Abstract
Purpose
Paclitaxel is associated with hypersensitivity reactions (HSRs). Intravenous premedication regimens have been devised to decrease the incidence and severity of HSRs. At our institution oral histamine 1 receptor antagonists (H1RA) and histamine 2 receptor antagonists (H2RA) were adopted as standard. Standardizations were implemented for consistent premedication use in all disease states. The purpose of this retrospective study was to compare the incidence and severity of HSRs before and after standardization.
Methods
Patients who received paclitaxel from 20 April 2018 to 8 December 2020 having an HSR were included in analysis. An infusion was flagged for review if a rescue medication was administered after the start of the paclitaxel infusion. The incidences of all HSR prior to and post-standardization were compared. A subgroup analysis of patients receiving paclitaxel for the first and second time was performed.
Results
There were 3499infusions in the pre-standardization group and 1159infusions in the post-standardization group. After review, 100 HSRs pre-standardization and 38 HSRs post-standardization were confirmed reactions. The rate of overall HSRs was 2.9% in the pre-standardization group and 3.3% in the post-standardization group (
Conclusions
This retrospective interventional study demonstrated that same-day intravenous dexamethasone, oral H1RA, and oral H2RA are safe premedication regimens for paclitaxel. No change in the severity of reactions was seen. Overall, better adherence to premedication administration was seen post-standardization.
Introduction
Paclitaxel (Taxol®) is FDA-approved and used for the treatment of common types of cancers such as breast cancer and ovarian cancer. 1 A black box warning for anaphylaxis and severe hypersensitivity reactions (HSRs) is listed in the package insert. Paclitaxel has poor solubility and is commercially compounded in Cremophor EL®, an emulsifying agent that plays a role in the pathogenesis of HSRs. 2 Cremophor EL® is thought to cause complement activation leading to HSRs and, hence the need for premedication. 3 Three premedications are required prior to paclitaxel infusion to reduce the severity of and prevent HSRs. A histamine 1 receptor antagonist (H1RA) and histamine 2 receptor antagonist (H2RA) should be administered at least 30 min prior to the paclitaxel infusion. The package insert recommends dexamethasone administration approximately 12 and 6 h prior to paclitaxel; however, intravenous dexamethasone on the day of infusion 30 min to paclitaxel infusion has been validated in the literature as effective and commonly employed in clinical practice.4,5 HSRs can still occur despite recommended premedication.
HSR can present with cutaneous eruptions, lower back pain, flushing, dyspnea, chest pain, hypotension, and throat tightness that vary in severity. 6 Management of hypersensitivity reactions involves stopping the infusion and employing symptom management.7,8 Agents used as premedications can be administered for reactive symptom management in paclitaxel HSRs. Further symptom management with vasopressors, fluid boluses, bronchodilators, and epinephrine can be used in severe cases. 9 In recent prospective trials, institutions have reported HSRs of all grades occurring in 6% or less of their population.10–12 Nearly 95% of reactions to the general class of taxanes occurred during the first or second infusion. 13 Additionally, patients who have had a history of reactions to previous courses of paclitaxel, the presence of respiratory dysfunction, obesity, and postmenopausal status are at risk for paclitaxel HSRs. 14
At our National Cancer Institute-Designated Cancer Center, a multidisciplinary group was developed to evaluate paclitaxel premedication regimens to identify opportunities for improvement after concerns were raised by nursing staff regarding unclear language in order sets leading to variable premedication practices. An internal review found inconsistencies among paclitaxel order sets between cancer disease groups on paclitaxel orders including variation in timing and route, varied nursing premedication practices for subsequent doses due to “may hold if no previous reaction” verbiage, and the use of syringe pumps to administer IV premedications leading to variable timing between the end of premedications and the start of paclitaxel. Changes were made to order sets based on published literature, institutional data, and recommendations of a multidisciplinary group that included clinical pharmacists, nursing, and providers. Our institution implemented oral administration of H1RAs and H2RAs as consistent timing would allow for the absorption and distribution of these medications. The purpose of this retrospective study was to compare the rates of reactions to pre- and post-standardization interventions to understand the impact of the changes.
Methods
This was a retrospective single-center study conducted at the Fred Hutchison Cancer Center in Seattle, WA, USA. This study was approved by the University of Washington Human Subjects Division Institutional Review Board with a waiver of consent granted.
Data collection
Patients who received paclitaxel from 20 April 2018 to 8 December 2020 identified by paclitaxel orders were included in the overall analysis. The standard practice is that all chemotherapies are ordered through an order set to ensure safe ordering, appropriate pharmacist and nursing verification, and documentation. HSRs were identified by the administration of H1RA (diphenhydramine), H2RA (famotidine), steroids (methylprednisolone or hydrocortisone), and/or epinephrine to the patient after the start of the paclitaxel infusion. Finally, a reaction to paclitaxel was confirmed through nursing infusion notes (all infusions are documented by a nurse in the medical chart even if no reaction occurred). System-wide changes were made between 20 April 2020 and 21 April 2020. The pre-standardization group was patients who received paclitaxel from 20 April 2018 to 20 April 2020. The post-standardization group was patients who received paclitaxel from 21 April 2020 to 8 December 2020. Patients were excluded if “rescue medication” was given for reasons other than hypersensitivity reactions. Patients were counted if they had HSR reactions, and repeat reactions identified from a repeat patient were excluded from analysis. Type of malignancy was obtained. Infusion reaction severity was analyzed and graded based on Common Terminology Criteria of Adverse Events 5.0. 15
Institutional interventions
Prior to standardization, there were differing time intervals between the end of the administration of premedications and the start of paclitaxel. In reviewing 42 paclitaxel order sets across the center, it was discovered that premedication timing varied between 15 and 30 min depending on the disease state group's decision. Additionally, the use of syringe pumps for non-standardized dilutions and rates created even more variability.
Post standardization, all order sets had clear language outlining the need for 30 min to elapse between the end of the administration of premedications and the start of paclitaxel infusion rates. Additionally, there were nursing in-services and computerized provider order entry changes to use oral administration for H1RAs and H2RAs as a default. Additionally, there was elimination of syringe pumps in infusion areas.
The efficacy of oral versus intravenous diphenhydramine for mitigating HSRs was not firmly established at the beginning of the study period. Due to a national intravenous diphenhydramine shortage, oral diphenhydramine was adopted as a premedication. Understanding this “natural experiment” was part of the rationale for this study. Our institution defaulted all order sets to the oral route of administration of diphenhydramine and famotidine. For dexamethasone, studies looking at oral dexamethasone have compared the two-dose oral strategy versus same-day intravenous.5,16 Dexamethasone was kept as a one-time intravenous dose. The nursing workload was decreased with decreased medication manipulation and stocking of all premedications in the automated dispensing cabinet. Additionally, there was a decreased cost to the institution, and cost savings were passed on to patients and insurers.
Implementation of mandatory premedications with the second infusion of weekly paclitaxel was clarified in nursing communications and defaulted in CPOE. Prior to standardization, the first paclitaxel infusion had mandatory premedications. On subsequent infusions, premedications were only mandated if the patient had reacted to previous infusions. On a preliminary, internal data review, 76% of patients who reacted to the second dose of paclitaxel did not react to their first infusion. Paclitaxel premedications are recommended for all paclitaxel infusions per the manufacturer. However, it has been established that the majority of HSRs will occur during the first and second paclitaxel infusions. 13 Based on the literature, our institutional policy mandated premedications for the first two paclitaxel administrations, and premedications were removed for patients without reactions as this had no impact on the incidence of HSR or increase of rescue medications in previous studies.12,17
Further clarification was made to verbiage in nursing order communication for premedication. The order stated “may hold if no reaction to previous doses” and was interpreted differently by nursing staff in different clinics leading to variable practices including holding all, none, or some premedications. Nursing order communications were changed to “hold if no reaction to previous doses of paclitaxel.” Conversely, throughout all clinics, the standard practice of premedication administration for any history of HSRs with paclitaxel was adopted. Additionally, paclitaxel's rate may be slowly titrated up depending on provider choice if previous infusion reaction was documented; without any reaction, paclitaxel is infused over 1 h if the dose is ≤ 90 mg/m2 and over 3 h if the dose is >90 mg/m2. Provider discretion was dictated by the severity and type of reaction. All these changes were made to reduce inter- and intrapatient variability of supportive care measures.
Results
The primary outcome was to compare the overall incidence of paclitaxel hypersensitivity reactions and the rate of reactions for first and second infusions prior to and after the implementation of the previously outlined standardization measures. Secondary outcomes included severity of hypersensitivity reactions, rescue medications administered, characterization of premedication regimen administered to those who reacted, and cost savings. Fisher's exact test was used to compare rates of hypersensitivity reactions pre- and post-intervention. Categorical variables were evaluated with the χ2 or Fisher's exact test as appropriate.
In total, there were 3499 infusions in the pre-standardization group and 1159 infusions in the post-standardization group. Of these, 159 HSRs in the pre-standardization group and 57 HSRs in the post-standardization group were flagged for review. After a retrospective chart review, 100 HSRs and 38 HSRs were confirmed, pre-standardization and post-standardization, respectively. A Fisher's exact test resulted in no statistically significant difference in HSRs between the pre-standardization and post-standardization groups (
Contingency table comparing all infusion reactions.

Fisher's exact two-tailed test
Contingency table comparing first and second infusion HSRs.

Fisher's exact two-tailed test
HSR infusion characteristics.

Other malignancies encompass colorectal, gastroesophageal, pancreatic, sarcoma, thyroid, neuroendocrine, small-cell lung cancer.
HSR infusion characteristics are listed in Table 3. After review, 100 (2.9%) HSRs pre-standardization and 38 (3.3%) HSRs post-standardization were confirmed with nursing administration notes. The mean age of patients who experienced HSRs was 52 and 53 years old, in the pre-standardization and post-standardization groups, respectively. There were 84 (9.7%) HSRs pre-standardization and 22 (8.2%) HSRs post-standardization that occurred in the first and second paclitaxel infusions. A majority of the patients received weekly paclitaxel for the treatment of breast cancer.
Most hypersensitivity reactions were grade 2 in both groups. There were four cases of grade 3 infusion reactions occurring in the prior-to-standardization group. There were two cases of grade 3 and one case of a grade 4 infusion reaction in the post-standardization group, as shown in Table 4. No reactions were reported as grade 1 or grade 5.
The severity of infusion reaction based on CTCAE 5.0.

Characterization of premedication prescribing habits during the first and second infusions are depicted in Table 5. Increased use of the oral formulations of diphenhydramine and famotidine was seen post-standardization. An increase in the utilization of oral diphenhydramine from 15.7% to 78.5% was found. Additionally, there was increased use of oral famotidine from 42% to 78.9% post-standardization. Fewer patients were given no premedication administration post-standardization. A cost comparison is presented in Table 6.
Characterization of administration of premedications for first and second infusions.
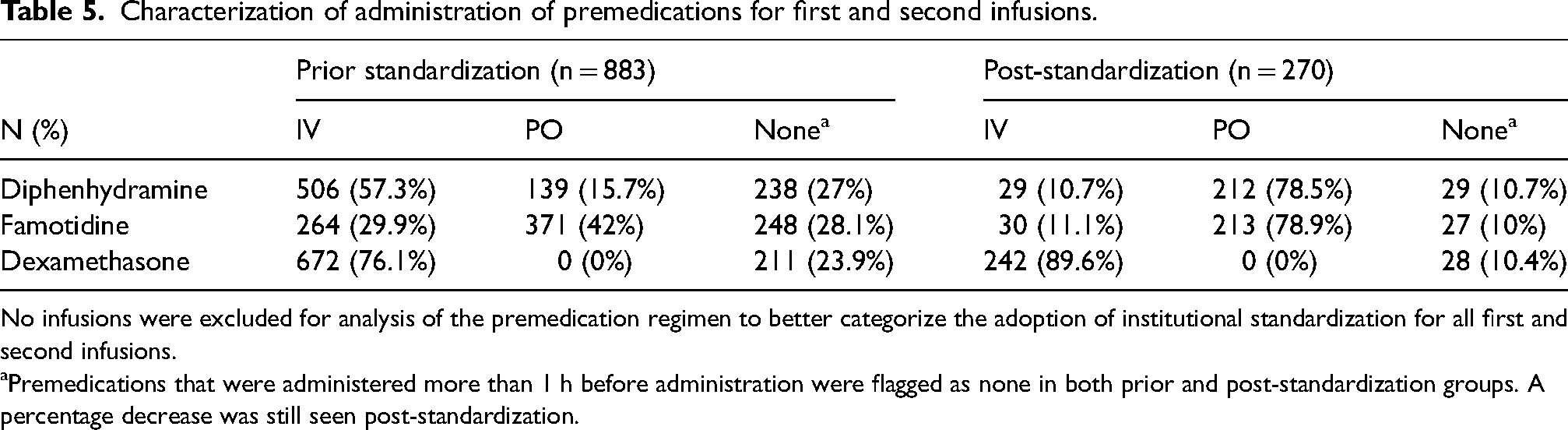
No infusions were excluded for analysis of the premedication regimen to better categorize the adoption of institutional standardization for all first and second infusions.
Premedications that were administered more than 1 h before administration were flagged as none in both prior and post-standardization groups. A percentage decrease was still seen post-standardization.
Average wholesale price (AWP) comparison of drug regimen prior to standardization and post.

Discussion
The use of oral H1RAs and H2RAs has not been studied for paclitaxel premedication regimens, and intravenous premedications are the standard of care. Our study shows no clinical or statistically significant difference in the severity or incidence of HSRs with oral diphenhydramine and famotidine. Institutional standardization resulted in decreased incidence of first and second-infusion HSRs, although not statistically significant.
There was increased use of oral premedications after the numerous interventions and standardization efforts. Oral administration prevents the use of syringe pumps and other means of infusing intravenous medications, improving safety through the ability to precisely time the premedications. This also serves as a cost-saving initiative and results in ease of administration for nursing staff. The cost of premedications can be seen in Table 6 with oral premedication being cheaper than intravenous administration. The multidisciplinary group mutually decided not to move to oral dexamethasone as the only premedication regimens studied for oral dexamethasone are 12 and 6 h prior to paclitaxel infusions at the time of the study. However, a recent paper by Lansinger et al. 18 that looked at steroid premedication in the first administration of any type of taxane found no correlation between route or dosing with incidence/severity of HSRs. This article in conjunction with our study may catalyze other institutions to implement an entire oral premedication regimen to aid in the reduction in infusion chair time and nursing demand. In addition to removing syringe pumps from infusion areas, the institution developed standard dilution guidelines for IV push medications, including dexamethasone. Standardizing the dilution and push time helped alleviate concerns about adverse effects.
Prior to this study, the institutional practice required premedications for only the first dose of weekly paclitaxel, and premedications for subsequent infusions were only required if a prior reaction was reported. Our change to requiring premedications for first and second doses demonstrated a downward trend in reaction rates. Although the trend is not statistically significant, we believe our sample size post-implementation is not large enough to demonstrate statistical significance. Stopping premedications after the first two infusions without demonstration of reaction in patients may reduce the exposure to systemic glucocorticoids, decreases appointment times/chair time, and aids in the convenience of administration of paclitaxel.
The implementation strategy was key to the successful deployment of the interventions. Where possible, the changes were built into workflow processes, removing the need for providers, nursing, or pharmacists to refer to guidelines or policies. To prevent variation in prescribing practices, the oral route has defaulted in the computerized order entry for diphenhydramine and famotidine. Premedications for the first and second doses have defaulted. Syringe pumps were removed from all infusion areas to prevent use. Nursing staff were educated about all the changes implemented including standard dilution practices. Dedicated nursing education days were used, and competency assessments were completed. A multidisciplinary approach provided a comprehensive outlook of workflow processes and stronger support to implement institutional changes.
Through this retrospective review, a comprehensive review of deviations from our standard practice was performed. There were some patients who received IV premedications in the post-standardization group. This may be explained by legacy order sets with non-standard premedication regimens. Additionally, there may be a subset of patients who are unable to take oral medications, necessitating the need for IV administration. There is a subset of patients in the post-standardization group who did not have documented premedication that may be explained by transferring from an outside institute or re-entry of an order set flagging as a “new” initiation.
A hypersensitivity trigger tool may serve others in identifying and characterizing hypersensitivity for other types of medications. Paclitaxel infusion reactions were flagged when rescue medications were provided to patients after the start of the paclitaxel infusion and confirmed by cross-referencing nursing notes. Nursing documentation is key to ensuring the interdisciplinary team's ability to formulate a plan for the patient, accounting for the severity and management of the HSR. A manual retrospective review was necessary to ensure that the trigger tool reflected a paclitaxel HSR, as paclitaxel is often administered with carboplatin. Carboplatin carries a risk for infusion reactions usually occurring after multiple exposures to the platinum agent.19,20 Additionally, this trigger tool is only able to capture infusion reactions grade 2 and above, because of the necessity for the administration of a supportive care agent. 15 Overall, using rescue medications as a trigger tool for adverse drug events can be implemented at infusion centers assesses the rates of HSRs to paclitaxel and other agents with unique rescue pharmacotherapeutic agents. This generalization to other medications can help improve workflow processes that may not be easily identified by staff.
There are some limitations to this retrospective trial. Clear documentation was necessary to grade HSRs. Additionally, some treatment plans may have continued past the post-intervention date, as an adoption grace period was not implemented. Another limitation is the small sample size of the post-intervention group due to the limited time frame compared pre-implementation group. Due to the small sample size of the population, caution should be taken to generalize these results to a larger population that may not be reflective of what was studied. Gynecologic malignancies have well-documented hypersensitivity reaction rates. 21 Patients living with breast cancer were the majority of the population studied. The results of this trial should be further investigated with a larger gynecologic population in order to further validate institutional interventions employed in this study.
In conclusion, this retrospective interventional study demonstrated that same-day intravenous dexamethasone, oral H1RA, and oral H2RA is a convenient and safe options for the prevention of paclitaxel HSRs. The study also showed a decline in first and second-dose HSRs with the implementation of required premedications, improved ability to time premedications with the change to oral administration, and cost savings for diphenhydramine, famotidine, and ondansetron. Dropping premedications after the second dose of weekly paclitaxel may prove useful in decreasing systemic exposure to corticosteroids. Additionally, there may be a theoretical benefit with the reduction of line manipulations using an oral premedication regimen. Through multidisciplinary collaboration, standardization of order sets, and education prescribing habits were effectively changed. This study demonstrated the usefulness of a trigger tool to identify workflow improvements in paclitaxel, and the possibility of generalization for other medications that are commonly associated with infusion reactions. More studies are needed to further elucidate if these changes could translate to other clinical outcomes such as infection risk or patient satisfaction.
Footnotes
Author contributions
All authors contributed to this retrospective review's design and conception. Manuscript preparation and data analysis were performed by the first author. The first draft of this manuscript was written by the first author and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the University of Washington Human Subjects Division Institutional Review Board with a waiver of consent granted due to the retrospective nature of this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
