Abstract
Objective
The lack of anticancer drugs for curative and supportive purposes is the critical reason for the low survival rate in low-and-middle-income countries. This study aims to analyze whether the National Essential Medicines List (NEML) and Registered Essential Medicines List (REML) are in concordance with the World Health Organization (WHO) Essential Medicines List (EML) and whether the formularies prevalent in the country are parallel to each other and to the NEML.
Method
An observational study design was used in which antineoplastic drugs from the 2021 NEML and REML were compared with 2021 WHO EML to evaluate their availability in Pakistan. Market access was determined. Moreover, the formularies of six different hospital types were compared with each other and with the NEML, and REML to estimate the availability within hospitals.
Results
There were 66 anticancer drugs in 2021 WHO EML and all were found in Pakistan's 2021 NEML but only 48 drugs (73%) were found in the REML. Hydroxycarbamide and dasatinib were two registered drugs absent in all hospitals’ formularies. The market access for anticancer medicines was 73% (48 of 66). Semigovernment hospital (86%) has the highest availability, followed by the government hospital (80%). All the hospitals have unregistered drugs including bortezomib, lenalidomide, and mesna.
Conclusion
Pakistan's NEML adopts WHO EML abruptly but all medicines are not registered. The hospitals are trying their best to increase availability but optimum drug regulations to revise NEML based on the country's requirements and emphasizing registration of anticancer medicines are needed to improve the country's availability of antineoplastic agents.
Introduction
The World Health Organization (WHO) began issuing a list of drugs called Essential Medicines list (EML) that deemed vital health systems components. The list was made through a complex process. Based on EML as a guideline, most regulatory authorities produced their National Essential Medicines List (NEML) to clarify, prioritize, procure, and finance their essential medicines. These NEMLs show the recognition and prioritization of the drugs to the public. The 2021 EML is the 22nd edition, focusing on cancer medicines.1,2
In developing countries, lack of access to affordable essential medicines is a significant health issue compared to developed countries. 3,4 WHO defined access to medicines as “available within the context of functioning health systems at all times in adequate amounts, in the appropriate dosage forms, with assured quality and adequate information, and at a price the individual and community can afford.” 5 Access included the availability and affordability. Availability is defined by WHO as “the presence of medicines in national formulary available to patients for free or for a fixed fee.” In contrast, affordability is defined as “for the health system—the proportion of spending on cancer medicines compared to existing expenditure on medicines or other health products and services. For individual patients—the number of days’ wages needed to pay for the cost of treatment.”6,7 The primary disease burden is remarkably transferring from infectious diseases to noncommunicable diseases in these countries, particularly cardiovascular and cancer diseases. 1 Cancer is an increasing threat to the public worldwide. The low-and-middle-income countries (LMICs) have specifically increased the incidence of cancer cases. So availability of all the anticancer drugs is critical for increasing survival rates, especially in LMICs 8 because the cancer treatment is comprised of multiple drugs involving different treatment strategies and interventions. 9 Access to essential medicines is decisive, calculated by market access, and patient access in many studies.10,11 Moreover, placing anticancer drug on NEML does not guarantee its availability to the patient. The availability is determined from the institutions’ formularies in the countries that show the actual access to the patient.2,11
Pakistan has a hybrid healthcare system comprise of government, semigovernment, private, and nonprofit organizational (nongovernmental organization, NGO) hospitals. Private sector caters the needs of 70% of the population of the country. 12 The availability and affordability of anticancer medicines are poor in Pakistan. The leading causes are the weak health system, uneven resource allocation, limited number of drugs in the formulary, and lack of communication among health professionals. Unavailability or unaffordability of anticancer drugs for patients could result in worsening patients’ disease state. 13
Lack of adequate anticancer drugs leads to lower survival rates, which will bring a great challenge for LMICs like Pakistan, where the availability and affordability of these drugs are poor. 13 A few research studies studied the accessibility to medicines, 14 but none of them provided enough information about essential cancer drugs. Recent few studies on shortages of oncology medicines mentioned the unavailability of anticancer medicines in the country.9,15 The primary objective of this research was to evaluate the concordance of anticancer drugs in the essential medicines list of country in comparison to the WHO.
Method
An observational study was conducted in which the anticancer drugs in the WHO EML were compared with the Pakistan's NEML, Registered Essential Medicines List (REML), and formularies of six different hospitals of Pakistan.
The WHO EMLs’ 20th, 21st, and 22nd editions of 2017, 16 2019, 17 and 2021 18 were extracted from WHO official website. 19 The Pakistan NEMLs of the year 2016, 20 2018, 2020, and 202121,22 were accessed from the official website of the Ministry of National Health Services Regulations & Coordination Islamabad. 23 The list of registered cancer medicines was accessed from the Drug Regulatory Authority of Pakistan (DRAP) 24 and formularies were taken from the hospital pharmacists.
In this study, WHO EMLs were compared with the following NEMLs of Pakistan to evaluate the adopting trend. The antineoplastic drugs in the NEMLs were also compared with each other. Then REML was compared with the recent 2021 NEML to analyze whether REML of Pakistan is parallel to NEML. Following this, a list of drugs from 2021 NEML was prepared and sent to the pharmacies of six hospitals across the country. The pharmacists from the respective hospital marked the drugs which were physically available in the hospital. Finally, the hospital drug lists were compared with the NEML to calculate patient access and then compared with the REML to identify the presence of registered and unregistered drugs within the hospitals.
In Pakistan, there are four types of hospitals: government, semigovernment, private, and not-for-profit organization hospitals. The formularies of one nonprofit organizational hospital, three private hospitals, one governmental hospital, and one semigovernment hospital were analyzed to evaluate the availability of anticancer medicines in hospitals. All the hospitals were tertiary care hospitals.
The market and patient access were calculated based on NEML and REML. Market access was calculated by the number of accessible active pharmaceutical ingredients (APIs) approved in Pakistan for treating all kinds of cancers. So market access was defined as “the number of anticancer APIs approved in Pakistan as the percentage (%) of number of APIs available in 2021 WHO EML.” 2 Patient access was calculated by the number of available anticancer medicines listed in the NEML for treating all kinds of cancer. So patient access was defined as “the percentage (%) of number of APIs available in the hospital to the number of anticancer APIs present in 2021 NEML.” 2 Microsoft Excel 2013 was used to analyze the data.
Results
There were 66 anticancer drugs (immunomodulators and antineoplastics) in 2021 WHO EML classified in 6 classes: 4 immunomodulators for nonmalignant disease, 35 cytotoxic medicines, 10 targeted therapies, 4 immunomodulators, 9 hormones/antihormones, and 4 supportive medicines. All the 66 drugs were present in 2021 NEML and 48 (73%) were found in registered medicines list of Pakistan. Out of 35 cytotoxic agents, 29 (83%) medicines were officially registered in Pakistan. From targeted therapies, 6 (60%) were registered (Table 1). The market access (%) for immunomodulators and antineoplastics was 73% (48 of 66) in Pakistan and the patient access (%) varied with the type of hospital.
Comparison among WHO EMLs, NEMLs, REML, and formularies of six facilities.
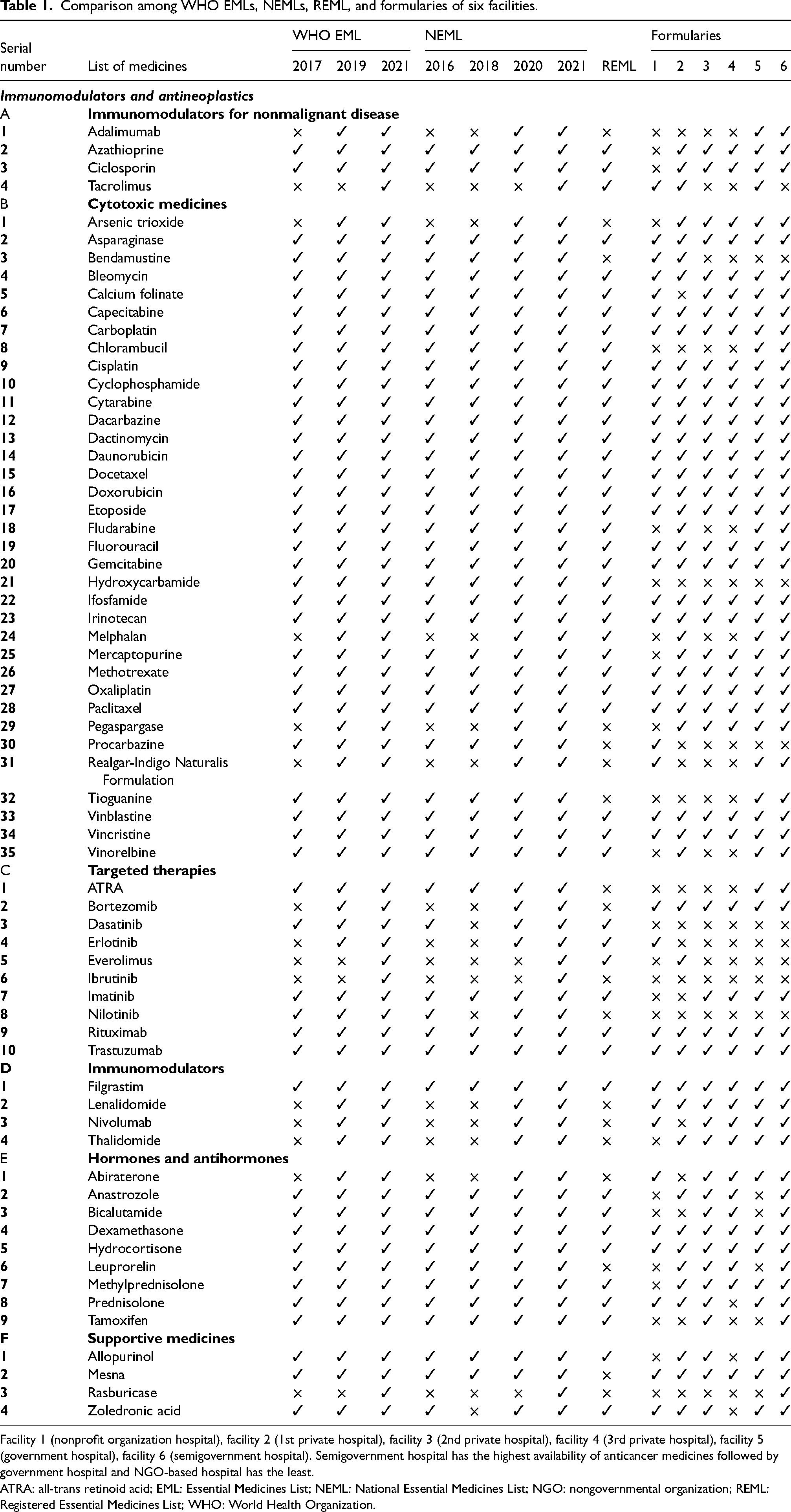
Facility 1 (nonprofit organization hospital), facility 2 (1st private hospital), facility 3 (2nd private hospital), facility 4 (3rd private hospital), facility 5 (government hospital), facility 6 (semigovernment hospital). Semigovernment hospital has the highest availability of anticancer medicines followed by government hospital and NGO-based hospital has the least.
ATRA: all-trans retinoid acid; EML: Essential Medicines List; NEML: National Essential Medicines List; NGO: nongovernmental organization; REML: Registered Essential Medicines List; WHO: World Health Organization.
Comparison of NEMLs (2016, 2018, 2020, 2021) and NEMLs versus WHO EMLs
The NEMLs of Pakistan were updated as the WHO EML as seen from 2016, 2019, and 2021 NEMLs but the 2018 NEML has 48 drugs. The drugs in some classes (immunomodulators for nonmalignant disease, cytotoxic drugs, immunomodulators, and hormones/antihormones) were the same in both NEML 2016 and 2018. But the number of targeted therapies and supportive medicines were different (dasatinib, nilotinib, and zoledronic acid). Pakistan's 2016 NEML has more cancer drugs than 2018 NEML. The 2020 NEML has 62 drugs with the addition of 10 drugs (adalimumab, arsenic trioxide, melphalan, pegaspargase, Realgar-Indigo Naturalis Formulation, bortezomib, erlotinib, lenalidomide, nivolumab, thalidomide, and abiraterone). The NEML 2021 is the most recent list with the addition of four drugs (tacrolimus, everolimus, ibrutinib, and rasburicase).
REML versus 2021 WHO EML/2021 NEML
Out of 66 medicines from 2021 WHO EML and 2021 NEML, 48 (73%) were registered in Pakistan (Figure 1). It included 29 cytotoxic medicines and 3 immunomodulators for nonmalignant disease. There were six targeted therapies, one immunomodulator, seven hormones/antihormones, and two supportive medicines. Among 48 registered agents, there were 2 drugs that were absent in all hospitals’ formularies. The first agent was hydroxycarbamide, a cytotoxic medicine, and the other was dasatinib, a targeted therapy agent (Table 1).
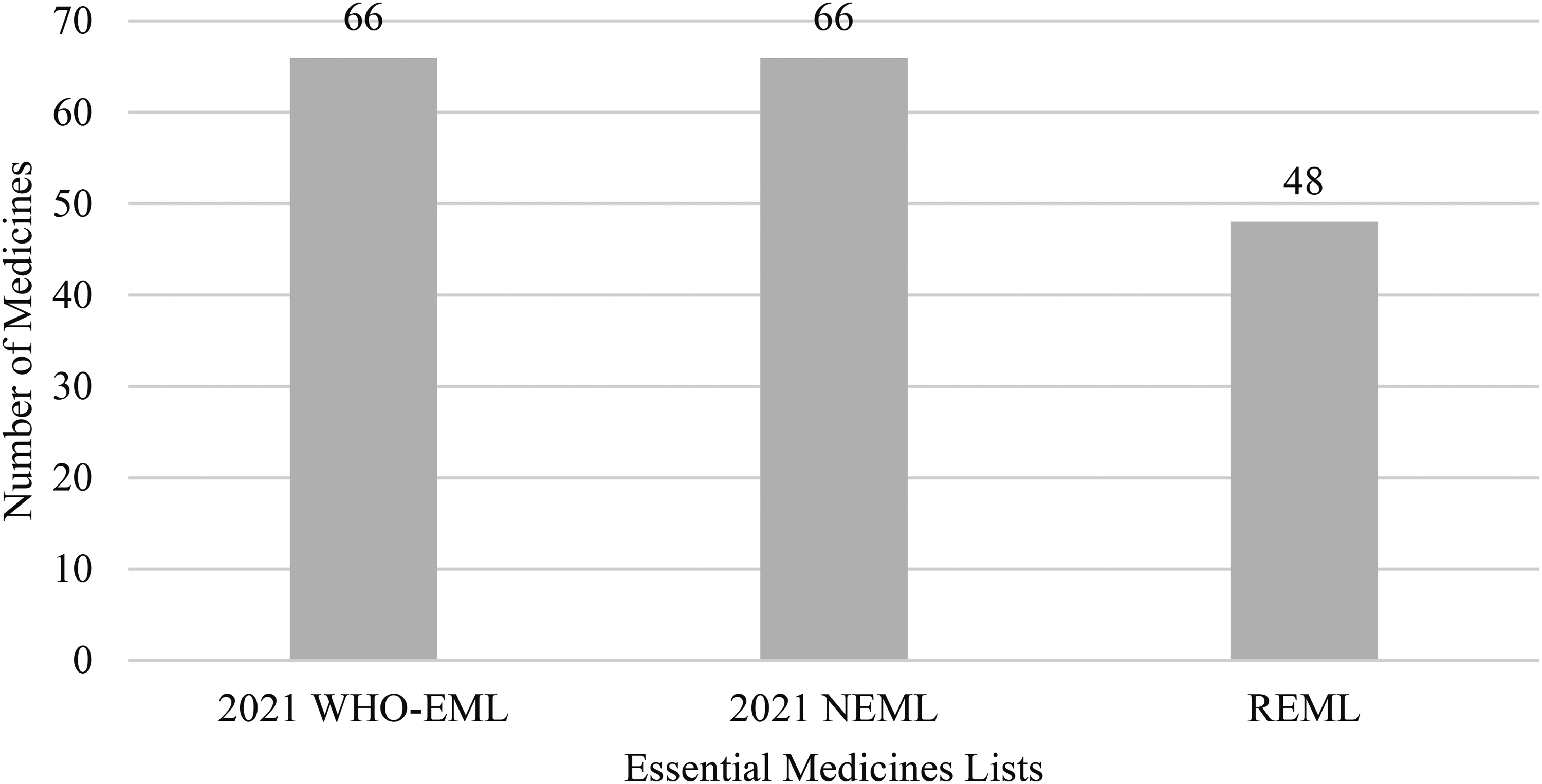
Comparison of number of drugs in 2021 WHO EML, 2021 NEML, and REML. EML: Essential Medicines List; NEML: National Essential Medicines List; REML: Registered Essential Medicines List; WHO: World Health Organization.
Patient access based on 2021 NEML
In Pakistan, based on NEML (whether registered or unregistered), the availability of medicines was depended on the type of hospital. The patient access was highest for semigovernment hospital (86%). For the 3 private hospitals, 48(73%), 48(73%), and 44(67%) medicines were available. In the government hospital, 53(80%) medicines were found, whereas, 40(61%) medicines were available in NGO-based hospital (Table 2). Figure 2 shows the comparison of availability of different classes of anticancer medicines in all six hospitals.
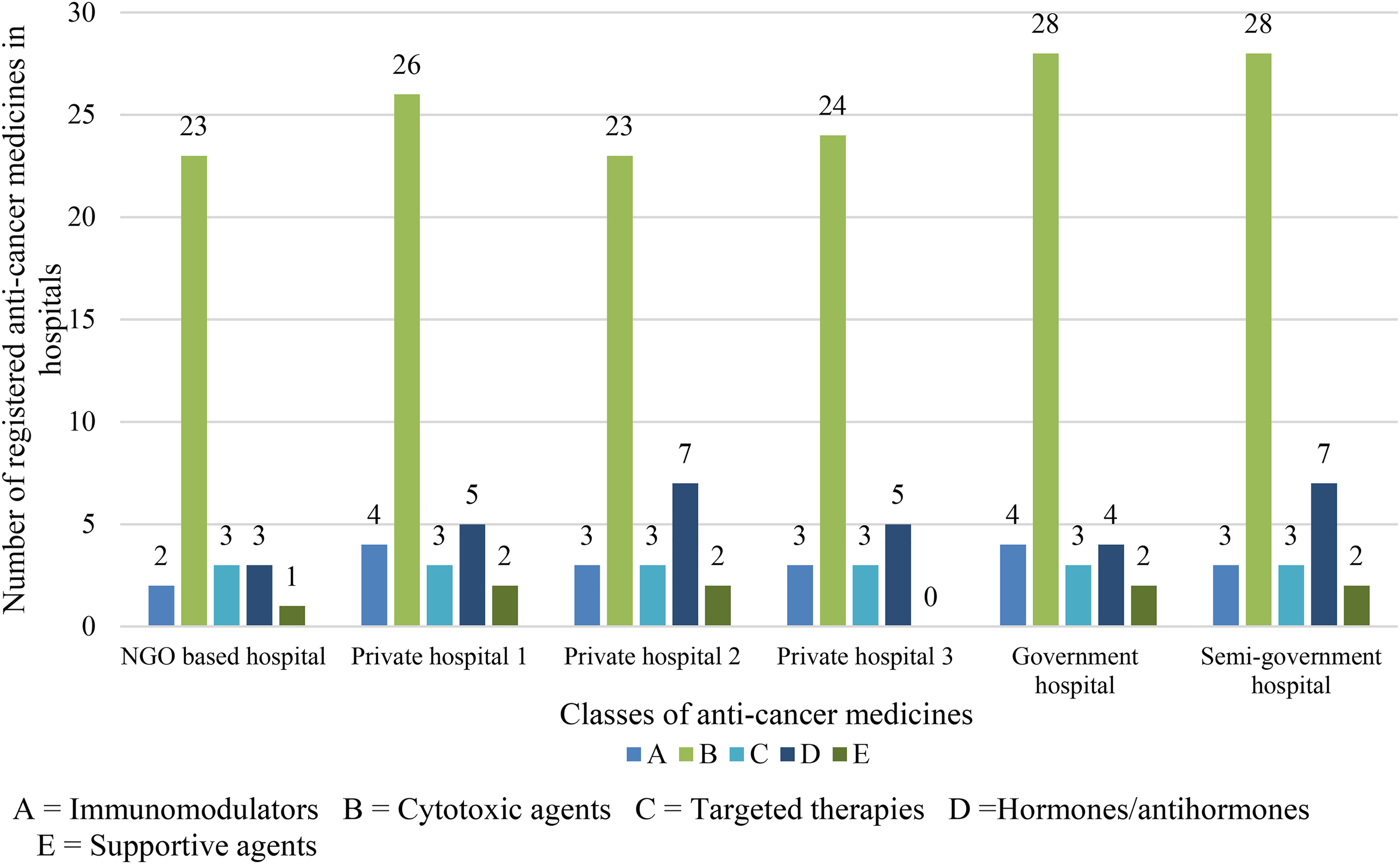
The availability of different anticancer medicines in different hospitals.
Medicine information in each hospital.
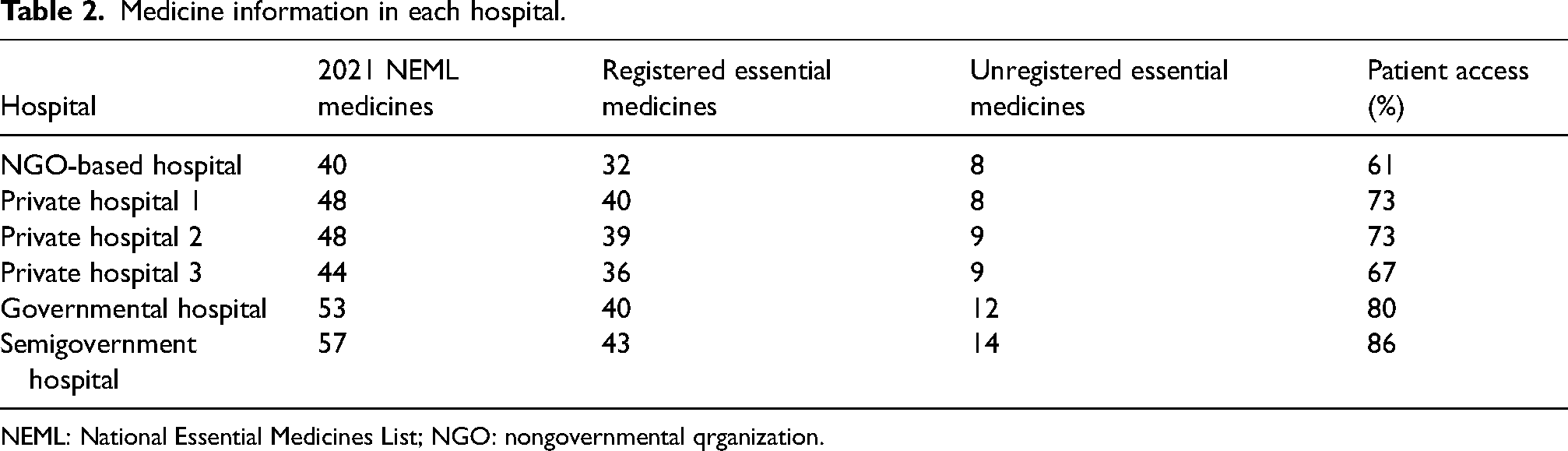
NEML: National Essential Medicines List; NGO: nongovernmental qrganization.
Presence of unregistered drugs from NEML in hospitals
When REML is compared to NEML, it was found that only 48 drugs were registered. Comparing to the hospital formularies, it was shown that unregistered NEML drugs were present in the hospital formularies. The semigovernment hospital has highest unregistered medicines followed by the government hospital (Table 2). The unregistered cytotoxic agents were arsenic trioxide, bendamustine, pegaspargase, procarbazine, Realgar-Indigo Naturalis Formulation, and tioguanine. The unregistered targeted therapies included all-trans retinoid acid, bortezomib, and nilotinib. The unregistered drugs which were present in all hospitals included bortezomib, lenalidomide, and mesna (Table 3).
List of unregistered anticancer drugs present at different hospitals.
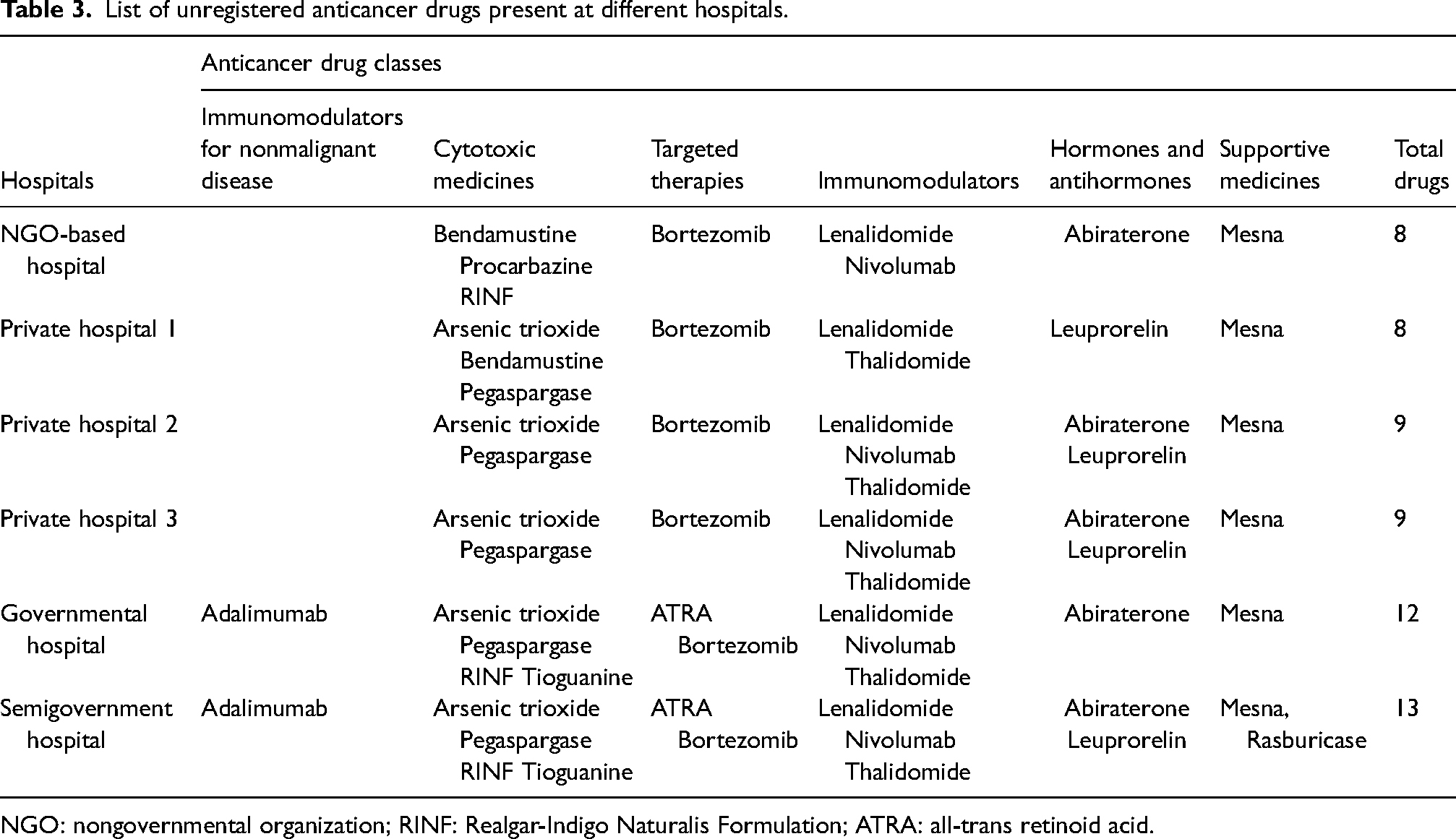
NGO: nongovernmental organization; RINF: Realgar-Indigo Naturalis Formulation; ATRA: all-trans retinoid acid.
Discussion
The cancer burden is increasing in developing countries with a growing population. In Pakistan, it is critical to ensure that the population has access to essential medicines and exceptionally affordable anticancer medicines. Because the NEML covers the healthcare needs of most of a country's population, it must be continuously updated to imitate new treatment alternatives and changing therapeutic demands followed by their registration. The study evaluates the availability of anticancer medicines in the country through the actual estimation of available data. As evidenced in the NEMLs of 2016, 2018, 2020, and 2021, Pakistan's healthcare system follows the WHO EML actively. In the current 2021 NEML, all the 2021WHO EML agents are present, as seen in upper-middle-income countries. 25
The country's NEML is being adopted from WHO EML. But only 48 (73%) medicines from 2021 NEML are registered. There are two main reasons: the first reason is the compromised role of the DRAP. Similar results are found in Armenia, with the registration of 73% anticancer medicines from the EML. 26 A study from Europe also recommended increasing awareness of regulators, policymakers, and health professionals. 6 Another study found that half of the world's population is deprived of essential medicines irrespective of NEMLs in many LMICs 27 whereas a survey in European countries found that 90% of the countries have access to 63% of the medicines from WHO EML. 6 Optimized role of regulatory authorities with drug registrations are needed because they ensure quality and safety by allowing the pharmaceutical market to regulate, monitor, and govern its supplies.
The compromised role of the regulatory authorities led to four other issues. Firstly, it led to divergent nature of NEML and the registered medicine list. On the one hand, the country is actively adopting the WHO EML, but registration of anticancer medicines is not dependent on that list. Therefore, the regulatory authorities should evaluate the need for essential anticancer medicines in the country and develop their NEML and registered medicines list accordingly. The second issue is the complexity of the registration process. The absence of sound and updated policies leads to an irritating registration process, and the investors found it challenging to fulfill rigid requirements for registration. 28 The third issue is the unorganized process of import. Lack of policies, good platforms, and incentives for importing anticancer drugs also lead to the lack of registered drugs. Registration also regulates their import in optimized storage conditions with the least financial burden.29,30 The fourth issue is the lack of local production. Pakistan imports most of the anticancer medicines from different countries. In contrast, countries fulfilling their requirements through local production face far fewer challenges, such as India and China.9,28 The regulatory authorities should pay attention to increase local production and the quality of locally produced drugs.
Due to the compromised role of regulatory authority, NEMLs of Pakistan are not designed according to the country's requirement, and drug import, registration, and licensing policies are very complex so the healthcare system of Pakistan is unaware of the most needed essential anticancer drugs. The hospitals prefer to import drugs on demand (if they have patients) rather than keep enough stock which is highly risky in case of cancer. In this scenario, the healthcare system of Pakistan is not prepared to face the expected 2040 cancer burden. 31
The second reason for the lack of registered medicines is financial instability. Economic factors like budget implications and cost-effectiveness are vital roadblocks to accepting novel cancer treatments into the Ministry of Health Medicines Formulary. This condition is also observed in other countries, where access to novel cancer treatments is uneven.26,32 Lack of cost-effectiveness has been identified as one of the primary reasons for the rejection of anticancer drugs. A study in Europe about access to essential anticancer medicines also mentioned that financial instability is a barrier to access to novel expensive drugs for developing countries. 6 The registration of all the essential molecules is the utmost need of the country due to the increasing burden of cancer cases. A study on the selection of oncology medicines in LMICs also stated that the role of drug regulators and international organizations in LMICs is needed to minimize the death toll due to cancer. This reduction in morbidity and mortality can only be made by increasing access, availability, and affordability of anticancer drugs. 25
Market access is related to presence of registered medicines in the market, whereas patient access is related to the availability of anticancer drugs under NEML within a hospital. The market access for immunomodulators and antineoplastics is 73% in Pakistan. According to a study conducted in Thailand, market access is 43% lower than in Pakistan because Thailand has a much smaller number of registered drugs than Pakistan does. However, all necessary molecules must be registered in both countries. Availability or patient access of anticancer medicines varies depending upon hospital type and its good news that although not all the essential medicines are not registered, but hospitals are trying their best to serve patients, purchasing more unregistered essential medicines. The highest patient access was 86% in semigovernment hospital, where its market access was 73%. A study in Thailand found opposite result of greater market access than patient access as in Pakistan. 2 The availability in semigovernment hospital is satisfactory followed by government hospital. The results are unpredictable because government and semigovernment hospitals mostly have budget issues whereas the private hospitals are self-sustaining with rare budget issues. A recent study from Pakistan highlighted that private and NGO-based hospitals have more access to anticancer drugs whereas the drugs from government and semigovernment hospitals have less availability but affordable to the patients. 15
All the hospitals have unregistered medicines showing that REML is unable to meet the demands. The hospitals have to import these unregistered medicines to meet their needs but the process is complex. When there is a patient need base/institution need base import for any unregistered medicine in the hospital, the hospital submits a special request to DRAP for import permission. This process also causes delayed treatment leading to increased morbidity and mortality. A study in Armenia found that 18% of registered medicines were absent in drug stores, 26 so our results showed that 27% drugs are not registered from NEML but most of the registered drugs are present in all types of hospitals. A study in Mexico found that the availability of anticancer drugs falls below WHO's 80% target in both public and private sectors. 8
As far as we knew, this is the first study to estimate the anticancer medicines in the NEML of Pakistan but it has limitations. Firstly the results cannot generalize to the whole country because of the small sample size as we measured the patient access based on the formularies of only six hospitals. However, to overcome this, all four types of hospitals from different regions of Pakistan were included. Moreover, three private hospitals were included. Secondly, the hospital pharmacists were requested to share the data about the availability of anticancer drugs within hospitals that could be biased as many drugs are imported on demand but they are not present in the stock. In this case, the patient access will be overestimated. To avoid the biasness, the hospital pharmacists were clarified to mark only those drugs which were physically present within the hospital.
Conclusion
The study highlighted the lack of availability to the essential anticancer drugs in the country because all medicines listed in 2020 NEML are not officially registered. On one hand, the regulatory authorities are updating the NEML in accordance with the WHO EML, but on other hand medicines are not registered parallel to the NEML. The market access is low but patient access is satisfactory depending upon hospital. The NEML needs to be revised based on the country's requirements, and all the essential anticancer medicines need to be registered based on the updated NEML. We believe that optimum drug regulations and emphasizing anticancer therapy are required to improve the situation.
Footnotes
Abbreviations
Authors’ contributions
SS and CY were involved in conceptualization; RA and IM in data retrieval; SS and CY in data analysis; and SS, JZ, and CY in writing the original draft. All author s were involved in critical revision and editing of the manuscript and approval of the final version of the manuscript. CY supervised the whole study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the National Natural Science Foundation of China (72174166).
