Abstract
Introduction
Niraparib, a strong poly(adenosine diphosphate-ribose) polymerase (PARP) inhibitor, contributed significantly to progression-free survival as a maintenance therapy in the platinum-sensitive period in both first-line and recurrent ovarian cancer, regardless of the BRCA mutation. Grade 3–4 anemia, which has a manageable side effect profile, especially hematological, is seen in almost 1 out of every 4 patients. To the best of our knowledge, there has been no reported case of pure red cell aplasia (PRCA) induced by niraparib treatment.
Case Report
A 65-year-old woman diagnosed with stage 3 serous carcinoma of the tuba received niraparib front-line maintenance treatment had grade 4 anemia after 3 months of niraparib treatment. She underwent bone marrow aspiration and biopsy because of refractory anemia, which needs red blood cell (RBC) transfusions despite interruption of treatment.
Management and Outcome
The patient was treated with 1 mg/kg methyl prednisolone, after histopathological assessment was consistent with PRCA. The hemoglobin count returned to the normal range with steroid treatment.
Discussion
In daily practice, it should be kept in mind that in the case of refractory anemia induced by niraparib, the underlying cause might be PRCA and can be improved with steroid administration.
Introduction
Niraparib, a poly(adenosine diphosphate-ribose) polymerase (PARP) inhibitor, is an ideal treatment option for both primary and recurrent ovarian cancer due to its significant progression-free survival (PFS) advantage, particularly in patients with BRCA mutations or homologous recombination deficiency (HRD)-positive disease.1,2 Hematologic abnormalities (anemia, thrombocytopenia, and leukopenia) and gastrointestinal problems (nausea and diarrhea) are the most frequent adverse effects of niraparib. 3 Grade 3 or 4 anemia affected 25.3% to 28.7% of the patients, and it is managed with dose reduction, interruption, discontinuation, or red blood cell (RBC) transfusion. 4 Anemia comprises events with preferred terms of hemoglobin decrease, macrocytic anemia, aplastic anemia, and normochromic normocytic anemia. 5 To the best of our knowledge, there have been no cases of pure red cell aplasia (PRCA) documented in randomized clinical trials or case reports in the scientific literature. We present PRCA, a rare adverse effect, and its successful management in a patient with BRCA-mutant advanced high-grade serous tuba cancer who received niraparib for front-line maintenance.
Case report
A 65-year-old woman underwent an urgent appendectomy, and appendix pathology was reported as serous carcinoma infiltration. Following the pathology report, computed tomography (CT) imaging was conducted, and 3 cm diameter omental implants were detected. The level of cancer antigen-125 (CA-125) was 192 U/mL. The patient had a total abdominal hysterectomy, bilateral salpingo-oophorectomy, and pelvic lymph node dissection. Because there were 2 cm of serous carcinoma in the right tube and one metastatic lymph node in the left external iliac, it was determined to be stage 3A tubal serous carcinoma. The patient’s ECOG performance status was 0. Complete blood count, liver, and kidney function tests were within the normal range, and the postoperative CA-125 level was 197 U/mL. BRCA-1 germline mutation was identified. At the postoperative fourth week, she was initiated on adjuvant chemotherapy and received six cycles of carboplatin area under the curve (AUC) 5 and paclitaxel 175 mg/m2. By the completion of chemotherapy, the CA-125 level was 13 U/mL, and imaging of the thorax, abdomen, and pelvis revealed minimal implants with considerable radiological regression. Niraparib was planned for maintenance treatment. Before initiating niraparib, laboratory parameters, electrocardiography (ECG), and blood pressure were evaluated. The variables in the laboratory parameters were confirmed to be within normal ranges. Heart rate and blood pressure were within normal limits. Since the patient weighed 71 kg and had a platelet count above 150 × 10 e3/μL, maintenance niraparib treatment was started at a dose of 200 mg/day.
The patient's complete blood cell count, liver function tests, kidney function tests, ECG, and blood pressure were measured every week for the first month and then monthly after that. Laboratory parameters, the ECG, and blood pressure were in the normal range in the first 2 months. At the third-month control, the hemoglobin level was measured at 4.7 g/dL. Hematocrit, platelet, and red and white blood cells were also low. Peripheral blood smear and anemia parameters were evaluated. Except for reticulocytopenia, nutritional and hemolytic anemia parameters were normal. The peripheral blood smear demonstrated normocytic, normochromic erythrocytes, and the reticulocyte count was low (4, 78 × 10 e9/L). Niraparib was interrupted because of grade 4 anemia. The patient underwent 1 unit of RBC transfusion once a week for 4 weeks. Afterwards, anemia was followed up with weekly hemogram control. A bone marrow biopsy was performed, and it revealed normal cellularity for age (70%–80%) and a scarcity of erythroid precursors (1% by manual count). In the immunohistochemical staining, the rare erythroid serial's element was observed with E-cadherin. Granulocytic and megakaryocytic lineages were normal in the bone marrow. The patient whose bone marrow biopsy was compatible with PRCA was entirely discontinued from receiving niraparib medication (Figure 1). Antinuclear antibody (ANA) test, serum complement levels, and romatoid factor (RF) were performed to exclude autoimmune etiologies, but no abnormalities were detected. Epstein–Barr virus (EBV), cytomegalovirus (CMV), parvovirus B-19, HIV, and hepatitis B and C were evaluated to exclude viral infections, and all were found negative with PCR and immunoglobulin. Initially, no treatment was planned for PRCA. A close hemogram follow-up was performed to determine RBC transfusion needs and red cell aplasia control. Since the RBC transfusion requirement persisted for 1 month, once or twice every week, the patient was administered a dose of 1 mg/kg of methyl prednisolone. One month after the initiation of methyl prednisolone, the hemoglobin level began to improve, and transfusion dependence disappeared. Following 1 month on 1 mg/kg of methylprednisolone, the dose was gradually reduced. Table 1 and Figure 2 show the laboratory parameters of the patient and the graphic of hemoglobin levels, respectively.
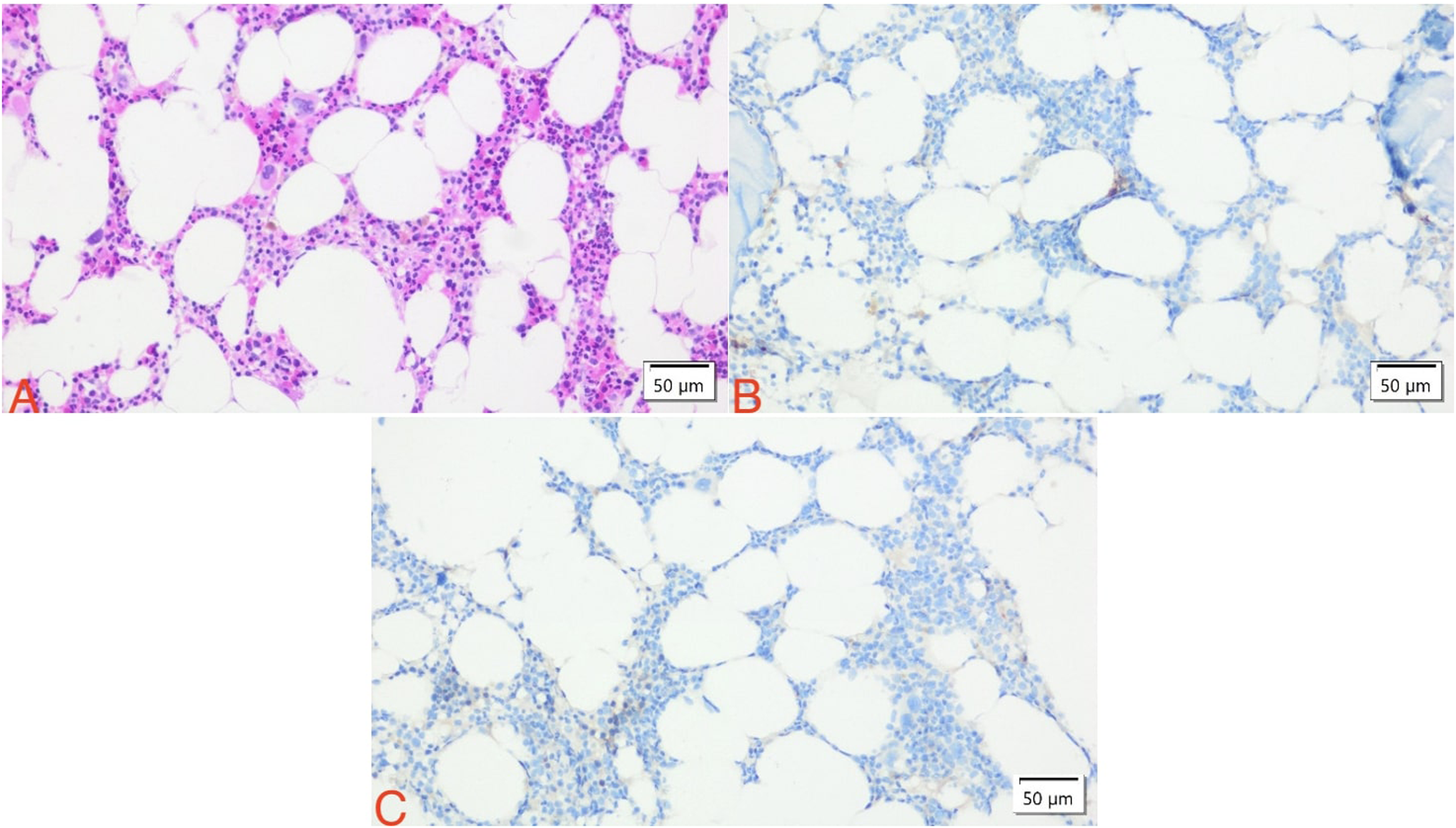
Bone marrow biopsy. (A) Megakaryocytes and myeloid cells at different maturation stages are present. However, erythroid cells are not seen (H&E 200×). (B) No erythroid precursor cells stained by E-cadherin immunohistochemistry (E-cadherin 200×). (C) No erythroid cells stained by CD71 immunohistochemistry (CD71 200×).
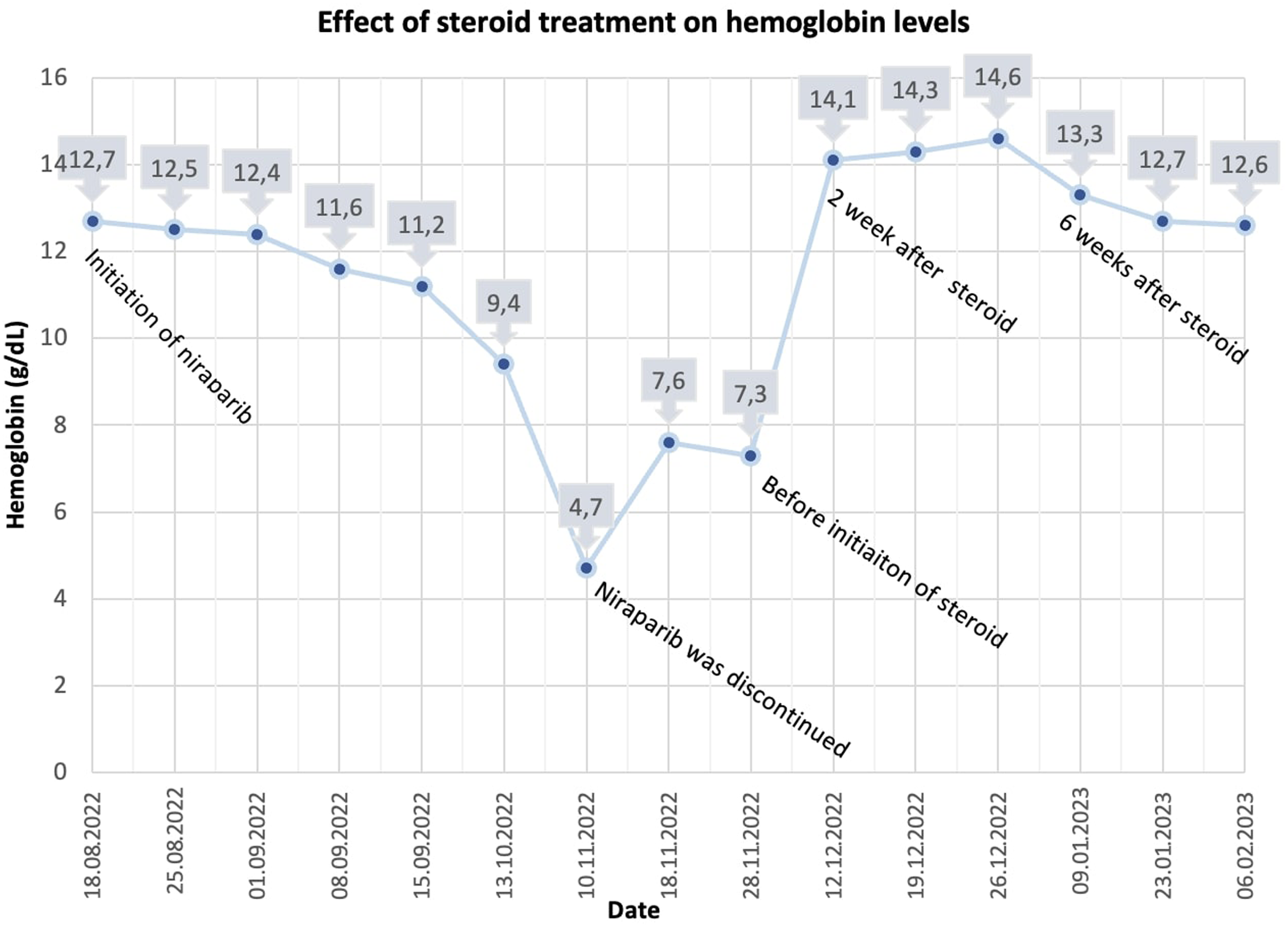
The trend of hemoglobin levels from the initiation of niraparib through the post-pure red cell aplasia period.
Laboratory parameters during the evaluation of anemia etiology.

Final Naranjo Score: 4 (please see Supplementary File S1).
Discussion
We reported a case of PRCA in a patient with serous tuba cancer who was treated with niraparib. As the severe anemia did not resolve and the patient required frequent RBC transfusions after discontinuation, steroid was administered, resulting in a rapid and permanent recovery.
In clinical practice, PARP inhibitors can also considerably increase the risk of organ system toxicities, including as hematological toxicities, in addition to the significant survival benefits.6,7 However, most of randomized controlled trials (RCTs) of PARP inhibitors identified clinical benefits rather than adverse events, and only a brief summary of the severe or even fatal adverse events was provided.8–10 In a recently published meta-analysis of 29 randomized control trials of PARP inhibitors hematological toxicity, it was reported that niraparib caused more hematological toxicity than other PARP inhibitors. This may be due to the stronger trapping effect of niraparib. 11 Additionally, PARP1 stimulates cell differentiation in the bone marrow or blood system, while PARP2 controls erythropoiesis. 12 According to preclinical studies, PARP inhibitors may cause hematological toxicity by suppressing the expression of PARP1 and PARP2. 13 The hematological mechanism of the PARP enzyme system in humans remains unclear; therefore, additional research is required. Furthermore, it is still unknown whether hematological toxicity is more frequent in BRCA-mutant patients than in BRCA wild-type patients.
PRCA is a condition characterized by normocytic, normochromic anemia with severe reticulocytopenia and a substantial reduction or absence of erythroid precursors in the bone marrow.14,15 Primary PRCA accounts for 40% of all PRCAs, and it is an idiopathic autoimmune disorder. Immune attack of erythroid precursor cells may be caused by an autoantibody or other autoimmune process. Secondary PRCAs, the most common subtype of PRCAs, include systemic lupus erythematosus, chronic lymphocytic leukemia or big granular lymphocyte leukemia, infection with parvovirus B-19, thymoma, and other solid malignancies, drugs, and toxic agents.16,17 Several drugs, including recombinant erythropoietin, diphenylhydantoin, fludarabine, and azathioprine, have been implicated as potential causes of PRCA.18,19 PARP inhibitor associated PRCA has not been reported in the literature to date. To our knowledge, our case report of niraparib-associated PRCA with a permanent response to immunosuppressive therapy is unique in the literature.
Complete withdrawal of the causative factor is the mainstay treatment for secondary PRCA. 20 Intensive immunosuppressive treatments achieve and maintain remission in most patients. Corticosteroids have been used to treat PRCA and have been regarded as the first-line therapy. 21 Although the response rate to steroid treatment ranges from 32% to 60%, the median duration of response to steroid treatment is 2 to 5 weeks. Relapse occurs in most patients with steroid dose reduction, in which case cyclosporine or cyclophosphamide is used. 22 If our patient experienced a relapse after dose reduction, cyclosporine would be administered as an alternative treatment. Additional treatment options such as cyclophosphamide, azathioprine, and intravenous immunoglobulin would be appropriate treatment alternatives if our case report became a refractory PRCA.
Conclusion
In daily practice, it should be kept in mind that in the case of refractory anemia induced by niraparib, the underlying cause might be PRCA and can be improved with steroid administration.
Supplemental Material
sj-tif-1-opp-10.1177_10781552231197808 - Supplemental material for Niraparib-induced pure red cell aplasia
Supplemental material, sj-tif-1-opp-10.1177_10781552231197808 for Niraparib-induced pure red cell aplasia by Kadriye Bir Yücel, Seyma Yıldız, Osman Sütcüoglu, Fatma Sena Güvercin, Pınar Uyar Göçün, Nuriye Özdemir, Ozan Yazıcı and Ahmet Özet in Journal of Oncology Pharmacy Practice
Footnotes
Author contributions
All authors meet the ICMJE authorship criteria. All authors have seen and approved the final version of the manuscript and contributed significantly to the work. Material preparation, data collection and analysis were performed by KBY, OS, FSG, PUG. The first draft of the manuscript was written by KBY, OS. Supervision was done by NY, OY, AÖ. All authors conducted the literature review and commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent to publish the case report was obtained from the patient.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
