Abstract
Background
Real-world safety outcomes between the two flat-dose nivolumab regimens demonstrated to be similar in a study of adjuvant nivolumab recipients for melanoma. However, this study was limited by a single oncology patient population, a small sample size, and insufficient study power. The primary objective of this study was to evaluate the incidence of immunotherapy-related adverse effects (irAEs) between nivolumab regimens with differing dosing patterns in various solid tumor patient populations.
Methods
Single-center retrospective cohort study of adult patients with solid tumor malignancies who received nivolumab 240 mg Q2W or 480 mg Q4W, or who were transitioned from 240 mg Q2W to 480 mg Q4W from March 1, 2018 to March 31, 2022 were selected for analysis from an electronic health record generated report. The primary endpoint evaluated was the incidence of irAEs. Secondary endpoints included the incidence of significant irAEs and reasons for treatment discontinuation. These endpoints were compared by univariate analysis between all three cohorts. A multivariate analysis was then conducted for the primary endpoint.
Results
Nivolumab 240 mg Q2W was associated with a statistically significant increase in the incidence of colitis whereas the 480 mg Q4W regimen was associated with a statistically significant increase in the incidence of pruritis. The incidence of irAEs was not different between the three cohorts, while the incidence of significant irAEs was higher in the 240 mg Q2W and 240 mg Q2W to 480 mg Q4W cohorts.
Conclusion
Clinicians ought to be aware of differences in the irAE profiles between nivolumab regimens with differing dosing patterns.
Introduction
Nivolumab, a programed cell death 1 (PD-1) inhibitor, is globally approved for the treatment of several cancer types, including but not limited to, melanoma, genitourinary cancer, nonsmall lung cancer, gastrointestinal cancer, and squamous cell carcinoma of the head and neck.1,2 The linear pharmacokinetics and wide therapeutic index associated with nivolumab encouraged the evaluation of a fixed dose option to reduce dosing errors, facilitate efficient drug preparation, and allow for ease of administration.3–5 Therefore, as a result of population pharmacokinetic modeling and exposure-response analyses demonstrating similar exposure-response, efficacy, and safety profile to the historical weight-based dose of 3 mg/kg in patients with advanced cancers, nivolumab is currently approved by the U.S. Food and Drug Administration as well as the European Medicines Agency for administration at a flat dose of 240 mg every 2 weeks (Q2W) and 480 mg every 4 weeks (Q4W).6–8
Real-world safety outcomes between the two flat dose regimens were demonstrated to be similar in a study of adjuvant nivolumab recipients for the treatment of melanoma. 9 However, the aforementioned study had notable limitations such as a single oncology patient population, small sample size, and insufficient study power. Therefore, the primary objective of this study was to evaluate the incidence of immunotherapy-related adverse effects (irAEs) between nivolumab regimens with differing dosing patterns (240 mg Q2W, 480 mg Q4W, transition from 240 Q2W to 480 mg Q4W) in a variety of solid tumor oncology patient populations. Secondary objectives included assessment of the incidence of significant irAEs, reasons for treatment discontinuation, and impact of prior ipilimumab use or BRAF mutation status on the rate of irAEs.
Methods
A single center retrospective cohort study of adult patients (age ≥ 18 years) who received either nivolumab 240 mg Q2W, 480 mg Q4W, or who were transitioned from 240 mg Q2W to 480 mg Q4W from March 1, 2018 to March 31, 2022 were selected for analysis from an electronic health record (EHR) generated report. Three EHR reports were generated reflecting patients belonging to the aforementioned cohorts. The cohort that transitioned from 240 mg Q2W to 480 mg Q4W were initially identified by searching for patients who had received both nivolumab regimens during a consecutive treatment period. Patients who were actively enrolled in a clinical trial or received investigational drugs and with incomplete medical records were excluded. Study data obtained from the EHR included baseline (defined as within 30 days prior to initiation of nivolumab) patient demographic and clinical characteristics (age, gender, race, Eastern Cooperative Oncology Group performance status (PS), lactate dehydrogenase (LDH), indication, cancer stage, body weight, number of cycles administered, duration of therapy, BRAF mutation status, receipt of ipilimumab within 90 days prior to nivolumab initiation), safety, and reasons for treatment discontinuation. Patients in each cohort were matched on a 1:1:1 basis for the following demographic and clinical characteristics: age, gender, and disease stage. Safety was assessed by the incidence of irAEs and significant irAEs from the date of nivolumab initiation to the end of follow-up or date of death, whichever came first. Significant irAEs were defined as any irAE that led to any of the following outcomes: withheld dose, altered dose and/or schedule, permanent treatment discontinuation, hospitalization, or emergency department visit. Safety endpoints were collected after the switch was made to 480 mg Q4W for the patients who were transitioned from 240 mg Q2W to 480 mg Q4W and no irAEs were reported prior to the switch. All the aforementioned data points were collected and stored in the REDCap web-based application. Approval was granted by the Roswell Park Institutional Review Board (IRB study #00002015) and research was conducted in accordance with the principles outlined in the Declaration of Helsinki. Patient consent was obtained in a manner appropriate for a retrospective cohort study of this magnitude.
The statistical analysis utilized in this study was modeled similarly to the one published by Samlowski et al. 9 The required sample size was calculated to be a minimum of 53 patients per cohort in order to have 80% power at a one-sided alpha of 0.025 to detect a relative difference of 0.5 between irAEs. A 0.5 difference was elected because based on published data, the anticipated irAE rate within 12 months would be approximately 0.59.
Demographic and clinical data were summarized by treatment cohort using the median and range for continuous variables; and frequencies and relative frequencies for categorical variables. Comparisons were made using the Kruskal–Wallis and Pearson chi-square tests, as appropriate. Multivariate logistic models, adjusting for cancer indication (selected a priori), were used to evaluate the association between irAEs and treatment. All model assumptions were verified graphically. Similar analyses were conducted to compare irAE rates by ipilimumab and BRAF status. All analyses were conducted in RStudio v4.0.2 at a significance level of 0.05.
Results
Of the 55 patients who were switched from nivolumab 240 mg Q2W to 480 mg Q4W, 53 met the inclusion criteria for review (Figure 1). Two patients from the aforementioned cohort were excluded due to incomplete medical records (n = 1) and active clinical trial enrollment (n = 1). A matched number (n = 53) of the 240 mg Q2W and 480 mg Q4W cohorts who met the inclusion criteria were randomly selected for review. Demographic and baseline characteristics were similar between the three cohorts except for a number of cycles, duration of therapy, LDH group, indication, and prior ipilimumab administration (Table 1). The 240 mg Q2W to 480 mg Q4W cohort received a higher median number of cycles (n = 17) compared to the 240 mg Q2W cohort (n = 13) and 480 mg Q4W cohort (n = 12). Likewise, the duration of therapy was greater in the 240 mg Q2W to 480 mg Q4W cohort as a result. The median number of 480 mg Q4W cycles administered in the 240 mg Q2W to 480 mg Q4W cohort was nine. A greater number of patients had an LDH above the upper limit of normal (ULN) in the 240 mg Q2W cohort (35.7%) in comparison to the 480 mg Q4W (7.5%) and 240 mg Q2W to 480 mg Q4W cohort (16.9%). The 240 mg Q2W and 480 mg Q4W cohorts had a greater number of patients who had an LDH that was within normal limits. Ipilimumab administration within 90 days prior to nivolumab initiation was greater in the 480 mg Q4W cohort (34.0%) when compared to the 240 mg Q2W (0%) and 240 Q2W to 480 mg Q4W cohort (11.3%). The 240 mg Q2W to 480 mg Q4W and 480 mg Q4W cohorts had a higher number of patients who received nivolumab for treatment of GU cancer when compared to the 240 mg Q2W cohort (20.8% vs 20.8% vs 5.6%). In addition, the 240 mg Q2W cohort had a greater number of patients who received nivolumab for the treatment of lung cancer when compared to the 480 mg Q4W and 240 mg Q2W to 480 mg Q4W cohorts (35.9% vs 9.4% vs 11.3%).
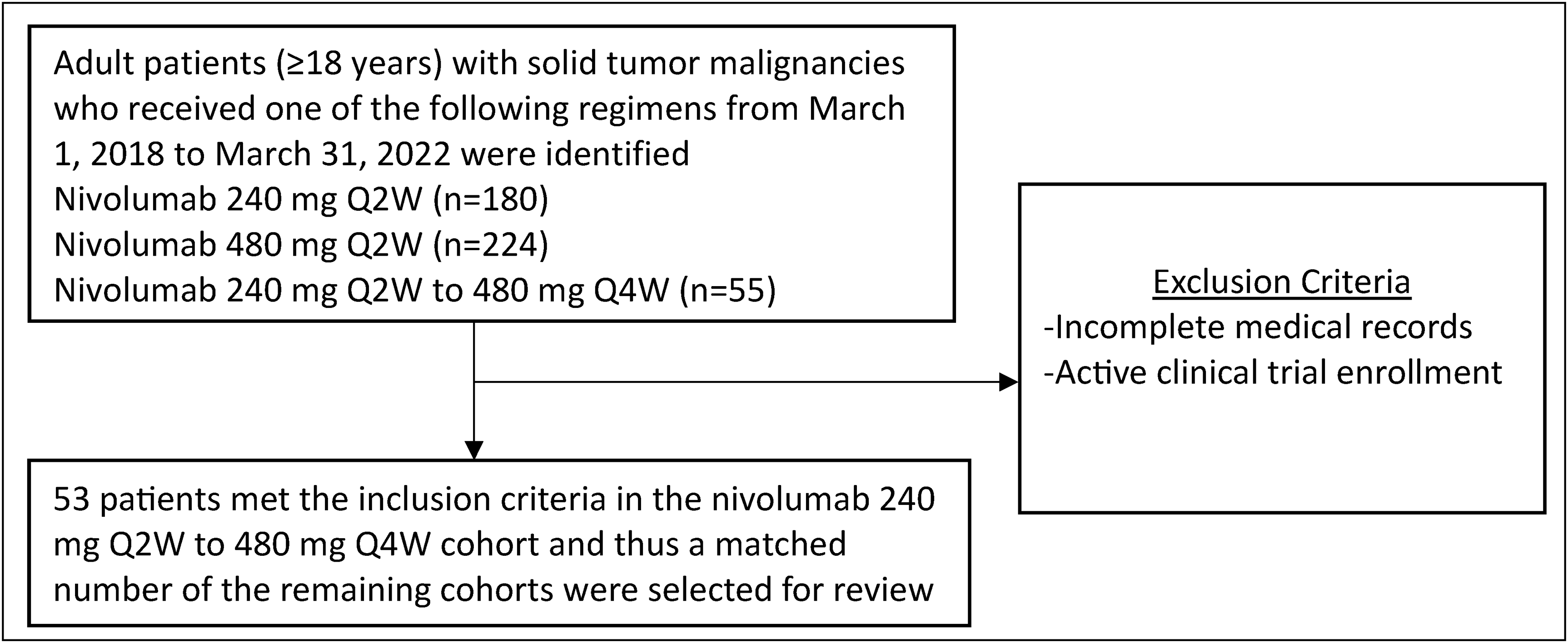
CONSORT diagram.
Baseline demographic and clinical characteristics.
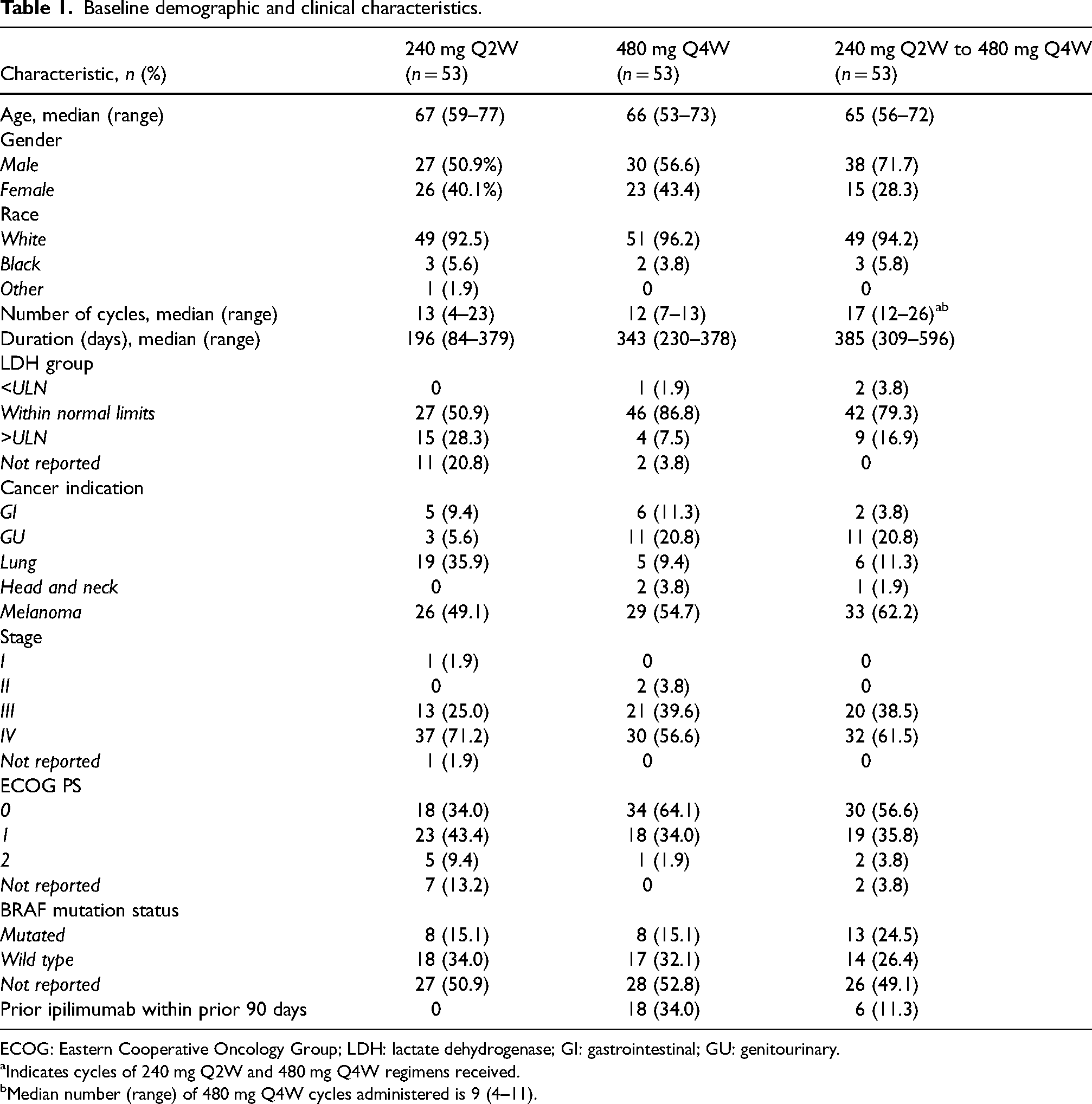
ECOG: Eastern Cooperative Oncology Group; LDH: lactate dehydrogenase; GI: gastrointestinal; GU: genitourinary.
Indicates cycles of 240 mg Q2W and 480 mg Q4W regimens received.
Median number (range) of 480 mg Q4W cycles administered is 9 (4–11).
There was no difference in the incidence of irAEs between the three cohorts except for pruritis and colitis (Table 2). Incidence of pruritis was significantly greater in the 480 mg Q4W cohort in comparison to the 240 mg Q2W and 240 mg Q2W to 480 mg Q4W cohorts (11.3% vs 0% vs 0%; p = .003). Likewise, the incidence of colitis was significantly higher in the 240 mg Q2W cohort when compared to the 480 mg Q4W and 240 mg Q2W to 480 mg Q4W cohorts (9.4% vs 1.9% vs 0%; p = .04). The incidence of any irAE was not found to be significantly different between the three cohorts (p = .56). However, the incidence of significant irAEs was significantly greater in the 240 mg Q2W and 240 mg Q2W to 480 mg Q4W cohorts when compared to the 480 mg Q4W (p = .03). The colitis, pruritis, and significant irAEs remained significantly different (p = .008, p = .004, p = .01) for the aforementioned cohorts on multivariate analysis when correcting for the difference in indications between all three cohorts (Table 2). A subgroup analysis was conducted to determine if a significant difference existed between patients in each cohort who were BRAF mutation positive or had previously received ipilimumab and experienced either any irAE or significant irAE. There was no difference observed in patients who previously received ipilimumab and the incidence of any irAE (p = .07) or significant irAE (p = .58) across all three cohorts. Likewise, no difference was observed in patients who were BRAF mutation-positive and the incidence of any irAE (p = 1.00) or significant irAE (p = .78) across all three cohorts. Premature discontinuation of nivolumab therapy as a result of toxicity was numerically highest in the 240 mg Q2W cohort (n = 13) when compared to the 480 mg Q4W (n = 9) and 240 mg Q2W to 480 mg Q4W cohort (n = 11) (Table 3).
Summary of irAEs and significant irAEs.
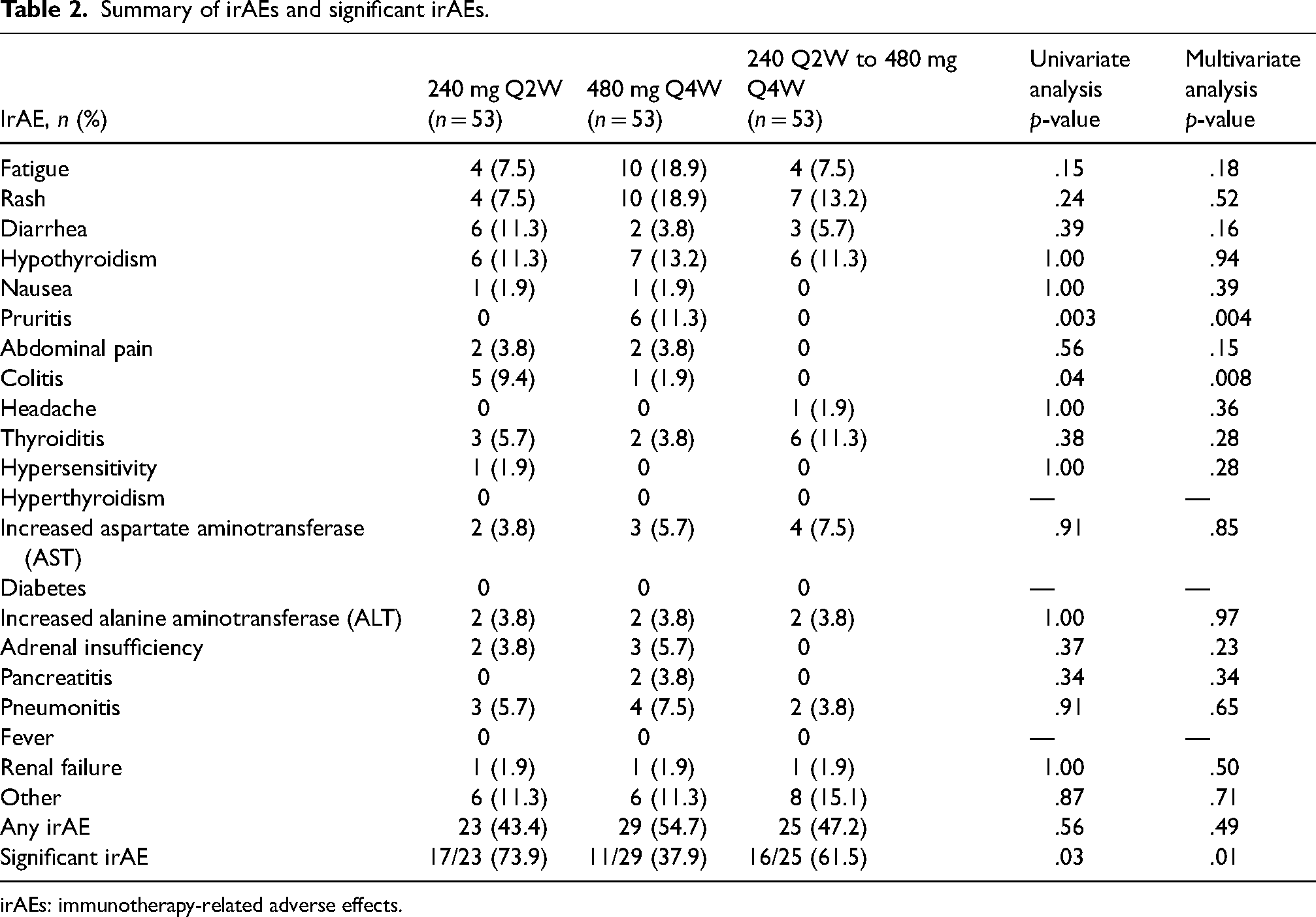
irAEs: immunotherapy-related adverse effects.
Reasons for treatment discontinuation.
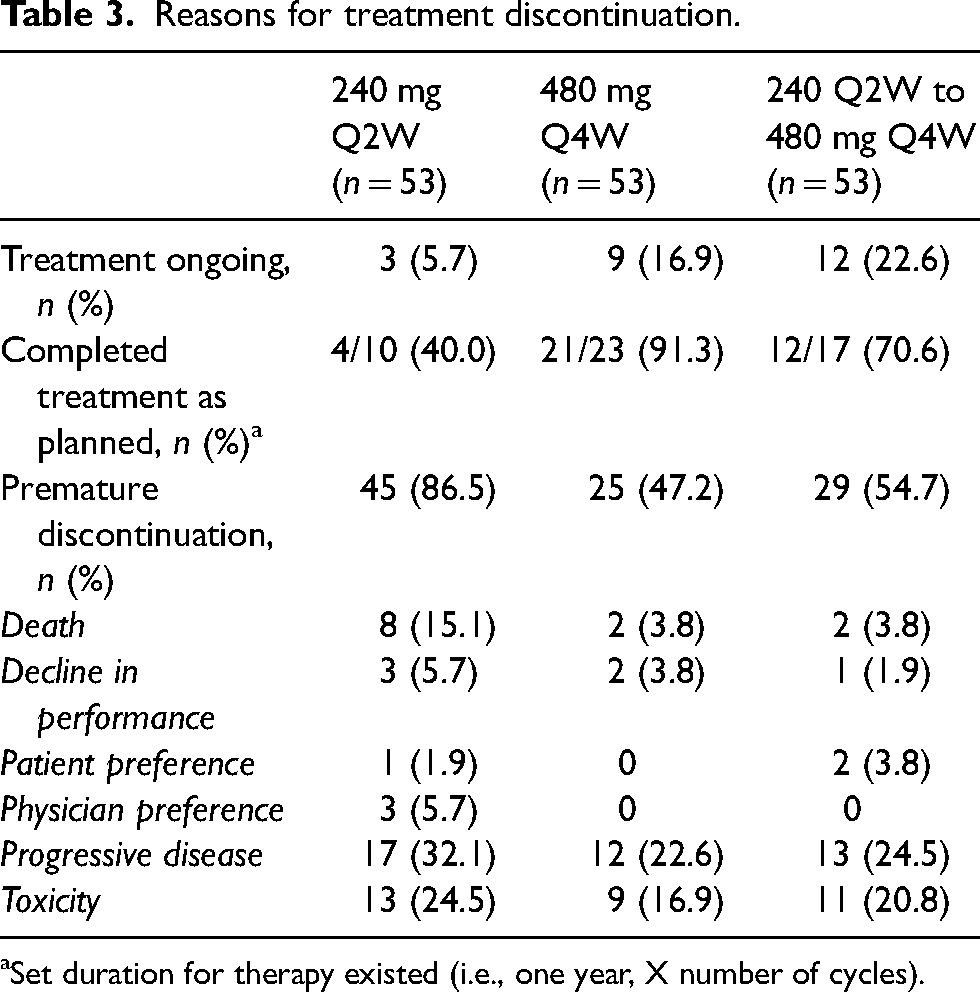
Set duration for therapy existed (i.e., one year, X number of cycles).
Discussion
The 240 mg Q2W and 480 mg Q4W flat dose regimens demonstrated to have a similar safety profile to the historical weight-based dose of 3 mg/kg in clinical studies.6–8 However, retrospective data of the safety profile between the different flat dose regimens in real-world patients receiving nivolumab for a variety of solid tumor cancers remains limited. The significantly higher incidence of pruritis in the 480 mg Q4W cohort and colitis in the 240 mg Q2W cohort was a finding that has yet to be reported in clinical studies. This finding was maintained even when correcting for the heterogeneity in indications for treatment with a multivariate analysis amongst the cohorts. In addition, there was a higher incidence of significant irAEs among the 240 mg Q2W and 240 mg Q2W to 480 mg Q4W cohorts when compared to the 480 mg Q4W cohort, which was maintained on a multivariate analysis similar to the aforementioned primary endpoints. The absence of colitis in the nivolumab 240 mg Q2W to 480 mg Q4W cohort in comparison to the 240 mg Q2W cohort may indicate that colitis as an irAE is more likely to occur from frequent lower dose rather than from less frequent higher dose nivolumab exposure. This hypothesis is supported by the higher median number (n = 13) of cycles administered to the 240 mg Q2W cohort when compared to the 240 mg Q2W to 480 mg Q4W cohort (median number of cycles for both doses = 17; median for just the 480 mg Q4W doses = 9) indicating overall less exposure to the 240 mg Q2W dose prior to switching. Although the study was not powered to detect a difference between the cohorts with regard to significant irAEs, it is still worth noting and potentially exploring as a primary outcome in a future study. In contrast, the incidence of any irAEs was not significantly different. This discordance between the rates of significant irAEs and any irAEs highlights an interesting paradigm: a similar incidence of irAEs may exist between different nivolumab regimens but of those irAEs, some may only be significant with certain flat dose regimens. This idea may also extend to the greater incidence of pruritis and colitis irAEs observed with certain nivolumab regimens as previously mentioned. Patients who harbor BRAF mutations or had received ipilimumab within 90 days prior to nivolumab initiation did not demonstrate a higher incidence of any irAEs or significant irAEs among all three cohorts. Even though this finding was based on a subgroup analysis that was insufficiently powered, patients who had received ipilimumab prior to maintenance nivolumab did not appear to experience a higher incidence of irAEs or significant irAEs regardless of the flat dose regimen selected. Another patient population-specific consideration with regard to BRAF mutation status not impacting safety outcomes with differing nivolumab regimens is patients with cutaneous melanomas. This finding is reassuring because the DREAMSeq trial demonstrated superior two-year overall survival rates when sequencing ipilimumab and nivolumab prior to BRAF/mitogen activated protein kinase (MEK) inhibitors for treatment naïve patients with BRAFV600 mutation-positive metastatic melanoma. 10 Although this subgroup analysis was insufficiently powered, the results suggest that nivolumab maintenance therapy in melanoma patients with BRAF mutations or after receipt of ipilimumab is not likely to precipitate a higher incidence of any irAEs or significant irAEs. Furthermore, toxicity as a reason for treatment discontinuation was numerically greatest in the 240 mg Q2W cohort in comparison to the 480 mg Q4W and the 240 mg Q2W to 480 mg Q4W cohorts, although this difference was not statistically significant. The 240 mg Q2W to 480 mg Q4W cohort did have a comparable number of discontinuations due to toxicity. The likely rationale for this regarding both cohorts is the higher incidence of significant irAEs observed with these two cohorts, which may have caused patients to discontinue treatment.
There have been two previous studies which have evaluated safety outcomes with differing nivolumab regimens. Morimoto, et al. published a single center study which assessed safety outcomes between nine patients who were initiated on the extended-interval dosing of nivolumab or pembrolizumab versus 60 patients who were switched to extended-interval dosing. 11 These patients were receiving nivolumab treatment for solid tumor cancers. The rate of irAEs was reduced from 51.7% (the rate prior to switch) to 21.7% in patients who were switched to extended dosing. 11 However, the incidence of irAEs was noted to be high in the early phase of extended-dose switching. 11 Nevertheless, the incidence of irAEs was lower in patients who were switched to extended-interval dosing (21.7%) versus those who received extended-interval dosing for the first time (33.3%). 11 This study was limited by small sample size, disproportionate cohort size (60 vs 9), and insufficiently powered to identify a difference in the incidence of irAEs between the two cohorts. In addition, the safety outcomes were evaluated for both nivolumab and pembrolizumab, which may confound safety outcomes if they differ between certain PD-1 inhibitors, as noted in our study. Also, the lack of difference between all three cohorts with regard to an incidence rate of any irAEs in our study contradicts the findings of the Miromoto et al. study. The lack of difference in incidence of any irAEs or significant irAEs between nivolumab regimens with differing dosing patterns (240 mg Q2W vs 480 mg Q4W vs 240 Q2W to 480 mg Q4W vs 3 mg/kg Q3W) was also demonstrated in recipients of adjuvant nivolumab for melanoma treatment. 9 Our study design was modeled similarly to the design in the aforementioned Samlowski et al. trial. In addition, the Samlowski et al. study reported the 240 mg Q2W to 480 mg Q4W cohort had the highest number (n = 8) of patients who had toxicity as a reason for premature discontinuation of treatment. This differs from our study since the 240 mg Q2W cohort experienced the highest incidence (numerically) of toxicity as a reason for treatment discontinuation, although this difference was not statistically significant. As previously mentioned, the Samlowski et al. study was restricted to a single oncology patient population, had a small sample size, and was not powered to detect a difference in the incidence of irAEs between the various nivolumab cohorts. These limitations were augmented in our study and are likely why we were able to identify a significant difference in the incidence of certain irAEs as well as significant irAEs. Limitations of this study encompass the previously mentioned retrospective and single-center nature as well as variability in EHR documentation among providers. Additionally, the small sample size was a limitation, primarily due to the low number of patients who transitioned from nivolumab 240 mg Q2W to 480 mg Q4W over the four-year period analyzed. Although this study has a larger sample size than previous studies, it is still not sufficiently powered for some subgroup analyses. Future studies with a similar study design and, if possible, larger sample sizes than included in this study would be ideal to confirm the findings and determine if the same results are replicated. The applicability of the results from this study into clinical practice remains limited at this time as the body of evidence regarding the difference in irAE rates between dosing patterns for nivolumab specifically is continuously evolving.
Conclusion
Evaluation of safety outcomes for different nivolumab regimens has been limited to studies with small sample sizes, insufficient study power, and a single oncology patient population. Therefore, this study was able to augment those limitations and utilize a previously published study design but expand the patient population to encompass numerous solid tumor malignancies and power the study to identify a difference in irAEs between various nivolumab dosing schemes. These study-related attributes likely allowed for the identification of a significantly greater incidence of pruritis in the 480 mg Q4W cohort and colitis in the 240 mg Q2W cohort. Although the incidence of any irAEs was not found to be different between the three cohorts, additional monitoring for signs and symptoms of colitis is necessary in patients who receive nivolumab 240 mg Q2W. Likewise, close monitoring for significant irAEs may be required for patients receiving nivolumab 240 mg Q2W and those who are transitioned from the 240 to the 480 mg Q4W regimen.
Footnotes
Author contribution
JE, IP, LH, and GR contributed to the preliminary literature review, study design, and manuscript development. JE and BC performed data collection. KA and KW assisted with the development of the statistical analysis plan as well as conducted the statistical analysis of the collected data. The final version of the manuscript was reviewed and approved by all authors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
