Abstract
Background
Luspatercept is a recombinant fusion protein that binds several endogenous TGF-β superfamily ligands, thereby decreasing Smad2/3 signaling. This mechanism promotes differentiation and proliferation of late-stage erythroid precursors and improves hematology parameters associated with ineffective erythropoiesis. 1 Luspatercept has approved indications for anemia due to beta-thalassemia and anemia failing an erythropoiesis-stimulating agents, requiring 2 or more red blood cell (RBC) transfusions over 8 weeks, in patients with very low-to intermediate-risk myelodysplastic syndromes with ring sideroblasts (MDS-RS) or with myelodysplastic/myeloproliferative neoplasms with ring sideroblasts and thrombocytosis (MDS/MPN-RS-T). 1 It is not indicated as a substitute for RBC transfusions in patients who require an acute need for transfusion.
Myelodysplastic syndromes (MDS) are comprised of a group of hematologic malignancies characterized by abnormal cellular maturation, cytopenias, and clonal hematopoiesis. Morbidities of this disease process include cytopenias which may include symptomatic anemia and the transformation to acute myeloid leukemia (AML). 2 MDS can be further complicated in elderly populations where chronic anemia is associated with cardiovascular complications, increased falls, bone fractures, and overall shorter survival. 2 Ineffective erythropoiesis and need for transfusions can result in iron overload resulting in endocrinopathies, liver, and cardiac dysfunction as well. 2 Dependence on red-cell transfusions can lead to treatment toxicities and decreased quality of life. Disease stratification is based on a multitude of factors classified by the Revised International Prognostic Scoring System (IPPS-R). 3 Patients are categorized as very-low, low, intermediate, and high risk. Considerations for the optimal treatment strategies include the patients’ IPSS-R score as well as age, performance status, and comorbidities.2,3
First-line treatment options for patients with lower-risk MDS include erythropoietin-stimulating agents (ESAs). Patients must meet pretreatment variables that are predictive of response to ESAs (EPO ≤ 500 IU/L). Lenalidomide is established for patients with lower-risk MDS with a del(5q) deletion who are dependent on transfusions, regardless of previous use with erythropoiesis-stimulating agents. Other therapeutic alternatives for patients who have failed ESAs (defined as: at least 12 weeks of therapy at maximal dose and unlikely to respond to ESA; endogenous serum EPO >500 U/L) include, granulocyte colony-stimulating factors, antithymocyte globulin, hypomethylating agents, cyclosporine A, eltombopag, and luspatercept. 4 Adjunct supportive care such as transfusions, psychosocial support, and iron chelation therapy should be considered throughout the continuum of treatment.
Treatment goals for patients with lower-risk MDS include transfusion independence, improvement in hemoglobin levels, and improvement of quality of life. A phase 2 study involving patients with lower-risk MDS found 63% of luspatercept treated patients had an erythroid response defined as a reduction in RBC transfusions of ≥ 4 units per 8 weeks and 38% had a transfusion independence of 8 weeks or longer. 5 MEDALIST, a phase 3 trial, evaluated the efficacy of luspatercept in patients with ring sideroblasts and found a reduction in the severity of anemia and transfusion independence for 8 weeks or longer observed in 38% of patients in the luspatercept group compared with 13% of those in the placebo group (p < 0.001). 6 Erythroid response is defined as a reduction in red-call transfusions of ≥ 4 units per 8 weeks in patients with a baseline transfusion burden of ≥ 4 units per 8 weeks or as an increase in the hemoglobin level of ≥ 1.5 g per deciliter over a period of 8 weeks in patients with a baseline transfusion burden of <4 units per 8 weeks. 7
The goal of this research is to evaluate if patients achieve optimization of dose escalations under the prospective review of a pharmacist at an outpatient center vs. no prospective review of a pharmacist at a large academic medical center's main campus. Dose optimization was measured by calculating the percentage of off-label doses ordered. 26% of patients in the MEDALIST trial had initial response at 1 mg/kg dose and 24% had subsequent response interval at the same or higher doses. 6 On-label dose escalations may optimize outcomes and provide improved response to treatment. Secondary endpoints include reporting real world outcomes of luspatercept efficacy.
Methods
Trial design
We performed a retrospective multi-center study involving patients at a major academic medical center main campus, which did not have a pharmacist prospectively review luspatercept doses, as well as a community satellite campus infusion center, who had a pharmacist prospectively review doses and who received at least one dose of luspatercept between January 1, 2017, and August 31, 2022.
Patients
Eligible patients were 18 years of age or older with MDS, receiving regular red-cell transfusions (≥ 2 units per 8 weeks), and had received at least one dose of luspatercept at either center. Patients were excluded if they received luspatercept for beta-thalassemia or a myeloproliferative neoplasm, started therapy at an outside institution, or had MDS and were transfusion-independent at baseline.
Baseline characteristics
The baseline erythropoietin, pre-dose hemoglobin, platelets, and neutrophil levels were defined as the closest level prior to first dose of luspatercept. Patients were defined as disease refractory to erythropoietin if they completed at least 12 weeks of therapy at maximal dose and were unlikely to respond to ESA (endogenous serum EPO >500 U/L). 4 Patients were defined as RBC refractory if they received regular red-cell transfusions (≥ 2 units per 8 weeks). 6 The normal range of folate was defined as 2.7 to 17 ng/mL per institutional lab range.
Trial endpoints
The primary endpoint was the percentage of off-label luspatercept doses not consistent with dose adjustment recommendations in the prescribing information. Off-label doses were defined as doses that should have been escalated that were not, doses that were escalated in patients that were transfusion independent, doses that were discontinued prior to a dose escalation, but the patient still required therapy, and doses that were continued when it should have been discontinued (three doses without the reduction in RBC transfusion requirements).
Secondary endpoints included the number of patients that underwent correct dose modifications for pre-dose hemoglobin levels or rapid hemoglobin rise. Other secondary endpoints evaluated real world efficacy analyses. Efficacy was defined as the number of patients that achieved transfusion independence for at least 8, 12, and 16 weeks, mean increase in hemoglobin levels by > 1 g/dL [defined as an increase from baseline of at least 1 g/dL (>14 days after the last red-cell transfusion or within 3 days before the next red-cell transfusion) that was sustained over any consecutive 56-day period in the absence of red-cell transfusions], and the percentage of patients who demonstrated efficacy to luspatercept prior to dose escalation (defined as: erythroid response or hematologic improvement. This is defined according to the International Working Group [IWG] 2006 criteria as a reduction in red-cell transfusions of ≥ 4 units per 8 weeks in patients with baseline transfusion burden of ≥ 4 units per 8 weeks or as an increase in the hemoglobin level of ≥ 1.5 g/dL over a period of 8 weeks in patients with a baseline transfusion burden of <4 units per 8 weeks or transfusion independent at baseline). 3
Safety analyses evaluated the number of patients that stopped luspatercept due to adverse effects and adverse effects as noted in progress notes. Adverse effects included injection site reactions, gastrointestinal disorders (nausea/vomiting/constipation/diarrhea), nervous system disorders (dizziness, headache, confusion), musculoskeletal (back pain, arthralgia), respiratory (dyspnea, cough), and infection.
Statistical analysis
REDCap was utilized to capture patient data collection from Epic®, the electronic health record database. Descriptive statistics were utilized to describe the population(s). 8 Comparisons were performed with Fisher's exact test for categorical variables and paired t-tests for continuous variables. Statistical significance was defined at a p < 0.05, and all tests were two-sided. Appropriate dose escalations were calculated by the number of off-label dose escalations divided by the total number of doses.
Results
Baseline characteristics of the patients
A total of 35 patients were reviewed with 17 meeting inclusion criteria. Reasons for exclusion included patients who received luspatercept for indications other than MDS (n = 10), started luspatercept at an outside hospital (n = 2), or were receiving less than 2 RBC transfusions in the 8 weeks prior to starting luspatercept (n = 6). There were 11 patients evaluated from the main campus, whose dosing was not prospectively reviewed by a pharmacist, vs. 6 patients from the community satellite, whose dosing was prospectively reviewed by a pharmacist. Overall, patients’ baseline characteristics were balanced between groups (Table 1). The median age of the patients was 79 (range, 78 to 86), and 77% of the patients were male. Regarding IPSS-R categories, 11.8%, 64.7%, 17.6%, and 5.9% had a myelodysplastic syndrome defined as very low risk, low risk, intermediate risk, and very high risk, respectively (Table 1).
Demographics and disease characteristics of the patients at baseline.
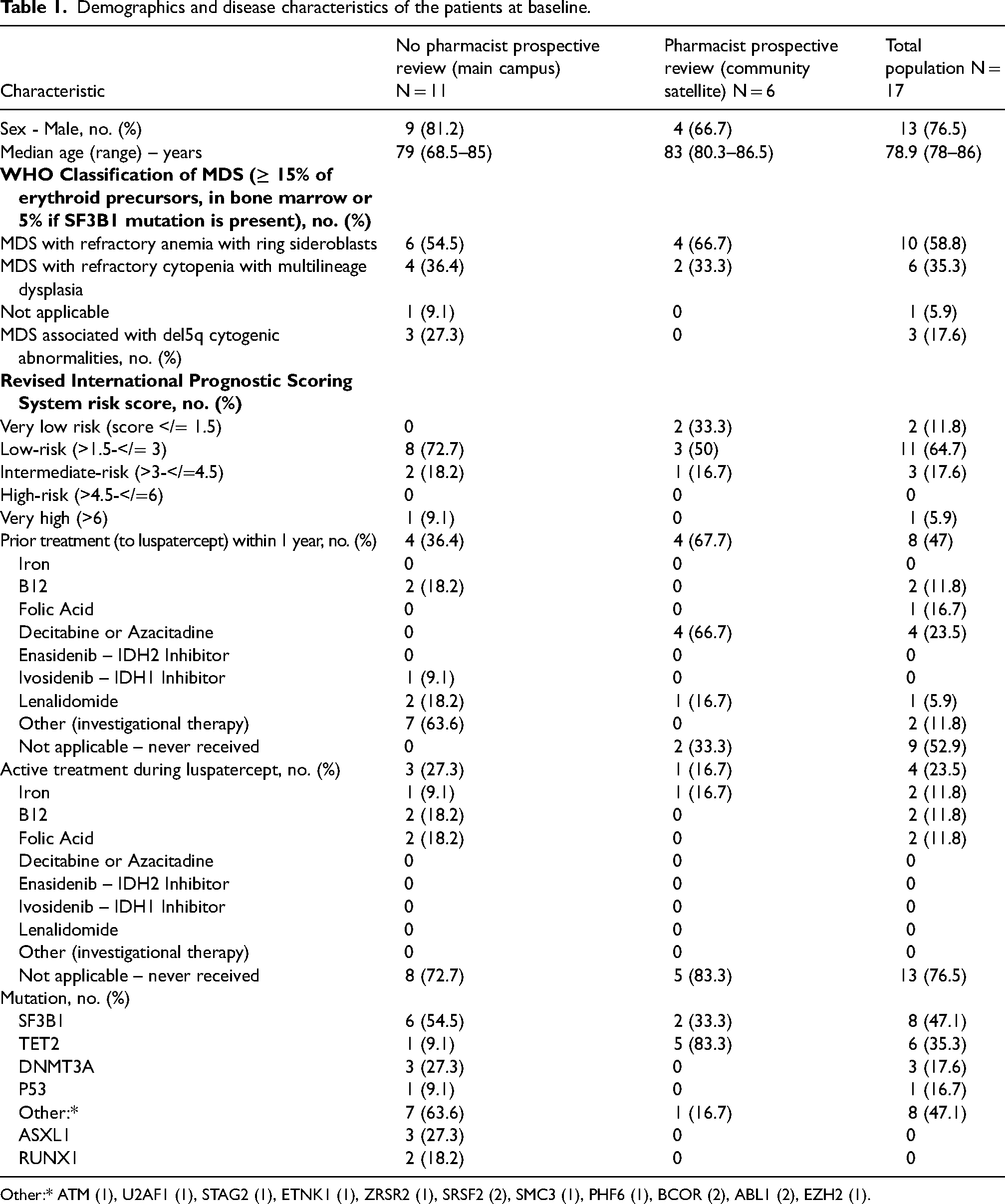
Other:* ATM (1), U2AF1 (1), STAG2 (1), ETNK1 (1), ZRSR2 (1), SRSF2 (2), SMC3 (1), PHF6 (1), BCOR (2), ABL1 (2), EZH2 (1).
The baseline serum erythropoietin levels were less than 100 units per liter, 100 to less than 200 units per liter, 200 to 500 units per liter and greater than 500 units per liter, in 17.6%, 17.6%, 23.5%, and 29.4% of patients, respectively. There were no serum erythropoietin levels in 11.8% of patients. The median RBC transfusion burden was 6.5 units (range, 4 to 8) in the 8 weeks prior to the first dose of luspatercept. The baseline transfusion burden was at least 6 units per 8 weeks in 52.9% of patients and less than 6 units per 8 weeks in 35.5%; 11.8% had a baseline burden of less than 4 units per 8 weeks. SF3B1 mutations were detected in 54.5% of patients in the main campus group and in 33.3% of patients in the community satellite group. There was a greater percentage of patients in the MGH North Shore group with TET2 mutations (83.3% vs. 9.1%) and a greater number of patients that had DNMT3A and P53 mutations in the main campus group (Table 2).
Baseline laboratory measurements.
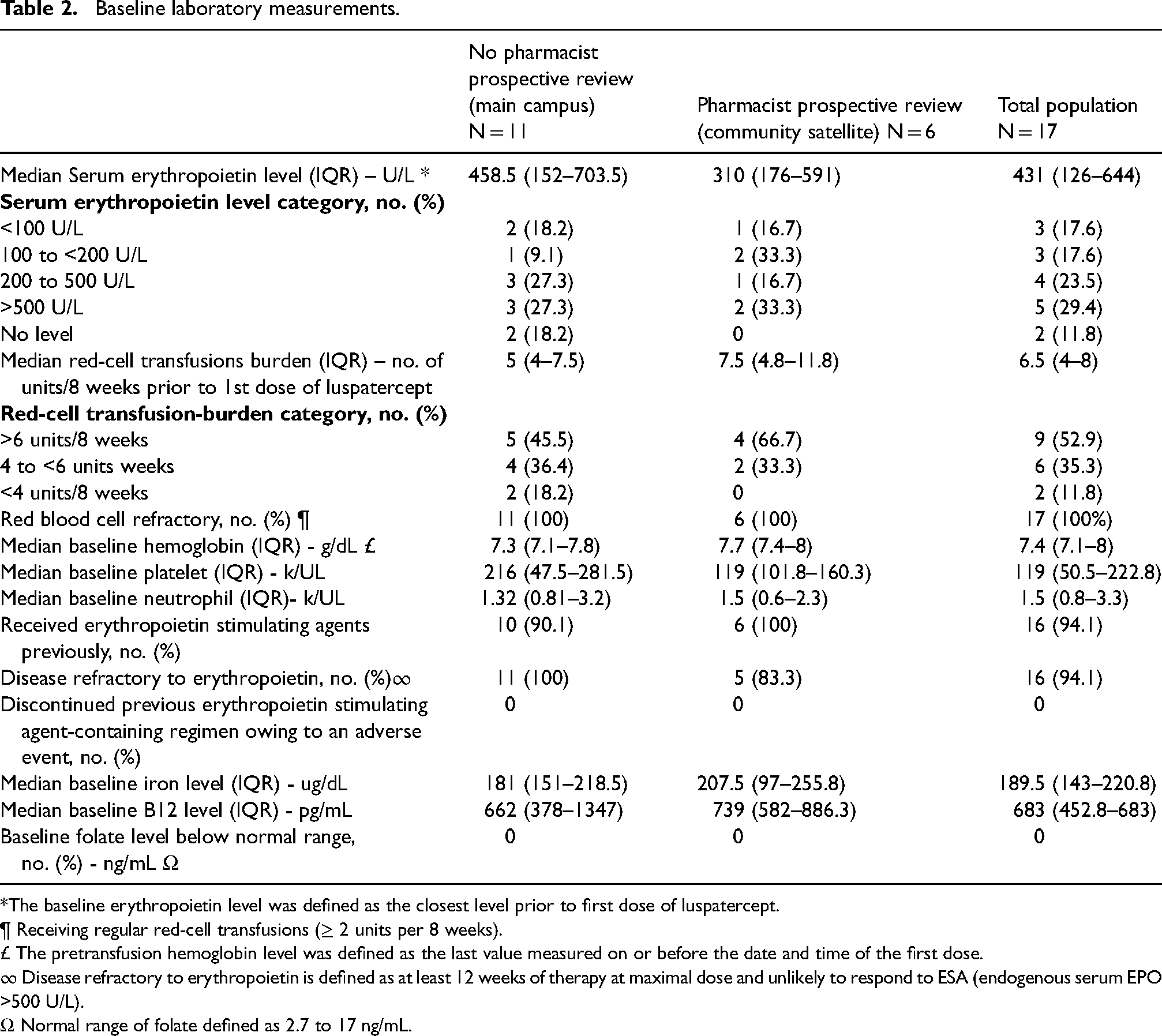
*The baseline erythropoietin level was defined as the closest level prior to first dose of luspatercept.
¶ Receiving regular red-cell transfusions (≥ 2 units per 8 weeks).
£ The pretransfusion hemoglobin level was defined as the last value measured on or before the date and time of the first dose.
∞ Disease refractory to erythropoietin is defined as at least 12 weeks of therapy at maximal dose and unlikely to respond to ESA (endogenous serum EPO >500 U/L).
Ω Normal range of folate defined as 2.7 to 17 ng/mL.
Overall, 94.1% of the patients received ESAs previously and were noted to be disease refractory. All patients had a normal folate level.
Primary endpoint
Of the 162 doses evaluated, 37 (23%) were defined as off-label. Off-label dosing at the center without a pharmacist conducting prospective review was more common than at a center with a pharmacist conducting prospective review of luspatercept dosing (29.6% vs. 2.4%; p < 0.003). Of the 37 doses that were not consistent with the prescribing information, 17 (10.5%) required a dose escalation, 4 (2.5%) did not require a dose escalation, and 12 (7.4%) met requirements for discontinuation, but received a dose. More doses at the main campus required a dose escalation but did not compared to the community satellite (28.1% vs. 2.4%; p < 0.006) (Table 3).
Off-label dosing and median pre-dose lab levels.
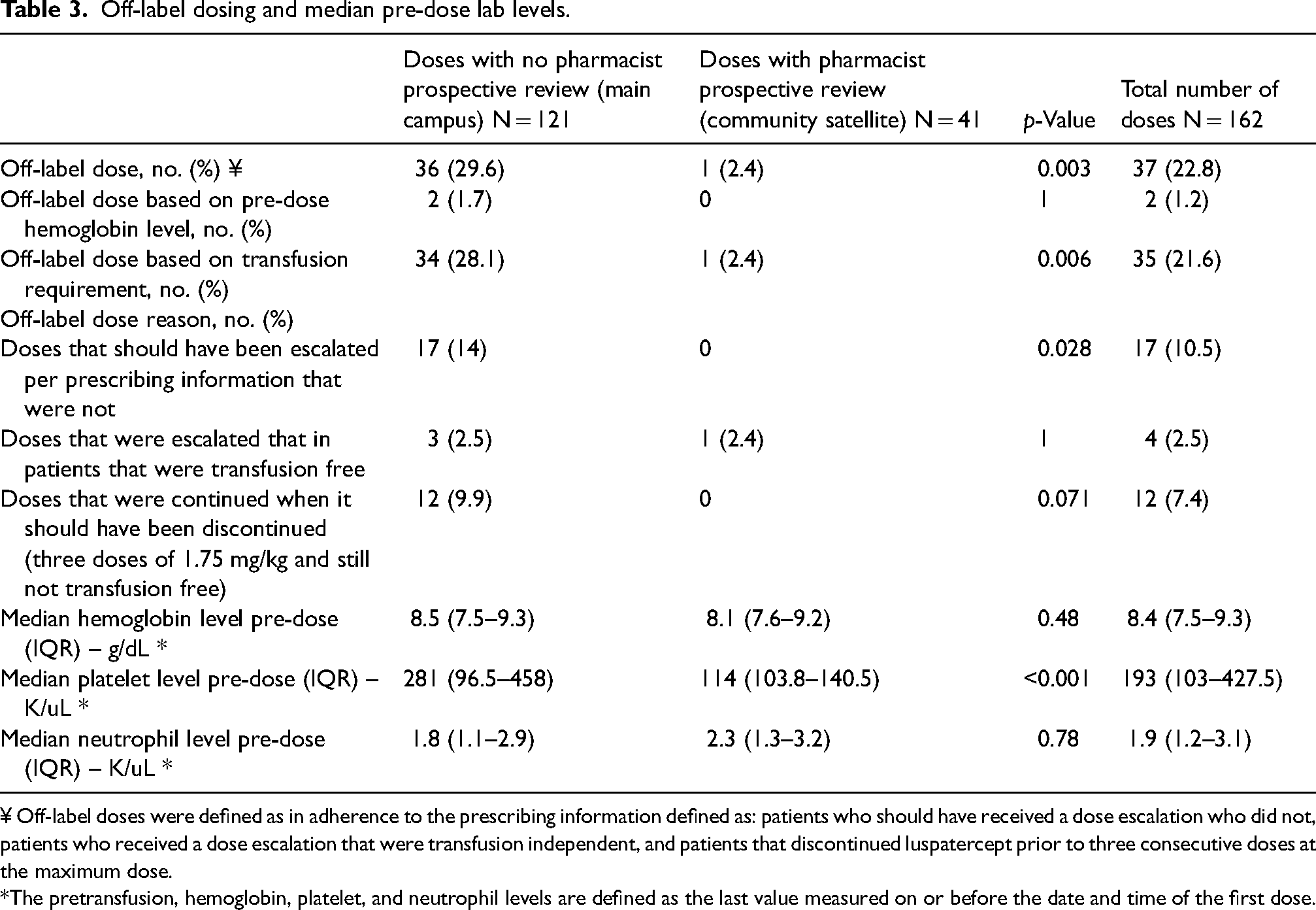
¥ Off-label doses were defined as in adherence to the prescribing information defined as: patients who should have received a dose escalation who did not, patients who received a dose escalation that were transfusion independent, and patients that discontinued luspatercept prior to three consecutive doses at the maximum dose.
*The pretransfusion, hemoglobin, platelet, and neutrophil levels are defined as the last value measured on or before the date and time of the first dose.
Secondary endpoints
Transfusion independence was achieved more frequently at a center with a pharmacist conducing prospective review vs. at the center without a pharmacist conducing prospective review (83.3% vs. 27.3; p < 0.389). Only patients at a center with prospective pharmacist review achieved a mean increase in hemoglobin of >1 g/dL (18.2%; p < 0.544). 18.2% of patients at the main campus demonstrated efficacy to luspatercept prior to dose escalation. 3 Luspatercept was discontinued prior to three consecutive administrations of the maximum dose more frequently at the main campus vs. community satellite (18.2% vs. 0%) (Table 4). Overall progression of AML was greater at the main campus vs. community satellite (9.1% vs. 0%).
Transfusion independence and increase in mean hemoglobin levels.

*Transfusion independence achieved defined as ≥2 weeks transfusion free.
€ Defined as an increase from baseline of at least 1 g/dL (>14 days after the last red-cell transfusion or within 3 days before the next red-cell transfusion) that was sustained over any consecutive 56-day period in the absence of red-cell transfusions.
± Luspatercept discontinued inappropriately/prematurely if a provider discontinues luspatercept due to lack of efficacy prior to the patient receiving seven correct doses.
Safety
The most frequent reported adverse events were dizziness, headache, and confusion (Figure 1). Three (17.7%) of the total patient population discontinued luspatercept due to these adverse events. One patient (5.8%) described symptoms of excessive fatigue causing luspatercept to be discontinued. Another patient described symptoms of heart palpitations; these were resolved after luspatercept was discontinued and another patient discontinued luspatercept for risk of increased clot burden attributing to treatment (Figure 1). Three (9.1%) of the patients in the main campus group had progression of myelodysplastic disease and developed AML. No patients in the community satellite group developed AML.

Adverse events.
Discussion
To our knowledge, this is one of the first real-world studies evaluating a pharmacist's prospective review on dosing luspatercept. This study highlighted an opportunity where pharmacist's prospective review may optimize patient outcomes. This evaluation included patients at a large academic medical center and a satellite campus. Most of the patients had low-risk MDS, were refractory to ESAs, and were transfusion dependent. There were a higher percentage of off-label dosing at a center without a pharmacist prospective review vs. a center with a pharmacist prospective review (29.6% vs. 2.4%; p < 0.003). There was a greater response to luspatercept in patients treated at the community satellite campus, which could be due to less off-label doses. Most off-label dosing was caused by a need for a dose increase, based on transfusion requirements. Secondarily, 12% of doses at the main campus should have been discontinued based on transfusion requirements, that were continued. Off-label dosing can be caused by difficulty in tracking RBC transfusions in the patient chart, specifically, original transfusion requirements. These values are not readily available during provider ordering; therefore, this step may get omitted leading to off-label dosing.
Similar to the MEDLIAST trial, the majority of our patients had lower-risk MDS with ring sideroblasts, had failed ESAs, and were receiving regular RBC transfusions demonstrated safety and efficacy to luspatercept. 6 Approximately half of our patient population achieved transfusion independence, aligning with the MEDLIAST trial where 38% of patients who were treated with luspatercept achieved transfusion independence vs. 13% in the placebo group (p < 0.001). 6 Although the study was underpowered to perform statistical analysis of off-label dosing and patient outcomes, our findings support more patients with prospective pharmacist review achieved transfusion independence. However, there was no difference in the duration of transfusion independence with a pharmacist's prospective review. Other reasons efficacy may have differed was difference in mutations with greater number of P53 mutations in the main campus group which has been associated with a poorer prognosis. 9
Luspatercept was associated with low-grade adverse effects that rarely led to treatment discontinuation. The most common adverse effect was fatigue and is consistent with the MEDALIST trial findings where more patients in the luspatercept arm experienced fatigue vs. those who received placebo. 6 Progression to AML was infrequent in the overall patient population. However, AML was greater at the main campus vs. at the community satellite (9.1% vs. 0%). These results may be due to a greater population of critically ill patients at the main campus vs. at the community satellite. The main campus had a greater incidence of P53 mutated disease which has been associated with a poorer prognosis and may have contributed to this finding. Overall, these rates were similar and consistent with data from phase 2 and phase 3 studies that showed no increased risk of progression to AML and minor side effects.5,6
Limitations to consider for this study are the retrospective design, small patient population captured, and the study was underpowered. Due to the nature of a retrospective review, some lab values could not be captured if they were obtained at an outside institution limiting the clinical reliability of our results.
Although a pharmacist's contribution to oncology pharmacy practice has not been fully understood and recognized, this study adds to documented literature highlighting the value of the oncology pharmacist as a frontline practitioner critical for enhancing patient care. 10 As the number of oncologic diagnoses increases, and the projection of shortages of oncologists in upcoming years, expanding pharmacy practice with initiatives such as, the expansion of practice settings, growth of medication therapy management programs, the development of independent prescribing protocols, and implementing institutional credentialing and privileging through collaborative practice agreements, may all lead to improved patient outcomes, decreased costs, and job satisfaction. 11 We hope to encourage our profession to continue to expand and enhance their roles in areas where pharmacist's involvement can optimize patient outcomes.
Most importantly, this study highlighted opportunities for enhancements in the current ordering and review process. Improvements include the involvement of a pharmacist's prospective involvement at both centers, inclusion of the number of transfusions and hemoglobin levels in the provider ordering screen, and implementations of lab monitoring in electronic medical record therapy plans. On-label dose optimization may lead to a higher percentage of patients achieving transfusion independence.
Footnotes
Contributor statement
JS, CB, and BR conceived the study. All authors research literature. MK was involved in protocol development, gaining ethical approval, and data analysis. MK wrote the drafts of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
