Abstract
Objective
Despite depression being a common comorbidity among adults with cancer, limited literature is available regarding pharmacologic depression treatment patterns and predictors in this population. This study aims to examine patterns and predictors of antidepressant prescribing among adults with cancer and depression in ambulatory care settings in the United States (US).
Methods
This retrospective, cross-sectional study utilized data collected from the 2014 to 2015 National Ambulatory Medical Care Survey (NAMCS). The study sample consisted of adults (age ≥ 18 years) with cancer and depression (unweighted N = 539; weighted N = 11,361,000). A multivariable logistic regression analysis was used to adjust for individual-level factors to identify predictors of antidepressant prescribing.
Results
Most patients were adults aged ≥ 65 years, female, and non-Hispanic whites. Thirty-seven percent of the study sample received antidepressant treatment. Multivariable logistic regression analysis revealed that race/ethnicity, physician specialty, and number of medications were significantly associated with receiving antidepressant(s). For example, non-Hispanic whites were two-and-half times more likely to receive an antidepressant [OR 2.43, 95% confidence interval 1.13–5.23] compared to other race/ethnic groups. Every unit increase in the number of prescribed medications increased the likelihood of receiving an antidepressant by 6% (OR 1.06, 95% CI: 1.01–1.11).
Conclusion
Among adults with a comorbid cancer and depression diagnosis and a recorded U.S. ambulatory care visit in 2014–2015, 37% received antidepressant treatment. This suggests most patients with cancer and depression do not receive pharmacologic treatment for depression. Future studies are needed to investigate the impact of antidepressant treatment on health outcomes in this patient population.
Introduction
According to the Centers for Disease Control and Prevention (CDC), cancer is the second leading cause of death in the United States (US). 1 Patients with cancer often incur a heavy burden of adverse effects from their treatment regimen. These adverse effects combined with the mental stressors of facing a life-threatening illness put cancer patients at risk of developing depression. 2 In fact, patients with cancer have up to a four times higher incidence of depression than the general population. 3 Common depressive symptoms such as reduced energy and hopelessness may worsen cancer treatment outcomes and increase mortality.4,5 Examples of poorer outcomes include reduced quality of life, 6 nonadherence with chemotherapy, 7 and increased risk of committing suicide. 8
The American Society of Clinical Oncology (ASCO) practice guidelines recommend that all adult patients with cancer be screened for depressive symptoms at their initial visit and at appropriate intervals, as clinically indicated. 9 Upon screening positive for depressive symptoms, it is then recommended that clinicians first treat any medical causes of depressive symptoms, followed by the use of pharmacological and/or non-pharmacological interventions. 9 Pharmacological interventions for depression treatment include selective serotonin receptor inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), atypical antidepressants such as trazodone, mirtazapine, vortioxetine, and bupropion, tricyclic antidepressants (TCAs) and monoamine oxidase inhibitors (MAOIs). However, the ASCO guidelines make no recommendations about the superiority or inferiority of specific antidepressant pharmacological regimens.
Data collected between March 2006 to May 2008 from the ECOG SOAPP trial found that an estimated 19.2% of patients with cancer are prescribed SSRIs and SNRIs in ambulatory care settings in the United States. 10 Previous studies comparing antidepressant therapy to placebo have yielded low-quality evidence to support definitive pharmacologic treatment recommendations for depression in patients with cancer.11,12 While this may partially explain the relatively low rates of antidepressant treatment reported in the ECOG SOAPP trial, it is unlikely this is the sole explanation since antidepressants are one of the most accessible and cost-affordable treatment options available for treating depression. For example, non-pharmacologic treatment options such as psychotherapy are not always available,13,14 and are less likely to be effective for the treatment of severe depression.15,16 As such, it's important to explore what individual and contextual factors may impact antidepressant prescribing patterns. 17
The literature is currently lacking both in scope and a number of studies regarding antidepressant treatment in patients with cancer with a comorbid diagnosis of depression.11,12 Therefore, this study aims to further elucidate patterns and predictors of antidepressant prescribing in patients with cancer by analyzing US national-level data from ambulatory visits.
Methods
Study design
Data from the 2014–2015 National Ambulatory Medical Care Survey (NAMCS) was collected and analyzed for this retrospective, cross-sectional study. The Institutional Review Board at the University of Arizona determined that Human Subjects Review for ethical approval was not required for this study since it did not involve interaction or intervention with human subjects or involve access to identifiable private information.
Data source
The CDC sponsors NAMCS data collection annually for ambulatory care services provided by physicians at non-federal physician offices that provide direct patient care. National-level estimates are determined by assigning weights to each visit in the survey. The 2014 survey had a response rate of 29.6% 18 and the 2015 survey had a response rate of 39%. 19 These were the most recent publicly available data from NAMCS at the time data analysis was conducted.
A physician–patient encounter is defined in NAMCS as a personal visit with the physician or healthcare worker supervised by the physician for the purpose of receiving health services. For data collection, NAMCS uses a multistage probability design. Primary sampling units (PSUs) (e.g.—counties, county equivalents) are used to generate initial probability samples. Patient visits from the sample were chosen using a probability sample of active physicians of each PSU. Initially, the entire sample of physicians is randomly divided into 52 subsamples that are closely identical in size. Then, each subsample has a random assignment to a week in the survey year out of 52 weeks. Next, physicians select a systematically randomized sample of patient encounters from the week assigned to them.
The Patient Record Form (PRF) is used by NAMCS to collect a diverse set of information such as characteristic data of patients and physicians, diagnoses (≤ 5 diagnoses), prescribed medications (≤ 30 medications), and therapeutic services received by the patient at the time of the patient encounter.
Study sample
There were 45,710 patient visits in 2014 and 28,332 patient visits in 2015 recorded by NAMCS. At a national level, the study sample represented 11,361,000 adult (unweighted n = 539; age ≥ 18 years) ambulatory care visits of patients with cancer and depression. A patient's diagnosis of cancer and depression was identified if the answer to the question “Regardless of the diagnoses previously entered, does the patient now have: … cancer” and “Regardless of the diagnoses previously entered, does the patient now have: … depression,” were both “yes.” Due to the use of only five diagnosis codes in NAMCS, these questions are also included by NAMCS to identify patients with these comorbid conditions and avoid underestimating the size of this patient group. NAMCS has used these questions for gathering data since 2005. 20
Dependent variable
Generic drug codes and Multum Lexicon Codes were used to determine if patients were using antidepressants at the time of the surveyed patient encounter. Antidepressants in the survey were categorized by drug class: monoamine oxidase inhibitors (MAOIs), selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), serotonin modulators, tricyclic antidepressants (TCAs), and miscellaneous antidepressants. TCAs consisted of amitriptyline, amoxapine, clomipramine, desipramine, doxepin, imipramine, maprotiline, nortriptyline, and protriptyline; MAOIs consisted of phenelzine and tranylcypromine; serotonin modulators consisted of trazodone, nefazodone, and vilazodone; SSRIs consisted of citalopram, escitalopram, fluoxetine, paroxetine, and sertraline; SNRIs consisted of duloxetine, venlafaxine, and desvenlafaxine; miscellaneous consisted of bupropion and mirtazapine.
Independent variable
The independent variables included in this study were grouped into three different categories by utilizing the Anderson Behavioral Model: (i) predisposing; (ii) enabling; and (iii) need factors. 17 Predisposing factors included age (18–64 years; ≥ 65 years), gender (female/male), and race/ethnicity (White Only Non-Hispanic; Other). Enabling factors consisted of health insurance status (Government-Medicaid/Medicare; Other), physician/clinic specialty (General, Family, and Internal Medicine practice; Other), and geographical region (South; Other). Need factors consisted of receiving new prescriptions during visit (yes/no), new patient visit (yes/no), reason for visit (chronic problem/other), total number of chronic conditions, and total number of medications used. Variables were dichotomized as necessary to avoid reporting any unweighted sample sizes < 30 as recommended by NAMCS. 21 Categorization of these independent variables is provided in Table 1.
Demographic and clinical characteristics of adults with cancer and depression NAMCS 2014–2015.
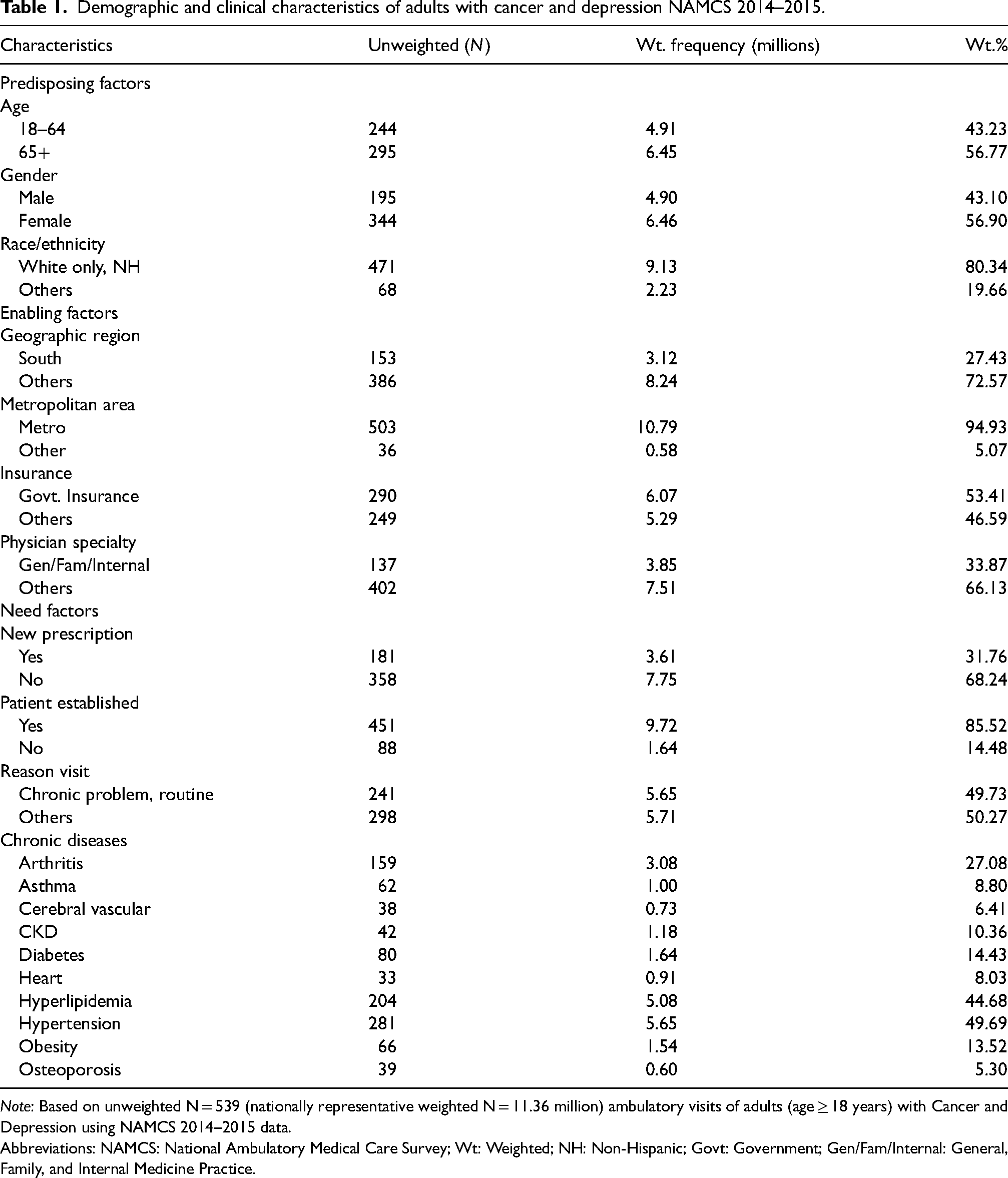
Note: Based on unweighted N = 539 (nationally representative weighted N = 11.36 million) ambulatory visits of adults (age ≥ 18 years) with Cancer and Depression using NAMCS 2014–2015 data.
Abbreviations: NAMCS: National Ambulatory Medical Care Survey; Wt: Weighted; NH: Non-Hispanic; Govt: Government; Gen/Fam/Internal: General, Family, and Internal Medicine Practice.
Statistical analysis
At the national level, patient visits to physicians at ambulatory settings were reported as weighted frequencies (in millions) and weighted percentages. This study used a multivariable logistic regression analysis to identify predictors of depression treatment in this study sample. In SAS version 9.4 (SAS institute Inc., Cary, NC, USA), survey procedures such as SURVEYFREQ, SURVEYMEANS, and SURVEYLOGISTIC were utilized to adjust for NAMCS’ complex survey design to obtain national-level estimates.
Results
Patient demographics and clinical characteristics
Of the 11,361,000 patients (unweighted N = 539) that had a recorded visit for depression with a cancer diagnosis, 4,285,000 were prescribed antidepressants (weighted percent = 37.7%, unweighted N = 224). Table 1 reports the demographic and clinical characteristics of the analyzed patients. Most visits occurred in adults aged ≥ 65 years (56.8%), females (56.9%), and Non-Hispanic Whites (80.3%).
Thirty-seven percent of the study sample received antidepressant treatment. The top three prescribed drug classes were SSRIs (24.9% of the weighted sample, 95% CI: 19.5–30.4%), SNRIs (7.7% of the weighted sample, 95% CI: 4.0–11.3%), and the miscellaneous group consisting of either mirtazapine or bupropion (6.2% of weighted sample, 95% CI: 3.8–8.6%) (Table 2). The two most commonly prescribed medications in the depression treatment group were citalopram (8.9% of weighted sample, 95% CI: 5.7–12%) and bupropion (4.7% of weighted sample, 95% CI: 2.7–6.7%).
Characteristics of antidepressant treatment in adults with cancer and depression NAMCS 2014–2015.
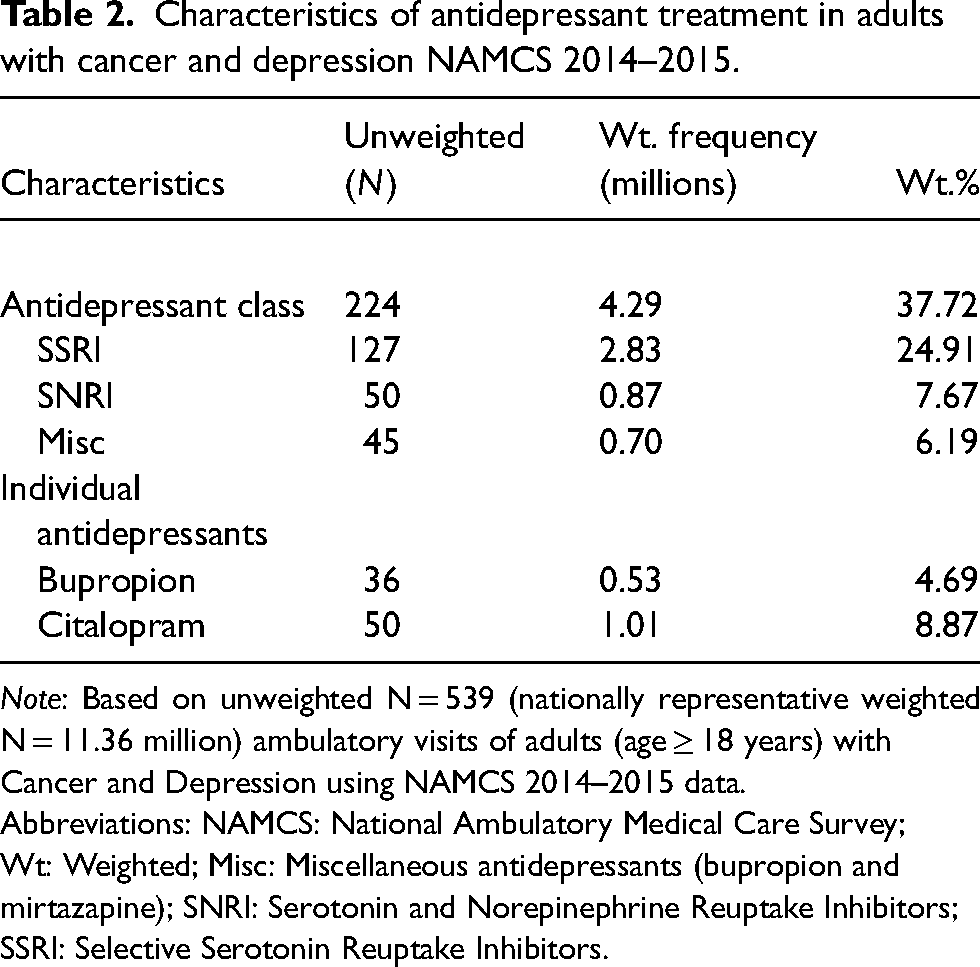
Note: Based on unweighted N = 539 (nationally representative weighted N = 11.36 million) ambulatory visits of adults (age ≥ 18 years) with Cancer and Depression using NAMCS 2014–2015 data.
Abbreviations: NAMCS: National Ambulatory Medical Care Survey; Wt: Weighted; Misc: Miscellaneous antidepressants (bupropion and mirtazapine); SNRI: Serotonin and Norepinephrine Reuptake Inhibitors; SSRI: Selective Serotonin Reuptake Inhibitors.
Predictors of depression treatment
Race/ethnicity, physician specialty, and number of medications were significantly associated with receiving antidepressant treatment based on multivariable logistic regression analysis (Table 3). Non-Hispanic Whites were 2.5 times more likely to receive antidepressant treatment (OR 2.43, 95% CI: 1.13–5.23) compared to other race/ethnic groups. Other physician specialties were 48% less likely to prescribe antidepressants compared to General, Family, or Internal medicine physicians (OR 0.52, 95% CI: 0.30–0.91). Every unit increase in the number of prescribed medications increased the likelihood of receiving an antidepressant by 6% (OR 1.06, 95% CI: 1.01–1.11).
Discussion
The results of this study reveal that less than half of patients with a diagnosis of cancer and depression receive antidepressant treatment. To the best of our knowledge, this is the first study to report patterns and predictors of antidepressant prescribing for this patient population using the NAMCS database. Our findings are similar to a 2015 prospective, multi-institutional, observational study that enrolled over 3,000 adults in US outpatient oncology clinics from 2006 to 2008, where only 25% of patients with cancer and depression received antidepressants. 10 However, this study from Fisch et al. did not include national-level estimates nor a generalizable sample, as it only enrolled patients with breast, colorectal, prostate, or lung cancer from 38 different institutions.
It is important to note that these findings are based on one patient visit with one provider, meaning patients may have received antidepressant treatment at other visits. On the other hand, clinicians may choose not to prescribe antidepressants since there is limited evidence regarding antidepressant treatment in patients with comorbid cancer. There are numerous barriers to conducting placebo-controlled studies in this population, 22 but of the very limited evidence available, findings have not demonstrated first-line antidepressants to be more efficacious than placebo. A 2017 meta-analysis involving eight randomized controlled trials (RCTs) on pharmacologic treatment for depression in cancer patients between 2005 and 2015 reported that trials of fluoxetine, paroxetine, and desipramine failed to show superiority in depression remission over placebo, while studies on second-generation antidepressants have been limited to open-label case studies. 12 Only one RCT reported mianserin as having strong evidence for depression remission in cancer patients. However, this medication is only available outside of the United States and carries a risk of bone marrow suppression.
Citalopram and bupropion were the most commonly prescribed antidepressants in our study sample. Published studies do not favor one antidepressant over another for effective pharmacologic treatment in cancer patients 12 ; however, these two medications may be more commonly prescribed because of their favorable side effect profile and off-label benefits. For example, citalopram can reduce the intensity and frequency of hot flashes,23,24 which may benefit breast cancer patients who commonly experience hot flashes from menopause induced by antiestrogen therapy. Bupropion also has advantages related to its side effect profile and efficacy in smoking cessation. In a double-blind, placebo-controlled RCT from 2010, cancer patients with depression taking bupropion had higher rates of abstinence from smoking, lower rates of withdrawals, and higher quality of life scores compared to cancer patients without depression. 25 Furthermore, an open-label study of 21 cancer patients suggested that this antidepressant may relieve cancer-related fatigue (CRF). 26 The first double-blinded RCT in the US to examine the effect of bupropion for CRF for over 400 cancer patients is currently ongoing. 27 Additionally, bupropion's lack of serotonergic activity results in a much lower incidence of nausea, diarrhea, sexual dysfunction, and somnolence compared to SSRIs,28–30 which is significant considering these are also common side-effects of various chemotherapy agents. 31 Both citalopram and bupropion are relatively inexpensive; thus, a multitude of factors such as cost, provider-preference, side-effect profile, and off-label benefits may explain the higher use of citalopram and bupropion compared to other first-line antidepressant options in cancer patients.
The number of medications a patient was taking was associated with whether a person received antidepressant therapy. Fisch et al. 10 reported similar findings; cancer patients taking >10 medications were more than three times as likely to be prescribed an antidepressant than patients taking < 10 medications. Patients prescribed a higher number of medications may be more likely to inquire about a medication for depression compared to those who wish to take fewer medications. Alternatively, patients with certain comorbidities may be more likely to experience depressive symptoms.32,33
Our results reveal racial disparities in antidepressant treatment among adults with comorbid cancer and depression. White, non-Hispanic patients were more likely to receive treatment than other racial/ethnic groups. Fisch et al. 10 reported similar findings, in which white, non-Hispanic adults were also twice as likely to receive antidepressants than other racial groups. Additional studies have also reported that non-Hispanic, white adults with depression and cancer are significantly more likely to receive antidepressant treatment than other races.34–36 For instance, non-Hispanic white adults were twice as likely as Hispanic, non-Hispanic black, and other racial/ethnic adults to receive antidepressants in a 2017 retrospective analysis of over 3,000 US cancer survivors in the National Health Interview Survey from 2010 to 2013. 34 Another retrospective analysis of over 1,600 US adults ≥ 66 years old with breast, colorectal, or prostate cancer and newly diagnosed depression in the Surveillance, Epidemiology, and End Results (SEER) and Medicare database from 2007 to 2011 found that 49.8% of non-Hispanic whites were on antidepressants versus 36.0% of African Americans. 35 Lastly, a 2012 retrospective study of over 800 US Medicare beneficiaries (age > 65 years) with cancer and depression in the Medicare Current Beneficiary Survey (MCBS) from 2000 to 2005 found that non-Hispanic whites were almost twice as likely than minorities to receive antidepressants. 36 Since depression can worsen health outcomes among adults with comorbid cancer and depression,4,5 future studies should examine how racial/ethnic disparities in antidepressant treatment may affect this population's health outcomes.
Primary care physicians (PCPs) prescribed antidepressants more than other specialty physicians. There are few studies that report on differences in antidepressant prescribing patterns for cancer patients between different practice specialties. In 2009, the Cancer Care Outcomes Research and Surveillance Consortium reported that over 90% of PCPs were involved in diagnosing/treating depression in cancer patients, while over 50% of cancer specialists provided referrals for or were not involved in depression assessment/treatment. 37 Our findings demonstrate that adults with cancer continue to be more likely to receive antidepressant treatment from PCPs than other physician specialties. This may be due to PCPs having more systems in place to screen for depression 38 or oncologists perceiving antidepressant treatment to be out of their scope of practice since practice guidelines from the ASCO recommend management of depressive symptoms be delivered by appropriately trained individuals. 9
There were limitations in this study that should be taken into consideration. Possible underreporting and misclassification of depression may have occurred. Physicians may not have reported a patient as having depression who was no longer acutely depressed but was receiving continuation or maintenance treatment with an antidepressant. This could lead to misleading results suggesting a lower prevalence of antidepressant treatment in patients with cancer and depression who were willing and able to receive treatment. Additionally, the number of patients receiving antidepressants may have been underreported since patients with reported depression may have been prescribed an antidepressant at a later patient encounter. The intended indication for prescribed antidepressants was not reported, meaning individuals prescribed these medications for reasons other than depression could not be identified. Therefore, data from this study may also report an inflated number of patients with cancer receiving pharmacologic treatment for depression. Antidepressant dosing information was also unavailable. Data pertaining to the duration, severity, and performance status of cancer was not available. This study only focused on patients within ambulatory care settings and did not include other settings, such as palliative care. We were unable to differentiate what percent of “Other” physician/clinical specialties that prescribed antidepressants consisted of oncologists. Additionally, we were unable to determine whether the surveyed physician was the one providing a prescription for an antidepressant, or whether they were seeing a patient who was already prescribed an antidepressant. Due to the small sample size, certain variables had to be dichotomized to avoid reporting subgroups with an unweighted sample size < 30. 21 The possibility of reporting errors, coding errors, and interviewer effect in identifying cancer or depression were not reported. Lastly, although recommendations from the ASCO practice guidelines for the care of depressive symptoms in adults with cancer have not changed since 2014, 9 antidepressant prescribing patterns for this population may have changed since 2014–2015. These were the most recent data available from NAMCS at the time the study took place and future studies are needed to continue evaluating prescribing trends.
Predictors of antidepressant treatment among adults with cancer and depression NAMCS 2014–2015.
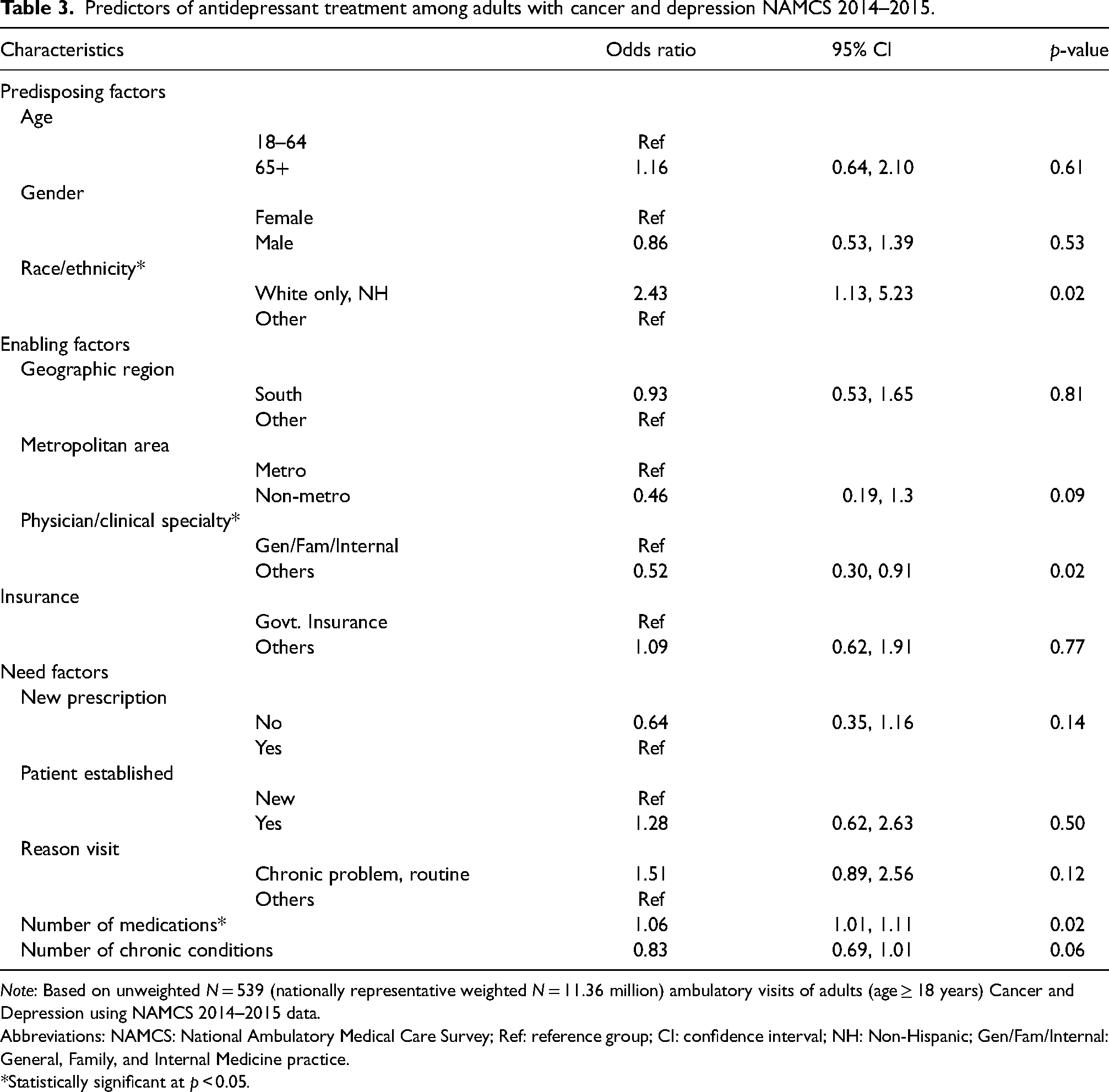
Note: Based on unweighted N = 539 (nationally representative weighted N = 11.36 million) ambulatory visits of adults (age ≥ 18 years) Cancer and Depression using NAMCS 2014–2015 data.
Abbreviations: NAMCS: National Ambulatory Medical Care Survey; Ref: reference group; CI: confidence interval; NH: Non-Hispanic; Gen/Fam/Internal: General, Family, and Internal Medicine practice.
*Statistically significant at p < 0.05.
Conclusion
Approximately 37% of adults that had a recorded U.S. ambulatory care visit from NAMCS in 2014–2015 with a comorbid cancer and depression diagnosis received antidepressant treatment. Citalopram and bupropion were the most commonly prescribed antidepressants. Non-Hispanic white patients and patients prescribed a higher total number of medications were more likely to receive antidepressant treatment. PCPs were more likely to prescribe antidepressants to patients with cancer than other physicians/clinical specialties. Future studies should seek to investigate the impact of antidepressant treatment on health outcomes for adults diagnosed with cancer and depression.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author contributions
All authors contributed to designing the study and drafting various sections of the manuscript. ALe collected the data and performed the analysis. All authors interpreted the results and reviewed and approved the final version of the manuscript.
