Abstract
Introduction
Extravasation is a potentially severe complication of intravenous administration of antineoplastic drugs. The limited data makes it difficult to develop an optimal management scheme. The objective of this study is to describe the clinical practice in the extravasation management of antineoplastic agents in Spanish centers.
Methods
An online survey was distributed to oncology pharmacists using the email distribution list of the Spanish Society of Hospital Pharmacists. Respondents were surveyed on the standard operational protocol (SOP) of extravasation, tissue damage risk classification, and specific measures of extravasation management.
Results
A total of 68 surveys were completed. A specific extravasation SOP was available in 82.4% centers. The pharmacist participates in the authorship (100%) and actively collaborates in extravasation management (76.5%). A tissue damage risk classification based on the three categories was mostly adopted (48.2%) and 73.2% applied specific criteria based on concentration and/or extravasated volume. Extravasation management was mainly performed with the application of physical measures and/or antidotes (91.2%). High variability in the choices of pharmacological and/or physical measures recommended is outstanding.
Conclusion
The results of this study highlight the involvement of Spanish pharmacists in extravasation management, the application of physical measures and/or pharmacological measures as the method of choice in extravasation management, as well as the existing discrepancies in tissue damage risk classification and management recommendations
Introduction
Extravasation is rare although potentially dangerous complication related to intravenous administration of antineoplastic agents. The severity of clinical consequences of extravasations depends mainly on the chemical nature of the extravasated drug, its concentration, and extravasated volume. 1 If left untreated, extravasation can cause progressive tissue destruction requiring surgical treatment.1,2 The injuries caused are often painful due to functional deterioration of the affected structures, they can require hospitalization and can affect considerably the patient's quality of life.
Data on the incidence of extravasations caused by antineoplastic drugs are scarce owing to the absence of national centralized registries and low rates of published extravasation reports, mainly those cases without clinical consequences. Different studies estimated the incidence of extravasation in a range between 0.1% and 6%.3–5 However, recent reports stated much lower rates around 0.01–0.05%, after years of intravenous administration improvements as well as better preventive measures.6,7
Numerous institutions, scientific or professional societies, have developed guidelines to improve early diagnosis and its prevention and treatment, with the ultimate goal to reduce the risk of extravasation and minimize the damage to the patient.8–14
The Spanish Group of Oncological Pharmacy (GEDEFO) of the Spanish Society of Hospital Pharmacy (SEFH) has also developed a specific consensus guide for all health personals involved in the care of oncological patients, as an updated operational guide for the prevention and management of antineoplastic drug extravasations to be adopted at a national level. 15 This guide incorporates the best available scientific evidence to date, including the consensus of a group of oncology pharmacist experts. After all the literature analysis process, great variability was unveiled both in the classification of tissue damage caused by antineoplastic drugs and in the specific treatment of extravasation, both in the type of physical and pharmacological measures recommended and in their administration guidelines. This variability was related to the low quality of the available information. Literature related to chemotherapy extravasations often comes from small uncontrolled studies, case series, and few studies in animal models. In addition, the results may be contradictory or incomplete, making the decision-making process even more complex.
The objective of this study is to describe the clinical practice in the management of extravasation of antineoplastic agents in Spanish centers, prior to the publication of the GEDEFO consensus document and focused on three key aspects: availability of a standard operational protocol (SOP) on the subject, classification of the risk of tissue damage and extravasation management (pharmacological and nonpharmacological measures).
Methods
An online survey on antineoplastic drug extravasation was designed using the REDCap®-SEFH web platform. The questionnaire was designed by the expert coordinators of the extravasation project of the GEDEFO group. Besides, the survey was approved by GEDEFO executive committee. This study did not require the approval of the Clinical Research Ethics Committee.
In April 2021, the survey was distributed to all oncology pharmacists using the email distribution list of the SEFH. Only one response per hospital was allowed. The survey was available for a period of six weeks (04/13/2021 to 05/23/2021).
The questionnaire was structured in two different sections, each of them divided into several parts.
Section 1: General data
− Demographic data − Hospital types (general, children, oncology, others) − Professional experience of the respondent − SOP of extravasation:
Availability, development, and updating of the SOP Pharmacist involvement, extravasation, documentation, and training − Tissue damage risk classification
Specific classification criteria List of drugs of interest and their classification − Specific measures in extravasation management (pharmacological and physical):
General criteria for applying measures Specific criteria for applying antidotes Type of measures, pharmacological measures (antidotes), and dosage regimen List of drugs of interest and measures used
Section 2: Specific data
The classification of tissue damage was adopted and the specific measures used in each center for 23 different antineoplastic agents, 17 in each section, were investigated. These drugs of interest were selected based on conflicting data previously observed in both classification and management.
Qualitative variables are presented as absolute values and relative frequencies by category. Statistical analysis was performed with the statistical program IBM SPSS v23.0 (IBM SPSS Statistics for Windows, IBM Corporation, Armonk, NY).
Results
A total of 68 surveys from 14 of the 17 autonomous communities and two autonomous cities of the Spanish national territory were completed. This represents data from about 84% of all Spanish territory. The geographical origin of one of the surveys received was not recorded.
The characteristics of the health care centers and participants are described in Table 1.
Characteristics of the health care settings and participants who participated in the survey.

a center specialized in rehabilitation and palliative care and a center that does not specify the type of hospital.
DMSO: dimethylsulfoxide.
Out of 68 respondents, 82.4% (56/68) had a specific extravasation SOP or reference document. The content was developed interdisciplinary in 78.6% (44/56) of the cases. Specific data of SOP are shown in Table 2.
Characteristics of extravasation standard operational protocol.

In 26.5% (18/68) of the surveys, training courses or sessions were held for health personnel on the action in the event of antineoplastic extravasation in the center.
In 76.5% (52/68) of the centers, the pharmacist actively participated in the extravasation management and treatment process; 100% (52/52) provided information, 32.7% (17/52) participated in extravasation follow-up, and 9.6% (5/52) participated in the clinical assessment of the patient.
Sixty-eight centers responded to the survey, 66.2% (45/68) of them had a specific record to document extravasation, and 22.2% (10/45) indicated that it was not used. The registry allowed statistical analysis in 64.4% (29/45) of the cases.
Focusing on the risk capacity to cause tissue damage, 48.2% (27/56) of the centers indicated that antineoplastic agents are classified into three categories in their SOP, 44.6% (25/56) into four categories, 5.4% (3/56) into five categories, in one center the answer was not valid (1/56). Applying specific classification criteria based on concentration and/or volume for some extravasated drugs was reported by 73.2% (41/56) of pharmacists. Cisplatin (92.7%), carboplatin (41.5%), etoposide (34.1%), and 5-fluorouracil (48.8%) were the drugs most frequently classified with specific criteria. The tissue damage classification results of the selected 17 drugs are shown in Table 3.
Classification of antineoplastic drugs according to their tissue damage risk.
About the management of extravasations, it was mainly performed by the application of physical measures (cold/heat) and/or pharmacological measures (antidotes), 62/68 (91.2%). Besides, observation was done in 3/68 (4.4%) or saline washout or Gault technique in 1/68 (1.5%).
Only 35.7% (20/56) of the hospitals have implemented specific recommendations for extravasation management based on the time elapsed between its occurrence and its diagnosis; in 32.1% of the centers (18/56) specific measures were only applied in the first 24–48 h, in 3.6% (2/56) these measures were never applied beyond 48 h since extravasation, in 21.4% (12/56) they were always applied regardless of the time elapsed and in most centers, 42.9% (24/56), there were no specific recommendations.
Concerning the existence of specific criteria for the use of antidotes and/or nonpharmacological measures, 48.2% (27/56) of the centers had implemented specific criteria for some antineoplastic according to extravasated volume or extension of extravasation, 42.9% (24/56) according to the concentration of extravasated drug, 21.4% (12/56) according to the extravasated route (peripheral vs. central), 7.1% (4/56) only according to the extravasated drug and 5.4% (3/56) according to patient characteristics, finally in 35.7% (20/56) of the centers none of these criteria were applied to any drug.
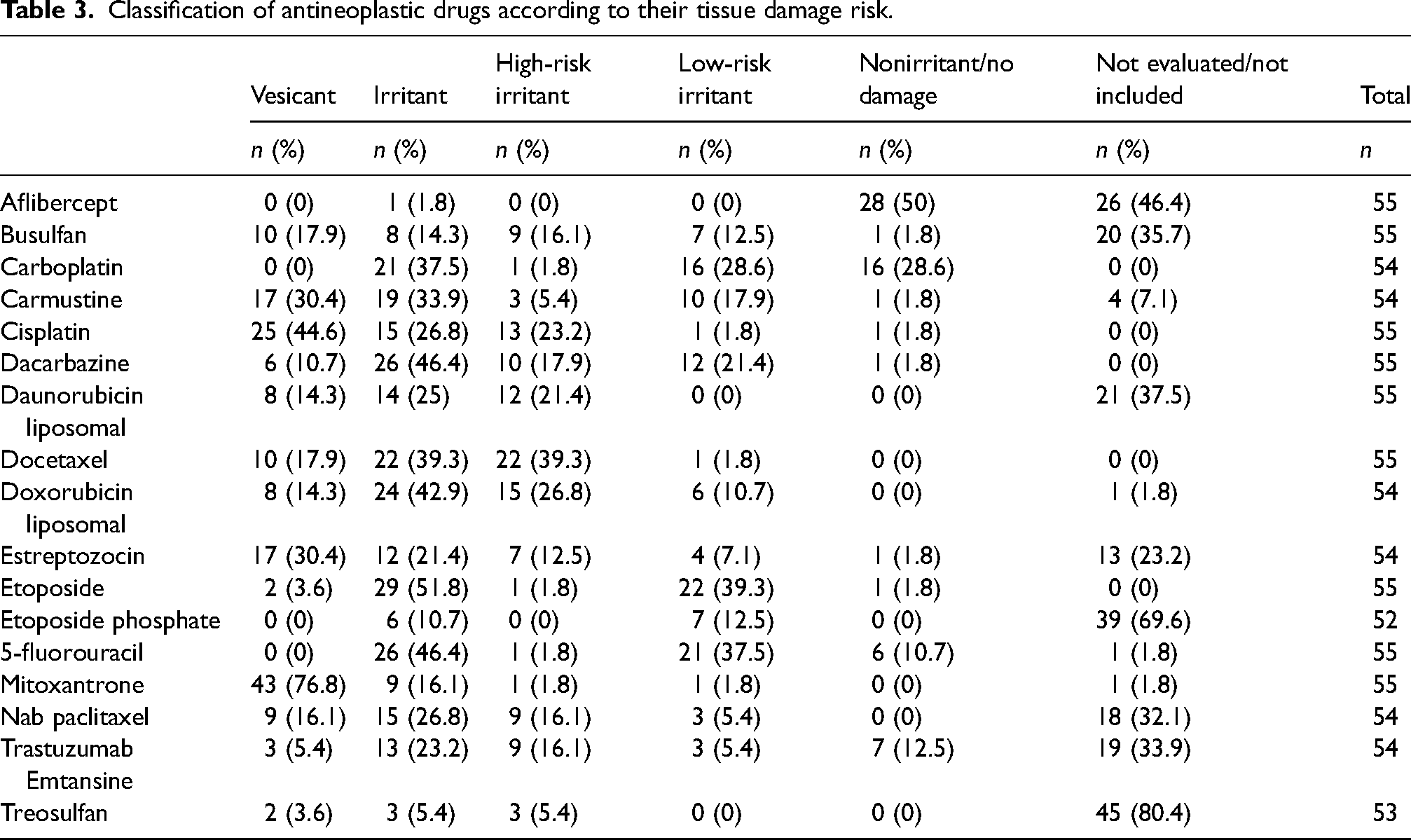
Figure 1 shows the antineoplastic drugs in which the use of pharmacological measures depended on the concentration of the extravasated drug.
Use of pharmacological measures recommendation according to the concentration of the extravasated drug.
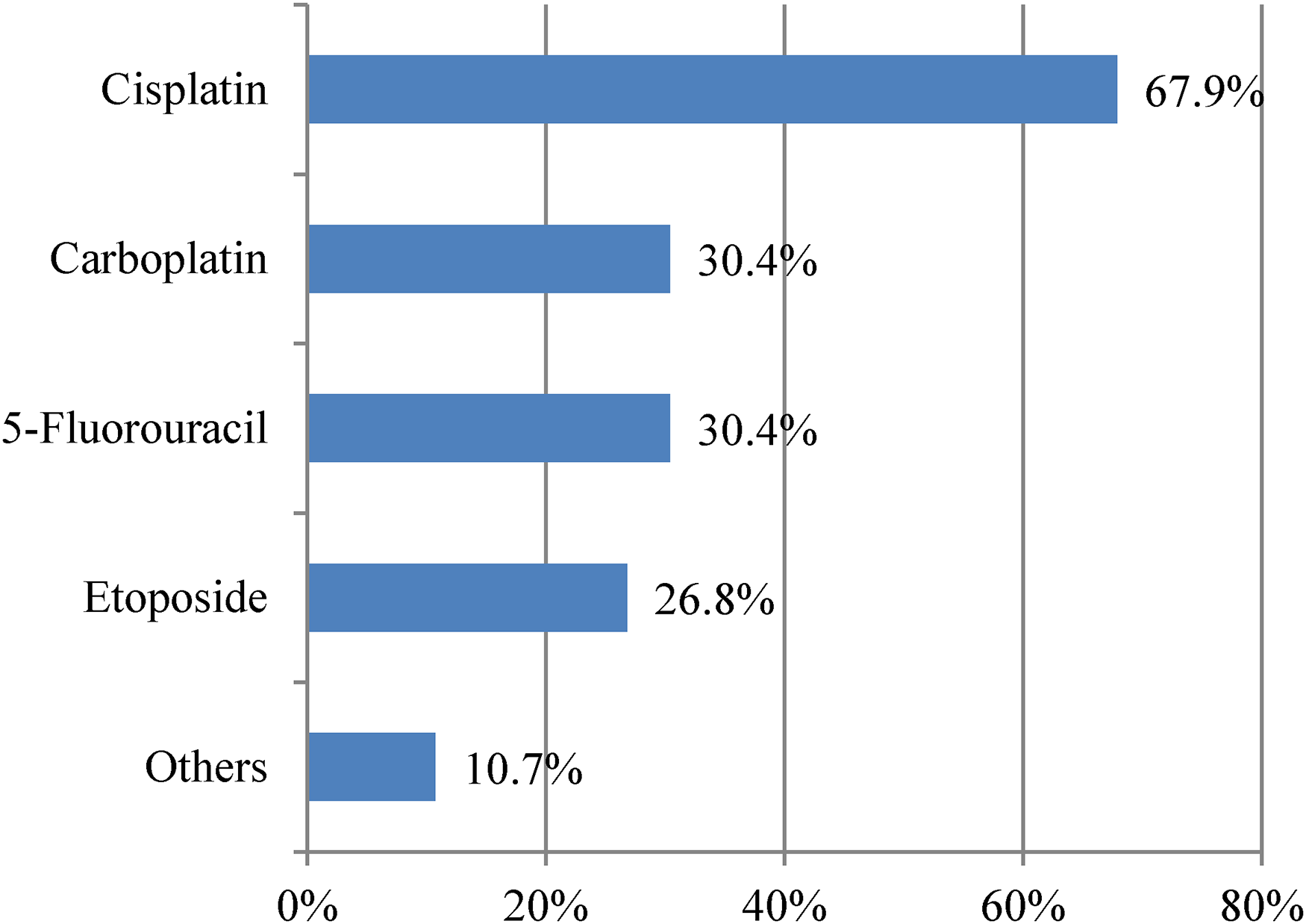
The antidotes mostly used in the centers that responded to the survey were hyaluronidase, sodium thiosulfate, dimethylsulfoxide 99% (DMSO), and dexrazoxane. One center reported the use of Burow water as a pharmacological measure. The frequency of use is shown in Figure 2.
Specific pharmacological measures use.
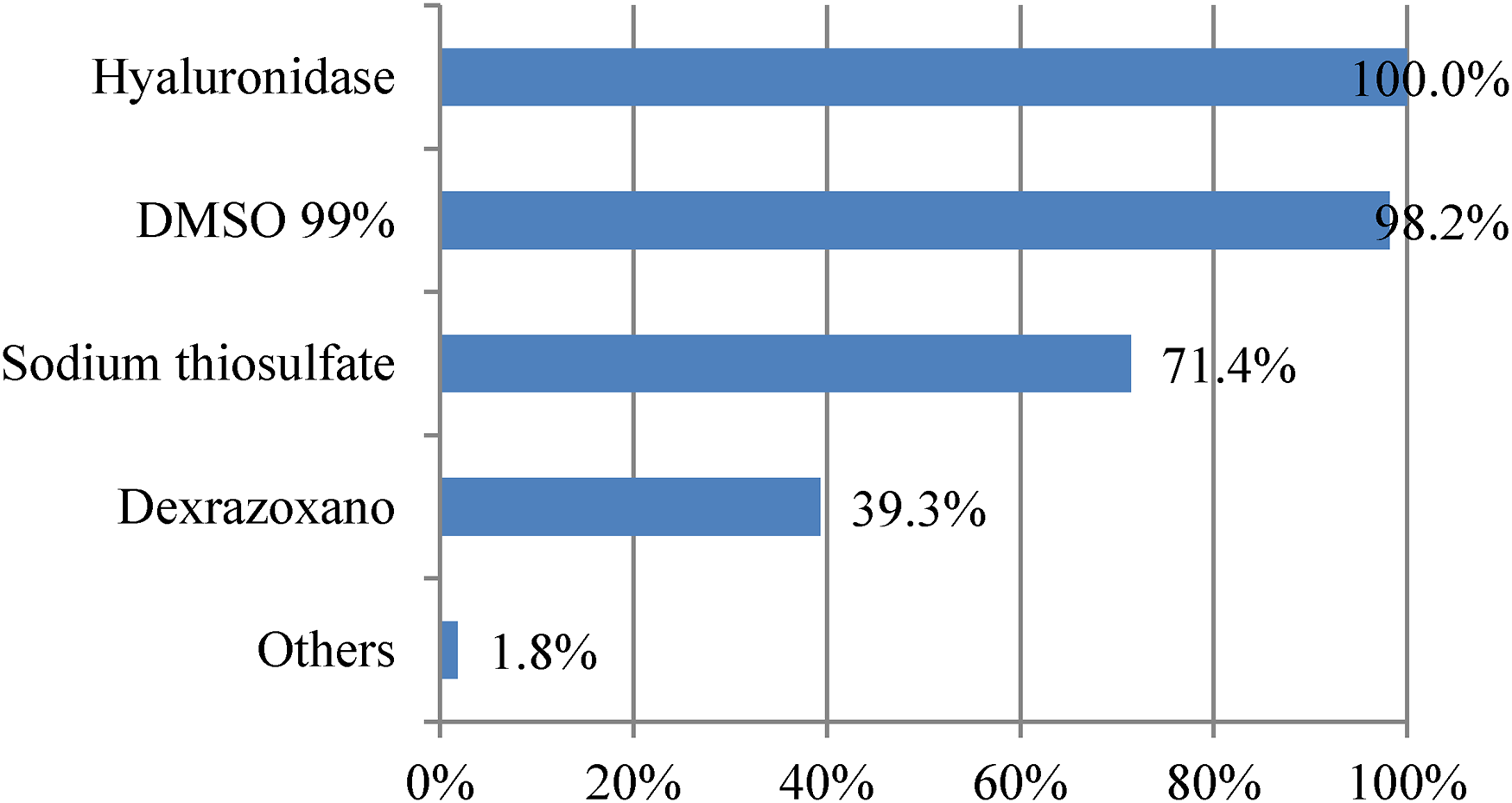
Regarding the use of hyaluronidase, the most frequently used presentations were vials of 100–150 IU (51.8%), as a compounding formula, and vials of 1500 IU (46.4%). One center (1.8%) did not specify this information. The concentration range of hyaluronidase administered was between 50 IU/ml and 1500 IU/ml (Figure 3). The dosage regimens for hyaluronidase administered subcutaneously are summarized in Table 4.
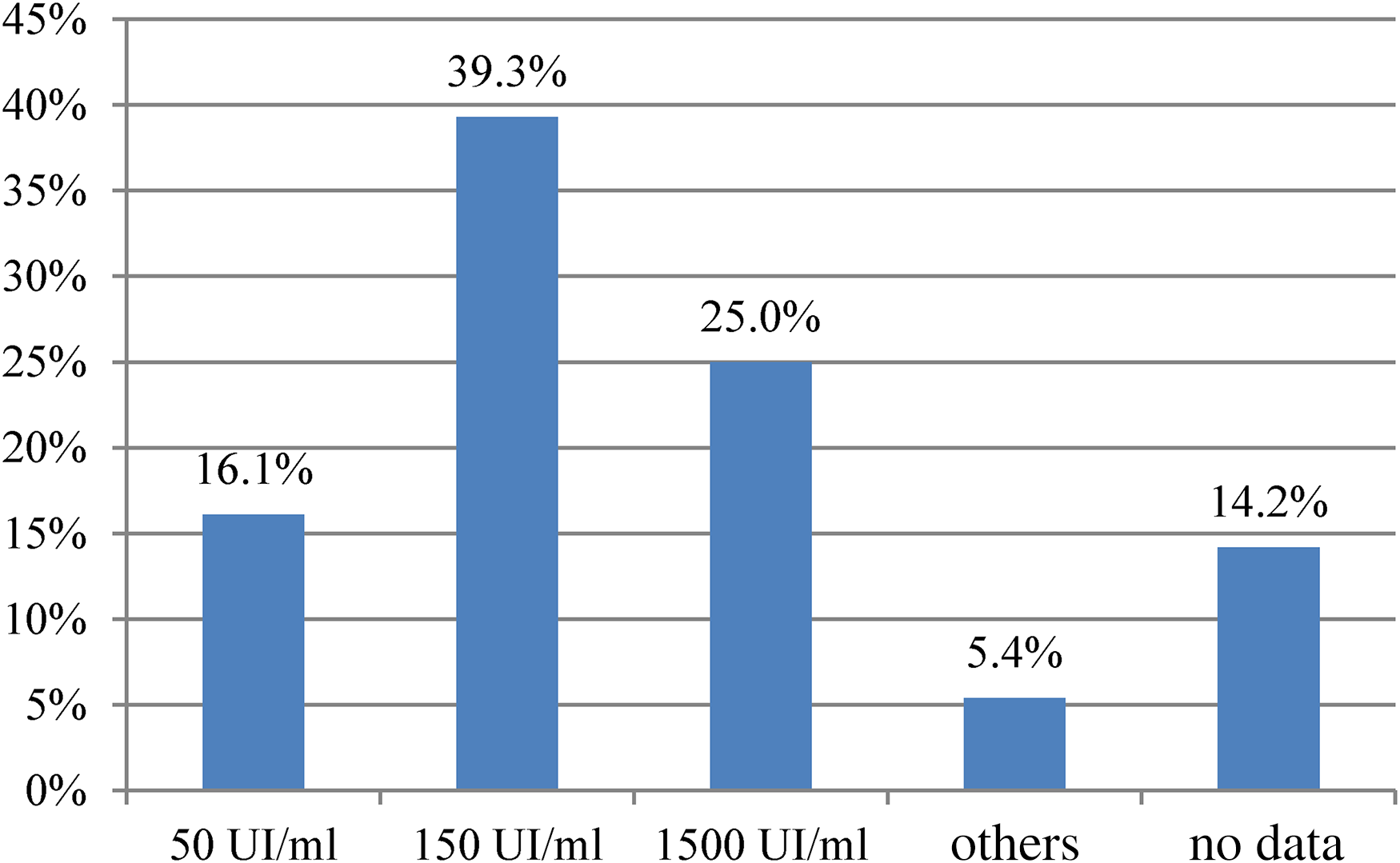
Hyaluronidase concentration (UI/ml) use in different hospitals.
Hyaluronidase dosage regimen recommended pattern of DMSO, specific nonpharmacological measures.

Abbreviations: N/A: Not applicable; DMSO: dimethylsulfoxide.
DMSO 99% was mainly recommended for mitomycin, mitoxantrone, and nonliposomal anthracycline extravasations (see Figure 4).
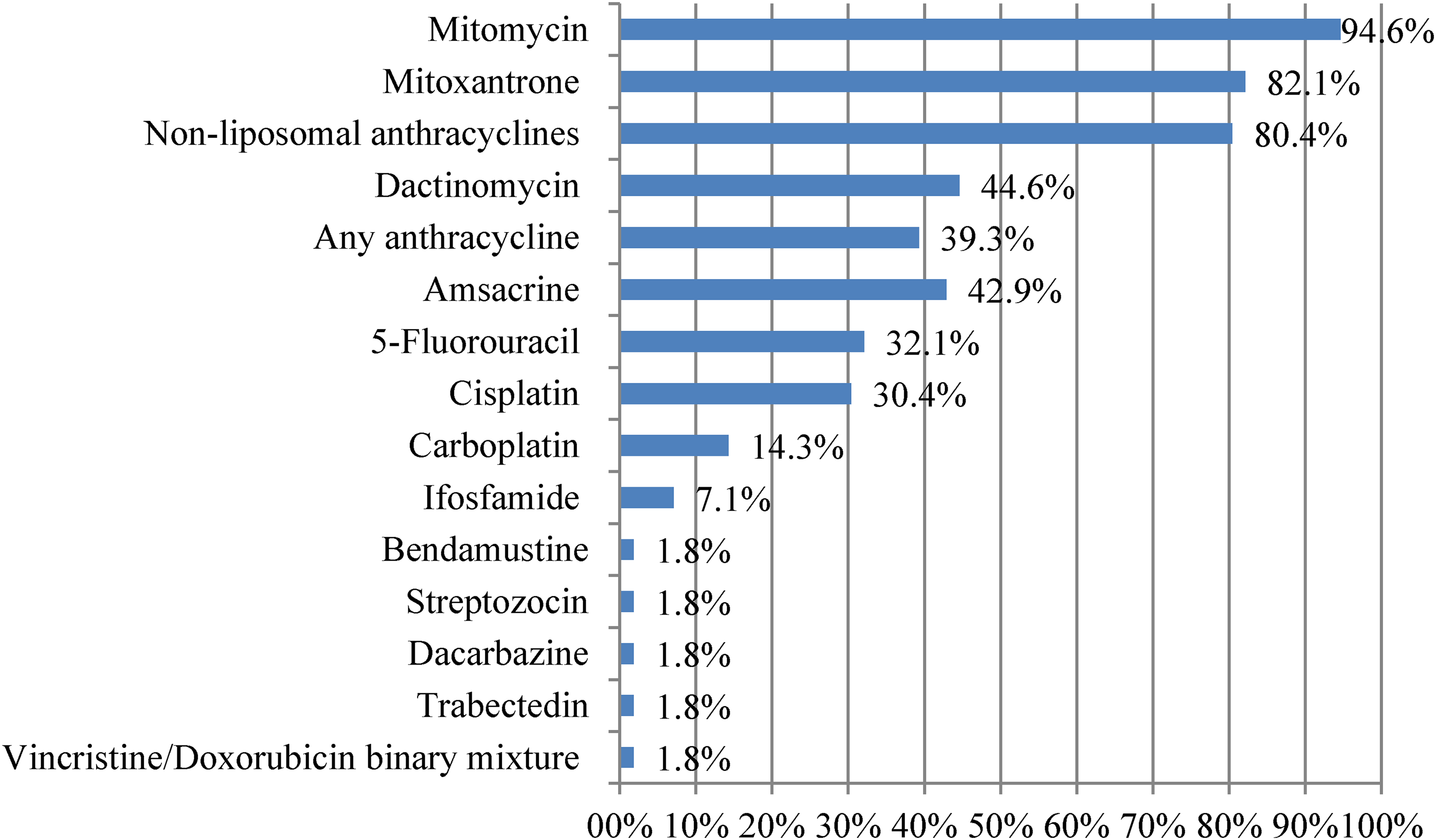
Extravasated drug and use of DMSO 99% recommended.
Different criteria were identified to establish the dosage of DMSO, 99%. By volume (1–2 ml), 39.3% (22/56), depending on the affected surface area (2 drops/4cm2 or 4 drops/10 cm2) 30.4% (17/56), by saturation (soaked gauze) 10.7% (6/56), and in 19.6% (11/56) were not specified. The frequency and duration of treatment are shown in Table 4.
Regarding systemic antidotes, 39.3% (22/56) of the centers reported the inclusion of dexrazoxane in their protocols. The criteria for the use of this drug were agreed upon by the Pharmacy and Therapeutic Committee (PTC), Departmental or Autonomous PTC in 31.8% (7/22) of the centers. Of these, 100% (7/7) recommended dexrazoxane for conventional anthracyclines and none for the treatment of liposomal anthracycline extravasation. Other criteria used were central line extravasation, 42.9% (3/7) and potentially serious extravasation, 57.1% (4/7).
About the use of topical corticosteroids in the treatment of extravasation, they are only used in case there is a medical prescription, 33.9% (19/56), also are recommended only in specific cases, 26.8% (15/56), they are recommended as a usual general measure, 21.4% (12/56), its use is not recommended, 16.1% (9/56) and it was not specified (1/56).
The application of cold as a nonpharmacological measure was recommended only for some vesicant and irritant drugs, 53.6% (30/56), for all vesicant and irritant drugs, 23.2% (13/56), for only vesicant drugs, 8.9% (5/56), only in specific situations, 7.1% (4/56) and for any extravasation, 5.4% (3/5). Concerning the recommendation of cold, the time of application, frequency, and duration were asked (Table 4).
Heat in the treatment of extravasation was applied for vinca alkaloids and oxaliplatin, 44.6% (25/56), only for vinca alkaloids, 23.2% (13/56), whenever hyaluronidase was used, 7.1% (4/56) and in other indications, 25% (14/56). Table 4 shows the time of application, frequency, and duration of heat.
It was recommended to avoid light exposure to the affected area in case of extravasation due to dacarbazine, 80.4% (45/56), 5-fluorouracil, 73.2% (41/56), mitomycin, 66% (37/56), cisplatin, 21.4% (12/56), and for other antineoplastics, 16.1% (9/56). Two of the 56 centers (3.6%) did not recommend this measure for any extravasated antineoplastic agent and it was not specified in 5/56 centers.
Table 5 summarizes the pharmacological and/or physical measures recommended for the treatment of extravasation of the 17 specific antineoplastic drugs selected.
Pharmacological and/or physical measures used in the extravasation of antineoplastic agents of interest.
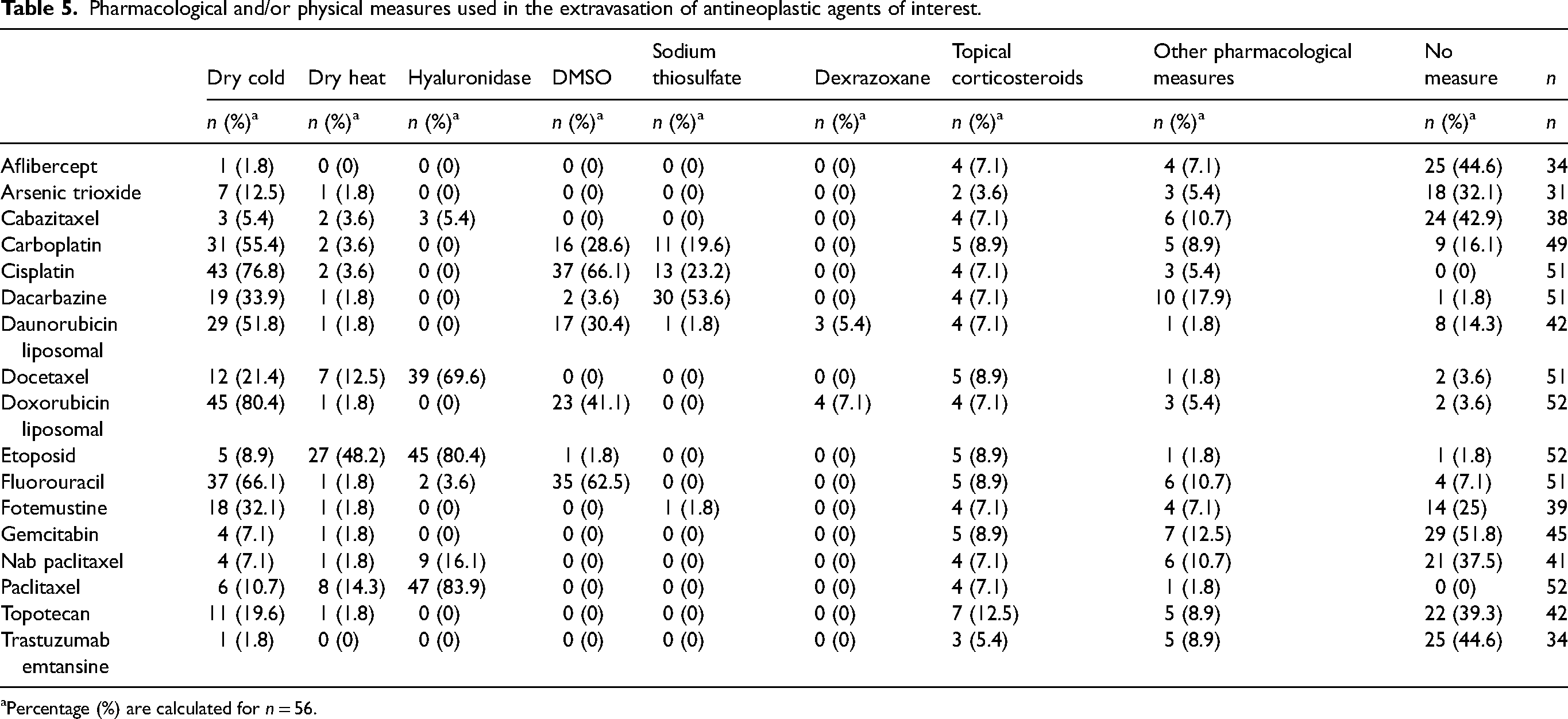
Percentage (%) are calculated for n = 56.
Discussion
This national study highlights that most health settings had protocols of antineoplastic extravasation management, as well as the high participation and commitment of pharmacists, both in their development and in the subsequent consensus process with other health professionals involved. These are important aspects because compliance with recommendations contained in clinical practice guidelines have been shown to be an effective method for optimizing patient care and improving health outcomes. 16 This fact becomes even more important with the high uncertainty due to the low quality of the available scientific evidence. Another highlight is the high number of centers that had a specific registry. Given the limited evidence available on the management of extravasation, these records constitute an important source of information, which, after being analyzed and published, would increase knowledge in this field.
Once the extravasation has occurred, the pharmacist maintains active participation in the process, mainly providing management information. These results are in accordance with the historical concerns of oncology pharmacists related to antineoplastic extravasation and its participation in multidisciplinary teams.17–19 However, some aspects to improve have been identified, such as periodicity of SOP updates (less than 50% in the last 2–5 years), implementation of professional training or agreement about clinical management and follow-up of extravasation.
In the present study, almost half of the settings classified tissue damage based on three categories (vesicants, irritants, and nonirritants). A slightly lower percentage of centers adopted a classification based on four categories, differentiating between high-risk irritants (HRI) and low-risk irritants (LRI), proposed by Conde et al. in 2012 and recently adopted by GEDEFO guidelines. 20 Up to 69.6% of the SOPs included specific tissue damage classification criteria, based on drug concentration or extravasated volume. In the 17 drugs with the greatest bibliographic variability in their classification, the results of the survey were similar to the results agreed by the GEDEFO group. 20 The potential for damage is mainly determined by the direct cellular toxicity of the antineoplastic agent in the affected tissues. However, different factors can contribute to its severity, such as drug concentration, extravasated volume, osmolarity, solution pH, and excipients.1,15,21 An irritating drug does not usually cause necrosis, but if extravasation of a large volume of a concentrated solution occurs, the risk increases and it can cause soft tissue ulceration. 22 In addition to these drug-related factors, those related to the patient or the procedure should be considered. In general, the classification discrepancies observed in this study are consistent with those published in the literature.
To reduce the severity of tissue damage, early extravasation diagnosis and management are crucial. There is controversy about the most appropriate management strategies due to the low quality of the published evidence. Numerous guidelines and expert consensus published by institutions and/or professional societies recommend the use of pharmacological and nonpharmacological measures as the main strategy to manage extravasations.8–12,15,23 Some authors and institutions opted for a surgical strategy based on saline lavage techniques.4,14,24,25 However, they require personnel specifically trained in these techniques and available, which could explain their lower implementation. The results of our national survey favor the use of pharmacological (91.2%) over surgical measures to prevent tissue damage.
Numerous pharmacological treatments have been described to manage extravasations. Some of them, such as antihistamines, sodium bicarbonate, ascorbic acid, heparin, lidocaine, or N-acetylcysteine, have been considered ineffective or with no proven effectivity. 1 Others, such as hyaluronidase, DMSO, sodium thiosulfate, or dexrazoxane, accumulate more evidence, based on nonrandomized studies and case series. Our survey shows the wide use of hyaluronidase (100%), DMSO (98%), and sodium thiosulfate (≈70%) in our country. Dexrazoxane is used in 39% of hospitals with specific criteria recommendations.
Dexrazoxane is the only drug approved for the treatment of anthracycline extravasations. However, there are concerns about its efficiency. The registered clinical trials lack a control arm and it has not been possible to establish the superiority of dexrazoxane over DMSO. 26 Therefore, its inclusion into treatment protocols is not generalized, as shown by the different positions adopted by scientific societies and health organizations. 27 Regarding the pattern of use in Spanish centers, the design of the survey prevents us from knowing this information, although in the small group of centers that agreed on the criteria for use, it was only used in the treatment of conventional anthracyclines as proposed in GEDEFO consensus.
Hyaluronidase can be obtained as a compounding product and as a foreign medication through the Spanish Agency for Medicines and Health Products. The recommended dosage regimen varies according to the sources consulted, and this may be a consequence of the different formulations available and the lack of comparative efficacy and safety studies between them, so the optimal regimen remains to be established. Depending on concentration, the maximum dose ranges from 1500 IU (puncture of 0.1–0.2 ml for the concentration of 1500 IU/ml) up to 75 IU (puncture of 0.2–1 ml for concentrations of 15 IU/ml).9,14,23,28,29It is not possible to establish a universal recommendation and usually, intermediate doses are used (150–450 IU). The survey reflects the wide variability observed in the published literature and shows that the usual practice in our country is based on the use of medium doses, endorsed by the ESMO-EONS clinical practice guidelines and by some national and European organizations such as GEDEFO and ASORS.10,11,15
Evidence for the use of DMSO in the treatment of extravasations comes from two prospective studies.30,31 The guideline proposed has been maintained in the current recommendations of the different institutions and professional societies consulted. Likewise, the results presented here are in accordance with these recommendations. About the use of DMSO, our survey shows that in Spain it is widely implemented to treat conventional anthracyclines (80.4%) and mitomycin (94.6%) extravasations, and there is a high international consensus supporting its use.8–11,13,15 About liposomal anthracyclines, it has been postulated that the liposomal complex remains stable after extravasation and DMSO could induce the release of anthracycline from the liposomes resulting in further damage. 1 For this reason, most authors and guidelines recommend against DMSO,9,15 but others recommend its use due to the potential severity associated with anthracyclines.14,32 For other anticancer drugs, the use of DMSO is more controversial. The GEDEFO group considers its use appropriate after extravasation of cisplatin, 5-fluorouracil, and carboplatin only in cases with specific criteria associated with a higher risk of damage, such as concentration or extravasated volume, that justifies the implementation of pharmacological measures. 33
Sodium thiosulfate is a widely incorporated antidote in Spanish centers, mainly for dacarbazine, cisplatin, and carboplatin. Historically, it has been used as an antidote after mechlorethamine extravasation and has also been suggested for dacarbazine or concentrated solutions of cisplatin,3,34 although paradoxically no published cases have been found. Due to the lack of evidence, the most recent guidelines do not include it or only recommend it for mechlorethamine, which is currently not available in Spain, so the GEDEFO group decided not to recommend its use.
Nonpharmacological measures can complement treatment, presenting the advantage of being noninvasive, easy to use, and with little toxicity. There is consensus in the literature about the recommendation of using topical cold in extravasations caused by some vesicant agents (like anthracyclines, mitomycin, and dactinomycin) and topical heat in those caused by vinca alkaloids. Most respondents used cold only for selected vesicant and irritant drugs (53.6%), while a smaller number of centers expanded their use to many vesicant or irritant agents (23.2%). The GEDEFO group recommends a cold regimen for vesicant and HRI, in the case of LRI it is recommended to apply cold only initially to relieve damage. Although there is no solid evidence of its use in many of the drugs, the panel decided to recommend it unless contraindicated. Regarding local heat, the survey shows a majority use for vinca alkaloids and oxaliplatin extravasations (44.6%). The benefit of physical measures after oxaliplatin extravasation remains uncertain. The application of cold has been documented, together with different pharmacological measures, with contradictory outcomes.35–38 The administration of cold could precipitate or worsen the peripheral neuropathy associated with oxaliplatin, so its use is not recommended. Some authors recommend the use of heat based on the theoretical increase in drug elimination by local vasodilation and relieving pain at the injection site.23,28,32,38
The survey reveals the lack of consensus about measures when extravasation is identified late. The general recommendation is to apply these measures as soon as possible after the identification of the extravasation; however, no time limit has been established after which its use would not be indicated and, in general, the published guidelines do not include specific recommendations. The GEDEFO group recommends an individual assessment when 24–48 h have elapsed between the extravasation and the detection. 33
Skin damage caused by extravasation could be aggravated by sun exposure, especially in the case of photosensitizing drugs such as dacarbazine, 5-fluorouracil, cisplatin, and mitomycin. Photoprotection is widely implemented in Spanish centers as shown by the results of this survey (>95%), in line with the GEDEFO recommendations and most guidelines and authors consulted.8,10,13,33
During the previous work of the GEDEFO extravasation group, wide variability was detected in the recommendations of specific measures for some of the drugs evaluated, so these were expressly included in the survey. 33 In the case of LRI extravasation, the trend observed reveals the absence of the use of any type of specific measures except for carboplatin, etoposide, and 5-fluorouracil. Although some authors and guidelines do not recommend the use of any antidote for these three drugs, the GEDEFO group recommends the use of DMSO in special situations, with the aim of preventing or slowing the progression of tissue damage in a high-risk situation. 33 In general, irritant drugs have a low harmful potential, so conservative strategies may be enough. For HRI drugs, the trend is reversed, with a higher proportion of centers opting for the use of specific measures, highlighting cisplatin, dacarbazine, liposomal doxorubicin, liposomal daunorubicin, and docetaxel. This group of drugs can cause local irritation, pain, and a burning sensation. However, cases of vesicant damage have been described justifying the adoption of more aggressive measures.39–41 The GEDEFO group does not recommend the use of specific pharmacological measures for HRI drugs, except for cisplatin and docetaxel, but recommends the use of physical measures, generally cold for 48–72 h or heat for oxaliplatin for two days. 33
The use of topical corticosteroids is a supported measure used in 70% of Spanish centers. Different studies have reported the intralesional use of corticosteroids with contradictory results and even at high doses or with multiple administrations a deleterious effect has been observed.5,23 They also can worsen skin damage caused by etoposide and vinca alkaloids (contraindicated).23,42 However, the topical use of corticosteroids can help reduce local inflammation and reduce erythema. For this reason, the GEDEFO group considered that the use of a low-potency topical corticosteroid could be considered a general measure.
In conclusion, the implementation of prevention measures for chemotherapy extravasations is essential, as well as having an institutional protocol that allows a rapid and homogeneous response to prevent serious damage associated with the event. The results of this study highlight the involvement of Spanish oncology pharmacists in extravasation management, the application of physical and/or pharmacological measures as the method of choice for their management, as well as the existing discrepancies in the classification and management recommendations, particularly for drugs classified as an irritant. The consensus document recently published by GEDEFO aims to be a guide for action in the treatment and prevention of extravasation for the whole interdisciplinary team and reduce the variability of clinical practice in our environment. Further research on the adoption of these recommendations is warranted.
Footnotes
Acknowledgments
The authors thank Spanish Oncology Pharmacy group members who care for oncological patients for participating in the GEDEFO-SEFH survey. Their contributions will increase the knowledge about the management of antineoplastic extravasation.
Author contributions
Inmaculada Jiménez-Pulido has contributed to all aspects of the preparation of the article: conception and design of the survey, bibliographic search, data analysis, manuscript writing, and approval of the final version. María Asunción Albert-Marí has contributed to the conception and design of the survey, bibliographic search, manuscript writing, and approval of the final version. David Conde-Estévez has contributed to the conception of the survey, critical review of the manuscript, approval of the final version and translation of the draft of the article into English. Begoña San José-Ruiz has contributed to the conception of the survey, critical review of the manuscript, and approval of the final version. María Ángles Gil-Lemus has contributed to the conception of the survey, critical review of the manuscript and approval of the final version. Ana Cristina Cercós-LLetí has contributed to the conception of the survey, critical review of the manuscript and approval of the final version. María Jesús Esteban-Mensua has contributed to the conception of the survey, critical review of the manuscript, and approval of the final version. María Sacramento Díaz-Carrasco has contributed to the conception and design of the survey, bibliographic search, manuscript writing and approval of the final version.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
