Abstract
Introduction
Camrelizumab is a novel anti-programed cell death-1 (PD-1) antibody that has been investigated for the treatment of various malignancies. Increasing immune-related adverse events have been reported in clinical practice, with CD4+ T-cell-mediated-reactive cutaneous capillary endothelial proliferation being the most common. Camrelizumab-induced oral lichenoid reaction (OLR) appears to be a rare adverse effect compared with other anti-PD therapies induced OLR, with the main pathogenesis of activated CD8+ T cells mediating autoimmune reactions. Herein, we report a rare case of camrelizumab-induced OLR and a possible pathogenic mechanism of subepithelial CD4+ T-cell infiltration.
Case report
A 57-year-old male patient, who was diagnosed with metastatic esophageal squamous cell carcinoma three years prior, presented with a two-month history of oral erosion that developed while under camrelizumab therapy. Diffuse erythematous and erosive lesions surrounded by bilateral white lesions on the buccal mucosa were detected in his physical examination. Hematoxylin and eosin staining of the lesions revealed the presence of basal keratinocyte degeneration and band-like subepithelial T-cell infiltration. The immunostaining for CD4 on T-cell was positive, while CD8 were sporadically positive. Flow cytometry showed a gradual increase in the CD4+ T-cell proportion in the peripheral blood, with the CD8+ T-cell percentage almost unchanged and in the normal range. We obtained a score of 6 based on the Naranjo algorithm, which means a probable adverse drug reaction.
Management and Outcome
The patient exhibited notable improvement after two weeks of treatment with topical glucocorticoid without regulating his immunotherapy, and remained in stable condition in the follow-up.
Discussion
This case may offer new insight to clinicians on the pathogenesis of anti-PD-1-induced OLR. More critically, it may provide some ideas for a more precise anti-PD therapy or corresponding combination therapy for patients becoming resistant to immunotherapy due to exhausted CD4+ T-cell responses in the tumor microenvironment.
Keywords
Introduction
The “normalization cancer immunotherapy” using anti-programed cell death-1 (PD-1) or programed cell death ligand 1 (PD-L1) antibodies by blocking the PD pathway to restore the antitumor activity of T cells has resulted in a positive therapeutic effect (collectively named anti-PD therapy). 1 It has been approved by the Food and Drug Administration to treat more than 10 malignancies, including esophageal cancer, head and neck squamous cell carcinoma, nonsmall cell lung cancer, etc.2,3 Camrelizumab (SHR-1210), a humanized high-affinity IgG4-kappa anti-PD-1 monoclonal antibody, can exert a significant effect on restoring immune function through the activation of T cells and cell-mediated immune responses against tumor cells, which leads to its treatment research in various advanced cancers, including classical Hodgkin lymphoma, esophageal squamous cell carcinoma, gastric/gastroesophageal junction cancer, etc. 4 Although various immune-related adverse events (irAEs) of camrelizumab were reported with CD4+ T-cell-mediated-reactive cutaneous capillary endothelial proliferation (RCCEP) being the most common (up to 80%),5,6 oral lichenoid reaction (OLR) was rarely reported. Herein, we report a rare case of camrelizumab-induced OLR with subepithelial CD4+ T-cell infiltration in a patient with metastatic esophageal squamous cell carcinoma after one year of camrelizumab therapy, different from other anti-PD therapies-induced OLR closely related to activating the CD8+ T-cell-mediated autoimmune reaction against basal keratinocytes.7,8 Two weeks of administration of topical glucocorticoid dramatically improved the lesions without altering his immunotherapy. This case may help clinicians more fully understand the pathogenesis mechanism of anti-PD-1-induced OLR. More importantly, it may provide a more precise anti-PD therapy or corresponding combined intervention for patients to overcome resistance to immunotherapy due to CD4+ T-cell exhaustion in the tumor microenvironment. 9
Case report
A 57-year-old man presented to our department in December 2021 with a two-month history of oral erosion. He was diagnosed with esophageal squamous cell carcinoma with bone metastasis in the first thoracic vertebrae three years prior. After capecitabine–nedaplatin combination radiotherapy failed, he received the camrelizumab (200 mg once intravenously every two to three weeks). He showed an objective response to the treatment. After one year of immunotherapy, the patient started complaining of oral erosion, which gradually expanded with obvious pain and seriously impacted his eating, speaking, and sleeping in the previous two months. He denied any oral or skin irAEs prior to the intervention with camrelizumab. Furthermore, the patient had no other medical history and had not received any medication change before the appearance of oral symptoms. He also did not take any other medications that could induce OLR such as nivolumab, atezolizumab, and pembrolizumab.
Physical examination revealed diffuse erythematous and erosive lesions surrounded by bilateral reticular white lesions on the buccal mucosa (Figure 1(A) and (B)). No vesicles or vesicle walls could be observed in his oral mucosa. Skin examinations were also negative. Laboratory tests, including routine blood tests, coagulation function tests, blood glucose tests, renal and hepatic clinical chemistry examinations, routine urine and stool tests, and HIV and syphilis tests, were negative or within the normal range. Serum autoantibodies against desmoglein (Dsg)1, Dsg3, bullous pemphigoid (BP) 180, and BP230 were also negative.
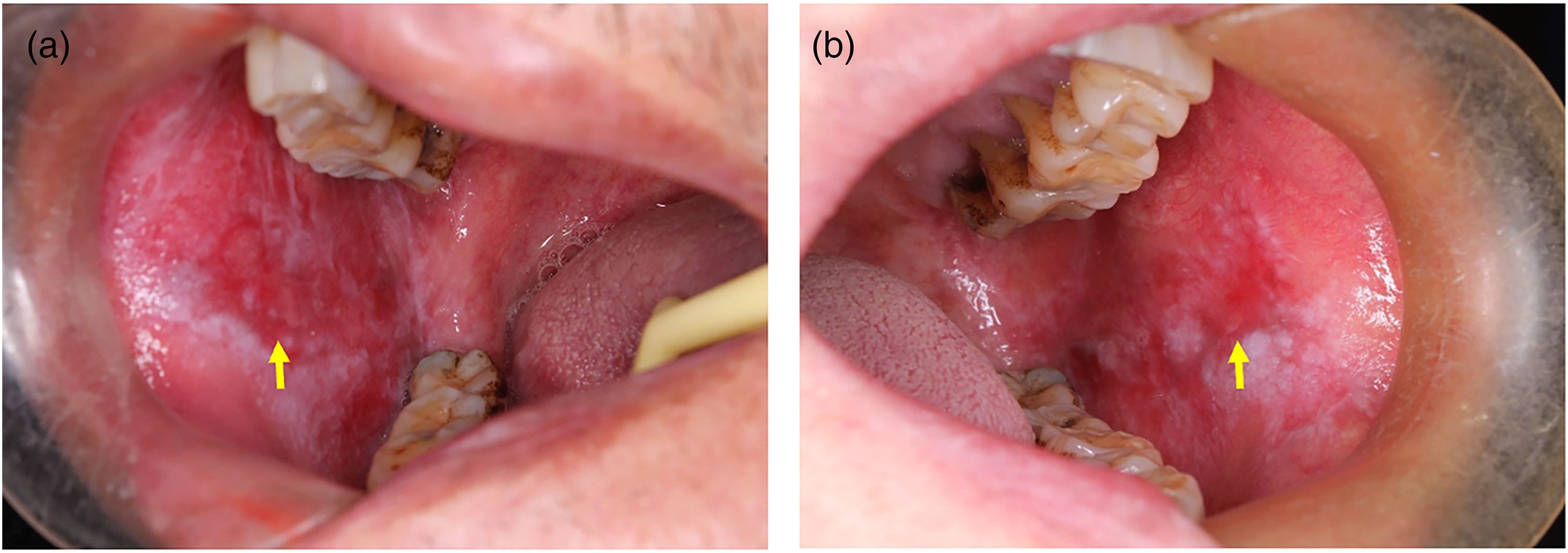
Oral manifestations of the patient with metastatic esophageal squamous cell carcinoma after immunotherapy with camrelizumab. (a–b) Diffuse erythematous and erosive lesions surrounded by reticular white lesions were noted on the bilateral buccal after one year of anti-PD-1 antibody therapy using camrelizumab. PD-1, programed cell death-1.
A biopsy specimen obtained from the left buccal mucosa showed basal keratinocyte degeneration and band-like subepithelial T-cell infiltration, which also extended to the epidermis (Figure 2(A) and (B)), typical of oral lichen planus (OLP). 10 Subsequently, immunostaining for CD4 on the T cells was positive (Figure 2(C)), while CD8 was sporadically positive (Figure 2(D)). A gradual increase in the CD4+ T-cell proportion in the peripheral blood could also be detected by flow cytometry six months before the onset of OLR and in the third, sixth, and ninth months after onset from 16.3% to 29.16% (normal range 27%–51%) (Figure 3(A) to (D)), with the CD8+ T-cell percentage almost unchanged and in the normal range (normal range 15%–44%) (Figure 3(E)–(H)). Direct immunofluorescence (DIF) assays producing negative deposits of immunoglobulin (lg)A, lgM, lgG, and complement component 3 (C3) at the intercellular and basement membrane zones excluded autoimmune bullous-related diseases, such as lichen planus pemphigoides, paraneoplastic pemphigus, and bullous pemphigoid. 11 A diagnosis of camrelizumab-induced OLR was made, which can be considered as a probable adverse reaction for the patient because he scored 6 points according to the Naranjo algorithm (Supplementary Table 1). 12
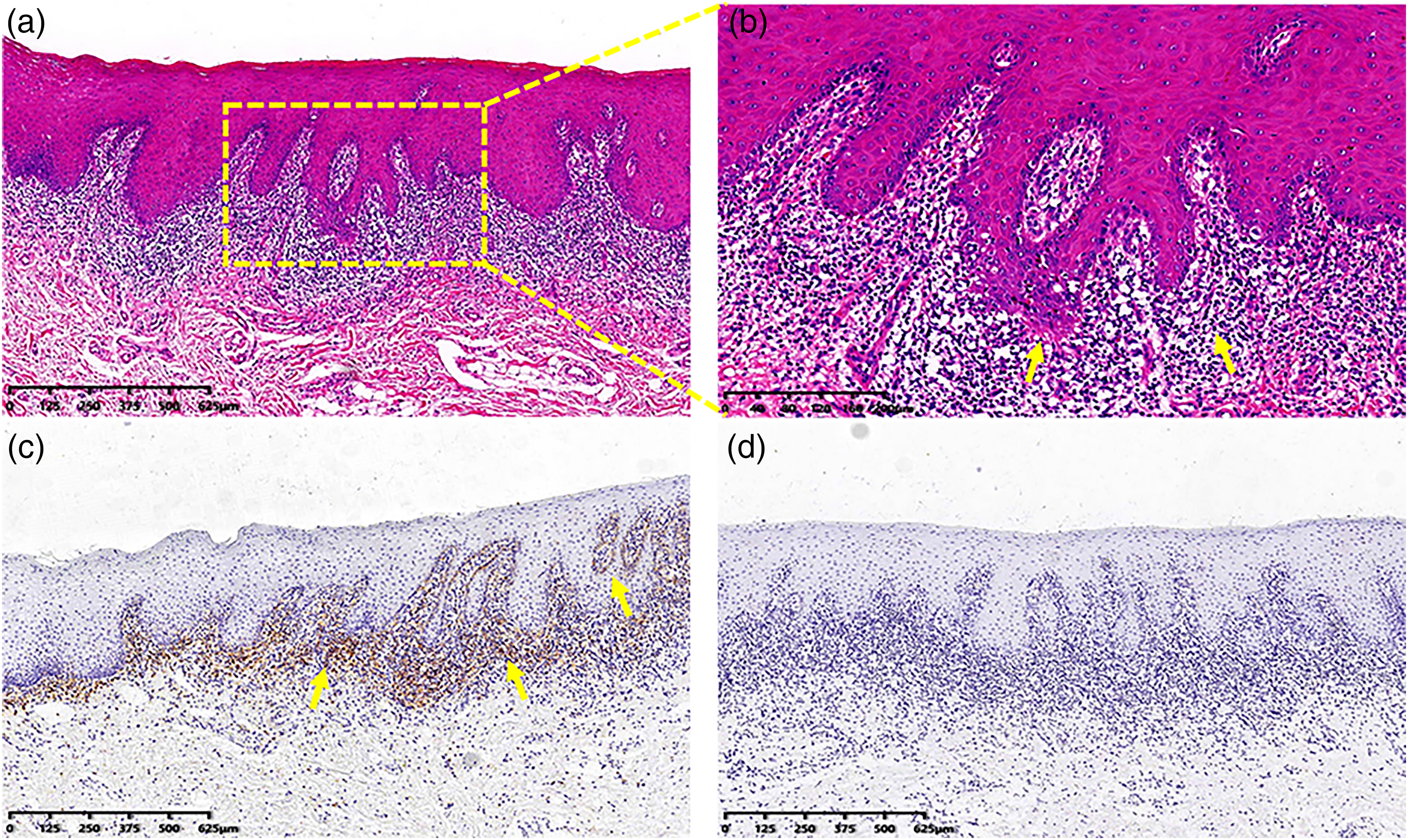
Camrelizumab induced oral lichenoid reaction: histopathologic features of the lesions from the left buccal mucosa. (a–b) Hematoxylin and eosin staining of the lesions revealed the presence of basal keratinocyte degeneration and band-like subepithelial T-cell infiltration, which also extended to the epidermis (arrow). T cells are strongly immunopositive for CD4 (c), whereas T cells are sporadically positive for CD8 (d).
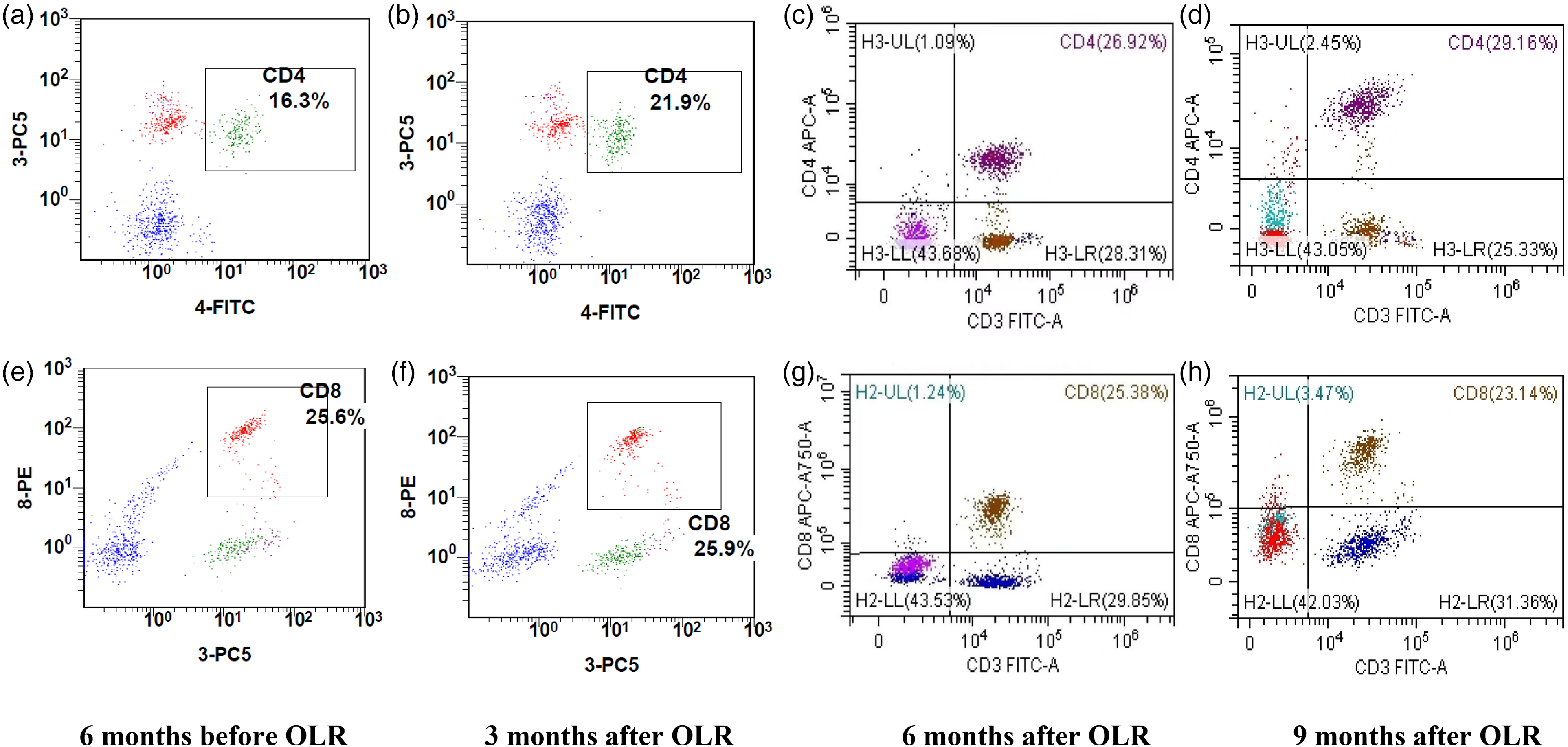
Proportion of CD4 + and CD8+ T cells in the peripheral blood of the patient with metastatic esophageal squamous cell carcinoma after immunotherapy with camrelizumab. Flow cytometry was performed, and the results showed a gradual increase in the CD4+ T-cell proportion in the peripheral blood from 16.3% to 29.16% (normal range 27%–51%) (a–d), with the CD8+ T-cell percentage almost unchanged and in the normal range (normal range 15%–44%) (e–h) at six months before the onset of OLR and in the third, sixth, and ninth months after onset. OLR, oral lichenoid reaction.
Management and outcome
The reports on the treatment of anti-PD-1-induced OLR have mainly focused on individual cases, and there is still no sufficient evidence-based medicine to help us select the best therapeutic decision for the patients. Hence, when referring to the National Cancer Institute's Common Terminology Criteria for Adverse Events (NCI-CTCAE criteria) (Version 4.0) for dermatologic toxicities, 13 we found that the patient exhibited observable improvement, with disappearance of the erosion and reduction of the erythema and white stripes after two weeks of treatment with 0.137 mg/mL betamethasone mouthwash (a kind of high potency topical glucocorticoid, which was prepared by diluting 1 mL of compound betamethasone injection containing 7 mg betamethasone with 50 mL of sterile water) three times a day, without altering his medication regimen of immunotherapy (Figure 4(A) and (B)). To avoid possible recurrence of the lesions, therapy was continued for another two months with the topical glucocorticoid (once every other day). Moreover, a 2% sodium bicarbonate solution was also given to prevent Candida infection. Clinical follow-up was performed at one, three, six, and nine months, and the patient remained in stable condition, without any reported effects on his eating, speaking, or sleeping. This study was conducted in accordance with ethical principles and approved by the Ethics Committee of our hospital (IRB#:2021-0944). The patient has given his written informed consent for publication.
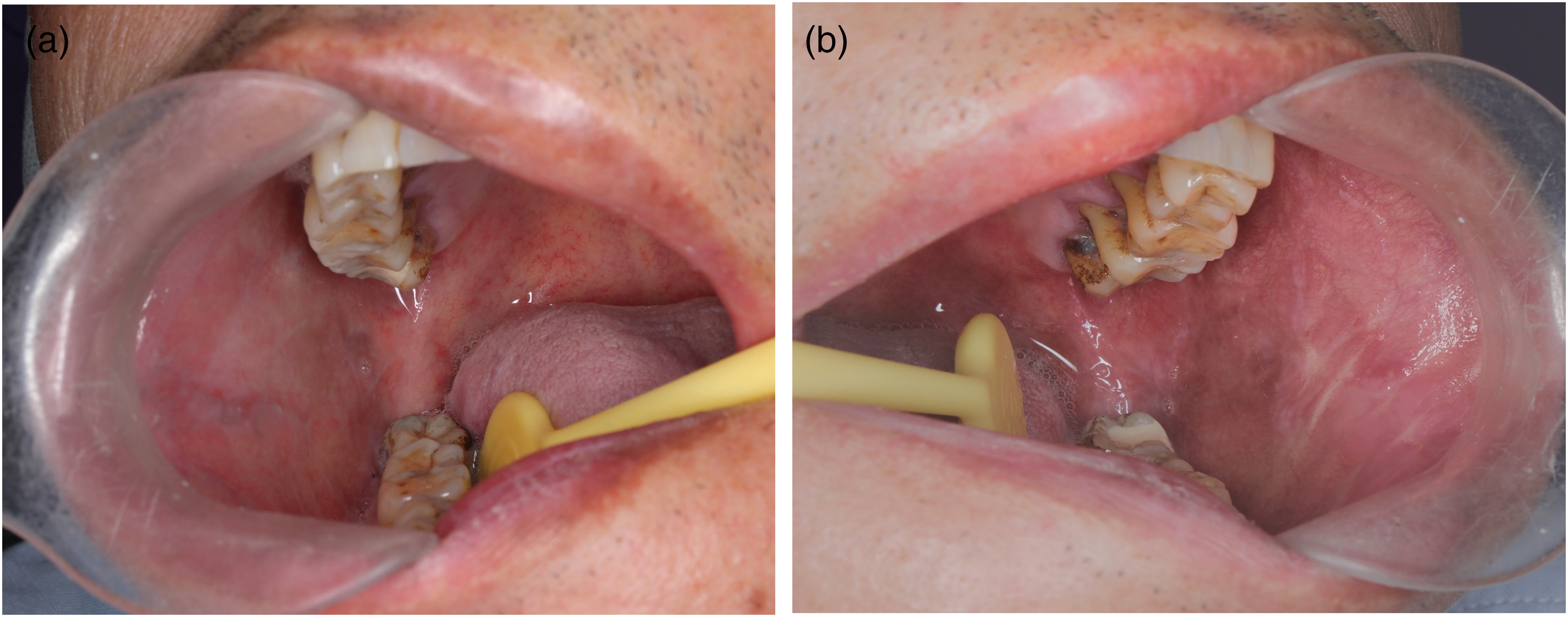
Clinical images for the camrelizumab-induced oral lichenoid reaction after a two-week treatment with 0.137 mg/mL betamethasone mouthwash. (a–b) Disappearance of the erosion, with reduction of the erythema and white stripes could be observed at the buccal mucosa without altering the immunotherapy.
Discussion
At present, the clinical application of PD-1 antibodies mainly includes nivolumab and pembrolizumab. PD-L1 antibodies include atezolizumab, avelumab, and durvalumab. The independently developed PD-1 antibody camrelizumab was approved globally for the first time in China on 31 May 2019, for patients with classical Hodgkin lymphoma. Several clinical studies have also reported promising activities of camrelizumab in patients with esophageal squamous cell carcinoma, gastric/gastroesophageal junction cancer, nasopharyngeal cancer, etc. 4
However, with the gradual promotion of anti-PD therapies in the field of cancer immunotherapy, irAEs involving the skin, gastrointestinal tract, liver, lung, endocrine system and other organs are attracting clinicians’ extensive attention, among which skin lesions can be observed in up to 40% of treated patients. 14 As the oral mucosa has a tissue structure similar to skin, it is also the most frequently involved, mainly including OLR, lichen planus pemphigoides, xerostomia, dysgeusia, erythema multiforme, ulcers and Stevens–Johnson syndrome.11,15 Among them, OLR is among the most prevalent irAE. Most patients (89%) receiving camrelizumab also experienced irAEs, with the most common being RCCEP, hypothyroidism, skin reaction, and hepatitis.4,5 Interestingly, OLR appears to be rare in camrelizumab treated-patients in a search across PubMed. OLR involving not only the oral mucosa but also the skin, nails and genitals was characterized by erythema, erosion, atrophy, or blisters associated with reticular white stripes, consistent with Wickham's striae. The histopathological characteristics are similar to those of OLP. lgM, lgG, and C3 colloid body deposition in the basement membrane area can be used in assisting the diagnosis of OLR. 16 In our case, the typical clinical manifestations of diffuse erythematous and erosive lesions surrounded by bilateral reticular white lesions on the buccal mucosa and the typical pathological manifestations of subepithelial T-cell infiltration and degeneration of basal keratinocytes with no epithelial blisters and no linear deposition of lgG and C3 in the basement membrane area supported the diagnosis of anti-PD-1-induced OLR. The negative expression of DIF in this case may be related to different pathogeneses of the lesions. 10
The specific pathogenesis mechanism of anti-PD-1-induced OLR has not yet been fully clarified and may be closely related to the autoimmune reaction mediated by direct or indirect T-cell activation. 17 In the tumor microenvironment, tumor cells can upregulate the expression of PD-L1 through multiple mechanisms (such as aberrant cell signaling or immunoediting), which results in local immune deficiency and protects them from the supervision and attack of effector T cells, thus mediating tumor immune escape. Therefore, the use of anti-PD-1/PD-L1 antibodies specifically blocking the correlation between PD-1 and PD-L1 can not only restore the antitumor activity of T cells but also induce apoptosis of epithelial keratinocytes mediated by activated T cells, thus leading to the occurrence of lesions. 8 In addition, anti-PD therapies have been found to be capable of strengthening organic immunity and exposing the original unmasked antigen or directly inducing the formation of new antigens on the surface of epithelial keratinocytes, thus activating the T-cell-mediated autoimmune response. 18 The CD8+ T-cell subtype has been found to play a synergistic role in the pathogenesis of drug-induced LR, which is closely related to the CD8+ T-cell release or expression of TNF-α, CD95 L and granzyme B, mediating the apoptosis signal pathway similar to the pathogenesis of OLP (i.e., antigen-specific mechanisms, including antigen presentation by basal keratinocytes and antigen-specific keratinocyte killing by CD8 + cytotoxic T cells).10,19 However, in addition to the above specific mechanisms, nonspecific mechanisms, including the destruction of the basement membrane caused by pro-MMP 9 activated by MMP-9 activator released by CD4+ T cells, the destruction of keratinocytes caused by CD4+ T cells migrating into the epithelium, or the recruitment of more lymphocytes, monocytes, natural killer cells, eosinophils and basophils through RANTES (regulated on activation, normal T-cell expressed and secreted) secreted by CD4+ T cells, could also mediate the occurrence of OLP. 10 Interestingly, the T cells infiltrated into the lamina propria in our case were mainly CD4+ T cells, and migrated CD4+ T cells could also be seen in the epidermis; a gradual increase in the peripheral blood CD4+ T-cell count was also found from 16.3% to 29.16%, while the CD8+ T cells remained within the normal range during immunotherapy, indicating that the nonspecific mechanism closely related to CD4+ T cells played a key role in the pathogenic mechanism of OLR in our case. In addition, a negative presentation of DIF indicated that camrelizumab did not induce exposure of the original hidden antigen or generation of a new antigen, which also indirectly indicated that anti-PD1-induced OLR in our case might be mediated by a nonspecific mechanism, especially considering the CD4+ T-cell-mediated pathogenesis of RCCEP associated with camrelizumab monotherapy. 6
There is no uniform treatment standard for anti-PD-1-induced OLR. The National Cancer Institute of the United States used the CTCAE criteria to grade the severity of skin irAEs and provided medication guidance, namely, that grade 1-2 irAEs (Grade 1, affecting less than 10% of the body surface area; Grade 2, 10–30% of the body surface area) can be controlled by topical moisturizers, moderate or high-potency topical steroids, oral antihistamines without stopping anti-PD therapy, or reassessment after two weeks and monitoring for change in severity. However, for high-grade (≥3) irAEs (more than 30% of the body surface area), topical moisturizers, high- or very high-potency topical steroids, oral antihistamines, and systemic steroids could be applied to control the symptoms. Temporary or permanent discontinuation of immunotherapy should be taken into account based on the results of reevaluation after several days/weeks for changes in severity. 13 In our case, we adopted a local treatment scheme based on topical betamethasone mouthwash without changing the dose of anti-PD-1 antibody because the localized lesions were in accordance with low-grade irAEs. In addition, to avoid the possible recurrence of the lesions because of CD4+ T cells migrating into epithelial tissue from peripheral blood, therapy with the topical glucocorticoid (once every other day) was continued for another two months. The results showed that the patient developing OLR with anti-PD-1 therapy had been successfully managed with local betamethasone without immunotherapy discontinuation.
The presence of oral or skin irAEs may represent a positive prognostic factor for the effectiveness of anti-PD therapy, with the improvement of the overall survival rate of the patients. 20 Stable disease or a partial/complete response has been reported in 80% of patients (12/15) who developed skin irAEs after receiving anti-PD therapy, 21 which was also supported by a meta-analysis demonstrating a noteworthy significant overall survival benefit in patients exhibiting irAEs. 22 LR usually appears approximately 17 weeks after starting immunotherapy, 21 and the clinical manifestations may gradually become serious over time, just as the extensive erythematous and erosive lesions in our case at the first visit after one year of immunotherapy, which has a negative impact on the health-related quality of life of patients, may even lead to delayed immunotherapy. 11 Therefore, early identification and management of such irAEs are believed to be critical in reducing the severity of the lesions.
The regression of tumor-specific effector T cells from lymphoid organs into peripheral blood and trafficking to tumor tissues is key to driving effective antitumor immunity. 1 CD4+ T-cell-mediated pathogenesis of OLR in our case, and CD4+ T cells infiltrating in the lesion of RCCEP, the most common irAE related to camrelizumab monotherapy, 6 suggested that the successful immunotherapy of camrelizumab might be attributed to its roles in homing of CD4+ T cells from lymphoid organs into peripheral blood and tumor tissues.
In conclusion, we demonstrated an unusual case of OLR as an irAE of camrelizumab monotherapy that was characterized by proliferative CD4+ T-cell infiltration in the superficial dermis. However, a possible immunotherapy strategy making use of camrelizumab's role in the homing of CD4+ T cells, especially for patients exhibiting tolerance to oncotherapy with a possible immunosuppressive mechanism of CD4+ T-cell exhaustion in the tumor microenvironment requires further clinical trials with larger sample size and more molecular biology experiment.
Supplemental Material
sj-docx-1-opp-10.1177_10781552231203723 - Supplemental material for Camrelizumab-induced oral lichenoid reaction with subepithelial CD4+ T-cell infiltration
Supplemental material, sj-docx-1-opp-10.1177_10781552231203723 for Camrelizumab-induced oral lichenoid reaction with subepithelial CD4+ T-cell infiltration by Xiangjian Wang, Tao Fu and Weilian Sun in Journal of Oncology Pharmacy Practice
Footnotes
Authors’ Contribution
XJW and WLS researched the literature and conceived the study. TF contributed to the data collection and analysis. XJW was involved in gaining ethical approval and funding. XJW and TF made contributions to drafting the manuscript. WLS revised and edited the manuscript multiple times. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 82102854); and the Medical Science and Technology Project of Zhejiang Province (grant number 2022RC166).
Patient Consent
Written informed consent was obtained from the patient for publication as a case report.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
