Abstract
Background
Premedication of cancer therapy against nausea and vomiting (NV) and hypersensitivity reaction (HS) is essential for good patient management. However, this prescription is not always optimal. Today, as a large part of cancer therapies are administered in day hospitals (DH), premedication taken on the day of the cancer treatment is injected as a 30-min infusion.
Objective
To assess compliance with recommendations for premedication prescription and intake; to analyse patient attitude about switching to exclusively oral forms taken at home.
Method
The study is conducted in the medical oncology DH of a French Hospital from 17 January to 25 February 2022. The data collection is carried out as an individual interview, associated with the distribution of two questionnaires. Data are coupled with the premedication set up on our software and the last medical report. Intakes are considered optimal when recommendations, tolerance, background, and adherence of the patient are taken into account.
Results
Seventy patients were included for interviews. Regarding software prescriptions, our configuration was consistent with recommendations in 100% of cases for HS and 37% for NV. Intakes were compliant in 51.4% of cases, non-compliant in 17.1% and debatable in 31.5%. Disparities between the practices of different physicians were identified. Regarding the feasibility of oral substitution, it could concern 63.5% of patients.
Conclusion
This work makes it possible to improve the management of all patients and to make the operation of the care unit more fluid.
Introduction
Advances in oncology have been constant and have led to better management of cancers and improved quality of life.1,2 However, both former and new treatments have adverse effects. Some of them are non-specific such as nausea and vomiting (NV) and hypersensitivity reactions (HS).3,4 These toxicities impair patients’ quality of life and may lead to postponement or even discontinuation of cancer therapy. 5 For example, unmanaged NV leads to delayed management of cancer in 20% of cases. 6 Treating properly and systematically these adverse events is essential.
Among the arsenal of management available to avoid these two toxicities, pre- and post-medications have shown their value.7,8 Many learned societies recommend specific combinations of drugs to prevent NV. Protocols differ according to the emetogenic risk of the cancer therapy (minimal, low, moderate, high) and the patient's condition. 9 Management will be increased if several risk factors are present, including young age, female gender, anxiety, sleep disorders before treatment, or a NV background. 10 The main drugs that can be used are serotonin antagonists (anti-5HT3), neurokinin type 1 receptor antagonists (anti-NK1), corticosteroids, short half-life anxiolytics and olanzapine.7,10 If recommended, pre- and post-medication for HS are summarised in the instructions of the cancer therapy. These are mainly H1 and H2 antihistamines and corticosteroids.8,11–15
Despite their proven value, premedication are often neglected. 16 According to Roeland and al in 2020, 35% of physicians who prescribe premedication for chemotherapy protocols including cisplatin are not doing it in an appropriate manner. 17 The prescription of premedication for chemotherapy with another emetogenic risk has also been shown to be suboptimal. 18 No study about the rest of the premedication for HS or for HS and NV both has been found.
Various factors may explain these inappropriate prescriptions: on the one hand, the correct prescription of premedication may seem to be a minor consideration compared to the complexity of the overall patient's management. On the other hand, it is very tedious, because of the large number of sources describing the management of toxicities and their relevance,9,19 and the many prescription supports. For example, a premedication may be set up as part of the chemotherapy protocol, or it may be added manually by the physician. It is not always easy to know the regulatory restrictions of these drugs: they can be available in pharmacies or only in hospitals, reimbursement is sometimes restricted to a particular indication, a specific form of prescription is needed. And there is an additional difficulty: beyond the standardised prescriptions recommended by pharmaceutical companies and learned societies, risk factors of patients should be consider to complete the management.20,21 However, the decision is entirely left up to the doctor's discretion.
For all these reasons, it is essential to improve our prescriptions and to ensure that the patient takes the premedication correctly to achieve optimal quality of care and life. This is particularly important in DH, as the patient goes home after the administration of cancer therapy, without prolonged monitoring. If delayed adverse effects occur, the adjustment of management is less easy than in conventional hospitalisation, and their reports to health professionals is lower. 16
Despite these difficulties, outpatient activity is constantly increasing and is reaching saturation: in our establishment, DH in medical oncology represents 90% of patient stays compared to conventional hospitalisation. In this context and keeping in mind that it is essential to maintain an optimal comfort for patients and to accompany them through any changes, we can ask ourselves whether it would be relevant to take all premedication at home and orally, if patients accept it and if the situation allows it.
Today, premedication taken on the day of cancer treatment is mostly administered by nurses by intravenous (IV) infusion over 30 min. The other pre- and post-medications, taking care of anticipated and delayed NV, are already taken orally, at home by the patient.
The aim of our study is to evaluate the relevance of patient prescriptions and intakes in medical oncology DH and to analyse patients’ attitude about a substitution towards oral forms taken at home exclusively. The objective is to ensure optimal and comfortable prescription of premedication, by adapting and harmonising protocols if necessary and by supporting patients in this change.
Method
Study design
This is a prospective, uncontrolled, single-centre observational study.
Study sample
The study is conducted in the medical oncology DH of a French University Hospital, treating almost exclusively cancers of digestive, urological and ears-nose-throat (ENT) location.
The data is collected by the same operator for five weeks, five half-days per week, from 17 January to 25 February 2022 (i.e. 25 half-days).
Giving the frequency of cancer treatment (often every 7, 14 or 21 days, sometimes monthly), the duration of five weeks for the study was found to be representative of the DH activity. This time slot gives the opportunity to include patients with all types of cancer treatment and to have most of que questionnaires back. Furthermore, this duration was short enough to keep the study easily reproducible.
Involvement in the study is presented to all patients scheduled and present in the department during these time slots. They must have IV premedication related to their current cancer treatment, be volunteers and able to communicate.
The following patients are excluded from the study: institutionalised or homeless, not speaking French, not having premedication, not wishing to participate in the study and included in clinical trial protocols.
Data collection
Data collection is carried out as an individual interview with patients during the administration of their cancer treatment.
A number is assigned to each patient who agrees to participate in the study to maintain their anonymity. All data collected are pseudo-anonymised and are detailed below.
- Socio-demographic characteristics: sex, age, travel time between home and hospital - Disease and treatment: nature of the cancer, cancer treatment and premedication, number of cycles’ treatment - Background and tolerance: history of NV (drug-induced, transport, anxiety) and HS (related or not to the administration of cancer therapy), presence of NV or HS during the previous cycle of treatment (48 h before administration and up to five days after), notion of a change in premedication since the last treatment - Adherence of the patient: assessment of the intake of pre- and post-medications already prescribed orally (if concerned), independent intake or presence of a caregiver - A Girerd questionnaire with six closed questions to assess the patient's overall compliance with their usual treatments - A questionnaire assessing patients’ attitude towards a substitution of premedication by oral forms, taken at home when the situation allows it. The questionnaire is composed of 11 statements for which the patient is asked to position himself on a four-point Likert scale: completely agree, agree, disagree and completely disagree. The questionnaire was drawn up by the pharmaceutical team and validated by the medical team. It is presented in Appendix 1.
At the same time, two questionnaires are given to patients, with the instruction to return them completed. They were used to complete the data collected during the interview.
The data collected for each patient was then coupled with their premedication set on our software and their last medical report.
Data analysis
To analyse the premedication set up in our software and those taken by patients in an uniform way, we decided that recommendations used as reference will be those of the AFSOS (French association of support care in oncology) for NV and of pharmaceutical companies’ instructions for HS. Cancer drugs particularly at risk of HS used in medical oncology are avelumab, carboplatin, cisplatin, cetuximab, docetaxel, oxaliplatin, paclitaxel and ramucirumab. This decision was made jointly by both pharmaceutical and medical team.
A quantitative analysis using data collected will be realised, with proportions, percentages and the conduction of statistical test at risk α = 5%. The objective is to obtain the following information:
- Concerning general characteristics of patients’ sample:
Profile of patients interviewed: sex, age, cancer site and stage Proportion of patients with risk factors or background, that is, young women, history of NV (transport, anxiety, medication) or history of HS (related or not to medication) Proportion of patients with NV (grade 1 and above) and HS reaction (grade 1 and above) during the previous cycle of treatment - Concerning the relevance of premedication prescriptions and intake:
Proportion of prescriptions properly configured on our software, that is, strictly in accordance with AFSOS recommendations for NV and laboratories instructions for HS, without considering neither the background nor the patient's tolerance. A prescription of this type constitutes the current configuration objective. If necessary, the physician can adapt the management of patients by prescribing additional premedication or removing them. This decision for configuration was made when the software was settled in our hospital to standardise the care. Proportion of patients who take premedication properly, considering the compliance with recommendations, the patients’ adherence to treatment, their background and their tolerance during the last treatment Impact of a potential under-prescription of premedication on the frequency of NV, with a Chi-square statistical test carried out at a α = 5% risk. - Concerning the harmonisation of premedication prescriptions between physicians: identification of possible differences in prescribing for patients with the same emetogenic and HS risk, proportions of the different practices. - Concerning a possible substitution of premedication by oral forms taken at home:
Proportion of patients claiming to have taken their oral premedication properly during the last cycle (if concerned) Proportion of patients declaring themselves autonomous in taking all their usual treatments (if concerned) Proportion of patients declaring to be globally adherent (score of 0 or 1 according to Girerd) Patients’ ability to swallow medications (especially for patients with ENT cancers) Patients’ attitude about substitution: proportion of patients in favour of change, nature of perceived obstacles and benefits
Ethical considerations
Patients are systematically given an information note on the study before any potential participation. Their informed consent is obtained orally.
The research protocol of the study was validated by a local group of ethics in the field of health on 14 January 2022, and registered with the clinical research department of the University Hospital.
Results
Sample studied
According to provisional schedule, 15–20 patients come to the medical oncology DH each day.
There were 532 visits between 17 January and 25 February 2022, of which 332 were related to the administration of a cancer drug requiring injectable premedication.
The details of the patients screened, included, and excluded from the study are presented in Figure 1. Finally, 70 patients were interviewed by our operator. Of these, 63 patients returned the Girerd questionnaire and the questionnaire assessing their attitude towards oral substitution, that is, 90% of the patients interviewed.
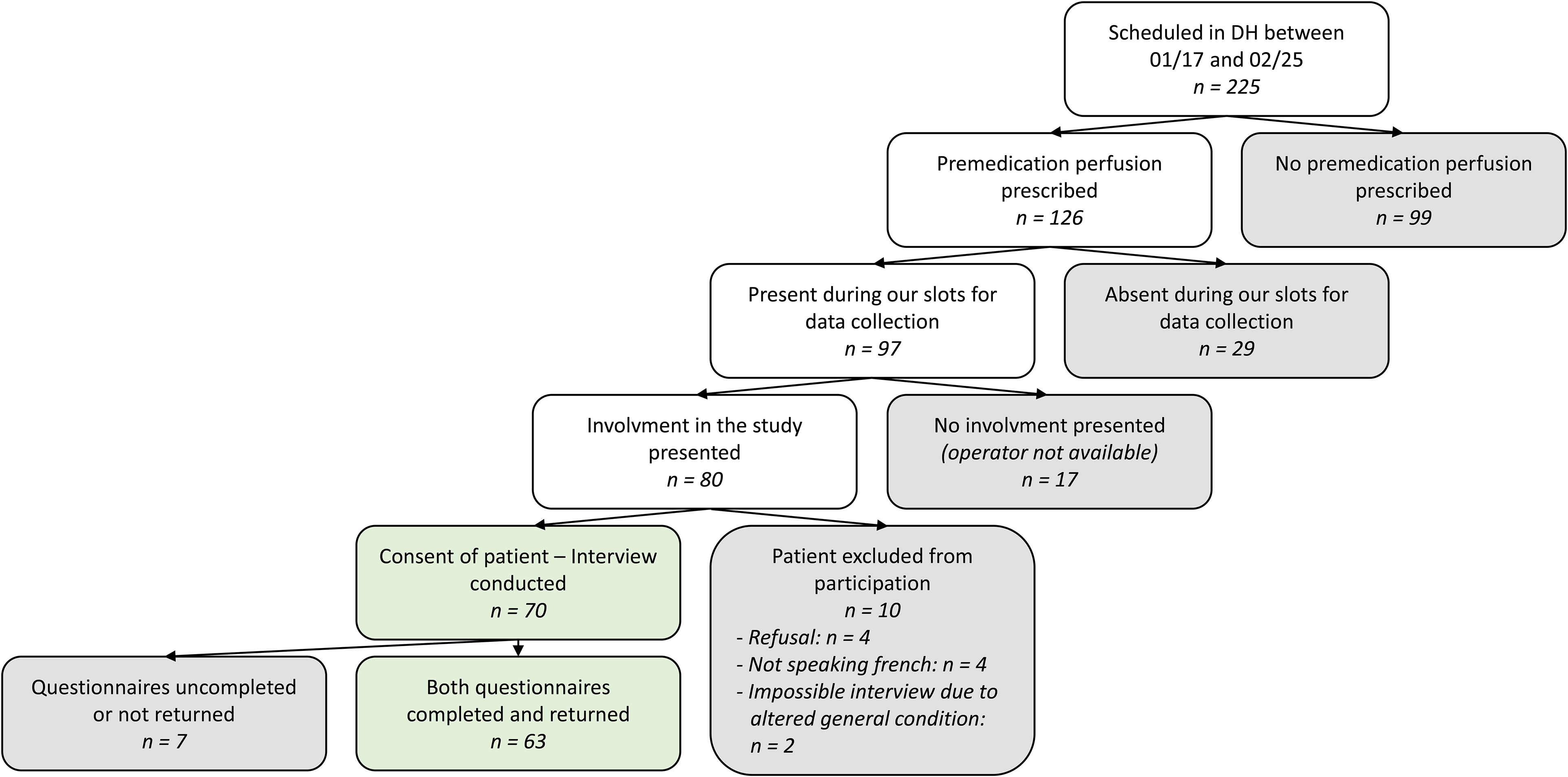
Selection of our sample – included and excluded patients.
The results presented are divided into several parts: first, general information about the patients, their treatment, their background, and their tolerance to cancer drugs; then an analysis of premedication prescriptions on our software and intakes by the patients; a review of the main disparities found among the prescriptions; and finally, a section on the feasibility of substituting premedication with oral forms taken at home.
General characteristics – background – tolerance
The age, sex, type, stage of cancer, risk level of treatment in terms of NV and HS, presence or absence of background and tolerance to the last treatment of the 70 patients interviewed are summarised in Table 1.
Summary of the general characteristics of the patients interviewed and their treatments.

Relevance of software prescriptions and premedication intake
Prescription on the software
The analysis of software prescriptions for patients allowed us to define proportions of consistency with recommendations for NV and HS (Table 2).
Consistency with AFSOS recommendations for NV and laboratory instructions for HS, without considering potential risk factors.

Among all these prescriptions, it is important to note that 100% of premedication settings for HS are compliant. For NV, only 37% of the premedication protocols are following the AFSOS recommendations. The anomalies are mainly due to poor configuration of moderately emetogenic chemotherapy (4% consistent) and highly emetogenic chemotherapy (14% consistent). The minimally emetogenic and lowly emetogenic protocols are quite well set up (100% and 60% consistent, respectively).
Actual intake of patients
The joint analysis of software prescriptions and data collected for each patient allow us to consider: AFSOS recommendations, laboratories instructions as well as the patient's tolerance and background. For each patient, the pre- and post-medication taken are considered to be appropriate, inappropriate or debatable. The summary of the results is described in Figure 2.
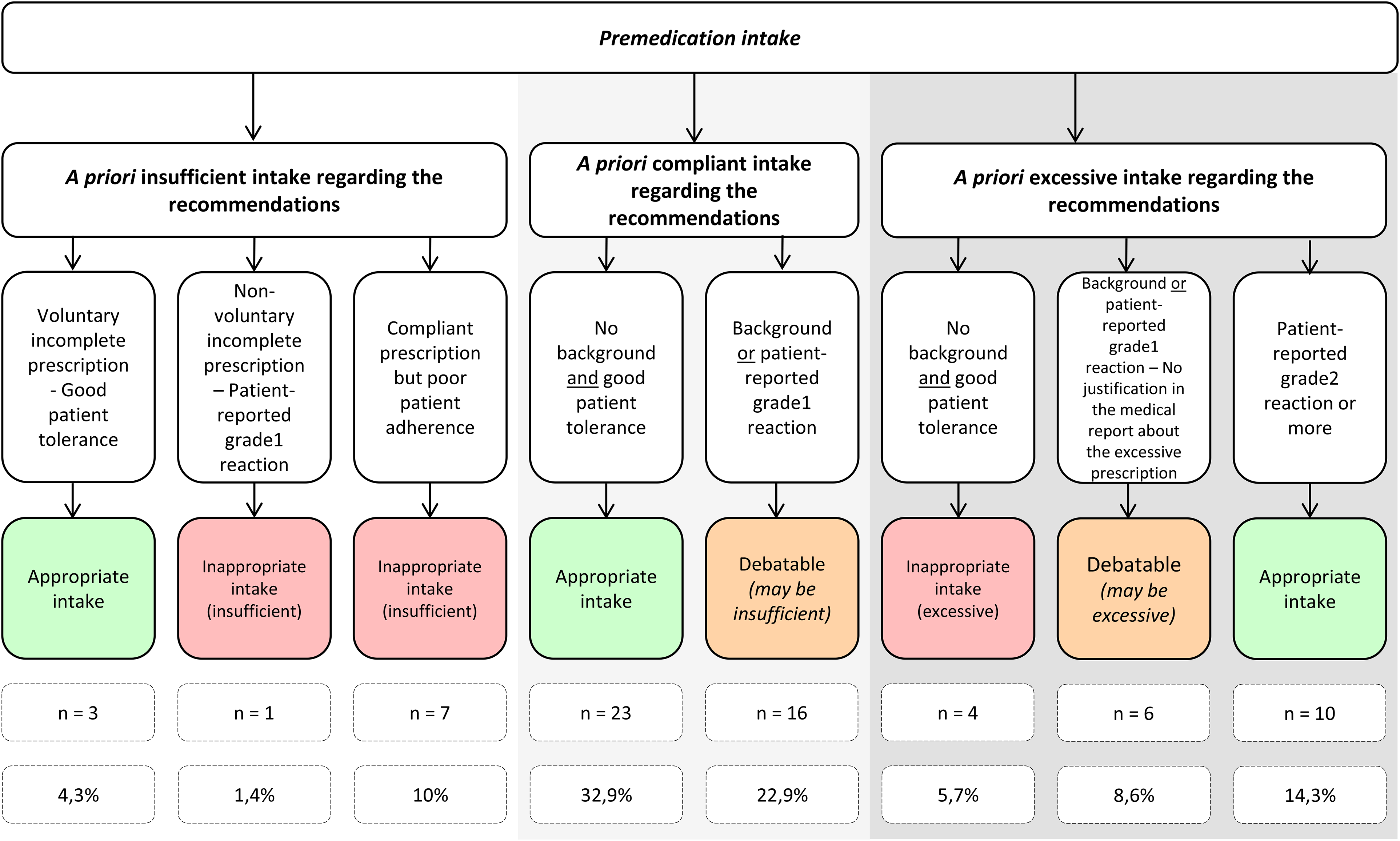
Summary of the relevance of premedication intake, considering recommendations (AFSOS and laboratories instructions), patient's tolerance and background.
All in all, 51.4% of cases were appropriate and 17.1% were not. In 31.5% of cases, the relevance of premedication can be discussed. In 18.75% of cases where the intake is debatable and potentially insufficient, the problem is caused by under-reporting by the patient to the physician. The prescriber cannot adapt to the management because he is not aware of a reaction.
Impact of under-prescribing premedication:
The aim was to determine whether under-prescribing NV medication alters the risk of NV compared to correct or over-prescribing. The results obtained are described in Table 3.
Number of patients affected by NV according to premedication intake (insufficient vs. compliant and excessive).

A Chi-square test with 1 degree of freedom was performed and interpreted at α = 5% risk. There appeared to be a bigger risk of NV when intake was insufficient compared to correct or excessive (69.6% vs. 29.8%; P < 0.01).
Variations in practices
Oxaliplatin
Prescriptions of premedication for HS reactions were compliant but not identical. For 4 out of 20 patients (20%), dexchlorpheniramine were prescribed to prevent HS reaction. For the remaining 16 patients, no specific management for HS risk is offered.
Ondansetron
For six patients, oral ondansetron 8 mg was prescribed for 5 days after the administration of cancer therapy. Three patients were taken it consistently and three when needed. Each time, they were complex patients with resistant NV. This additional prescription of ondansetron did not eliminate NV in any case.
Corticosteroids
Corticosteroids prescribed were IV methylprednisolone and oral prednisolone. Taking AFSOS recommendations as reference, only 4 patients out of 61 (6.6%) were prescribed a corticosteroid dosage consistent with these recommendations. For 17 patients (27.9%), the prescribed doses were superior to those recommended by the AFSOS, but this could be explained by the concomitant administration of a cancer drug with a known HS risk. Given this questionable situation, these prescriptions were considered to be appropriate, even if a work of harmonisation and research about relevance is needed. For 40 patients (65.5%), the doses prescribed were superior to those recommended by AFSOS, without justification.
Lowly emetogenic protocols with associated HS risk requiring corticosteroid premedication
Fourteen patients were concerned: of these, eight were prescribed corticosteroids as the only premedication, which played the role of anti-emetic and anti-allergic both. For the remaining six patients, the management was distinguished with two premedications: one for HS and another for emetogenic risk (Figure 3). All these prescriptions are considered to be appropriate, but a work of harmonisation is needed.
Premedication for NV in low emetogenic protocols with associated HS risk.
Lowly emetogenic protocols with no specific risk of HS
In case of a lowly emetogenic protocol, only one premedication should be chosen. This medication changes from patient to patient. Of the 30 patients concerned, 10 were taking an anti-5HT3 (ondansetron), 8 corticosteroids. For the other 12 patients, an irrelevant prescription of two associated drugs was found (Figure 4).
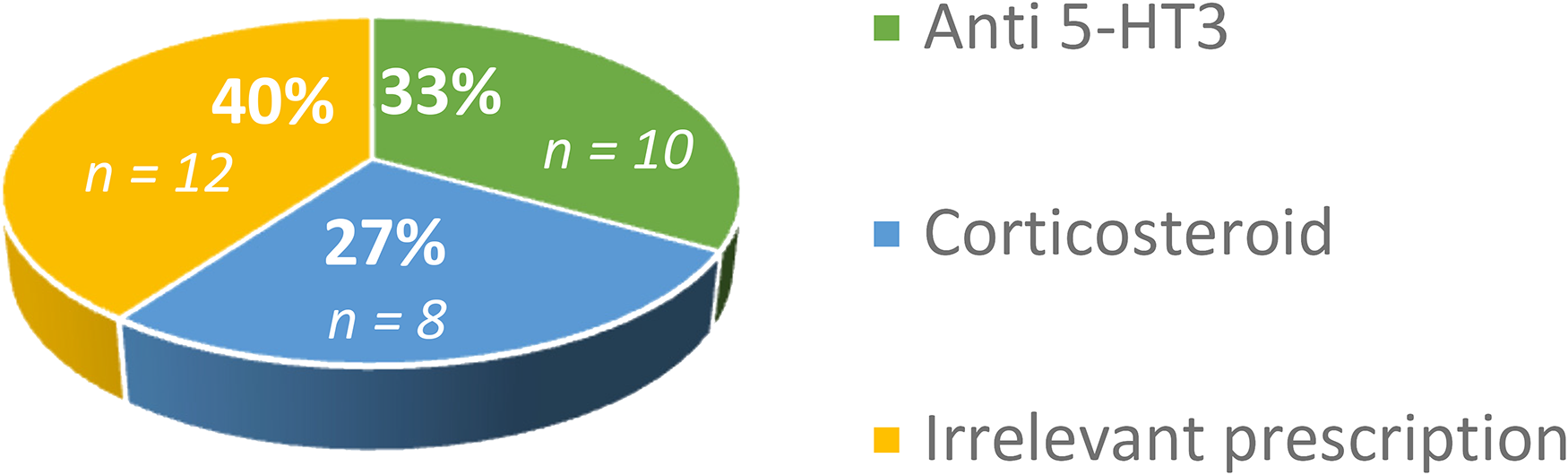
Premedication against NV in case of lowly emetogenic protocols without associated HS risk.
Compliance and attitude with oral substitution
In our study, substitution of premedication to oral forms is considered achievable by the medical and pharmaceutical team if four criteria are met.
- If concerned, the patient must declare that he/she had taken his/her pre- and post-medication properly during the previous cycle of treatment; - their Girerd score must be 0 or 1 (corresponding respectively to very good compliance and a minimal compliance problem); - they must be usually independent with taking their treatments - they must be able to swallow medications (especially patients with ENT cancers) and they must be in favour of taking oral premedication at home (they must have completely agreed or agreed to the question “Would you be in favour of taking oral premedication at home before going to the hospital?”).
The results are detailed in Table 4.
Number and proportion of patients meeting each of the four criteria for oral substitution of premedication, n = 63 patients.

Among the perceived advantages of substitution, 86.8% of patients consider that taking oral medications is more comfortable, 92.1% perceive the reduction in the workload of the nurses currently preparing and monitoring IV premedication as positive, 88.9% are in favour because it would allow them to spend less time in hospital. Among the potential obstacles, 26.9% of patients are worried about forgetting their treatments, and two patients added that they do not wish to shorten their hospital visit because they are reassured to be taken care by health professionals.
Discussion
Profile of patients interviewed and relevance of premedication
According to the epidemiology of cancers in France, our sample is representative of population, in terms of the male/female ratio, age and type of cancer. 22
According to scientific literature, the proper use of premedication allows the elimination of NV in 70–80% of cases, that is, NV persists for 20–30% of patients. 16 In our study, about 40% of patients had NV during the last cycle of treatment. The frequency of HS varies widely between studies and cancer drugs administered. 23 Following our results, approximately 15% of patients reported HS at the last treatment, which is compatible with the data usually observed in the literature.
The configuration of premedication prescriptions in our software must be improved. Our goal is to comply with recommendations without considering the patients’ possible risk factors. The physician must then complete his management by computer to adapt treatment to the patient's background and tolerance. In our study, only 37% of premedication for NV was set up properly, which means that pharmacists and doctors need to work on re-setting these premedications. Particular attention will be paid to the highly and moderately emetogenic protocols, as well as the setting of Aprepitant, since these three elements represent a large proportion of the non-compliances in setting.
This poor configuration is nevertheless partially rectified with manual prescriptions. Finally, more than half of premedication prescriptions are appropriate, that is, comply with recommendations and are adapted to patient's situation.
One-third of the intake of premedication are considered debatable. Indeed, some reactions (NV or HS) are sometimes considered minor by the physician. The prescription is then strictly in accordance with the recommendations and does not consider this problem of tolerance. On the contrary, in some cases, the treatment is upgraded, even though the patient does not declare any background and no justification is found in the medical file. In both cases, these treatments can be justified, but there is still a need to harmonise practices.
Premedication were non-compliant in approximately one case out of six. This non-adapted intake is linked to poor adherence on the part of the patient in half of the cases, which underlines the importance of follow-ups and therapeutic support.
Our study also linked an increased risk of NV to insufficient premedication (69.6% v 29.8%; P < 0.01). The correct use and prescription of premedication is therefore essential and not only allows a reduction in adverse effects, but also an improvement in quality of life, a reduction in visits to emergency department and decrease the costs associated with hospitalisation. 24 In the same way, good management of HS can make it possible to continue a first-line treatment instead of downgrading to another cancer therapy, potentially less effective. 8
Harmonizing practices
Oxaliplatin
Oxaliplatin is moderately emetogenic and has a known HS risk. The mechanism involved remains poorly understood. It seems to be IgE-related, and a few cases of IgG-mediated reactions have been described. 23 The reaction usually occurs after the 6th infusion. 25 Data in the literature are sometimes inconsistent. It would appear that premedication is only useful in limiting grade 1 or 2 reactions but has no effect on more severe reactions.26–28 Given the questionable usefulness of these premedication, the presence or absence of premedication is considered appropriate in our study. In the future, our goal is to harmonise our prescriptions by removing premedication against HS in the case of oxaliplatin administration. They will only be offered to patients with a high-risk background or a history of poor tolerance.
Ondansetron
Ondansetron may be prescribed for the management of delayed NV up to 5 days after administration of the cancer medication. This prescription is therefore considered appropriate in our work. However, Dranistaris et al. 29 state that it is preferable to prescribe it only as a single dose during the first 24 h after administration of the cancer medication, as its efficacy is too low compared to its price. 29
In our study, ondansetron was taken over five days by six patients, each time in complex situations, in case of resistant NV, after trials and failures of other therapies and with no alternative in the therapeutic arsenal of our institution. However, this type of prescription did not eliminate NV in any of our patients. We can ask ourselves whether it would be relevant to offer palonosetron for these patients and therefore to include it in our arsenal. Palonosetron has been shown to be more effective than ondansetron in case of resistant NV with highly emetogenic chemotherapies.30,31
Corticosteroids
Doses found in our study are not harmonised and vary from 40 to 120 mg methylprednisolone (i.e. from approximately 7.5 to 22.5 mg dexamethasone equivalent). Prescribing the optimal dose is not always straightforward as the recommended dose varies according to both the risk of emesis and HS.
For the management of NV, a precise dose is specified in the guidelines (12 mg dexamethasone or equivalent for the first day of a highly or moderately emetogenic protocol, 8 mg in all other cases).
For HS, the recommended dose is 20 mg for paclitaxel and 16 mg for docetaxel. The recommended dose for ramucirumab and cetuximab are not specified in laboratories instructions and are left to the physician's discretion. Lansinger et al. 32 compared the occurrence of HS following taxane administration when the dose was 10 mg or 40 mg of dexamethasone. No difference in the occurrence of adverse events was found. Therefore, we have decided to harmonise all HS management with dexamethasone 10 mg (or equivalent) for the four cancer drugs listed above. 32 The dose will be adjusted if the patient has a high-risk background.
If the protocol requires corticosteroid management for both NV and HS, the higher dose of these two risks should be prescribed and the two doses should not be added together. 33 If the protocol is lowly emetogenic, a single prescription of corticosteroids is sufficient. The addition of another antiemetic is unnecessary. 34
Lowly emetogenic protocols
After discussion with the medical team involved, it was decided that corticosteroids would be systematically preferred in the case where a single antiemetic would have to be chosen. This choice will harmonise practices and reduce the risk of HS.
Oral substitution of premedication
Oral substitution is achievable according to our four previously established criteria for approximately 60% of patients. Based on the same number of patients as at the time of our data collection, this would represent a saving of 30 min in the management of seven to eight patients per day, that is, a save of 3.5–4 h over a full day. Time for preparation and monitoring by nurses and cost of medications and medical devices necessary for IV infusion will also be spared.
Medications proposed for the oral substitution are: cetirizine as an anti-H1 instead of dexchlorpheniramine, which has shown its iso-efficacy while being less sedative35–37; ondansetron as an anti-5HT3 because it is the best balance between efficacy and cost, 38 prednisolone or dexamethasone depending on the protocol.
However, this oral switch should not be made at the expense of patient comfort. Patients for whom the four criteria are not met should remain on IV therapy. The same applies to patients treated with paclitaxel. Indeed, two premedication regimens are proposed in the laboratory instruction for this drug: an oral regimen with corticosteroids taken 12 and 6 h before the dose, and an IV regimen, with a single injection 30 min before the infusion of the cancer medication. After discussion, we decided to keep premedication on IV form in order to avoid altering patients’ comfort. 39 The oral form is slightly more effective but the balance is not in favour of oral administration, as the risk of non-adherence is very high, because patients would have to wake up the night before their appointment to take their oral premedication. 40
It is also essential to have a standardised back-up solution if patients forget to take their premedication before their appointments. Indeed, more than a quarter of patients were concerned about their tolerance if they forget it. Hsu et al. 41 propose a systematic regimen of dexamethasone 8 mg in case of forgetfulness with good efficacy and minimal time loss. 41 Similar solutions will be proposed for missed cetirizine or ondansetron.
Role of the different actors
This project to improve premedication practices should involve all actors who have a role in the management of premedication, whether it be the patients themselves, the physicians, the nurses, or the pharmacists.
Patients should be involved in their care. Health professionals should encourage them to report their adverse events. In our study, 20% of the potential under-use of premedication was related to a lack of reporting to the nurses or the medical team. Similarly, more than half of the non-compliances were related to lack of adherence by the patient and not to an inappropriate prescription. It has been studied that patients with NV often have a poor adherence, 42 which underlines the need of therapeutic support and the interest of pharmaceutical interviews.
Nurses are the health professionals who spend the most time with patients. They are therefore in good position to identify patients at risk regarding the tolerance, the misunderstanding of their treatment and the poor adherence. They are currently in first line for re-explaining the treatment regimen.43,44 In our study, the nurses were made aware of the project: presentation of the initial work to the whole team, data collection at the time of chemotherapy administration and monitoring, opinion about substitution collected to ensure the feasibility of the project, feedback afterwards.
Pharmacists also have a role to play in improving practices. Greaves et al. 42 looked at the impact of a pharmaceutical presence directly in the care unit. Promoting the proper use of premedication and providing therapeutic advices directly to patients led to an increase in compliance with recommendations, a decrease in premedication consumption, a decrease in the occurrence of adverse events to be managed by doctors, and thus enabled other patients to be taking care of. 42
It is also the pharmacists who set up the premedication and cancer protocols in our institution. These protocols are then validated and if necessary, adjusted by the physicians. When the prescribing software was deployed in our institution a few years ago, the change had to be made quickly and the first need was to set up the cancer drugs properly. Premedication was not a priority at the time, which explains why compliance with the recommendations against NV is only 37% in our data collection. It is therefore up to pharmacists to offer a new basis for work, in line with recommendations and adapted to the practices of our centre. It would be relevant to propose some tools to facilitate an optimal prescription.
Indeed, Roila et al. 45 showed that a simple audit of premedication with feedback to the department is not sufficient to significantly improve practices. Only real support for change has made it possible to improve them.45,46 This support can be a therapeutic follow-up programme or the implementation of standardised management protocols in the prescription software, adapted according to the nature of the cancer drug administered. Additional premedication would then be proposed directly in our software to upgrade the management in case of poor tolerance and high-risk background. The aim is to make the prescription of premedication as safe as possible, and it has been shown that prescribing with a software reduces the risk of error.47,48
In addition, Nolte et al. 49 studied the impact of creating a checklist to standardise the prescription of premedication. This action enabled compliance with the recommendations to rise from 73% to at least 87%, and the premedication budget to be reduced by more than two. Our objective is to carry out similar work but to integrate it directly into the prescription software.
Conclusion
Optimising the use of premedication is a multidisciplinary task involving doctors, pharmacists, nurses, and patients themselves. It is essential to seek the support of all these actors to improve practices. Our study has highlighted the importance of therapeutic support and pharmaceutical interviews to strengthen patients’ adherence to their treatments. It has also proved to us the need to upgrade the configuration of premedication in our software to propose a standardised basis that can be adapted to patients’ needs and circumstances. This “re” configuration will make it possible to propose exclusively oral forms for patients for whom substitution is feasible. This work will allow us to improve the management of all patients and to make the operation of the care unit more fluid.
Footnotes
Author Contributions
AG researched literature. NC, AG, EP, CF, CLG and DC conceived and developed the study (including the data that should be collected, the questionnaire and the ethical protocol). LL and AG were involved in patients recruitment, interviews and data analysis. AG wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and aproved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1: Questionnaire assessing patients’ attitude towards a substitution of premedication by oral forms,taken at home when the situation allows it
