Abstract
Elotuzumab in combination with dexamethasone and immunomodulating agents (IMiDs) lenalidomide or pomalidomide is 2nd to 4th line therapy for multiple myeloma. The labelled dosage of dexamethasone for use in conjunction with elotuzumab and IMiDs splits the dexamethasone dose into two administrations, one oral and one intravenous, on the days of each elotuzumab infusion. Administration of split dose dexamethasone on days of elotuzumab administration is based on the registration trials submitted for drug approval and was intended to ensure standard well-timed immunotherapy premedication using pharmacologically equivalent dexamethasone doses for both study arms. Administration of dexamethasone in the manner delineated by the elotuzumab product label adds complexity to the delivery of care. This commentary provides an empirical assessment of established medication safety and effectiveness which supports administration of dexamethasone standard intermittent dose instead of the split dose approach delineated on elotuzumab package insert. Simplification of regimen administration improves medication adherence, reduces the risk of inadvertent omission or duplication of medication therapy, and improves the workflow required for delivery of care.
Elotuzumab is a humanized IgG monoclonal antibody directed against SLAMF7 (signaling lymphocytic activation molecule family member 7) that activates natural killer cells and promotes antibody-directed cellular cytotoxicity against myeloma cells.1,2 Elotuzumab is Food and Drug Administration (FDA) approved in the United States for administration in conjunction with dexamethasone and lenalidomide or pomalidomide as 2nd to 4th line therapy for multiple myeloma.
The labelled dosage of dexamethasone for use in conjunction with elotuzumab and the immunomodulating agents (IMiDs) lenalidomide or pomalidomide splits the dexamethasone dose into two administrations, one oral and one intravenous, on the days of each elotuzumab infusion (Figure 1). 1 This differs from the standard approach in myeloma therapy which administers dexamethasone as a single dose intermittently (e.g. weekly) or once daily intermittently (e.g. days 1, 2, 8, 9, 15, 16 every 21 days, or days 1–4, 9–12, 17–21 every 28 days).3–5 Administration of dexamethasone in the manner delineated by the elotuzumab product label adds complexity to the delivery of care. Simplifying the delivery of patient care improves medication safety and regimen adherence. 6 The purpose of this commentary is to evaluate an empirical rationale for dexamethasone administration according to a standard schedule with elotuzumab-containing myeloma regimens.
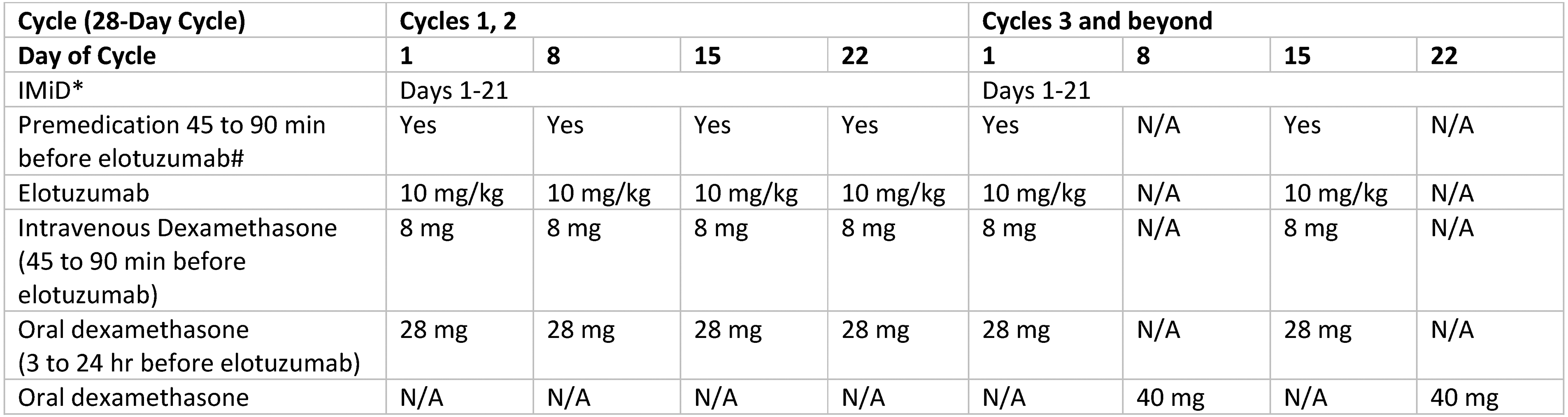
Dexamethasone split dose and ancillary medication administration for elotuzumab. Footnotes: *Labelled oral IMiD therapy is lenalidomide 25 mg daily or pomalidomide 4 mg daily; #usual premedication consists of oral or intravenous H1-blocker equivalent to diphenhydramine 25 to 50 mg plus H2-blocker equivalent to intravenous ranitidine 50 mg plus oral acetaminophen 650 to 1000 mg in addition to intravenous dexamethasone 8 mg.
Dexamethasone is a longstanding cornerstone of therapy for multiple myeloma.3–5,7 Dexamethasone administered with immunotherapy for treatment of multiple myeloma serves two purposes: (a) antimyeloma activity; (b) infusion reaction premediction.7–11 The labelled dexamethasone administration schedule for elotuzumab therapy is based on the registration trials supporting FDA approval of these treatments.9–11 Historically, a prominently administered dosage of dexamethasone 40 mg or 20 mg/m2 daily in 4-day increments was used for treatment of multiple myeloma.7,8 Over time, lower dosages of dexamethasone have been effectively incorporated into myeloma therapy.4,12,13 Streamlining dexamethasone administration for elotuzumab from the product label-delineated schedule to a standard approach should evaluate any potential effects on regimen-based efficacy and safety before considering convenience of medication administration.
Registration trials for elotuzumab (ELOQUENT-2, ELOQUENT-3) compared efficacy and safety of an IMiD and dexamethasone with or without elotuzumab for treatment of relapsed or refractory multiple myeloma.9–11 Lenalidomide and pomalidomide were the control arm IMiDs for the ELOQUENT-2 and ELOQUENT-3 trials, respectively.9,10 The primary objective was progression-free survival for both studies and with secondary objectives including efficacy and safety outcome measures.9,10 The ELOQUENT-2 trial included objective response rate as a primary objective. 9 Stringent scientific comparison of the elotuzumab and control treatment arms required equivalent IMiD and dexamethasone treatments for both cohorts. The control arms received orally administered dexamethasone 40 mg weekly. The intervention arms received oral dexamethasone weekly on weeks without elotuzumab administration and split dose dexamethasone (28 mg orally 3–24 hours prior and 8 mg intravenously 45 minutes prior to start of elotuzumab) on weeks with elotuzumab administration (Figure 1).14,15 The protocol-stated rationale for administration of split dose dexamethasone on days of elotuzumab infusion was to ensure standard well-timed immunotherapy premedication using pharmacologically equivalent dexamethasone doses for both study arms. The registration trials assumed bioequivalency of 76% for oral dexamethasone.14–16
Therapeutic efficacy of elotuzumab-based immunotherapy for multiple myeloma is unlikely to be skewed by administration of dexamethasone using a standard intermittent dose instead of the split dose method described in the product's package insert. This assertion is supported by use of the standard dexamethasone dose as a control for the split dose method in registration trials, which were based on the hypothesis that elotuzumab adds to the efficacy of immunotherapy for relapsed or refractory multiple myeloma.14,15 Clinical trials conducted for drug licensing undergo intensive review prior to implementation and additional stringent review following Biologic License Application submission to the FDA to ensure that study methodology sufficiently tests the fundamental hypothesis. 17 Equivalency of dexamethasone standard intermittent and split doses is pharmacologically plausible. Although the exact pharmacologic mechanism of action for dexamethasone's antimyeloma effect is not fully elucidated, it is seemingly secondary to drug-induced glucocorticoid receptor-mediated gene transcription.18–20 It is conceivable that systemic exposure to threshold drug concentration with repeated administrations is fundamental to achieve this effect which supports utility of either the split dose or standard schedule of dexamethasone administration.
Dexamethasone in addition to ancillary premedications (H2 blocker, H1 blocker, antipyretic) and infusion rate titration are intended to reduce the risk and severity of infusion reactions with elotuzumab administration. The elotuzumab package insert warns of infusion reactions most commonly occurring with the first treatment cycle and generally with administration of the first dose. The rate of infusion reactions in the ELOQUENT-2 and ELOQUENT-3 trials was 10% and 3.3%, respectively. All infusion reactions were severity grade 1–3; most reactions were grade 1–2.1,11 (Standard adverse event severity grading designates grades 1–2 as mild-to-moderate and grade 3 as severe or medically significant without being immediately life-threatening. 21 ) One consideration for patient safety is the time interval between dexamethasone administration and start of elotuzumab infusion on days of concurrent use, especially during the first treatment cycle. Overall, the dosage and duration of corticosteroid premedication for anticancer treatments (biologic therapy, chemotherapy, immunotherapy) varies with some regimens starting corticosteroid administration the day before and others the day of treatment administration.22–24 Notably, registration trial safety data was contingent on administration of intravenous dexamethasone 8 mg 45- to 90- minutes prior to start of elotuzumab infusion. 11 It seems prudent to maintain the 45- to 90-minute interval between dexamethasone administration using a standard dose amount and start of elotuzumab infusion, particularly with the first treatment cycle.
Corticosteroid administration is associated with a broad array of acute and chronic adverse effects to the immune-, endocrine-, neurologic-, musculoskeletal-, and cardiovascular systems which are related to dose and duration of therapy. Administration of dexamethasone using pharmacologically active dosages (at or exceeding 0.75 mg daily) is associated with acute and delayed adverse effects that are generally manageable within the context of acceptable risks of therapy.25,26 The impact of standard versus split dose dexamethasone administration with elotuzumab-based immunotherapy does not perceptibly change drug intensity according to accepted measures of systemic drug exposure such as daily dose, dose intensity, or duration of therapy. The treatment of multiple myeloma and management of disease- and treatment-related adverse events mandates close patient monitoring throughout the duration of their disease. 27 Adverse effects of dexamethasone should be monitored for and addressed by dose interruption, dose reduction, and appropriate supportive care measures.25,26 The dose of dexamethasone is commonly reduced in myeloma therapy for frailty and adverse effects. 27
Empiric assessment of established medication safety and effectiveness supports administration of dexamethasone standard intermittent dose (Figure 2) instead of the split dose approach delineated on elotuzumab package insert (Figure 1). Simplification of regimen administration improves medication adherence, reduces the risk of inadvertent omission or duplication of medication therapy, and improves the workflow required for delivery of care.
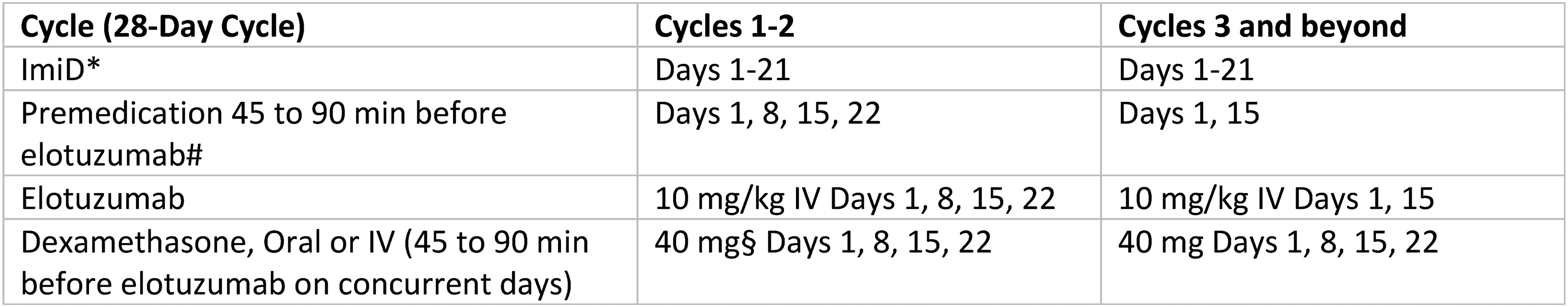
Corticosteroid administration for elotuzumab with standard intermittent dexamethasone dose. Footnotes: *Labelled oral IMiD therapy is lenalidomide 25 mg daily or pomalidomide 4 mg daily; #usual premedication consists of oral or intravenous H1-blocker equivalent to diphenhydramine 25 to 50 mg plus H2-blocker equivalent to intravenous ranitidine 50 mg plus oral acetaminophen 650 to 1000 mg §specific standard dexamethasone dose per oncologist as appropriate for patient and regimen, e.g. 20 mg days 1, 8, 15, 22; 10 or 20 mg days 1, 2, 8, 9, 15, 16, 22, 23.
Footnotes
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
