Abstract
Introduction
The complexity of treatment and extended therapy duration associated with bone marrow transplantation directly affect the psychological well-being of the patients, create anxiety, and reduce their quality of life. The aim of our study was to evaluate the quality of life of patients admitted to the bone marrow transplantation unit.
Methods
This prospective and descriptive study was conducted between January and June 2021 in an adult BMT unit in Turkey. The sociodemographic characteristics of the patients were recorded. The patient's quality of life was measured twice using the Functional Assessment of Cancer Therapy-Bone Marrow Transplantation (FACT-BMT) scale at the beginning of the study and 30 days later. SPSS 15 was used for the analysis.
Results
A total of 40 patients were included in the study. The mean age was 46 years. Most of the patients were diagnosed with multiple myeloma and 58% had at least one comorbid disease. Most of the patients (78%) were receiving myeloablative therapy. High dose melphalan regimen was the most commonly applied regimen (25%). Thrombocytopenia was the most common side effect (14%). Although there was no change in the quality of life, it was found that the social/family well-being scores increased (p < 0.05).
Conclusions
In our study, it was observed that the number of comorbid diseases was higher in patients with bone marrow transplantation. The incidence of side effects may be high in these patients. We believe that clinical pharmacists have an important role in monitoring adverse effects and improving the quality of life in bone marrow transplantation units.
Introduction
Bone marrow transplantation (BMT) is a procedure performed to treat various types of cancer and some other diseases. It is preferred in the treatment of cancers that directly affect the bone marrow, such as leukemia, lymphoma, Hodgkin's lymphoma, and multiple myeloma.1,2 It is a complex procedure, and it is associated with a lot of complications some of which are medication related. Some complications include severe and continuous bleeding, infection, liver disorders, skin rash, diarrhea, infertility, muscle spasms, numbness in the arms and legs, secondary cancers, tissue and organ rejection, graft versus host disease, and death. 2 Clinical pharmacists take their place as important members of the multidisciplinary team that provides care to hematopoietic cell transplantation patients. They play an active role in the management of antineoplastic and antimicrobial drugs, therapeutic drug monitoring, prediction and prevention of possible drug toxicities, and provide drug-related information to other team members, patients, and family members. 3
Quality of life (QOL) is defined by the World Health Organization as how individuals perceive their position in life in terms of their goals, expectations, interests, concerns, and living standards in the context of their culture and the value systems within which they live. 4 The presence of serious diseases that need complex therapy regimens affects the QOL of both patients and others around them. Assessing and improving the QOL of the patients at different stages of their treatment will also increase the quality of (treatment) care. The Functional Assessment of Cancer Therapy-Bone Marrow scale (FACT-BMT) 5 is a reliable measurement system used in clinical and academic research to determine the QOL in patients with BMT.
The aim of our study is to evaluate the QOL of patients treated in adult the BMT unit and determine possible predictive factors that affect patients’ QOL.
Methods
This prospective cross-sectional study was approved by the Marmara University Faculty of Medicine Clinical Research Ethics Committee with the protocol numbered 09.2020.1374.
This study was conducted in the “Adult Bone Marrow Transplant Unit” of a private hospital in Istanbul between 1 January 2021 and 31 July 2021. Adult patients >18 years patients who gave approval and signed the consent forms admitted to the BMT unit were included. In our study, 40 patients with BMT treatment were included in the clinic.
The sociodemographic characteristics of the patients were recorded using a patient profile form. The laboratory data and medication-related side effects of patients were recorded only beginning of the study. The QOL questionnaire FACT-BMT was administered to patients the day the patients were included in the study (baseline) and one month later.6,7 The necessary permissions for the use of the survey are obtained according to the directions on the website of facit.org.
Evaluation of QOL survey results
FACT-BMT is a 50-questions tool composed of a 27-question FACT-General (FACT-G) and a 23-question BMT-specific subscale (BMTS). 8 FACT-G considers the effects of cancer treatment on the patient in four categories: physical condition score, social/family well-being score, emotional well-being score, and functional well-being score.5,9 Patients are expected to respond to questions considering their situation in the last seven days and the completion time of the questionnaire was foreseen as 10–15 min. The item format consists of a Likert-type scale ranging from 0 to 4 (0 = not at all; 1 = a little bit; 2 = somewhat, 3 = quite a bit; and 4 = very much). The physical condition score consists of questions 1–7. The score range is from 0 to 28. The social/family well-being score consists of questions 8–14 and the score range is 0–28. The emotional well-being score consists of questions 14–20 and the score range is from 0 to 24. The functional well-being score consists of questions 20–27 and the score range is between 0 and 28. The BMT scale score consists of questions 28–50 and the score range is between 0 and 40. FACT-G total score; It is obtained by collecting the physical well-being score, social/family well-being score, emotional well-being score, and functional well-being score. The score range is from 0 to 108, and the higher the score, the higher the patient's QOL. The FACT-BMT total score is obtained by summing the FACT-G and BMTS score. The score range is 0–148, and the higher the score, the higher the patient's QOL. 10
Statistical analysis
Statistical Package for the Social Sciences (SPSS) 15.0 statistical program was used for the analysis. Data on sociodemographic characteristics, BMT treatments, side effects, and drug interactions of the patients were expressed as “%.” Categorical data were analyzed by chi-square tests (Pearson and Fisher exact tests). The data on the QOL questionnaire and side effects were expressed as “%.” The data of the study were evaluated using tests of normality (Shapiro-Wilk test of normality and Kolmogorov-Smirnov), and it was determined that the data was not normally distributed. The Wilcoxon-signed rank test (nonparametric test) was used to compare the difference between the means of the QOL, and laboratory data. A p-value of <0.05 at a 95% confidence interval was considered significant.
Results
The mean age of the 40 patients included in the study was 46.4 ± 2.5 [median 48 (19–76)] and 55% of the patients were male. The sociodemographic characteristics of the patients are shown in Table 1.
Sociodemographic characteristics of the patients.
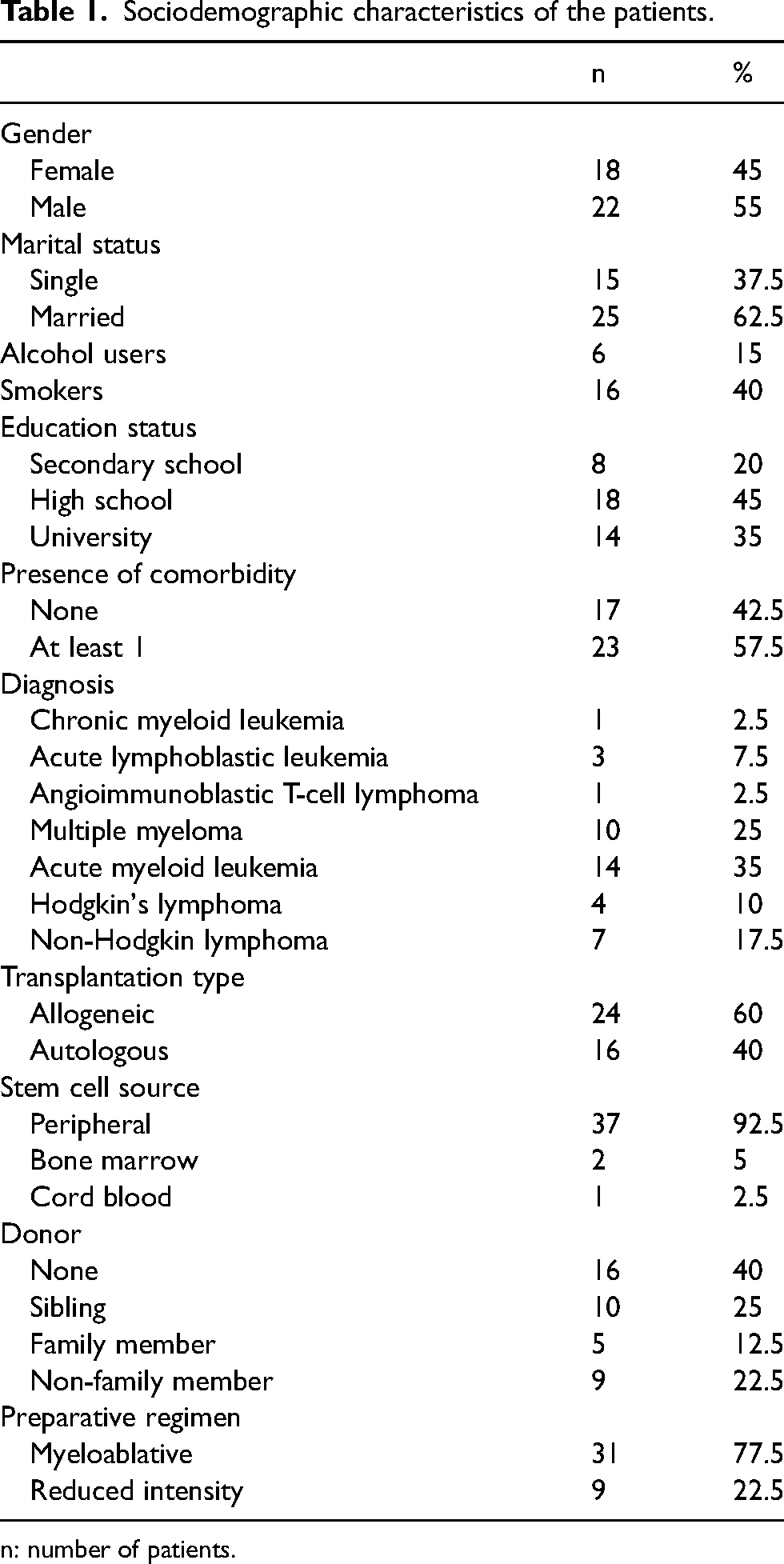
n: number of patients.
The mean hemoglobin, hematocrit, leukocyte, and procalcitonin values of patients were low. The C reactive protein value on the other hand was high (Table 2).
The laboratory data of the patients.
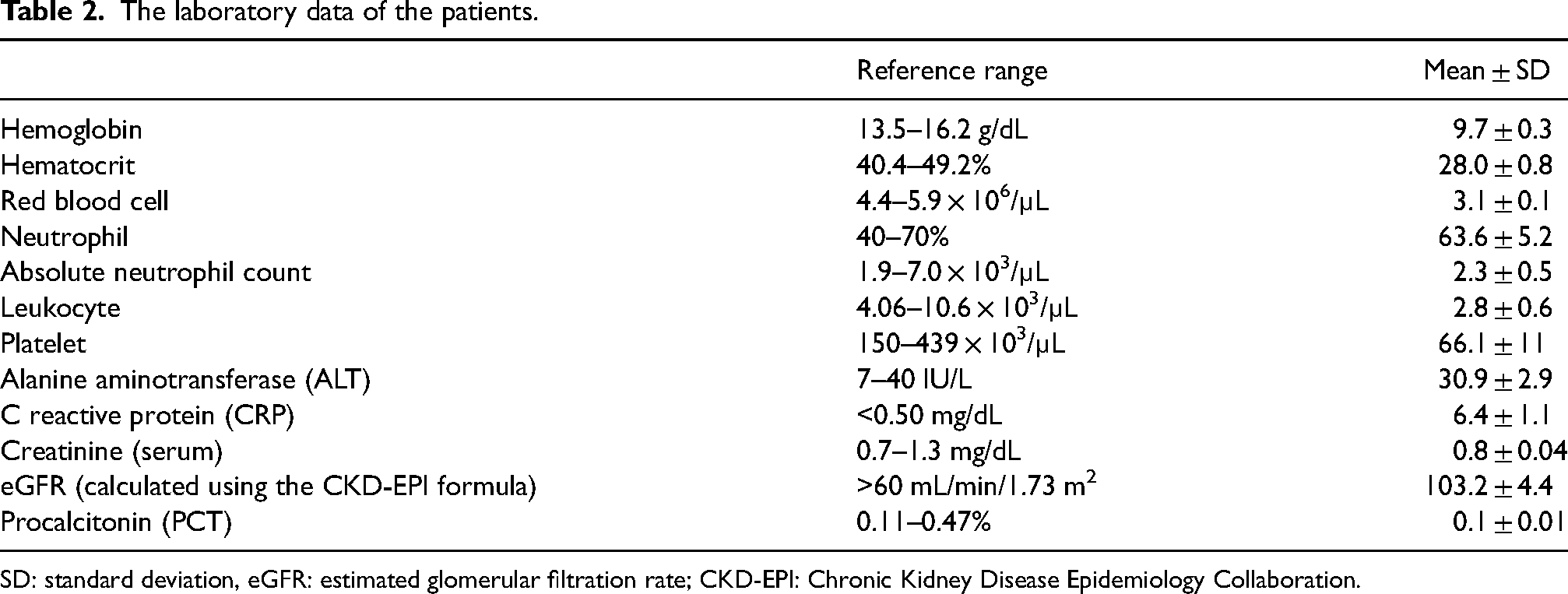
SD: standard deviation, eGFR: estimated glomerular filtration rate; CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration.
A total of 229 side effects were detected in the patients with an average of 5.7 ± 0.3 (2–14) side effects per prescription. Common side effects include thrombocytopenia (14%), weakness (13.1%), alopecia (13.1%), and oral mucositis (13.1%) (Figure 1).
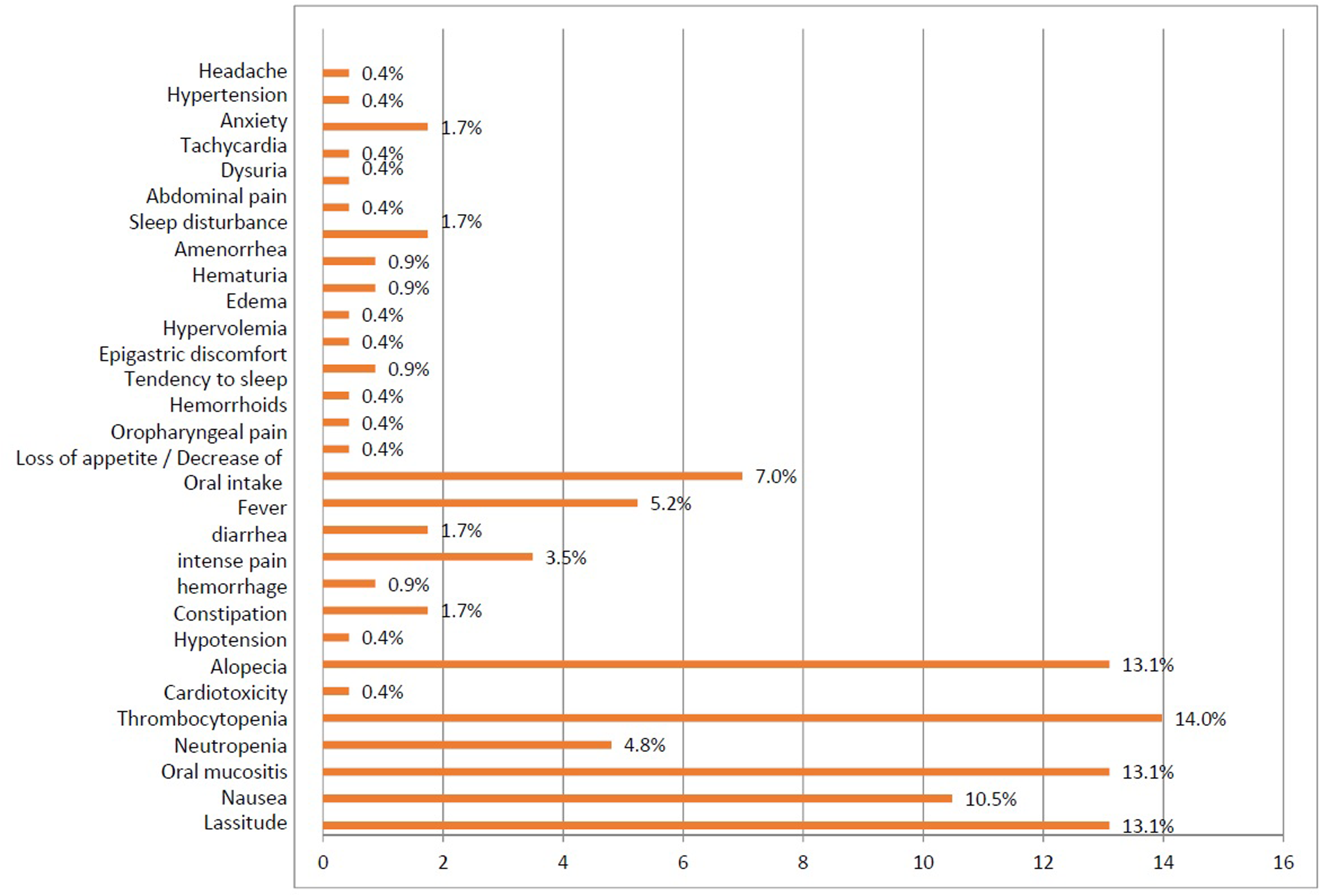
Distribution of side effects.
More side effects were reported by female patients (p < 0.05). There was no relationship between side effects and the patients’ diagnosis, stem cell donor, and source (p > 0.05). However, there were five side effects in a patient with angioimmunoblastic T-cell lymphoma and eight in a patient with chronic myeloid leukemia. Side effects in other diagnosed patients are presented in Table 3.
Evaluation of sociodemographic characteristics and a number of side effects reported.
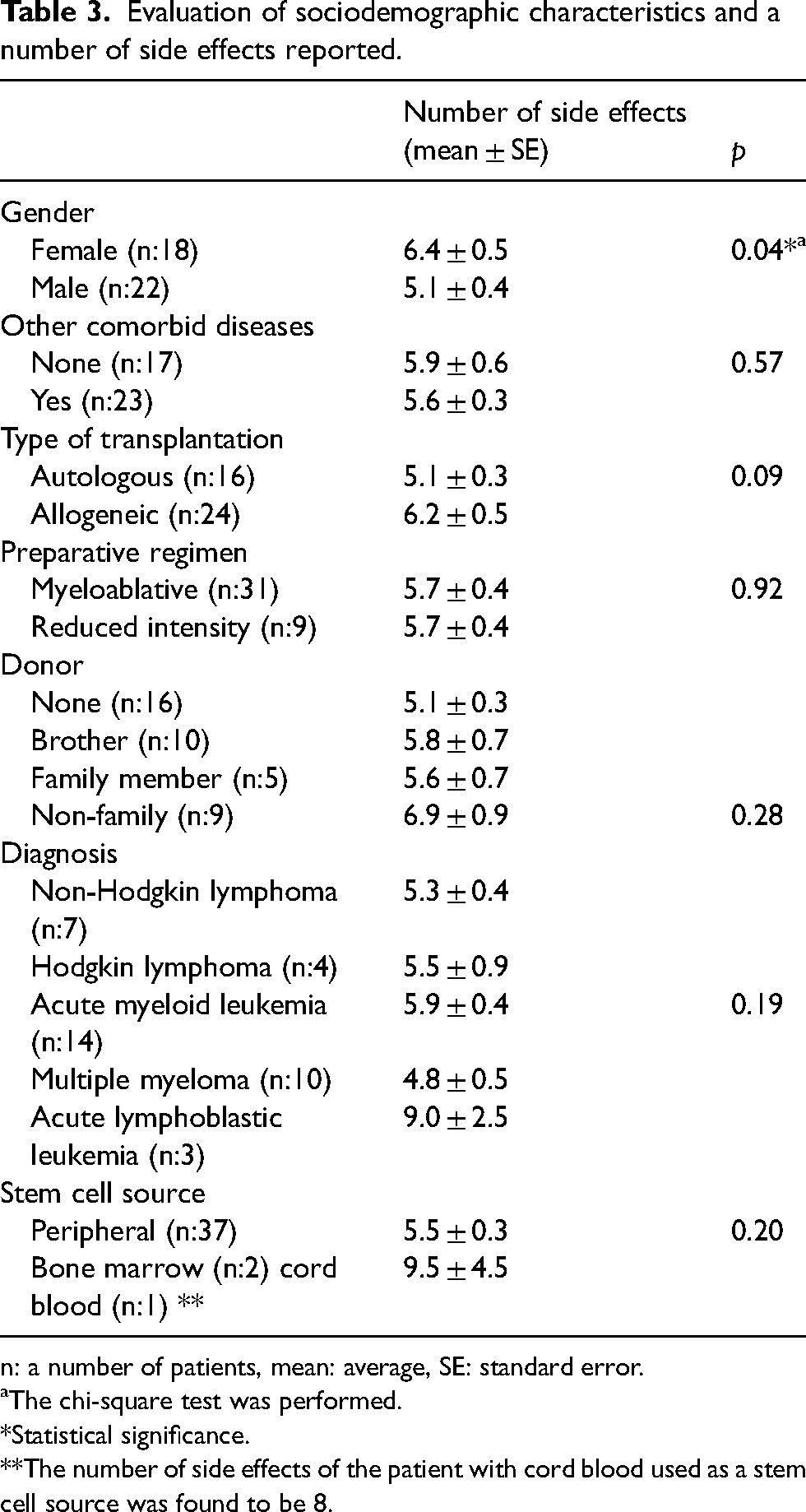
n: a number of patients, mean: average, SE: standard error.
aThe chi-square test was performed.
*Statistical significance.
**The number of side effects of the patient with cord blood used as a stem cell source was found to be 8.
The scores of all patients obtained one month apart were evaluated. It was found that only the social well-being score increased while no difference was observed in other sections (Table 4).
Quality of life scores.
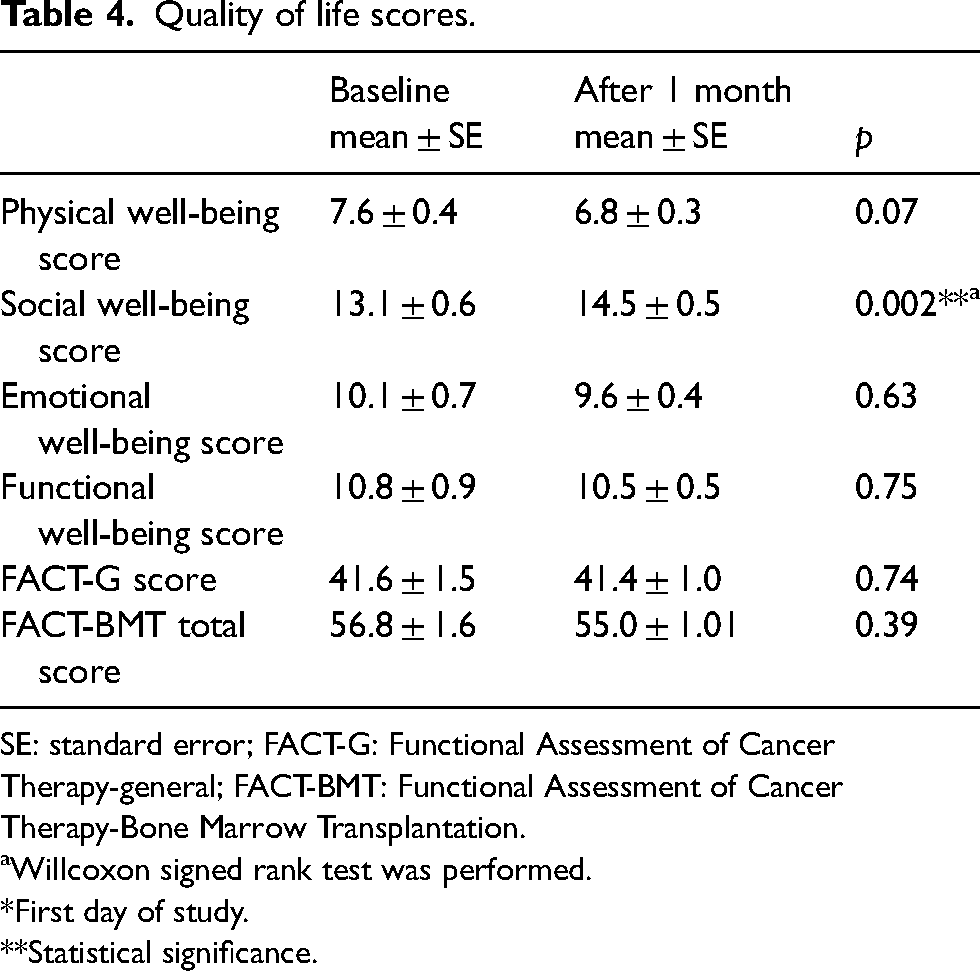
SE: standard error; FACT-G: Functional Assessment of Cancer Therapy-general; FACT-BMT: Functional Assessment of Cancer Therapy-Bone Marrow Transplantation.
aWillcoxon signed rank test was performed.
*First day of study.
**Statistical significance.
Possible predictors of QOL
The QOL scores were not correlated to gender, educational status, and presence of comorbid diseases (p > 0.05). There was a statistical significance between the FACT-G score at 1 month and the mean age of the patients (Spearman rho = 0.32, p < 0.05). There was also a statistical significance between the mean age of the patients and FACT-BMT scores at 1 month (Spearman rho = 0.32, p < 0.04).
There was statistical significance between emotional well-being score at 1 month and marital status (p < 0.001) and type of cancer (p = 0.05) (Table 5).
Evaluation of patients’ emotional well-being scores.
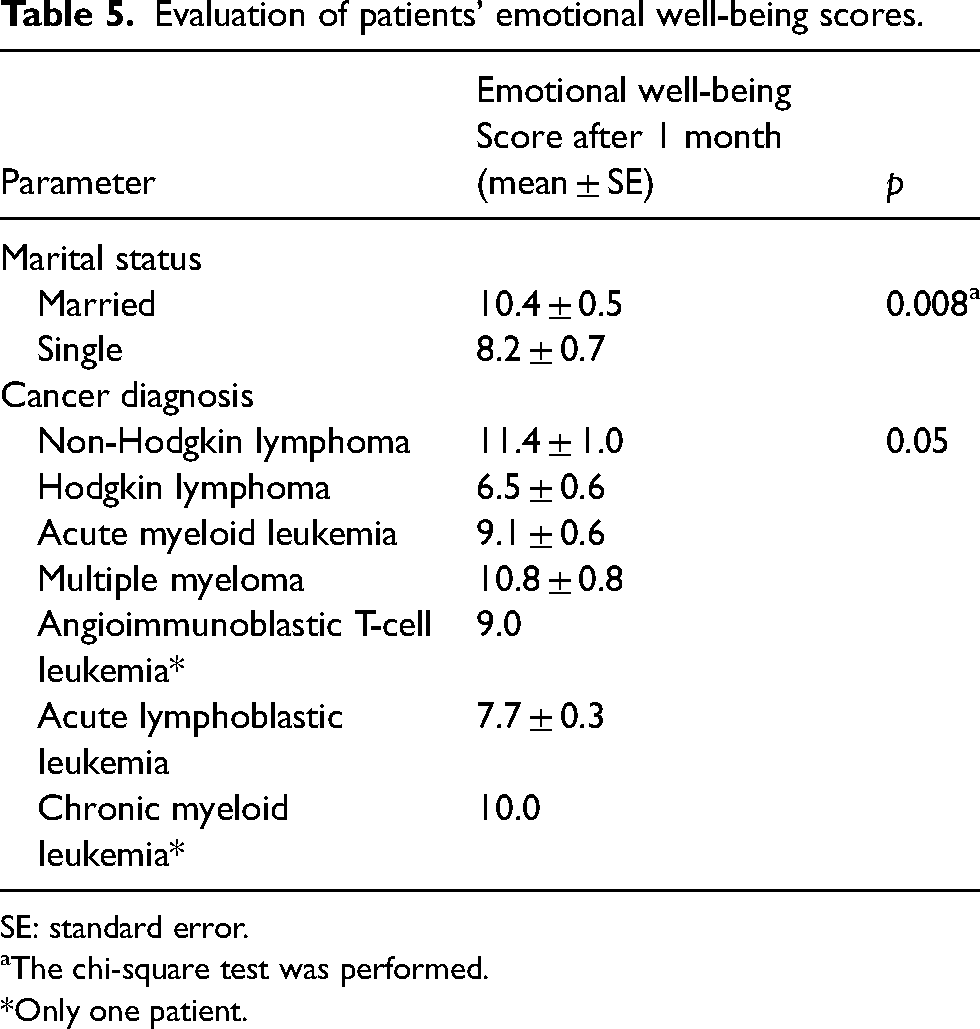
SE: standard error.
aThe chi-square test was performed.
*Only one patient.
The preparative regimen was correlated to the FACT-G score at baseline (p < 0.05). There was also a statistical significance between the type of transplantation and the FACT-G scores at 1 month (autologous 43.8 ± 1.3 and allogeneic 39.9 ± 1.4, p < 0.05). There was a statistical significance between the preparation regimen was correlated with functional well-being (at baseline) and FACT-G (at baseline) (p < 0.05). It was found that the patients treated with a myeloablative regimen have higher QOL scores than those treated with a reduced intensity regimen (the functional well-being (at baseline) scores: 11.9 ± 1.0 vs 7.2 ± 1.3; FACT-G scores: 43.3 ± 1.8 vs 35.8 ± 2.6, respectively). Evaluation of FACT-G and functional well-being scores. There was no difference between QOL scores and stem cell source and donor type (p > 0.05).
Discussion
There is a wide range of clinical pharmacy services provided in BMT units ranging from patent profile creation to providing solutions to drug-related problems. Complex drug regimens are involved in the BMT procedure and the drugs used vary in different regimens. The presence of other comorbid diseases can complicate the treatment further. In our study, the QOL scores of the patients have evaluated insight into possible drug-related problems.
The median age of the study population was 48 (19–76), 55% of which were male. In their study, Mishra et al. 11 examined the profiles of 430 patients treated in the adult BMT unit between 2013 and 2017. They reported median age was 50 years (18–86 years) and 62% of their patients were male. Similar values have also been reported in other studies.11–14
A study by Trevisan et al. 15 in 40 hospitalized hematopoietic stem cell transplant patients, reported the presence of comorbid diseases in 52.5% of their patients; with hypertension (94.4%), dyslipidemia (33.3%), hypothyroidism (16.7%), chronic renal failure not dependent on dialysis (16.7%), heart failure (11.1%), and type 2 diabetes (11.1%) being the most common comorbidities. There is a similar prevalence of comorbidities in our study as 57.5% of the patients had at least a comorbid disease. Similarly, hypertension (52.2%), diabetes (26%), obesity (17.4%), congestive heart failure (13%), chronic renal failure (13%), and hypothyroidism (8.7%), were the most recorded.
In our study, BMT indications included acute myeloid leukemia (35%), multiple myeloma (25%), and non-Hodgkin lymphoma (17.5%). Similar indications have been reported in other studies done in adult BMT units.11,13,16,17
In their study, Visani et al. 18 found moderate oral mucositis in 26%, gastroenteritis in 35%, and moderate vomiting and nausea in all patients who underwent autologous stem cell transplantation. Nephrotoxicity, cardiotoxicity, and interstitial or idiopathic pneumonia were not detected in any of their patients. 18 In our study, 86% of the patients underwent autologous transplantation. Similarly, moderate oral mucositis was detected in 86% of our patients, nausea and vomiting in all, appetite loss and decrease in oral intake in 43%, and cardiotoxicity in 14%. Nephrotoxicity and interstitial or idiopathic pneumonia were not detected in any of our patients. Also, in a retrospective study involving 47 patients with advanced hematological malignancies, stomatitis, nausea, vomiting, and diarrhea were reported in 32% of 37 patients, mental deterioration in 8%, and advanced nephrotoxicity in 5%. 19 In our study, nausea and vomiting were detected in 80% of the patients who underwent the preparative regimen, loss of appetite and decrease in oral intake in 60%, oral mucositis in 20%, and diarrhea in 20%.
Yasar and Akin 6 evaluated the effects of transplantation on patients’ QOL between March and October 2015. They included 100 adult patients, with an average age of 45 years, treated in the hematopoietic stem cell transplantation unit of a university hospital the majority of whom were in the process of allogeneic transplantation due to acute myeloid leukemia. The QOL of the patients was evaluated using the FACT-BMT scale and the overall QOL score was found to be 81.38 ± 21.91. They concluded that it had a moderate effect on the QOL of the patients. 6 The average score of patients in this study is higher than that of our study patients as we recorded an average of 56.8 ± 1.6, on the other hand, our scores were high than that recorded in another study, where the FACT-BMT scale questionnaire was filled by multiple myeloma patients on the day of stem cell apheresis administration and the on the day intensive toxicity is presumed, that is, approximately 10 days after hematopoietic stem cell transplantation. The QOL score was found to be 25.2 ± 5.9 and 23.8 ± 6.6 on the day of apheresis and post-transplantation, respectively. 20 In our study, the scale was applied 30 days apart with no consideration of the stage of the treatment process and there was no difference between overall QOL scores (41.6 and 41.4) and BMT total QOL scores (56.8 and 55) as well.
FACT-BMT QOL scale consists of four sub-dimensions as physical, social, emotional, and functional well-being. Of the patients included in our study one and 30. QOL sub-dimensional scores on days were, respectively; physical well-being (7.6 and 6.8); social well-being (13.1 and 14.5), emotional well-being (10.1 and 9.6), and functional well-being (10.8 and 10.5), and no difference was observed. Sherman et al. 20 found that FACT-BMT sub-dimension scores were scored as follows; physical well-being (16.5 and 15.1), social well-being (24.0 and 23.6), emotional well-being (18.2 and 18.3), and functional well-being (15.4 and 14.4). According to the study results of Yasar and Akin, 6 patients’ QOL sub-dimension scores were as follows: physical well-being (12.1), social well-being (20.8), emotional well-being (12.7), and functional well-being (14). In our study and in the other two studies, it was determined that the social well-being sub-dimension score was higher after transplantation. However, the QOL scores of the patients included in the study were found to be lower than in other studies. We think that the fact that our work was carried out during the pandemic period may have contributed to this situation.
In our study, it was found that chronic disease and the male gender reduced emotional well-being sub-dimension scores in patients. In addition, it was found that the social well-being sub-dimension score was low in young patients and the BMT QOL score was high in married patients. Sherman et al. stated that the demographic (gender, marital status, etc.) characteristics did not have an effect on the QOL outcomes. However, in the same study, FACT-G sub-dimension scores were found to be lower in patients who received thalidomide.
Limitations
Our study was a master of science thesis on Clinical pharmacy and was conducted during the COVID-19 pandemic. For this reason, the frequency of interviewing the pharmacist with the patients was very low, and this caused the treatment-related side effects to be evaluated only at the beginning of the study. Another limitation was the small size of the study. As we have limited time in a master's thesis program, only 40 patients were included in the study.
Conclusions
In conclusion, the QOL of transplant patients is affected by the treatment process. According to the results of our study, the QOL scores of the patients with BMT were found to be lower than the other studies. We think that the COVID-19 pandemic may contribute to a lower QOL compared to other studies. We think that clinical pharmacists will make positive contributions to improving treatment outcomes in patients with BMT.
Footnotes
Acknowledgements
The authors received no specific funding for this work.
Author contributions
All authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Songul Tezcan and Gozde Seray Ozturk. The first draft of the manuscript was written by Songul Tezcan and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of University Marmara (09.2020.1374).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
