Abstract
Introduction
Erlotinib is the main stay treatment of non-small cell lung cancer (NSCLC) in recent years. Though, interstitial lung disease following erlotinib use is rarely develop, it is a fatal adverse event if not immediately treat.
Case Report
We report the first case of erlotinib-induced interstitial pneumonitis with successful readministration. A Thai 64-year-old male patient with NSCLC, information includes patient presentation, laboratory findings, chest x-ray, computed tomography (CT) of chest, corticosteroid dose and duration.
Management & Outcome
The patient readministrated erlotinib after developed interstitial pneumonitis 3 weeks without developing second adverse event. Evaluation of disease after 2 months of treatment is stable disease per RECIST v1.1.
Discussion
We assumed that the mechanism for interstitial pneumonitis are diverse and some mechanism is not related with drug directly but rather transient condition and the drug can be readministrated without developing second adverse event. This could lead to change in practice of erlotinib readministration in the future.
Keywords
Introduction
Lung cancer is categorized as one of the most leading causes of death from malignancies. An estimate of 2 million patients were diagnosed with lung cancer and 1.8 million died from lung cancer. The rising incidence of lung cancer and its mortality are due to an increasing access of tobacco and industrialization in developing nations. 1 The challenge of diagnosed lung cancer is to detect it at an early stage as it has a poor prognosis and it is often incidentally found in the study of other diseases. Nowadays, there are various types of treatment for lung cancer such as surgery, chemotherapy, radiation therapy and targeted therapy which depend on staging of the disease. 2
Around 40%−80% of patients with non-small cell lung cancer (NSCLC) were presented with epidermal growth factor receptor inhibitors (EGFR) overexpression which is associated with a poor prognosis. Thus, the use of targeted therapy becomes significant. 2 Erlotinib is EGFR tyrosine kinase inhibitor drug which was approved by the FDA for specifically treated NSCLC with EGFR mutation in 2016. It was approved as a first-line treatment for patient with EGFR exon 19 deletions and exon 21 substitution mutation.3,4 The adverse side effect of erlotinib is similar to other drugs in EGFR TKI family, the most common side effects are diarrhea (20.3%) and rash (49%). The serious adverse effects events are very low but it must be noted that the use of erlotinib can resulted in acute renal failure and renal insufficiency, cardiac arrhythmia (if take erlotinib with gemcitabine), hepatotoxicity and hepatorenal syndrome, INR elevation (if take erlotinib with warfarin), exfoliative skin disorders, GI perforation, corneal perforation, cardiovascular events and hematologic adverse reaction in patient with comorbid pancreatic cancer and interstitial lung disease.5,6
This report presents the case of a middle-aged patient, with recent diagnosis of advanced staged adenocarcinoma of the lung (T3N3M1) with brain metastasis. Patient was treated with erlotinib (150 mg PO/day) as a first-line treatment. After 2 weeks of erlotinib, the patient presented with dyspnoea, dry cough, and diffuse ground glass opacity of both lungs which was diagnosed with interstitial lung disease. It was suspected to be Erlotinib-associated interstitial lung disease.
Case report
A 64-year-old man with no significant past medical history presented with progressive right hemiparesis for 9 days prior to admission. The sensation loss is prominent on the upper body than lower body. Despite this, patient also complained of a history of poor appetite and 2-kg weight loss over a month. Patient denied a history of nausea, vomiting, and headache. Physical examination showed motor power grade IV at both upper and lower extremities of right sided, no cervical, axillary, or inguinal lymphadenopathy was observed. Patient was sent for magnetic resonance imaging (MRI) of the brain and computed tomography (CT) of the chest for further investigation. CT of the chest revealed several bilateral paratracheal nodes and few bilateral hilar nodes up to 0.8 cm. The largest mass on the right lung is 4.0 × 4.3 × 3.6 cm and the largest nodule on the left lung size 2 cm which suggests multiple pulmonary metastasis. MRI brain revealed multiple nodular and rim-enhancing lesions at bilateral hemisphere with the size of 1.3 × 1.4 cm at left centrum ovale, 1.4 cm right parietal, and 1.6 × 1.1 cm at right occipital region and right cerebellar hemisphere. Histopathology of sample obtained via transbronchial biopsy showed non-small cell lung cancer (NSCLC) favoring adenocarcinoma. Mutation analysis of the sample was positive for EGFR mutation (exon-19 deletion). Patient was diagnosed with advanced staged adenocarcinoma of the lung (T3N3M1) with brain metastasis and was sent for whole brain radiotherapy (WBRT) doses of 30Gy. Soon after completion of whole brain radiation, the patient was starting on erlotinib 150 mg per day.
Fourteen days after starting Erlotinib, patient presented at Emergency department due to dyspnoea and dry cough for 2 days. Physical examination was body temperature 39.3°C, blood pressure 124/78 mmHg, pulse rate 95 beats/min, respiratory rate 20/min, oxygen saturation 87% room air which can be corrected with oxygen canula 3 LPM. Chest examination revealed diffuse rhonchi in both lung fields. A chest radiography showed patchy infiltration at left upper lung and right middle lung without pleural effusion (Figure 1). Initial investigation revealed haemoglobin 11.7 mg/dL, white blood cell 6,690, neutrophil 87%, lymphocyte 9%, platelet 131,000, BUN 20, creatinine 0.76, sodium 128, potassium 3.4, chloride 98, and bicarbonate 21. Two bottles of hemoculture were taken and empiric treatment with antibiotics was administrated. Dose of ceftazidime and azithromycin intravenous were given 2 g every 8 h and 500 mg every 24 h for 5 days, respectively. Erlotinib was continue during admission. Within 24 h, the patient developed respiratory failure which required endotracheal intubation and mechanical ventilation. Repeat chest radiography revealed diffuse markedly increased ground-glass infiltration of both lungs (Figure 2). Arterial blood gas after intubation showed pH 7.49, PCO2 30.1 mmHg, PO2 138.3 mmHg, BE 1.0 mmol/L. Ceftazidime was changed to piperacillin/tazobactam 4.5-g intravenous every 6 h. Erlotinib was discontinued due to suspected Erlotinib-associated pneumonitis and dexamethasone 5 mg intravenous every 6 h was started. Computed tomography (CT) scan revealed diffuse peribronchovascular patchy ground-glass opacity in both lungs with superimposed interlobular and intralobular septal thickening (crazy-paving pattern) in bilateral apical lungs without evidence of acute pulmonary embolism. There is moderate amount of bilateral pleural effusion (Figure 3). Laboratory evaluation revealed troponin T of and pro-BNP of 2143 pg/mL. He gained full conscious after 1 day of dexamethasone. Intravenous dexamethasone was given for 4 days then switched to oral prednisolone 45 mg/day. The patient can extubate after 6 days and on room air with oxygen saturation 99%. Hemocultures returned to no growth and antibiotics were discontinued after 7 days. He was discharged late on day 15 due to rehabilitation program with prednisolone 10 mg/day without oxygen required.
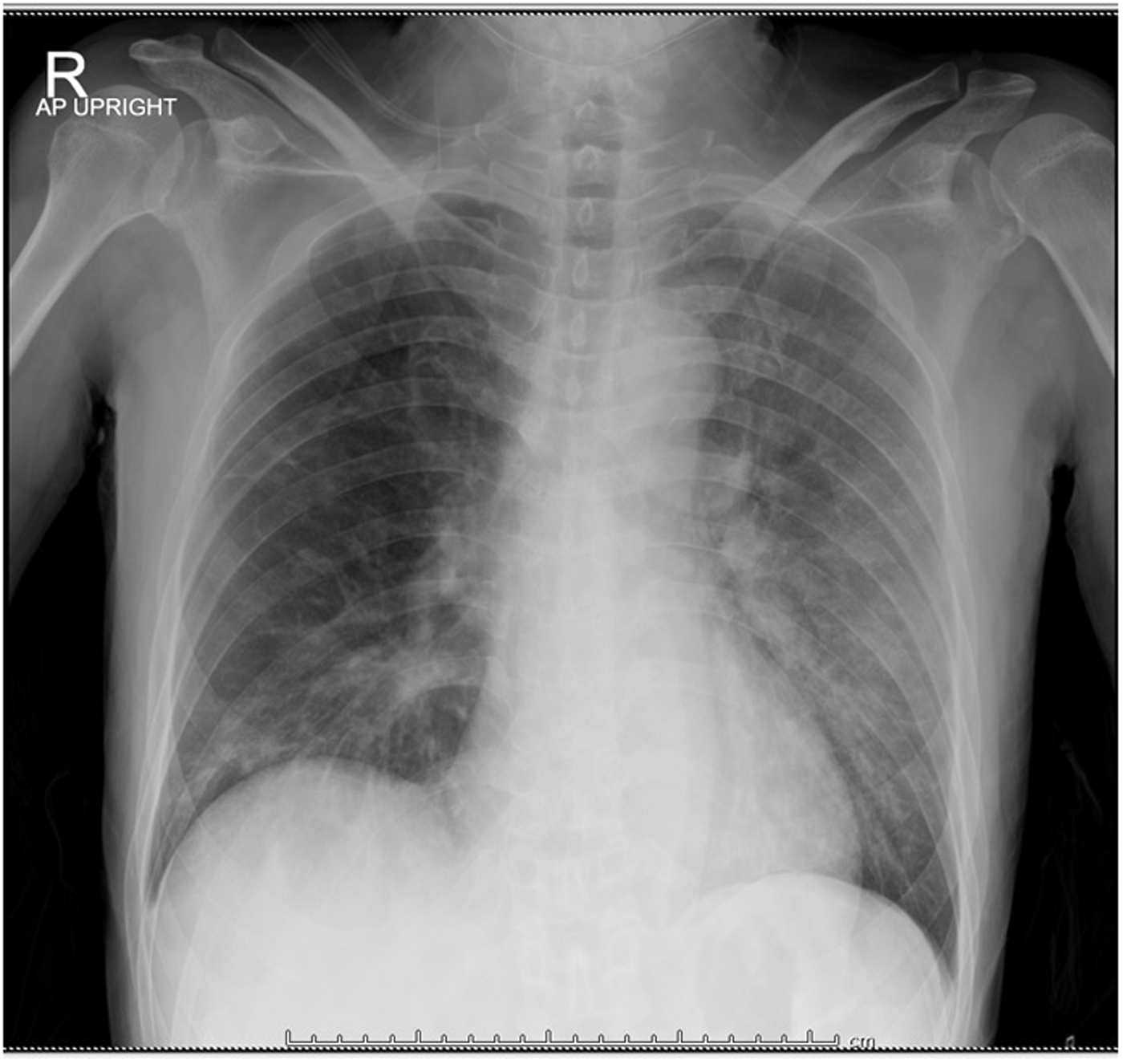
Chest X-ray showed patchy infiltration at left upper lung and right middle lung.
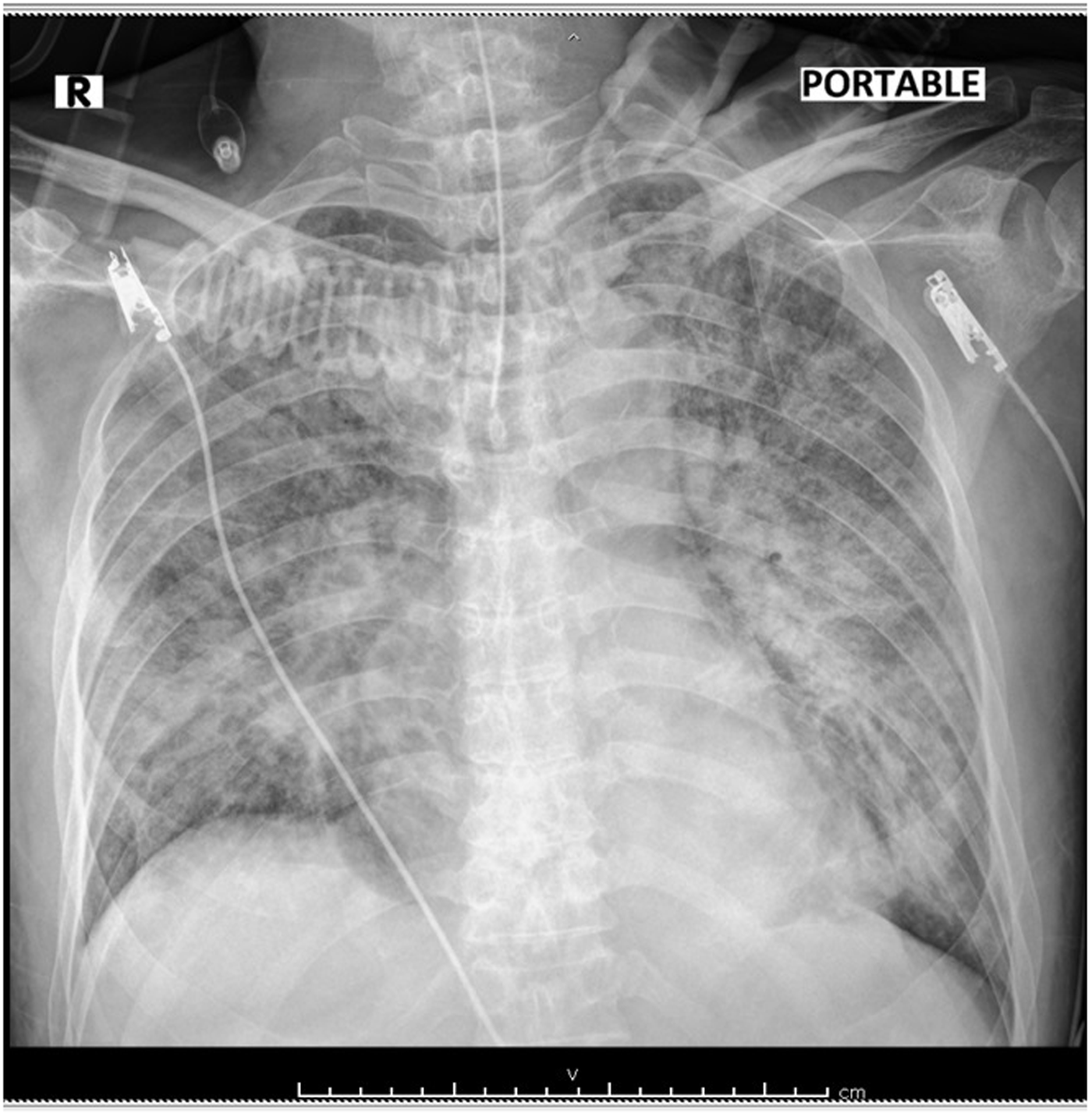
Chest X-ray showed ground-glass infiltration of both lungs.
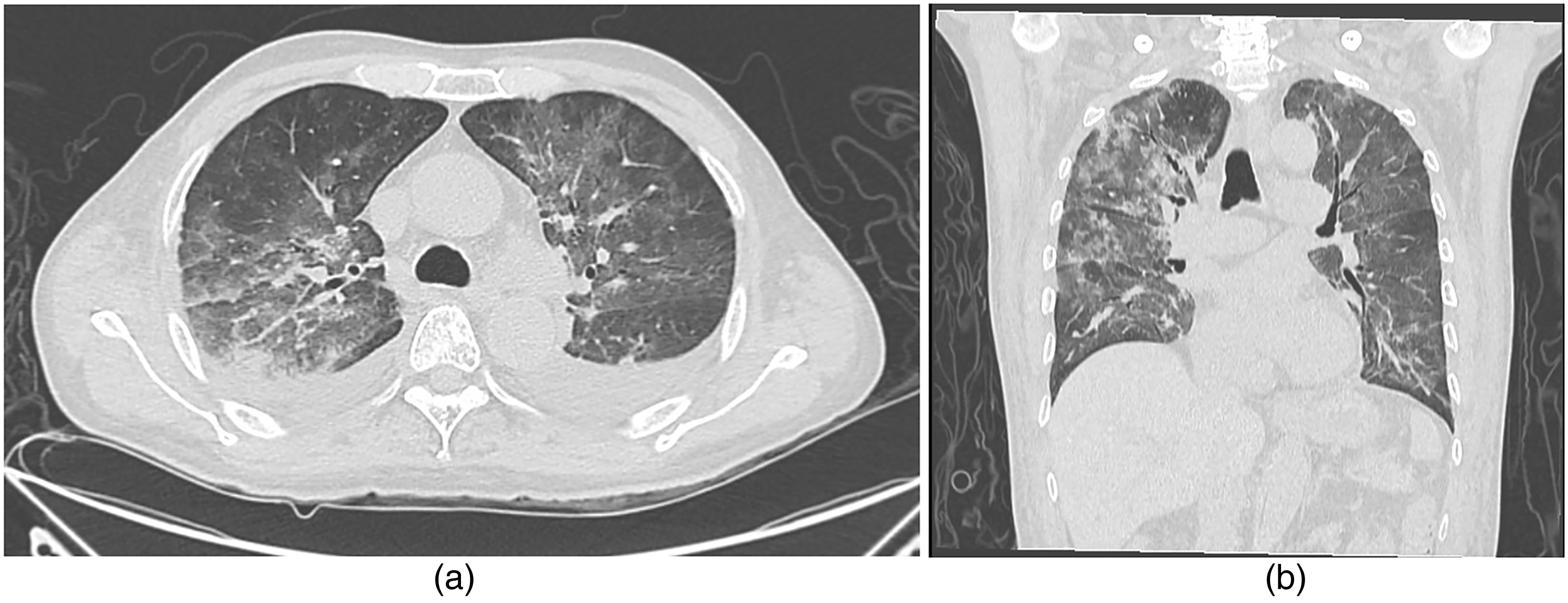
CT scan showed diffuse peribronchovascular patchy ground-glass opacity in both lungs.
Management & outcome
Due to limited coverage of insurance over certain targeted therapy, patient and relatives decided to continue the erlotinib under close observation after discussion of the risks and benefits of treatment. Erlotinib 150 mg per day was restarted 7 days after the patient was discharged at oncology clinic without complication. CT chest at 2 months revealed decreased size of malignant lesion and its adjacent pleural involvement in posterior basal segment of RLL, size 3.0 × 3.4 × 2.9 cm without pleural effusion, compatible with stable disease per RECIST v1.1.
Discussion
In recent years, targeted therapy has become the choice of treatment that is preferred and used more widely in cancer patients. Erlotinib is the first-line treatment for NSLC patient with EGFR exon 19 deletions and exon 21 substitution mutation. 3 There are several reports on the adverse reactions of erlotinib. Erlotinib-associated with fatal pulmonary toxicity was first reported in 2007.6,7 In 2008, Lind reported the case of two patients who were diagnosed with fatal interstitial lung disease after administration of erlotinib for 4 to 6 weeks which cause both to die from diffuse alveolar hemorrhage. 8 In 2010, Del Castillo Y reported a case of interstitial pneumonitis with successful treatment of corticosteroid. 9 In 2013, Wang reported a case of potential gefitinib and erlotinib inducing ILD. 10 Mangla A reported a case of interstitial disease after erlotinib use for 3 weeks in 2016. 11 Other than erlotinib, there are other tyrosine kinase inhibitor drugs that are associated with pulmonary toxicities such as imatinib and gefitinib. 12 Diagnosed mainly by history taking, clinical presentation, and radiological features which ruled out other potential causes such as pulmonary embolism and heart failure.
We report the first case of erlotinib-associated interstitial pneumonitis with successful rechallenging without second interstitial pneumonitis. The success may relate to the short timing of intravenous corticosteroid and erlotinib cessation within 48 h of hospitalization. After 2 months of re-administration, the disease evaluation is stable disease per RECIST v1.1.
Although the molecular mechanism of erlotinib-induced interstitial lung disease remains unknown, several significant risk factors have been identified such as male gender, history of smoking, coexistence pulmonary disease or pre-existence of pulmonary fibrosis, history of chemotherapy, and poor PS. Tsubata also describes association between extreme elevation of the erlotinib plasma concentration and the adverse events. 13 We assumed that the mechanism for interstitial pneumonitis is diverse and some mechanisms are not related with drug directly but rather a transient condition and the drug can be readministrated without developing second adverse event. This could lead to change in practice of erlotinib readministration in the future.
Conclusion
This case describes successful report of readministration of erlotinib after developing interstitial pneumonitis in a patient with advanced staged adenocarcinoma of the lung (T3N3M1) with brain metastasis.
Footnotes
Author contributors
ST wrote the first draft of the manuscript. NB researched literature and revised the manuscript. JJ participated in data collection and analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The patient and his family were informed about this publication and consent form was signed.
