Abstract
Introduction
Prescribing immune checkpoint inhibitors (ICIs) to cancer patients with an autoimmune disease (AID) is presumed safe when cautious adverse event management is applied. However, guidelines on immunosuppressant (IS) adaptations are limited and real-world evidence is scarce.
Methods
Current practice of IS adaptations is described in a case series of AID patients treated with ICIs in a tertiary university hospital in Belgium (1/1/2016–31/12/2021). Patient, drug and disease-related data were documented using retrospective chart review. A systematic search of the PubMed database was performed to identify similar cases (1/1/2010–30/11/2022).
Results
Sixteen patients were described in the case series (62% with active AID). Systemic IS were changed before ICI initiation in 5/9 patients. Four patients continued therapy, of which one achieved partial remission. Patients who had IS (partially) stopped before ICI start (n = 4) had AID flares in two cases; immune-related adverse events in three cases. In the systematic review, 37 cases were identified in 9 articles. Corticosteroids (n = 12) and non-selective IS (n = 27) were continued in, respectively, 66% and 68% of patients. Methotrexate was frequently discontinued (13/21). Biologicals, excluding tocilizumab and vedolizumab, were withheld during ICI treatment. Out of all patients with flares (n = 15), 47% had stopped IS therapy before ICI start and 53% had continued their AID drugs.
Conclusions
A detailed overview of IS management in patients with AID receiving ICI therapy is presented. Expanding the knowledge base germane to IS management with ICI therapy in the diverse population is essential to evaluate their mutual impact, thus advancing responsible patient care.
Introduction
Immune checkpoint inhibitors (ICIs) have gained an important role in cancer treatment due to their success in patients’ clinical outcomes. Tumor cells can induce signaling pathways involving immune checkpoints such as cytotoxic T-lymphocyte antigen-4 (CTLA-4) and programmed cell-death-1 (PD-1), in order to circumvent a natural T-cell immune response, and allow cancerous tissue to grow and spread uncontrollably. ICIs are monoclonal antibodies that block the immune checkpoints, reversing the inhibitory signal and facilitating T-lymphocyte proliferation and activation.1,2 In a subset of patients, this unrestrained effect on the immune system comes with dysimmune toxicities, called “immune-related adverse events” (irAEs). 3
An extensive immune system activation can be harmful for patients with autoimmune diseases (AIDs), which has led to their exclusion from the ICI registration trials, 4 and thus a knowledge gap exists concerning ICI efficacy and safety in this patient group. Fortunately, experience in daily practice is increasing and the use of ICIs in AID patients is presumed to be safe, provided cautious adverse event management is applied. 5 Conversely, there is still conflicting evidence on the occurrence of AID flares,6–9 that is, a reactivation or worsening of AID symptoms. Furthermore, patients with active AIDs often require immunosuppressants (IS) at the time of ICI initiation (20–43%).7,9–11 Recent data in non-small cell lung cancer patients starting PD-(Ligand, L)-1 inhibitor therapy with a baseline use of corticosteroids (≥10 mg per day), showed a lower response rate and decreased overall survival (OS), 12 urging a critical evaluation of baseline IS use prior to ICI therapy.
Practice guidelines on immunosuppressive therapy adaptations prior to ICI treatment in order to ensure ICI efficacy are limited and mainly based on management strategies in AIDs. 13 To objectify this need for consensus on the management of immunosuppressive drugs in cancer patients treated with ICI therapy, we describe a single-center case series combined with a systematic literature review of similar cases and discuss their IS management in order to report the current knowledge base.
Methods
Retrospective case series
The medical records of cancer patients treated with an ICI between May 1, 2016 and December 31, 2021 at the Universitair Ziekenhuis Brussel, a 721-bed tertiary hospital in Belgium, were retrospectively screened to identify patients with a history of AID. The patient records were fully accessible in the electronic hospital information system.
The following inclusion criteria were applied: diagnosis of a histologically confirmed malignant solid tumor, a history of AID treated with immunosuppressive therapy (limited to rheumatoid arthritis, ankylosing spondylitis, psoriasis, ulcerative colitis, or Crohn's disease), age 18 years or older at the time of ICI initiation. Patients diagnosed with a malignant hematological disease, chronic viral infection, pediatric patients, or patients receiving an investigational drug were excluded.
Literature review
PubMed was searched for relevant articles, published in English, on autoimmune flare-ups or irAEs in cancer patients with pre-existing autoimmune pathologies and ICI therapy. Different from the case series, we only included patients with active AID, treated with systemic IS. This systematic review included studies published from January 1, 2010 until November 30, 2022.
The search strategy is included in the Supplementary File. After duplicate removal, titles and abstracts were screened on relevance by two authors, followed by a full-text evaluation. Cited manuscripts were included if deemed relevant for the review. Identical inclusion criteria were applied as for the case series. In the case of combination ICI therapy, the article was excluded if the sequence of administration was not clear. A third reviewer made the final decision in case of disagreement.
Data collection
Demographics, disease-related data were collected by chart review of the electronic patient record for the case series, or as described in the article. IS management was documented (start/stop). In the case series, the occurrence, type, and management of AID flares and irAEs were collected starting from the ICI initiation date (baseline) until one year (or until no more data were available) after the final ICI administration or disease progression resulting in treatment change, whichever came first
Identified events were defined as AID flares when symptoms were consistent with the AID while irAEs were events with symptoms other than those of the AID. When ICI therapy was discontinued because of an adverse event, this was documented. Information concerning the patient's best overall response was obtained as known at the time of this study or as published in the included article.
Ethical considerations
This retrospective study was approved by the Committee of Medical Ethics of the Universitair Ziekenhuis Brussel - BUN: 1432021000707. All living patients reported in the case series provided informed consent. Informed consent was waived for patients who were deceased at the time of the study.
Data analysis
All extracted data were entered into a REDCap® database (Vanderbilt University, Nashville, TN, USA) and analyzed using descriptive statistics in Microsoft© Excel (Microsoft, Redmond, WA, USA). Percentages and frequencies were used for the presentation of nominal data and continuous variables were shown as median and interquartile range (IQR).
Results
Case series
Of 578 patients eligible for inclusion in the case series, 16 (2.8%) were retained. The median age was 66 years, and 75% (n = 12) were male. The main cancer diagnoses were melanoma (n = 6) and non-small-cell lung cancer (n = 4). Twelve patients were treated with anti-PD-(L)1, two patients received dual ICI therapy (ipilimumab + nivolumab), and another two patients were treated with an anti-PD-1 antibody and concomitant chemotherapy. Patient characteristics are summarized in Table 1. The individual case data are provided in Table 2. Progressive disease was noted in 8 patients (in 4 who received immunosuppressive treatment at baseline). Of the patients who continued IS during cancer treatment, the cancer progressed in 2 patients; 1 patient achieved partial remission and 1 patient died shortly after an ICI infusion, however, the cause of death was not related to the ICI therapy. Patients, in whom IS therapy was interrupted before ICI start (n = 4), showed either cancer disease progression (n = 2) or stable disease (n = 1). The fourth patient decided to be treated in another hospital and thus, outcome data were not available. The most frequent AID type in the case series were rheumatological AIDs (n = 8, 50%).
Characteristics of patients with active autoimmune disease before initiation of immune checkpoint inhibition.
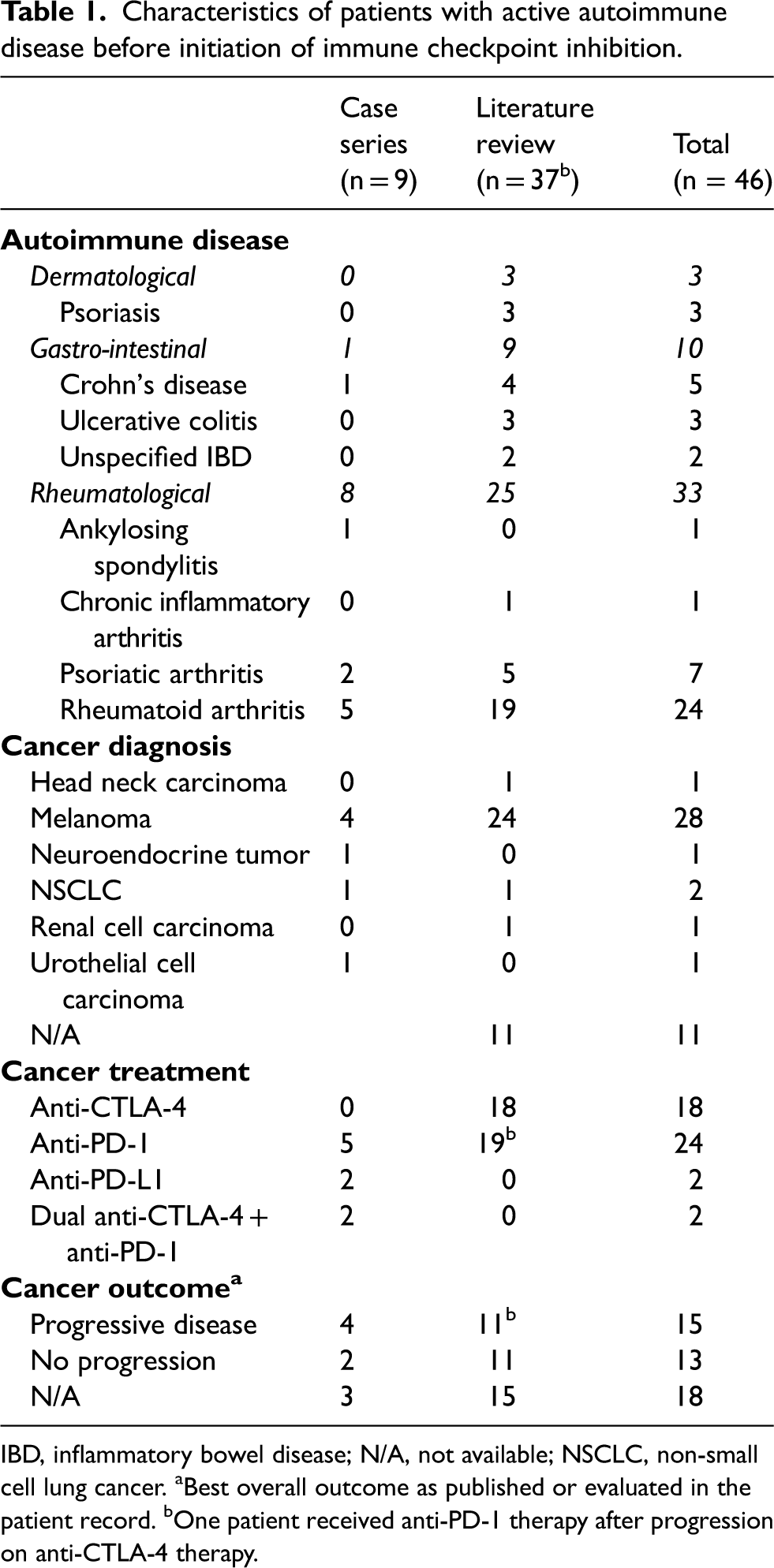
IBD, inflammatory bowel disease; N/A, not available; NSCLC, non-small cell lung cancer. aBest overall outcome as published or evaluated in the patient record. bOne patient received anti-PD-1 therapy after progression on anti-CTLA-4 therapy.
Baseline and adverse event management in cancer patients with autoimmune disease included in the case series.
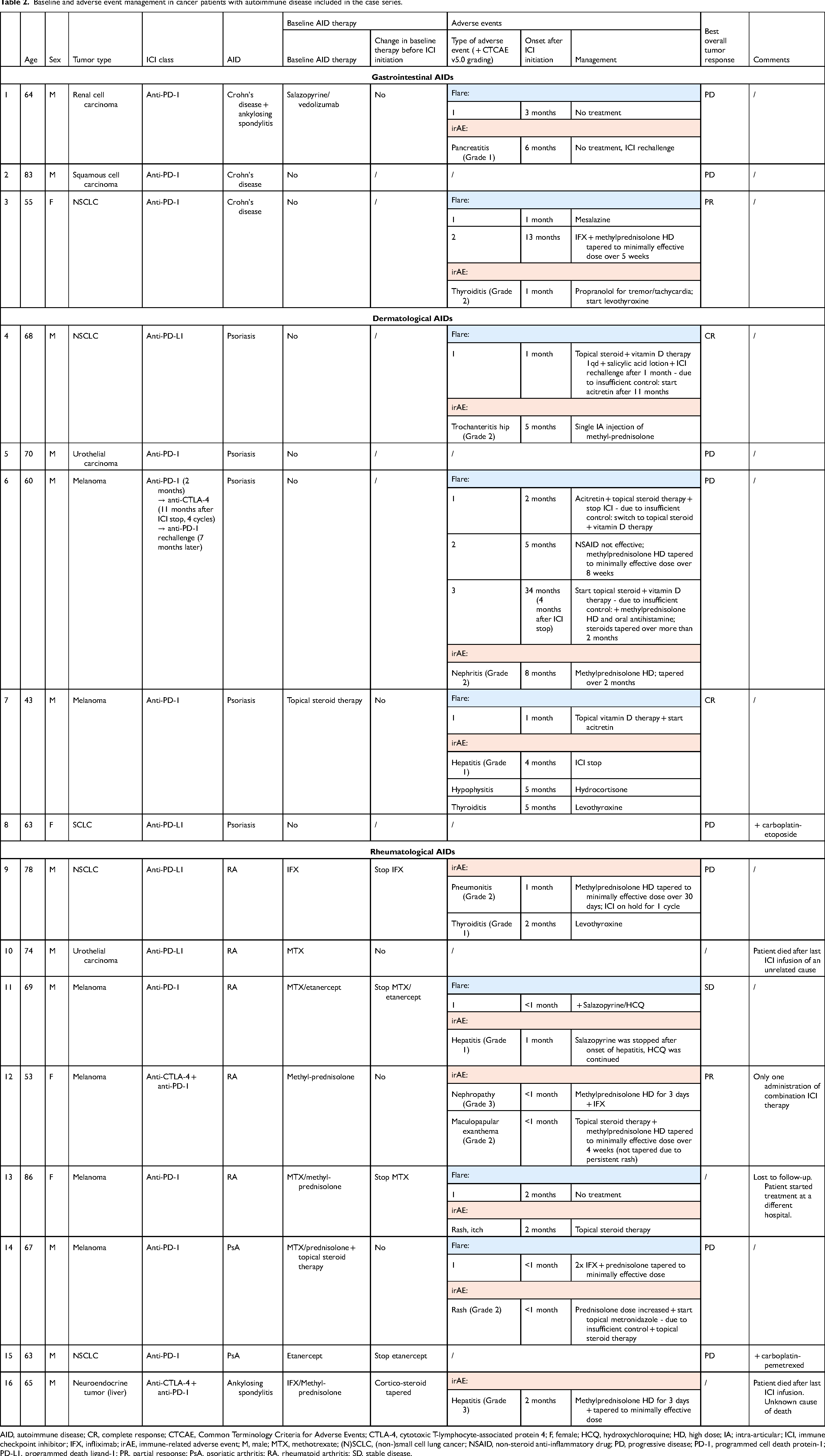
AID, autoimmune disease; CR, complete response; CTCAE, Common Terminology Criteria for Adverse Events; CTLA-4, cytotoxic T-lymphocyte-associated protein 4; F, female; HCQ, hydroxychloroquine; HD, high dose; IA; intra-articular; ICI, immune checkpoint inhibitor; IFX, infliximab; irAE, immune-related adverse event; M, male; MTX, methotrexate; (N)SCLC, (non-)small cell lung cancer; NSAID, non-steroid anti-inflammatory drug; PD, progressive disease; PD-1, programmed cell death protein-1; PD-L1, programmed death ligand-1; PR, partial response; PsA, psoriatic arthritis; RA, rheumatoid arthritis; SD, stable disease.
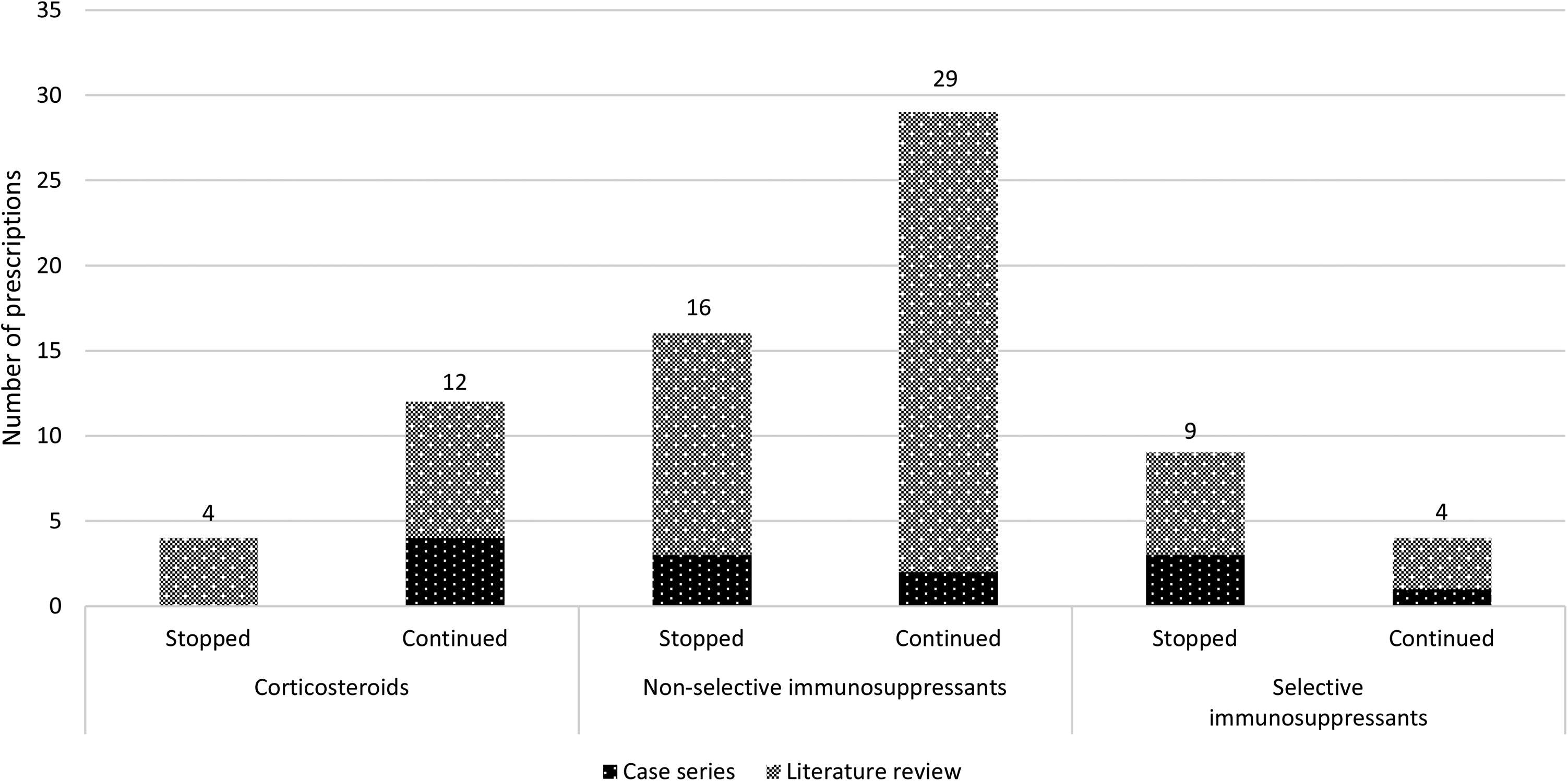
Ten patients (62%) had an active AID, of which 9 were treated with systemic IS. In 56% of these cases (n = 5), therapy was changed before start of ICI therapy (IS stop in 4 patients, corticosteroid taper to a minimally effective dose in 1 patient). Five patients continued their maintenance treatment (4 systemic IS, 1 topical IS) during ICI therapy. Total IS therapy changes are shown in Figure 1, details for the case series are presented in Table 3.
Immunosuppressive drug treatment changes before initiation of immune checkpoint inhibitor, combined data from case series and literature review.
Immunosuppressive drug treatment changes for actively treated patients with autoimmune disease (AID), before initiation of immune checkpoint inhibition.
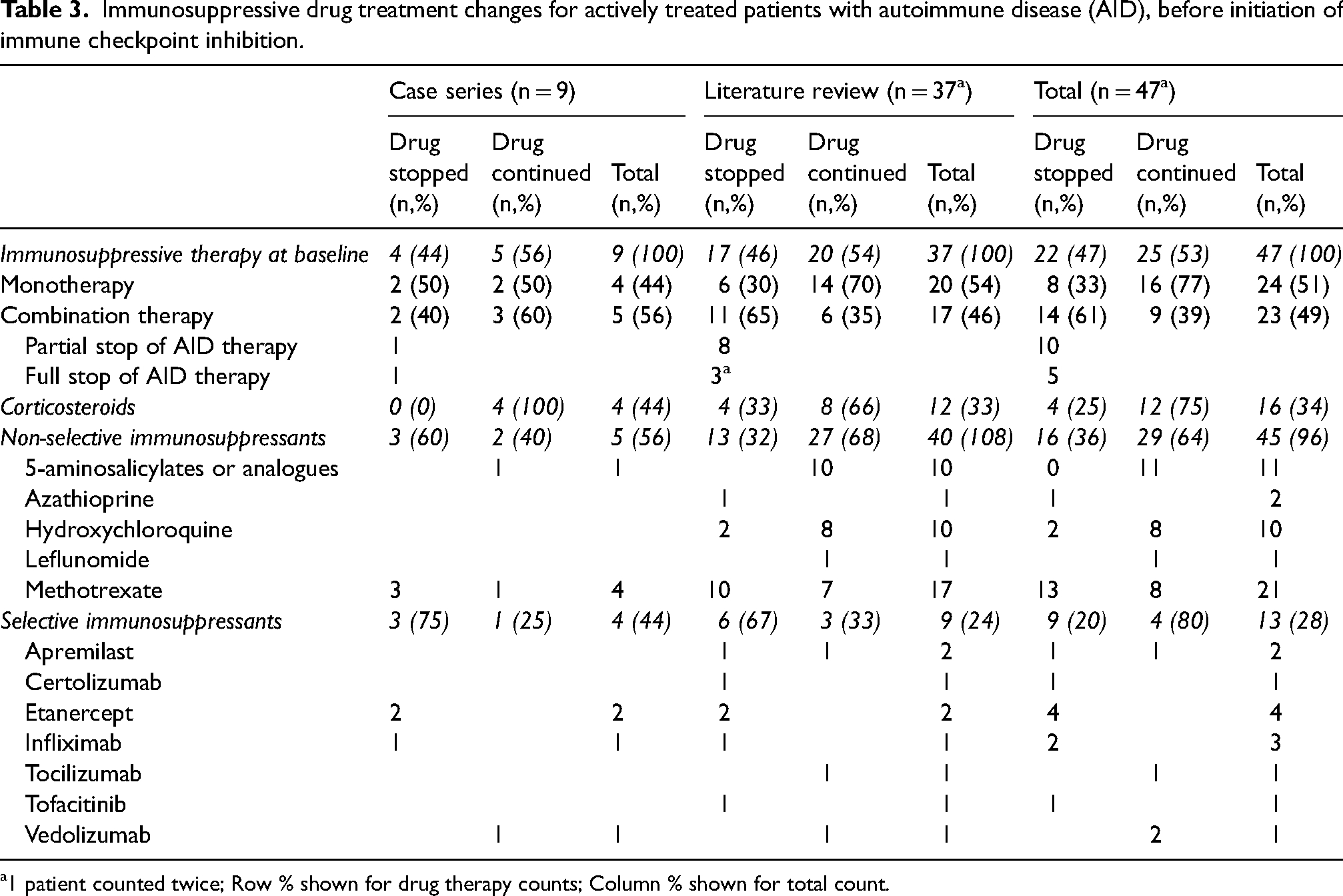
1 patient counted twice; Row % shown for drug therapy counts; Column % shown for total count.
AID flares or irAEs occurred in 11 patients (flare = 8; irAE = 11). The median number of flare episodes was 0.5 (IQR 0–1) and irAEs 1 (IQR 0–1). Time until first flare and irAE onset were respectively, a median of 1 (IQR 0.9–2) and 2 months (IQR 1–4.5). Every patient who had an AID flare, also experienced one or more irAEs. Five patients did not have any documented adverse event, of which 2 continued active IS treatment during ICI therapy. Patients who had their IS treatment (partially) stopped (n = 4), had flares in 2 cases and irAEs in 3 cases. The number of patients with AID flare and/or irAE occurrence is presented in Table S2, as a function of IS baseline treatment changes.
Most adverse events identified in this case series were managed successfully with systemic corticosteroids. Psoriasis flares were mostly controlled using topical ointments. ICI therapy was rarely withheld (n = 2).
Literature review
The search strategy resulted in the identification of 1035 references. The first two authors disagreed on three articles, leading to intervention of the third author. As a result, 9 articles were included reporting on 36 patients.11,14–22 Figure 2 shows a flow chart of the inclusion procedure.
Inclusion process of literature review.
The primary tumor type was melanoma (24/36, 67%). Of the 36 patients, 19 were treated with anti-PD-1 (53%) and 18 with anti-CTLA-4 antibodies (50%). One patient was initially treated with anti-CTLA-4 therapy, but was progressive and started on anti-PD-1 therapy. 19 The two periods of ICI administration were evaluated as two separate patient cases resulting in a total of 37 cases. The full list of the included patients can be found in the Supplemental Digital Content (Table S1). Unfortunately, 14 cases had missing data on the cancer outcome following ICI therapy. For the 23 other cases, 11 (48%) did not present progressive cancer disease, as known at the time of publication. Of patients who had continued IS therapy (n = 19), 9 (47%) had achieved a significant response to the cancer treatment.
At baseline, most patients were treated with IS, either in monotherapy (n = 20) or combination therapy (n = 17). Combined IS therapy was partially discontinued in eight cases, or completely stopped (n = 3). In Figure 2, all IS treatment changes are presented (more details in Table 3). Patients on corticosteroids (n = 12) and non-selective IS (n = 27) for their AID, continued intake in, respectively, 66% and 68%. Methotrexate (MTX) was discontinued more frequently than that is was continued (8/21). All biologicals, except tocilizumab and vedolizumab, were withheld during ICI treatment.
In the selected cases from the literature, 15 (42%) patients experienced one or more AID flares, and 15 (42%) patients encountered irAEs. Two patients had episodes of both types of adverse events. Out of all patients with flares, 7 (47%) had stopped IS therapy before ICI start and 8 (53%) had continued their AID drugs. Those who had stopped had a higher number of irAEs (n = 10, 67%), compared to those who persisted (n = 5, 33%). Common Terminology Criteria for Adverse Events (CTCAE) grading of irAEs was frequently missing and therefore not reported. Elaboration on IS treatment changes and AID flare/irAE occurrence can be found in Table S2 (Supplemental Digital Content).
Discussion
In this retrospective study, immunosuppressive management strategies were objectified in patients with AIDs treated with ICIs by means of a single-center case series and a supplementary literature review. In light of these results, pharmacological perspectives on both ICI efficacy and safety, and AID drug treatment were formulated.
Decision for ICI therapy in patients with AID
Not all AID patients have an active disease at the time of ICI initiation.7,9–11 In this study, only 48% of patients with active AID had changes in their IS treatment before ICI initiation. To date, little is known about the effect of concurrent intake of IS on ICI efficacy. For example, it appears that chronic corticosteroid use decreases OS in cancer patients,23,24 however, these results may be driven by a poor-prognosis subgroup requiring corticosteroids for palliative, cancer-related indications such as fatigue, cachexia or edema around brain metastases. 25 Cai et al. have reported no difference in progression-free survival (PFS) (n = 4293) and OS (n = 8053) between patients with AID, and patients without AID in their meta-analysis. Additionally, cancer or ICI type did not influence PFS or OS in the subgroup analyses. 26 Nevertheless, the influence of specific IS therapy on PFS and OS is yet to be determined. In the retrospective cohort study by Tison et al., AID patients without IS use showed a trend of better outcomes. 27 In the presented cases, results on cancer outcomes were often missing, which complicated further analysis. Nonetheless, as 52% of patients continued their immunosuppressive therapy after ICI initiation, these data are very relevant. The same counts for AID flare and irAE drug management. IS intensity during ICI therapy may be of interest to guarantee improved OS. Van Not et al. showed that melanoma patients requiring steroids and second-line IS for irAE treatment may be at higher risk for cancer progression or death, compared to patients treated with steroids alone when combination ICI therapy is administered. 28
Although ICI adverse events appear to be mild and manageable, close observation of patients with AIDs and ICI cancer treatment is required for early recognition and adverse event treatment.5,9 There is conflicting evidence to answer the question whether baseline systemic IS therapy is protective for AID flares and irAEs. There might be a trend that patients without treatment have less irAEs, 6 however, differences in AID type and activity status may result in different autoimmune responses. 29 As the included literature is still recent, it is expected that physicians will have more guidance on the effects of concomitant IS and ICI on patient outcomes in the upcoming years.
Management of immunosuppressive therapy
To this day, only one practice-based guideline has been published on immunosuppressive drug management in cancer patients with pre-existing AIDs treated with ICIs, suggesting a rotation phase to introduce selective IS (e.g. biological disease-modifying antirheumatic drugs, bDMARDs) rather than non-selective IS (e.g. conventional synthetic DMARDs such as MTX) following interdisciplinary discussion prior to ICI initiation. 13 These recommendations are mostly based on AID management guidelines in the absence of prospectively collected data of ICI therapy in cancer patients with AID. Additionally, a rotation phase is not always feasible due to aggressive tumors. Holstroem et al. have recently published efficacy and safety results of concomitant IS and ICI treatment in their COLAR trial, 30 indicating the feasibility of prospective studies in this subgroup of cancer patients treated with ICIs.
Although selective immunosuppression is preferred according to the Haanen et al. guideline, 13 therapy was discontinued in 9/13 (69%) of patients included in this study who were prescribed a selective immunosuppressive drug. Short-term use of TNF inhibitors does not negatively affect anti-tumor activity of the immune activity system, 31 but more clinical data of their long-term administration on ICI efficacy are needed. Moreover, and reassuringly, the use of TNF inhibitors are being explored for their potentially synergistic antitumor characteristics in combination with ICI therapy. 32 Additional safety results on the use of tocilizumab, an anti-IL-6 drug prescribed in immune-related colitis and arthritis, increases confidence in this selective IS for AID management.30,33,34 In inflammatory bowel diseases (IBD), the biological vedolizumab appears to be safe as well. This monoclonal antibody targeting α4β7 integrin is applied in the treatment of glucocorticoid-refractory ICI-induced colitis, 35 and can be an approach to avoid broad-spectrum, non-selective IS when considering ICI initiation. 13 However, in case 1 of the case series, continuing vedolizumab during anti-PD-1 therapy did not prevent AID flare or irAE occurrence.
On the other hand, nonselective immunosuppressive therapies were registered in 47 cases, in mono- or combination therapy, such as MTX (n = 21, 62% stopped) and hydroxychloroquine (HCQ) (n = 10, 80% stopped). All azathioprine (AZA) therapy was stopped at the time of ICI initiation (n = 1). There are currently no published data evaluating the impact of MTX on ICI therapy and/or tumor response. Only one patient with Crohn's disease and melanoma had a treatment conversion from AZA and infliximab to vedolizumab prior to ICI treatment, 14 which is concurrent with the recommendations of Haanen and colleagues. 13 However, the conversion was performed at the time of cancer diagnosis, long before ICI initiation, because of a known risk of non-melanoma skin cancer in patients on AZA therapy.36,37 Additional caution is required in IS and ICI adverse event management as there may be overlapping toxicities, such as AZA hepatotoxicity and immune-related hepatitis.38,39 The same was observed for HCQ. HCQ therapy was discontinued in some cases identified in the literature review, in an effort to prevent drug-associated myopathy. Myopathy is a severe adverse event of HCQ, seen in 12% of patients treated for rheumatic diseases. 40 Recent in vitro/in vivo studies highlight HCQ's potentially negative impact on ICI effectiveness.41,42 Yet, rotating from HCQ to a selective IS prior to ICI initiation may be complicated, taking into account the very long half-life of 30–50 days of HCQ. 43 The same holds true for leflunomide. 44 Besides its effect on the immune system, 45 an evaluation of the drug's pharmacokinetic parameters is necessary before adapting AID drug therapy. In IBD management, physicians did not alter baseline therapy with 5-aminosalicylates (5-ASA) analogs in the described patients. The impact of 5-ASA on ICI effectiveness is not clear but one can hypothesize that drugs with a local effect have a lower risk for systemic immunosuppressive effects. It is clear that the drug administration route should be taken into account as an additional factor in AID drug management in cancer patients treated with ICI, such as topical steroid treatment of psoriasis and 5-ASA in IBD, in order to adhere to ICI treatment.
As every AID has its specific characteristics, guidelines should be tailored to the type of AID and baseline immunosuppressive regimen. Although currently available guidelines indicate a preference for selective IS, much is still unknown on AID drugs and their impact on ICI efficacy and safety.
Limitations
Case reports are a means to improve pharmacovigilance in fragile patient groups that are excluded in clinical trials. However, these reports have several limitations, such as publication bias, failure to generalize the data, lack of a cause-effect relationship, and a retrospective design. 46 Unfortunately, we observed a consistent absence of essential patient and disease-related data in both the case series and the published case reports. Thorough documentation in the patient records is crucial to achieve high-quality case reports, preferably written according to the CARE guidelines. 47 Additionally, selection and referral bias cannot be excluded. Only cases with dermatological, gastrointestinal and rheumatological AID were included which may have affected the AID flare and irAE rate. Other AIDs, such as systemic lupus erythematosus, have a more systemic impact due to the production of multiple autoantibodies, compared to, psoriasis which remains localized to the skin.
The development of local and/or national registries is therefore recommended to provide solid databases for future research. The French REISAMIC registry is an example of such a relevant initiative that has already resulted in evidence on ICI treatment in cancer patients with pre-existing AID. 48 In addition, more study results are being published, showing encouraging effects of ICI therapy in patients with AID.49,50 Currently, the AIM-NIVO trial (Clinicaltrials.gov, NCT03816345) is collecting valuable evidence on the use of nivolumab in cancer patients with different types of AID. Nevertheless, researchers need to be encouraged to publish prospectively collected, structured data on IS treatment, as this will facilitate the development of evidence-based guidelines on IS management in AID patients with cancer. However, as AIDs present in various forms and require tailored IS management, individual case discussion with both a clinical and pharmacological approach may remain indispensable for adverse event management in this distinct patient group. A multidisciplinary collaboration between oncologist, organ specialist and clinical pharmacists/pharmacologists are therefore highly recommended to provide optimal AID and adverse event control, as well as adequate tumor suppression. In Belgium, the BITOX initiative, which is a multidisciplinary board to discuss those patient cases (www.BSMO.be/bitox) is fulfilling this role.
Conclusions
A detailed overview of IS management in cancer patients with AID receiving ICI therapy is presented. The lack of data on AID medication management in this patient group stresses the need for structured pharmacovigilance registration and publication of IS treatment strategies. As the amount of uncertainties in these patients is still, and may remain high, close monitoring and individual case management is required upon AID flare or irAE occurrence to evaluate the safety and effectiveness of combined IS-ICI therapy, thus ensuring responsible patient care.
Supplemental Material
sj-pdf-1-opp-10.1177_10781552231167824 - Supplemental material for Immunosuppressive therapy management in cancer patients with autoimmune diseases treated with immune checkpoint inhibitors: A case series and systematic literature review
Supplemental material, sj-pdf-1-opp-10.1177_10781552231167824 for Immunosuppressive therapy management in cancer patients with autoimmune diseases treated with immune checkpoint inhibitors: A case series and systematic literature review by Stephanie C.M. Wuyts, Charlotte A.H. Cappelle, Marthe Verhaert, Bert Bravenboer and Sandrine Aspeslagh in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
Not applicable.
Author Contributions
SW, MV, and SA were involved in the conceptualization of this study. SW and CC collected the patient data. SW, CC, and MV participated in the search and inclusion of articles for literature review. SW and CC analyzed the data with help of MV and SA. SW and CC wrote the first draft in cooperation with MV, under supervision of BB and SA. All authors reviewed and approved the final version of the manuscript.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
This retrospective study was approved by the Committee of Medical Ethics of the Universitair Ziekenhuis Brussel (UZ Brussel) - BUN: 1432021000707.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Patient Consent Statement
All living patients reported in the case series provided informed consent. Informed consent was waived for patients who were deceased at the time of the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
