Abstract
Background
Cytokine release syndrome (CRS) and immune effector cell-associated neurologic syndrome (ICANS) are well-documented toxicities of CAR T-cell therapy. To mitigate excessive toxicity, our center has formulated treatment protocols (early vs. standard) for timely management of CRS and ICANS with tocilizumab and/or corticosteroids.
Methods
This retrospective, single-center analysis included patients treated with CAR T-cell therapy. The goal was to describe the association of two management protocols with toxicity and efficacy outcomes.
Results
Fifty-five percent of the 40 patients assigned to early management, out of which 5% and 9% developed grade 3+ CRS and ICANS, respectively. Seventy-seven percent and 41% of these patients received tocilizumab and corticosteroids, respectively. Forty-five percent of patients were stratified as standard management, out of which 0% and 11% developed grade 3+ CRS and ICANS, respectively. Seventeen percent and 28% of these patients received tocilizumab and corticosteroids, respectively. The day +90 overall response rate (ORR) for all patients was 63%, with an ORR of 89% for those managed per early management versus 50% for those managed per standard protocol.
Conclusion
Early use of tocilizumab and corticosteroids is effective in preventing excessive CAR-T-related toxicities with no negative impact on efficacy.
Introduction
Chimeric antigen receptor (CAR) T-cell therapy has emerged as an important treatment option for patients with relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma (MCL), follicular lymphoma (FL), acute lymphoblastic leukemia (ALL), and multiple myeloma (MM).1–8 There are currently four CD19 targeting CAR T products approved for ALL and lymphoma and two B-cell maturation agent (BCMA) targeting CAR T products approved for R/R multiple myeloma.
Two common and potentially severe treatment-related adverse events are cytokine release syndrome (CRS) and immune effector cell-associated neurologic syndrome (ICANS). 9 CRS, characterized by fevers and fatigue, has been reported in 35–100% of patients treated with CAR T and may be severe (grade 3 or higher) in nearly 25% of patients.3,6,10–13 Severe cases of CRS may lead to vasodilatory shock, hypoxia, end-organ dysfunction, and ultimately death. First-line therapy for CRS outside of supportive care is anti-cytokine-directed therapy, most commonly tocilizumab, an interleukin-6 receptor antagonist. ICANS presents with symptoms ranging from mild delirium to seizures and has been reported in 10–50% of recipients of CAR T-cell therapy.3,6,10–13 Tocilizumab is ineffective in the amelioration of neurotoxicity (unless associated with CRS) as it cannot readily cross the blood–brain barrier; therefore, corticosteroids are considered front-line therapy due to their anti-inflammatory effects.
Literature regarding optimal timing of CRS and ICANS management is sparse and institutional management strategies vary. A single-center study by Caimi et al., suggests that the use of prophylactic tocilizumab may decrease the overall incidence and severity of CRS. 14 Results of cohort 4 in the ZUMA-1 trial demonstrate that early or prophylactic use of corticosteroids may decrease incidence of grade 3+ CRS and ICANS as well as overall steroid exposure when compared to those who did not receive corticosteroids until clinically indicated for the management of ICANS.15,16 Decreasing a patient's overall exposure to tocilizumab and corticosteroids may be beneficial as the long-term implications of the use of anti-inflammatory agents for the treatment of CRS and ICANS are not well understood. Concerns remain that the use of corticosteroids may induce T-cell apoptosis or suppress T-cell function, ultimately decreasing the effectiveness, durability, and persistence of CAR T-cells.3,5,10,16
The balance between the safety and efficacy of tocilizumab and corticosteroids use continues to be an area of interest. As an attempt to mitigate CAR T-related toxicities while assessing the optimal timing of intervention, our center has formulated treatment protocols (Early vs. Standard) for timely management of CRS and ICANS with tocilizumab and corticosteroids. We conducted a retrospective analysis of effect of the Early and Standard treatment protocols on the safety and efficacy of CAR T-cell therapy.
Material and methods
We performed a single-center retrospective chart review of patients with various hematologic malignancies (non-Hodgkin lymphoma, ALL, multiple myeloma) treated with CAR T-cell therapy over a three-year period from March 2019 through March 2022 at the Medical University of South Carolina (MUSC). All patients received lymphodepleting chemotherapy with fludarabine and cyclophosphamide prior to CAR T-cell infusion. Patients, if infused outpatient, were assessed daily for the first 30 days post CAR T infusion and admitted to the inpatient unit for monitoring either on day of infusion or if/when toxicities (CRS/ICANS) occurred. Prior to CAR T-cell infusion, patients were risk stratified for either Early versus Standard (EM and SM) toxicity management (Table 1) based on known risk factors for increased toxicity including toxicity profile of the product, CAR T construct/costimulatory domain, and disease burden (as determined by elevated ferritin and lactate dehydrogenase (LDH) biomarkers), per physician discretion. The primary difference between the EM and SM protocols is the approach toward grade 1 CRS and ICANS. EM protocol recommends tocilizumab (8 mg/kg, maximum of 800 mg/dose) if grade 1 CRS persists for 24 h whereas SM protocol recommends the first dose of tocilizumab at 72 h of persistent symptoms. EM protocol recommends dexamethasone 10 mg be given at the first sign of grade 1 ICANS whereas SM protocol allows the physician to “consider” dexamethasone but does not recommend a dose be routinely given until grade 2. The details of both EM and SM protocols are presented in Table 1.
Early versus standard management protocol for CAR T-related toxicities.

CRS: cytokine release syndrome; toci: tocilizumab; CAR T: chimeric antigen receptor T; IL-6: interleukin-6; IV: intravenous; ATG: anti-thymocyte globulin.
We evaluated the association of protocol selection and steroid use with toxicity, described as the incidence and severity of CRS and ICANS as graded by the American Society for Transplantation and Cellular Therapy consensus criteria, and efficacy as determined by overall response rate (ORR), complete response (CR), and treatment-related mortality at day +30 and +90 post-CAR T infusion. 17 The time to first dose of tocilizumab or corticosteroids and the cumulative exposure to each agent are also described.
Results
A total of 40 patients were treated with CAR T-cell therapy over the three-year period. A majority of patients had a lymphoma diagnosis and an Eastern Cooperative Oncology Group performance status of 0–1. Half of the patients received a CD28 costimulatory domain product. In the entire patient population, more patients with multiple myeloma were treated per EM versus SM protocol (27% versus 5%, respectively). Additionally, more (64% vs. 33%) patients in the EM group received a CD28 CAR T product as compared with a 41BB product (64% v 33%). In contrast, more patients in the SM group received a 4-1BB product (89% vs. 55%). There were no other significant differences between the two groups at baseline. Detailed patient characteristics are presented in Table 2.
Patient characteristics.
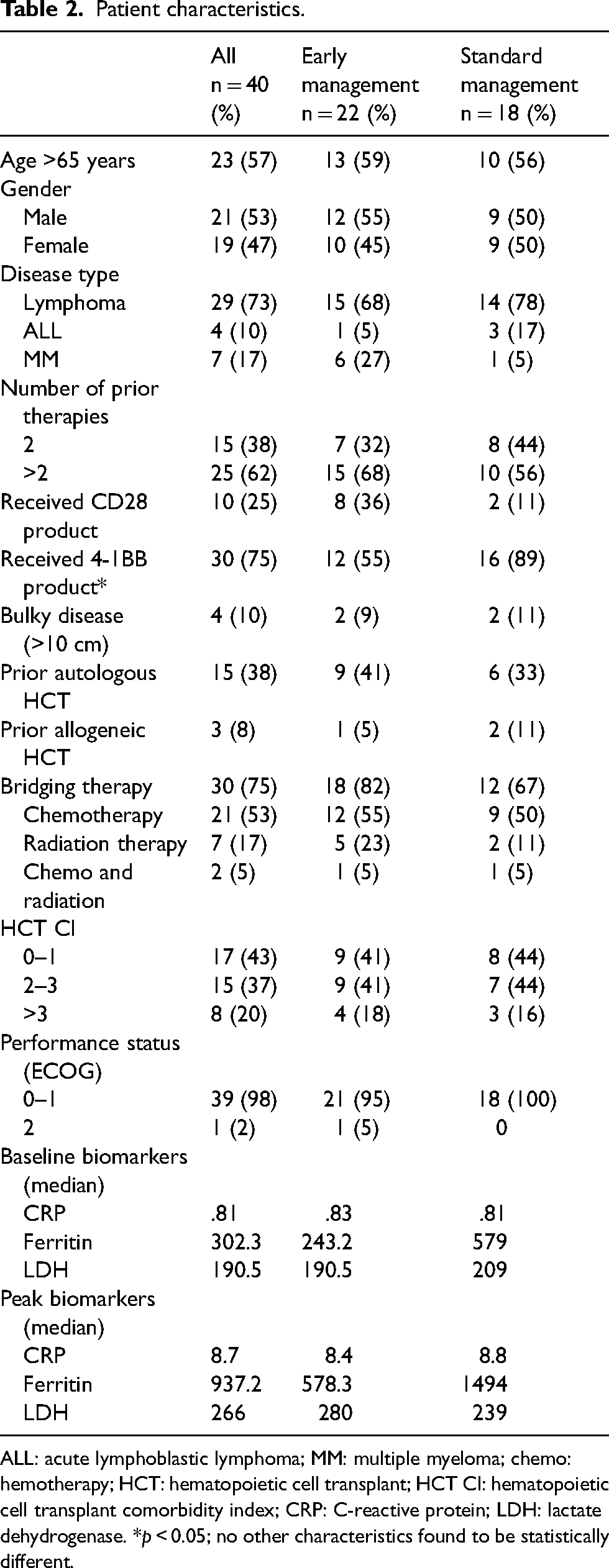
ALL: acute lymphoblastic lymphoma; MM: multiple myeloma; chemo: hemotherapy; HCT: hematopoietic cell transplant; HCT CI: hematopoietic cell transplant comorbidity index; CRP: C-reactive protein; LDH: lactate dehydrogenase. *p < 0.05; no other characteristics found to be statistically different.
The overall incidence of CRS was 75% of which 3% were considered severe (grade 3+). Half of patients received tocilizumab with a median time to first dose of two days (range 1–8 days). One patient received siltuximab (an interleukin-1 antagonist) for refractory CRS. The overall incidence of ICANS was 33% of which 10% were severe (grade 3+). Thirty-five percent of patients were managed with corticosteroids with a median time to first dose of six days. Two patients received anakinra (an interleukin-1 receptor antagonist) for refractory ICANS. There were no major differences in the incidence or severity of CRS or ICANS between those treated with EM versus SM protocols. A higher number of patients treated per EM protocol received tocilizumab than those treated per SM protocol (77% vs. 17%). Although the median time to first dose of tocilizumab was similar between the two groups (two days), the median number of tocilizumab doses received by those in the EM protocol was 2, as compared with 3 in the SM protocol. Similarly, a numerically higher number of patients treated per EM protocol received corticosteroids for ICANS versus those treated by SM protocol (41% vs. 28%). However, among patients who required corticosteroid intervention, those treated per EM protocol received less cumulative corticosteroids (40 mg dexamethasone equivalent vs. 103 mg) than the SM group.
Day +30 ORR and CR rates were better in those treated by EM protocol (77% vs. 56% and 64% vs. 33%, respectively). Day +90 ORR and CR rates for all evaluable patients were 63% and 50%, respectively. Day +90 ORR and CR rates were numerically higher for patients treated per EM versus SM protocol (89 and 50%, p = 0.14 vs. 61% and 42%, respectively). There was no major difference in survival or relapse-free survival between groups (Figures 1 and 2).
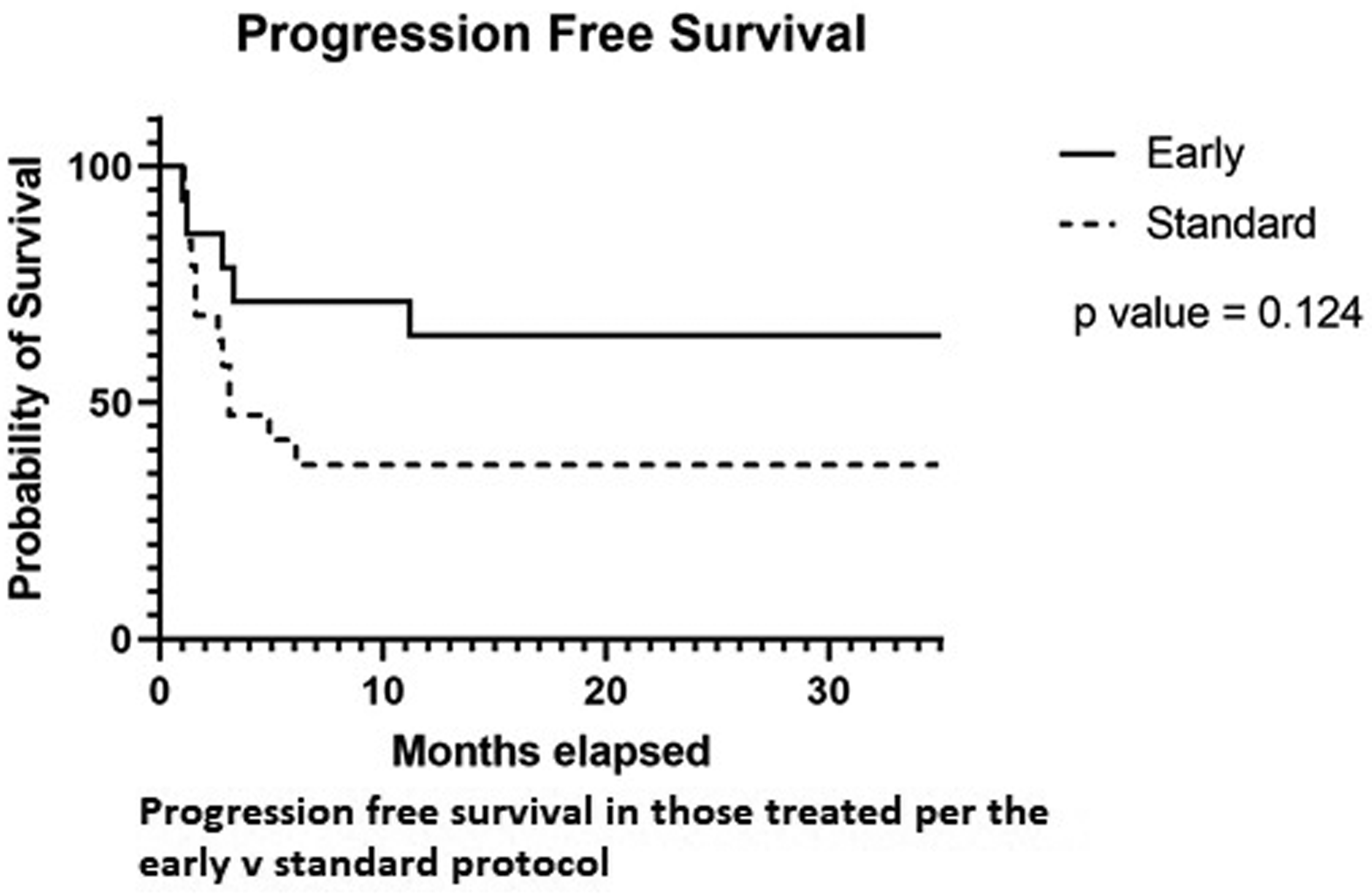
Progression free survival.
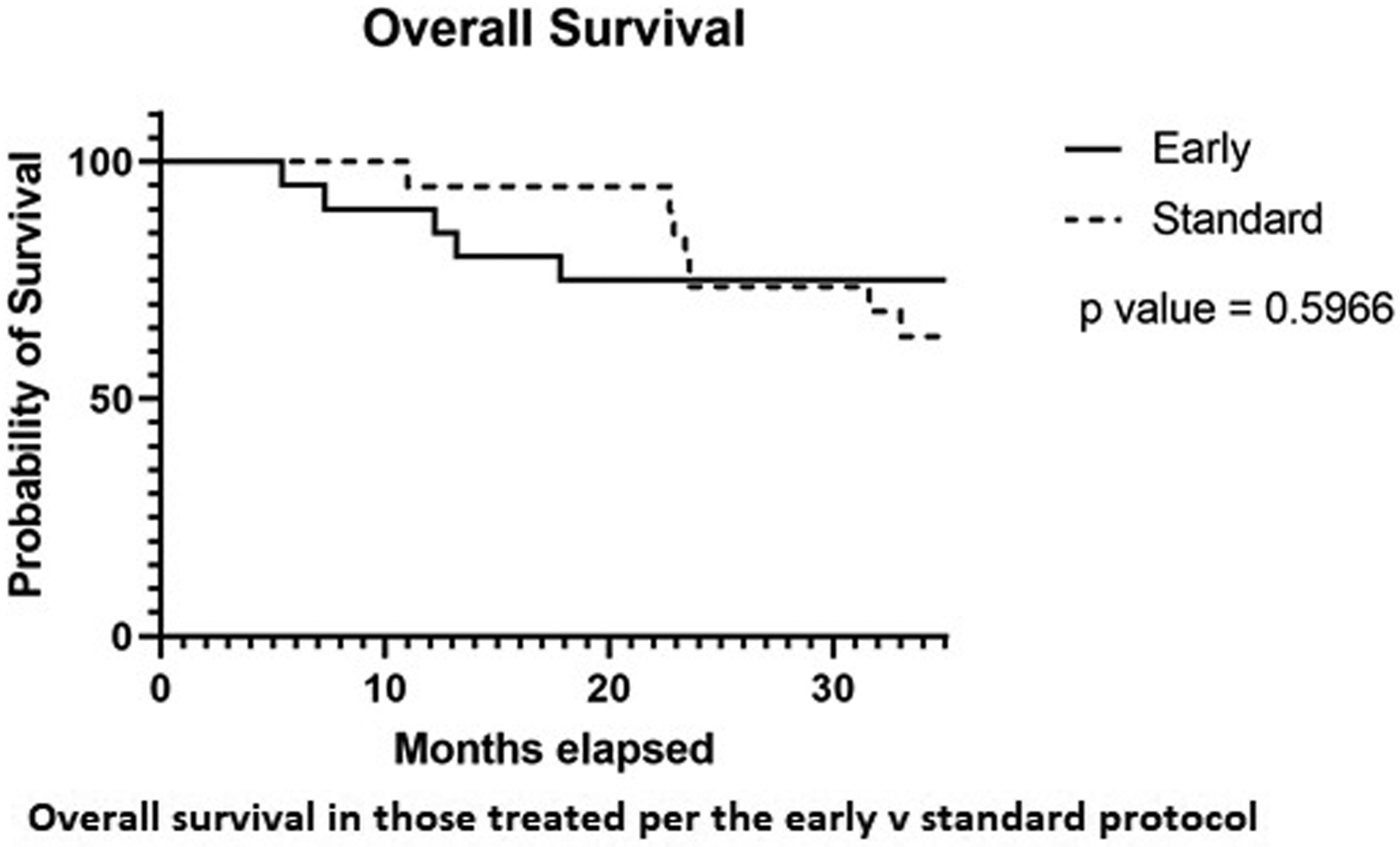
Overall survival.
No other significant differences in outcomes were observed between those managed per EM versus SM protocol including length of hospital stay and incidence of intensive care unit transfer. Additional details of toxicity and efficacy outcomes are presented in Table 3.
Safety and efficacy outcomes in patients with early and standard management of CAR T-related toxicities.
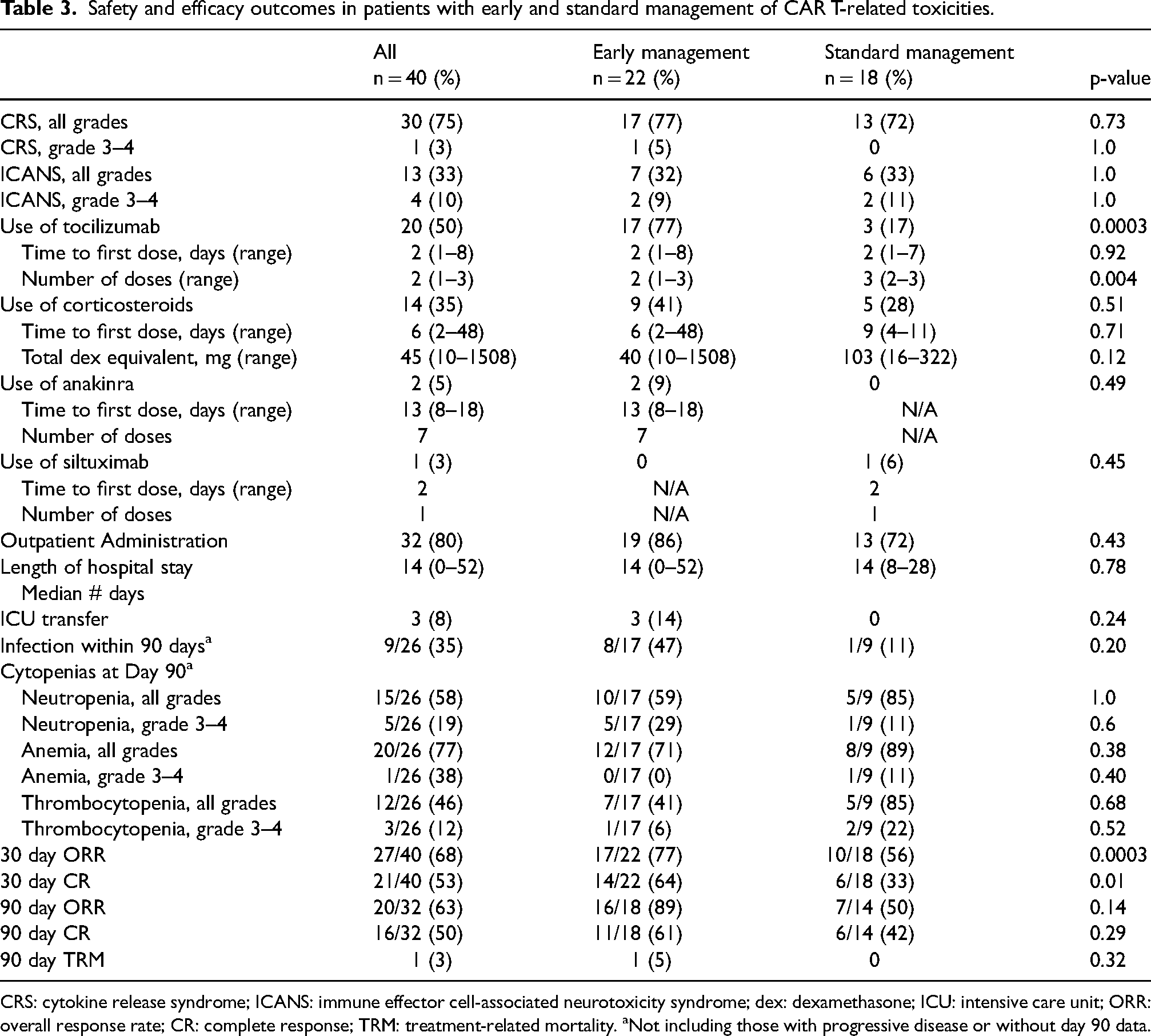
CRS: cytokine release syndrome; ICANS: immune effector cell-associated neurotoxicity syndrome; dex: dexamethasone; ICU: intensive care unit; ORR: overall response rate; CR: complete response; TRM: treatment-related mortality. aNot including those with progressive disease or without day 90 data.
There was a major difference in the incidence of both CRS and ICANS, as well as the severity of ICANS, between those who received corticosteroids versus those who did not (CRS: 100% vs. 62%, and ICANS: 71% vs. 12%, respectively). A higher number of patients who required corticosteroid treatment also required more tocilizumab than those who did not receive corticosteroids (86% vs. 31%). There was no significant difference between groups for other toxicity management strategies such as anakinra or siltuximab. Those who required corticosteroid treatment had longer length of hospital stay (21 days vs. 14 days) and were more likely to require an escalation of level of care through transfer to an intensive care unit (21% vs. 0%). Despite this, there was no difference in ORR, CR, or treatment-related mortality between groups. Additional details of toxicity and efficacy outcomes based on corticosteroid exposure are presented in Table 4.
Safety and efficacy outcomes in patients who received corticosteroids versus those who did not corticosteroids.
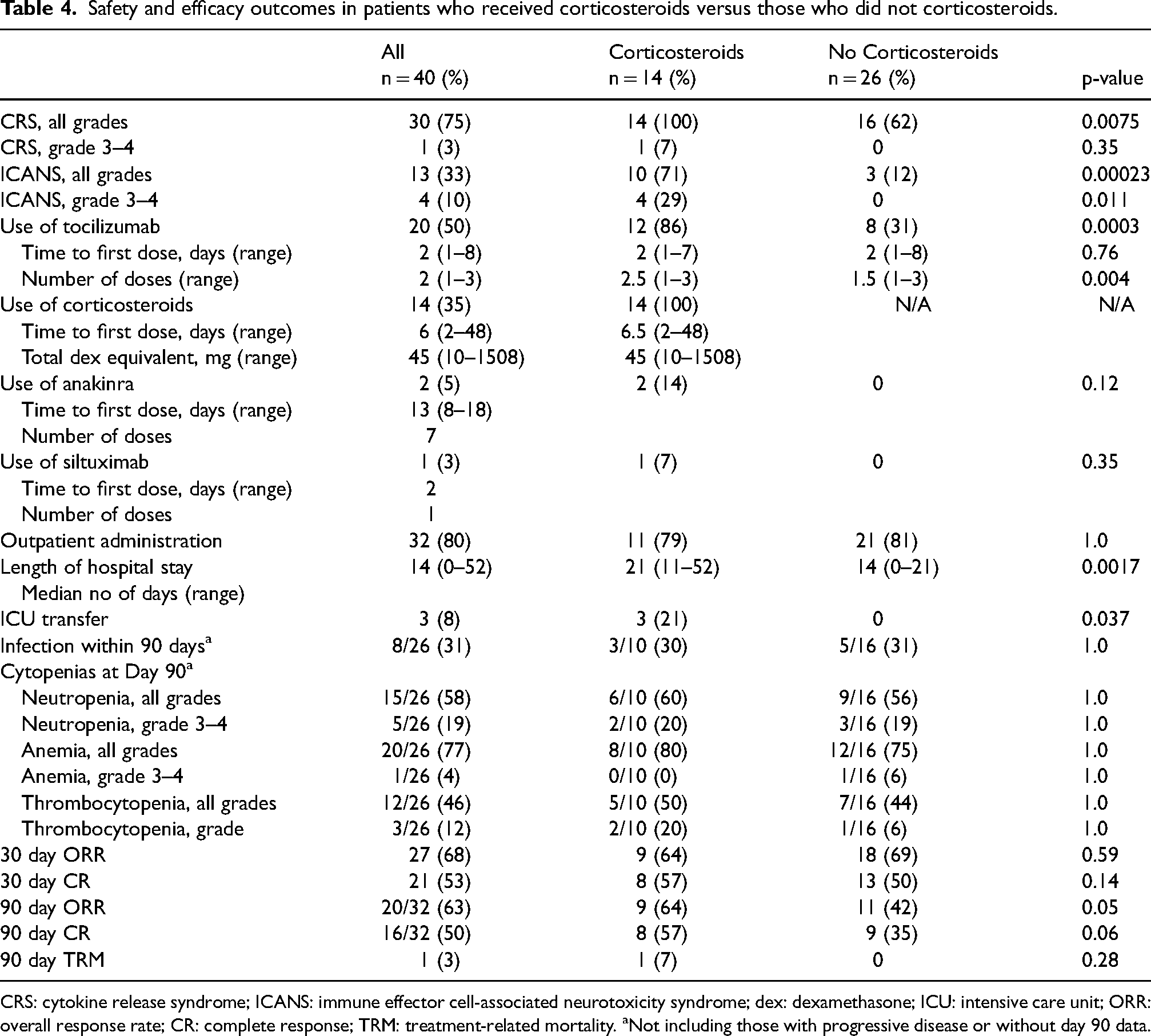
CRS: cytokine release syndrome; ICANS: immune effector cell-associated neurotoxicity syndrome; dex: dexamethasone; ICU: intensive care unit; ORR: overall response rate; CR: complete response; TRM: treatment-related mortality. aNot including those with progressive disease or without day 90 data.
Discussion
This study highlights CRS and ICANS as common toxicities after CAR T-cell therapy. With all-grade and grade 3+ CRS (75%, 3%) and ICANS (33%, 10%), the incidence of CRS and neurotoxicity reported here appear similar to other reports. In the ELIANA and JULIET trials, 58–77% of patients treated with tisagenlecleucel experienced CRS, with 22–47% experiencing grade 3 or higher toxicity.3,8 Similarly, 21–40% of patients experienced ICANS, of which approximately 10% were graded as severe (grade 3+). Other CAR T products report similar incidence and severity; for example, patients treated with axicabtagene autoleucel experienced CRS and ICANS in 93% and 28% of cases, respectively and those treated with lisocabtagene maraleucel experienced CRS and ICANS at rates of 42% and 30%, respectively.1,5,18 For patients with multiple myeloma, the KarMMa study showed that 85% of the patients experienced CRS and 18% experienced ICANS, with grade 3+ CRS or ICANS seen in less than 5% of the patients. Moreover, our center's overall use of tocilizumab and corticosteroids was also similar to that of other studies (14–48% and 9–27%, respectively). 16
It is important to highlight that patient-related factors of age greater than 65 years, comorbidity index, and performance status were equally distributed between the two groups without being associated with excessive risk of severe CRS or ICANS in either group. Numerically more patients in the EM group received bridging therapy, explaining lower baseline and peak ferritin (marker of inflammation) levels. Whether that could have a significant impact on post-CAR-T toxicities of CRS and ICANS remains an important question. Patients at higher risk for CAR T-cell associated toxicities, based on CAR T construct/costimulatory domain, were often selected to be treated per EM protocol. As these patients were inherently at a higher risk of toxicities, they were more likely to receive tocilizumab and/or corticosteroids. 19 In the absence of a multivariate analysis, it is hard to determine whether there were any statistically significant differences in incidence and severity of toxicities for patients treated with CD28 costimulatory domain on SM protocol. However, it is important to highlight here that if patients were to receive treatment with either tocilizumab or corticosteroid, those treated per EM protocol would have significantly less total exposure to each treatment versus those treated per the SM protocol. This supports the idea of a potential benefit of treating CAR T-cell-associated toxicities of CRS and ICANS at the earliest onset of symptoms rather than deferring treatment until more severe symptoms are experienced. The prophylactic or early use of tocilizumab and corticosteroids has been investigated at other centers and have not shown an increase in incidence of therapy-related toxicities or compromised response.13,19
Those treated per EM protocol were more likely to require an escalation of level of care (i.e. transfer to intensive care unit); however, this difference was small (3 vs. 0 patients) and did not translate to an extended length of hospital stay (14 days each) or higher treatment-related mortality (1 in each group). Hence, earlier use of tocilizumab and/or corticosteroids does not appear to increase the costs of care associated with CAR T-cell therapy. Those treated per EM protocol, although thought to be at higher risk of toxicities, appeared to have better ORR, including CR at day +30, compared to those treated per the SM protocol.
Despite the trend toward more frequent use of corticosteroids in the EM group, incidence of infections and cytopenias, both of which have been attributed to the use of corticosteroids after CAR T-cell infusion, was similar between the two groups (Table 4). 20 Additionally, the use of corticosteroids did not have a statistically significant effect on ORR at day +30 or day +90 post-CAR T. Use of corticosteroids was associated with longer length of hospital stay and need for intensive care, likely related to the higher incidence of grade 3+ ICANS requiring close monitoring.
There are several limitations of our analysis. First, this was a single-center retrospective study with a small number of patients. Additionally, the study population was heterogeneous and included patients with different underlying diagnoses and CAR T products. However, safety and efficacy outcomes reported here appear similar to previously reported studies in literature.10,19 Although, the EM or SM protocols were used as guidelines and physicians had the option of making clinical decisions outside of these protocols, no deviations from the institutional protocols were observed. Prospective, randomized, and multicenter studies with larger patient population are needed to support the findings of this report.
Conclusion
This analysis supports that early intervention with tocilizumab and corticosteroids may overcome the increased risk of severe CRS and ICANS associated with CAR T-cell therapy. Early use of tocilizumab and corticosteroids may also decrease the duration and dose of these medications for toxicity management. There seems to be no significant negative impact on efficacy with early management of toxicities.
Footnotes
Authors Contribution
KG and HH researched literature and conceived the study. MG, JD, KG, HH, and BH were involved in protocol development, data collection, and patient analysis. KG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Author note
Kathy Edwards is currently affiliated with Merck Corporation, NJ, USA.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Hamza Hashmi has consulted for Sanofi and BMS and received compensation. Dr Kathy Edwards has consulted for Omeros and received compensation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
