Abstract
Background
Cancer care is posing immense challenges to healthcare systems globally. Advances in screening, monitoring, and treating cancer improved patient outcomes and survival rates yet amplified the disease burden. Multiple barriers might impede early access to innovative therapies. We thoroughly examined the current challenges in oncology medication access in Saudi Arabia and provided consensus recommendations to revitalize the process.
Methods
A focus group discussion was conducted. Expert healthcare providers (pharmacists and physicians) were invited to participate based on prespecified criteria. The research team conducted a qualitative analysis of the discussion to identify themes and formulate recommendations.
Results
Fourteen experts were equally distributed into two groups, limiting the number in each group to 7. Pharmacists were 12 (∼86%), and physicians were 2 (∼14%). Ten were practicing in governmental hospitals, four representing different sectors; regulatory bodies, including Ministry of Health, National Unified Procurement Company, and Saudi Food and Drug Authority. Five themes were identified: national cancer burden, local data availability, pharmacoeconomic evaluation, patients reported outcomes, administration, and procurement. Consensus recommendations were formulated to optimize the formulary management process, enabling informed decision-making and facilitating early medication access for cancer patients.
Conclusions
The formulary management process can be enhanced by addressing the national cancer burden, promoting local data availability, conducting pharmacoeconomic evaluations, focusing on patient outcomes, and improving administration and procurement procedures. Implementing these recommendations can improve access to oncology medications and improve patient care outcomes in Saudi Arabia.
Introduction
Cancer care is an impending global challenge to healthcare systems. In 2020, the incidences of death and new cases globally were approximately 10 and 19.3 million, respectively. 1 In Saudi Arabia, there has been an acceleration in the trajectory of cancer cases over the last 5 years, with over a 50% increase since 2016.2,3 Several factors might contribute to the rise in incidences, including enhanced access to diagnostics and advances in national reporting and documentation systems.
The rise in cancer burden has adversely impacted national health budgets worldwide.4–6 Cancer care expenditures vary widely due to population differences, healthcare delivery systems, and mortality rates. Recent advances in cancer pharmacotherapy, particularly high-cost targeted therapies, contributed to the high economic burden of disease. In 2021, a study evaluated the economic burden of managing breast cancer in Saudi Arabia. 7 Most expenditures were related to medications, with targeted therapy representing 67%.
Access to oncology medications is a complex issue influenced by economic, geographical, policy, and patient-centric factors. For decision-makers, early access to oncology medications is impeded by the quality of the available evidence. Unfortunately, newly approved costly targeted therapies lack long-term efficacy and safety data in a larger population.8–10 Additionally, patient-reported outcomes are often not studied, and adherence to The Consolidated Standards of Reporting Trials guidelines was not optimal. 11 The high cost of such medications and the lack of long-term efficacy and safety data delay their access, especially at the time of new market entry, and ultimately affect the healthcare payer's decision-making. 12
On an institutional level, delay in access can result from a poorly structured Formulary Management System (FMS). 13 Establishing an appropriate FMS is crucial to ensure proper therapies and medication utilization policies and procedures are well-organized and consistent within the healthcare institution. It also maintains a comprehensive list of the needed oncology medications for cancer patients based on multiple factors, including the pharmacological class, indications, cost-effectiveness of therapies, and supply-chain issues. 14 Another major element of medication access is the procurement process of formulary and non-formulary oncology medications. Procurement strategies should focus on meeting the demands and providing an estimated supply through procurement companies such as the National Unified Procurement Company (NUPCO), which is responsible for medication acquisition and distribution in Saudi Arabia.
Addressing access barriers becomes paramount for more effective and equitable cancer care. Ongoing research and concerted efforts are essential to ensure that innovative oncology medications reach all those in need. To tackle these challenges, a group of experts representing different healthcare sectors collaborated to address and identify significant obstacles to oncology medication access in Saudi Arabia and provide national consensus recommendations to improve patient outcomes.
Methods
Participants
Pharmacists meeting predefined criteria were invited to participate in a focused group discussion. The criteria included (a) current involvement in oncology or oncology-related roles, (b) years of experience, (c) leadership roles, and (d) current employment in governmental institutions, hospitals, or procurement companies.
Study design and procedure
A focus group discussion was conducted with a group of experts. The facilitators guided the group throughout the sessions by asking predefined questions to guide the discussion. The focused group meeting was conducted for 6 h in a private meeting room. The sessions consisted of 1-h discussion for each theme with breaks in between. Each group was limited to 7 members in addition to the facilitator. The sessions started with an introduction of all the participants, followed by an explanation of the purpose and procedure of the focus group discussion. Group discussions were conducted in accordance with guidelines on focused group research. 15
Data analysis
The sessions were audio-recorded after obtaining written permission obtained from all the participants. The research team transcribed the discussion to identify the challenges and analyze the recommendations. The analysis followed the F Rabiee eight-criteria framework. 16 The research team reviewed the transcripts, identified themes, and formulated recommendations. An email was sent to the experts to review the analysis and provide feedback.
Results
Participants
A total of (14) experts agreed to participate, representing different sectors. The participants were equally distributed into two groups, limiting the number in each group to 7. Pharmacists were 12 (∼86%), and physicians were 2 (∼14%). Representing different sectors, 10 were practicing in governmental hospitals, and four were working in regulatory bodies, including Ministry of Health (MOH), NUPCO, and Saudi Food and Drug Authority (SFDA). The following themes were identified by the study team and confirmed by the experts: national cancer burden, local data availability, pharmacoeconomic evaluation, patients reported outcomes, administration, and procurement.
Analysis of the current challenges
National cancer burden
The cancer burden in Saudi Arabia is perceived to be low, based on the currently available data from the Saudi Cancer Registry, with a total number of 17,631 newly diagnosed cases. This perception is partially due to several factors that contribute to the inadequacy of reflective statistics, such as limited efficient local registries, population-based clinical trials, and less engagement in clinical research at many institutions within Saudi Arabia. As a result, the lack of accurate, locally generated data is a major challenge that can impact the proper estimation of disease burden and the need for a particular targeted treatment. The estimated numbers are historically based on patients’ duplication due to several institutional visits, which is considered a predicament that potentially gives false epidemiological data. Research is warranted to minimize the gap and uncover the unknown information related to the effect of new medications on the Saudi population. The sporadic adoption of molecular and genetic testing was identified as another major obstacle when approving medications that target specific genetic mutations and molecular abnormalities. This is especially challenging considering that there is no concrete local data on the number of patients with specific mutations or targets for a particular malignancy that could help in making decisions related to approving new targeted agents. Overall survivorship data are also lacking.
Local data availability
The availability of clinical data plays a major role in decision-making when introducing a medication to the formulary. One of the major challenges in this theme is the lack of clear preset rules for screening and filtering oncology-related medications by the committee. This becomes even more challenging when the approval of particular breakthrough cancer medications is based on phase II trials with missing clinically meaningful outcomes and patient-reported outcomes that are critical to support clinical and regulatory access decision-making. Additionally, most institutions do not have a standardized framework for Medication Use Evaluations (MUEs) that should be consistently used to aid the ongoing evaluation of medication utilization and identify medication-related issues.
Pharmacoeconomic evaluation
Inadequate local data pertaining to pharmacoeconomics and, more specifically, comparative-effectiveness studies for cancer medications was identified as a major challenge. As a result of the insufficient local data, the application of international pharmacoeconomics-based studies has been adopted by certain institutions, which may lead to inaccurate results. The lack of data related to patient-reported outcomes, including quality of life (QoL), was recognized as a significant disadvantage to conducting comparative-effectiveness studies, including direct, indirect, and social costs to alternative therapeutic regimens. Many institutions consider affordability and not necessarily cost-effectiveness during new medication assessment, resulting in some setting a ceiling price for each medication, which could adversely impact the patient's access to medications. A medium between the two is conducting budget impact analysis. However, that cannot be easily implemented due to either the unavailability of costing data or the great variability of such information between the different sectors or even different settings within one sector. Local pharmacoeconomic studies are difficult to implement due to the inability to reliably estimate the direct/indirect costs associated with cancer care stemming from side effect incidence, disease consequences, and/or the different costing structures, in addition to the lack of expertise in the field of health technology assessment.
Patients reported outcomes
Patients and caregivers play a crucial indirect role in the accessibility and evaluation of oncology medications. Health literacy and competency of cancer patients or their caregivers regarding disease awareness, medical concerns, treatment plans, and expected outcomes is a considerable component in oncology medication accessibility. For instance, patient-reported outcomes are merely dependent on patients/caregivers. Thus, having patients through societies to be present during Pharmacy and Therapeutic Committee (PTC) meetings is paramount. Unfortunately, scientific information and medical terminologies may be difficult for patients to comprehend. Additionally, choosing the appropriate patient representative is considered challenging.
Administration and procurement
Medication regulatory systems such as FMS, along with evaluating committees, including PTC, which comprise representatives from different departments and medical specialties, have a direct impact on the management of the entry and utilization of medications within the institution. However, since oncology clinical trials are especially complex, the consensus of the authors is that the average member of the PTC lacks the knowledge and expertise to analyze such studies. As a result, this may create obstacles in the assessment and introduction of innovative oncology medication into the formulary. In 2020, the MOH contracted experts to support national health insurance coverage. Delay in medication registration by pharmaceutical companies at the SFDA is considered another challenge that potentially postpones hospital formulary inclusion of new medications and can also cause delays in PTC approvals. The cost of cancer treatments is extremely limiting for many patients and may limit access to potentially life-saving treatments. The purchasing cycle, according to the national tender-based purchasing system, may also cause a significant delay in the availability of oncology medications due to certain governmental processes/restrictions. Procurement of oncology medications poses many challenges. The registration status of medications can also be another hurdle that may lead to delay. The approval and registration of medications by SFDA takes approximately 18 months, which can greatly impact procurement and subsequent listing by NUPCO. Usually, unregistered drugs are not added to formularies until registration is granted by the SFDA, which explains procurement delays. Additionally, supply-chains of certain formulary medications are deemed to influence procurement due to tender restrictions negatively. Another major drawback is medication shortage, especially for those with no alternatives. Procurement is also highly reliant on the hospital's budget. Certain hospitals have limited budgets, with the major part of the budget being utilized for treating chronic diseases such as diabetes and hypertension. Along with budget limitations, financial challenges due to the increase in the number of oncology patients and the availability of many new medications can pose an extreme reduction in oncology medication availability. These procurement issues can greatly affect the patient's disease course and treatment plan, and thus, the dynamics around these factors mandate accommodation to these changes regularly.
Discussion
In this qualitative study, we described the current status and challenges impeding access to oncology medications, and we reached a consensus on strategies to mitigate these challenges. Five themes were identified and discussed: national cancer burden, local data availability, pharmacoeconomic evaluation, patient-reported outcomes, and administration and procurement. This study has some limitations inherent to its focus group design. First, the limited number of participants restricts the breadth of perspectives on access obstacles represented. Second, the recommendations are based primarily on the expert's opinion, which could introduce some subjectivity. Lastly, the limited time allocated for each topic might have curtailed the discussion on complex themes. However, several strengths were also identified. This is the first national qualitative investigation to identify the major obstacles hindering access to oncology medications. Despite the design of the study, it successfully encompasses a diverse representation across healthcare sectors, augmenting the generalizability of the recommendations. Finally, a meticulous set of national and institutional recommendations was concluded to address the identified access obstacles and improve cancer care and patient outcomes.

Conclusion
This study delineates critical obstacles and pivotal recommendations across five themes influencing access to oncology medication in Saudi Arabia. The Saudi Oncology Pharmacy Assembly, in collaboration with experts representing multiple sectors, addressed the unique challenges within the national healthcare landscape. The consensus recommendations were designed to guide policymakers and stakeholders in enhancing oncology medication accessibility, ultimately aiming to improve cancer patient outcomes and the overall quality of care.
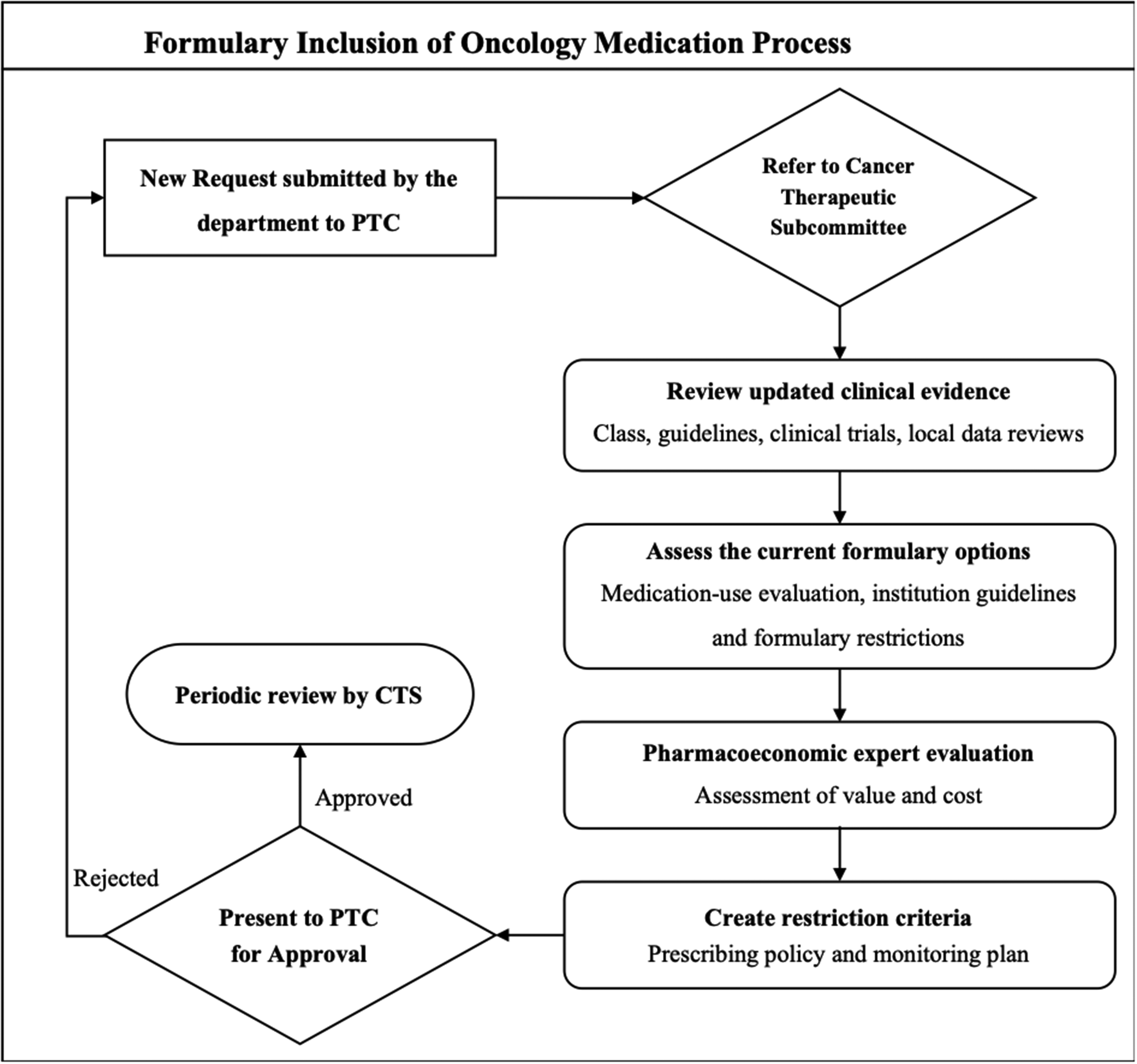
Formulary inclusion of oncology medications process.
Footnotes
Acknowledgment
We would like to thank King Saud University, Riyadh, Saudi Arabia, for supporting this research project, Researchers Supporting Project number (RSPD2023R1033).
Author Contributions
NK, MS, AH, and AR contributed to the conceptualization, data curation, and funding acquisition. NK and AR contributed to the formal analysis, investigation, project administration, and writing of the original draft. JH contributed to the review & editing of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by an unrestricted educational grant from Hikma Pharmaceuticals, KSA.
