding factors, as mentioned earlier..7,8,14,19 In contrast to Brown et al., 6 who proposed tapering the mucositis treatment with increasing symptom burden based on mucositis score grades, our results suggest that lidocaine should be the first choice, irrespective of symptom burden in treating oral mucositis. Bergamaschi et al. 9 summarised the use of lidocaine (a local anaesthetic) in combination with other components. Dexamethasone (an anti-inflammatory), in particular, was also effective.7,14 The same study also put topical lidocaine on par with topical morphine mouthwash, but highlights its limited availability in most centres. Further study is needed to establish whether the lidocaine mouthwash regimen, either used alone or in combination, provides greater effectiveness, or if it also serves to prevent the progression of mucositis.
Benzydamine mouthwash use for mucositis prevention is well documented, but its use for mucositis treatment is still under much debate.13,20 In particular, our study, which used benzydamine for both prevention and treatment, was overall less effective than lidocaine. Figure 1 shows that benzydamine gargle had the greatest negative values of pain score difference, indicating subjects had worsening pain when using benzydamine mouthwash at different periods of treatment. Benzydamine, on its own, causes burning and stinging effects, 21 but was likely exponentiated in our study because it contained 10% w/v ethanol. Alcohol-based mouthwashes are known to cause a stinging effect. 11 This supports the rationale of other studies recommending the use of non-alcohol-based mouthwash. This study, however, cannot rule out the effectiveness of other preparation forms of benzydamine without alcohol content that may be effective. Given that some of the data points in Figure 1 indicate strong positive pain reduction scores, and that benzydamine was superior to sodium bicarbonate (Table 3), our study implies potential benefits for benzydamine use in treating oral mucositis. More studies of benzydamine alcohol-free preparations are needed to evaluate further its efficacy for oral mucositis pain treatment.1,12,13,22
Sodium bicarbonate as a bland rinse turned out to be significantly inferior to the lidocaine and benzydamine mouthwash in the treatment of oral mucositis pain, which echoes the findings by Di Fede. 23 However, its use as a bland rinse to prevent mucositis remains uncertain.1,11
Our studies did not reveal a significant difference in any mouthwash used in mucositis xerostomia treatment. Despite sodium bicarbonate being known to keep the mouth moist and reduce dryness, it did not symptomatically prove to make any difference in our study population.11,24 We acknowledge that our Modified Xerostomia Score Scale was likely inadequate in capturing the individual differences concerning xerostomia symptoms. More studies are needed to justify using mouthwash to combat oral mucositis xerostomia.
Duration, dose and frequency of mouthwash usage
Table 2 reveals that the study population complied with the practice standard for dose and frequency of mouthwash but fell short. We theorised that subjects were on a median 10 seconds duration because of the practical challenges of gargling for longer periods. Patients with trismus, an aggravation in pain scores by mouthwash, and the added burden of multiple mouthwashes might find themselves resorting to a shorter gargling duration. More studies are needed to investigate the minimum accepted gargle duration as it varies according to practice settings. 13
Strengths and limitations of study
Strength
The strength of this study lies in its prospective longitudinal design, which carefully considers the mouthwash's performance across different time points throughout the treatment process. This approach accommodates potential ongoing changes during weeks 3, 5 and 7 of chemoradiotherapy.
Limitations
This study did not include an opioid-based agent like morphine mouthwash as a comparison because this mouthwash is unavailable in our setting.
Regarding the standard practice, the dose and frequency were met, but the duration of gargling did not meet the median mark; whether this affects the outcome measures remains uncertain. Practically, patients find it challenging to adhere to a 30-second duration, especially if they have many mouthwashes, start experiencing discomfort for a longer duration or do not feel the benefit of gargling for a longer duration.
It is important to note that the practical application of various mouthwash regimens often deviates from both manufacturers’ recommendations and established practice standards, leading to inconsistency. More research is essential in ascertaining the ideal regimen for each mouthwash, setting a gold standard for effective usage.
We did not include possible ongoing oral mucosal infections and side effects by individual mouthwash other than worsening pain or xerostomia.
Lastly, we used an internally accepted simplified xerostomia score scale, which might not capture a reduction in the xerostomia score. More studies are needed to validate scoring for xerostomia score scales.
Conclusions
Lidocaine mouthwash was superior in managing oral mucositis pain at all phases throughout the entire chemoradiotherapy treatment in advanced NPC. This study suggests that lidocaine mouthwash was the overall preferred choice for treating oral mucositis pain throughout CCRT. Sodium bicarbonate mouthwash and alcohol-based mouthwash were not recommended to treat mucositis pain. There is no evidence to suggest the preferred mouthwash for mucositis xerostomia treatment. This study is unable to recommend mouthwash for the treatment of mucositis xerostomia.
Supplemental Material
sj-docx-1-opp-10.1177_10781552241265933 - Supplemental material for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy
Supplemental material, sj-docx-1-opp-10.1177_10781552241265933 for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy by Rhubain Mageswaran and Zen Yang Ang in Journal of Oncology Pharmacy Practice
Supplemental Material
sj-docx-2-opp-10.1177_10781552241265933 - Supplemental material for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy
Supplemental material, sj-docx-2-opp-10.1177_10781552241265933 for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy by Rhubain Mageswaran and Zen Yang Ang in Journal of Oncology Pharmacy Practice
Supplemental Material
sj-docx-3-opp-10.1177_10781552241265933 - Supplemental material for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy
Supplemental material, sj-docx-3-opp-10.1177_10781552241265933 for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy by Rhubain Mageswaran and Zen Yang Ang in Journal of Oncology Pharmacy Practice
Supplemental Material
sj-docx-4-opp-10.1177_10781552241265933 - Supplemental material for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy
Supplemental material, sj-docx-4-opp-10.1177_10781552241265933 for A longitudinal assessment of major mouthwashes used in alleviating pain and xerostomia among advanced nasopharyngeal carcinoma patients receiving chemoradiotherapy by Rhubain Mageswaran and Zen Yang Ang in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
The authors would like to thank the Director-General of Health Malaysia for his permission to publish this article. We also like to express our gratitude to Chuah Paik Ling, Lenny Lee Cheann, Elaine Kan Mei Ying, Sia Wei Wei, Len Yi Won and Safawati Samsuri for their contribution in the data collection process and to Dr Ros Suzanna Bustamam, the National Head of Services for Radiotherapy and Oncology for the MOH Malaysia, for reviewing the paper prior to publication.
Author contributions
RM conceptualised and design the study, collected data, interpreted the results, and drafted the manuscript; ZYA collected data, performed the analysis, and interpreted the results. RM and ZYA were the co-first authors of this manuscript. All authors reviewed and approved the final version of the manuscript.
Data availability
The data supporting the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was registered in the National Medical Research Register of Malaysia (NMRR ID: NMRR-18-3457-45095), approved by the Medical Research and Ethics Committee, Ministry of Health Malaysia (KKM/NIHSEC/P19-245(7)), and in accordance with the Declaration of Helsinki. All participants provided written informed consent before participating.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
