Abstract
Introduction
Occupational exposure to antineoplastic drugs can lead to long-term adverse effects on workers’ health. A reproducible Canadian surface monitoring program was established in 2010. The objective was to describe contamination with 11 antineoplastic drugs measured on 12 surfaces among hospitals participating in this annual monitoring program.
Methods
Each hospital sampled six standardized sites in oncology pharmacies and six in outpatient clinics. Ultra-performance liquid chromatography coupled with tandem mass spectrometry was used for cyclophosphamide, docetaxel, doxorubicin, etoposide, 5-fluorouracil, gemcitabine, irinotecan, methotrexate, paclitaxel, and vinorelbine. Platinum-based drugs were analyzed by inductively coupled plasma mass spectrometry; this excludes inorganic platinum from the environment. Hospitals filled out an online questionnaire about their practices; a Kolmogorov–Smirnov test was used for some practices.
Results
One hundred and twenty-four Canadian hospitals participated. Cyclophosphamide (405/1445, 28%), gemcitabine (347/1445, 24%), and platinum (71/756, 9%) were the most frequent. The 90th percentile of surface concentration was 0.01 ng/cm² for cyclophosphamide and 0.003 ng/cm² for gemcitabine. Centers that prepared 5000 or more antineoplastic per year had higher concentrations of cyclophosphamide and gemcitabine on their surfaces (p = 0.0001). Almost half maintained a hazardous drugs committee (46/119, 39%), but this did not influence the cyclophosphamide contamination (p = 0.051). Hazardous drugs training was more frequent for oncology pharmacy and nursing staff than for hygiene and sanitation staff.
Conclusions
This monitoring program allowed centers to benchmark their contamination with pragmatic contamination thresholds derived from the Canadian 90th percentiles. Regular participation and local hazardous drug committee involvement provide an opportunity to review practices, identify risk areas, and refresh training.
Introduction
Healthcare workers are occupationally exposed to a large array of chemicals, including hazardous drugs. 1 The well-known hazardous drug list from the National Institute for Occupational Safety and Health (NIOSH) is not updated in real-time; the latest was published as a draft in 2020 and only encompasses new drug approvals received up to December 2015. 2 This organization encourages centers to maintain a local list of hazardous drugs. 2 However, the vast number of new drugs commercialized and the lack of easy-to-use criteria complicates the task of individual hospitals. There is even confusion with the use of the term “cytotoxic.” 3 Khaira and Guy emphasized that the growing hazardous drug list now encompasses a wide range of molecules and some, such as oncology biologics, may present specific compatibility challenges. 4
Environmental monitoring can help to identify issues and trends over time. The United States Pharmacopeia (USP) requires surface sampling every six months. 5 Likewise, Canadian guidelines recommend it once or twice a year, or after a practice change.6,7 Although there is flexibility in the implementation of a surveillance protocol, most instances recommend monitoring multiple surfaces and using more than one tracer drug. Different groups have adopted various strategies to identify contamination thresholds, as there is no safe exposure limit for antineoplastic drugs.8–11 The current Canadian monitoring program uses drug-specific 75th and 90th percentiles as pragmatic limits that facilitate an annual benchmarking of surface contamination. 8
Even so, surface contamination represents a proxy to estimate the risks to workers. A recent review reported a high degree of variability in biological monitoring studies, spanning from no contamination to almost all workers exposed. 12 The authors identified nurses as the most at risk. Despite this risk, a good proportion of nurses from another study indicated “never or sometimes using” various exposure controls and personal protective equipment (PPE) such as chemotherapy gloves (23%) or gowns (56%). 13 These factors were associated strongly with an increased hazard ratio of miscarriage. 13 In a French study, Benoist et al. reported that nearly half of the respondents to their survey felt that their risk of exposure was very low. 14
Training is of the utmost importance to make sure that staff is correctly handling antineoplastic drugs, and that they are well aware of the rationale and the risks involved. The USP 800 requires training and competency assessments. 5 Stage 2 of implementing Washington State's hazardous drug rule mandated employers to provide hazardous drug training. 15 Surface monitoring is a relatively low-cost measure to document potential occupational exposure; it also provides an opportunity to increase awareness. Periodic feedback on the current contamination to workers is a useful reminder that can be effective in association with a training program. For example, some authors monitored their surface contamination after training sessions were done.16,17
The primary objective of this study was to describe contamination with 11 antineoplastic drugs measured on 12 surfaces among Canadian hospitals participating in a monitoring program. The secondary objective was to assess general, training, and cleaning practices used by the centers.
Methods
Participating centers
The program consists of cross-sectional monitoring conducted once a year with voluntary Canadian centers. Pharmacy Department directors of Canadian centers with at least 50 beds were invited to participate. An email was sent on 30 November 2021 to a contact list of 257 people, including 138 pharmacy directors from 220 centers (in alphabetical order: Alberta (n = 3 centers contacted), British Columbia (n = 19), Manitoba (n = 13), New Brunswick (n = 9), Newfoundland and Labrador (n = 3), Northwest Territories (n = 1), Nova Scotia (n = 10), Ontario (n = 66), Prince Edward Island (n = 2), Quebec (n = 74), Saskatchewan (n = 19), and Yukon (n = 1)). Several follow-up email invitations were sent to centers that did not respond to the first invitation. The centers that consented to participate filled out an online questionnaire describing their centers’ characteristics and their usual practices with antineoplastic drugs (e.g. safe handling, hygiene and sanitation, training, results communication). Study data were collected and managed using Research Electronic Data Capture (REDCap) tools hosted at CHU Sainte-Justine. REDCap is a secure, web-based software platform designed to support data capture for research studies. 18
Sampling
Centers were asked to sample 12 standardized sites according to a prespecified method. Any site sampled that did not meet the inclusion criteria was excluded from the analysis. Sites were sampled at the end of a working day or before the beginning of the next shift so that data would reflect as best as possible a workers’ potential exposure.
The sampling was performed by a single research assistant for nearby centers (n = 20). Other centers were provided with a written method, photographs, and a video to train the person who would be performing their sampling. A research assistant shipped the sampling kits to the centers according to a specific schedule. Kits were shipped at room temperature and kept refrigerated on each center’s premises. Sampling kits contained 12 preidentified tubes, one control tube, 12 pairs of gloves, a written method, and a sampling template. Each tube contained one 6 cm × 8 cm Wypall® X60 wipe (Kimberly Clark Professional, Newton Square, Pennsylvania). Wipes were moistened with 1 mL of solution (5 mmol/L ammonium acetate in 10% v/v methanol) and stored refrigerated. Each 600 cm² surface was wiped four times: once horizontally and once vertically with each side of the wipe. Centers uploaded photographs and information about the sites sampled via the REDCap platform.
Analytical method
Samples were analyzed by the Centre de toxicologie du Québec, a public services laboratory. Each center paid the Centre de toxicologie du Québec for the drug quantification. Nine antineoplastic drugs were quantified: cyclophosphamide, methotrexate, gemcitabine, 5-fluorouracil, irinotecan, docetaxel, paclitaxel, vinorelbine, and etoposide. One additional antineoplastic drug was detected, but not quantified: doxorubicin. For an optional additional cost, centers could opt to quantify total soluble platinum, which consisted of cisplatin, carboplatin, and oxaliplatin. The method allows to quantify soluble organic platinum and excludes inorganic platinum found in the environment from other sources, as this last one is not soluble. This monitoring program is a collaboration between the analytical laboratory and the research team. The analysis of results and their interpretation was provided free of charge by the Unité de recherche en pratique pharmaceutique.
The limits of detection (LODs) and the limits of quantification (LOQ) are presented in Table 1. The LOQ and LOD used for this study were validated with the International Conference on Harmonization requirements. 19 In short, the repeated measurement of a low sample's response was used to establish the standard deviation (SD), and then to calculate the limits (where LOD = 3 × SD and LOQ = 10 × SD). For some analytes, the matrix effect was deemed problematic or the signal-to-noise ratio of the least intense diagnostic ion was not greater than 3:1 (see confirmatory criteria). In these cases, the lowest calibrator's concentration or the LOQ was selected as the functional LOD for the study. For practical purposes, this functional LOD is simply referred to as LOD for the rest of the article.
Limits of detection and quantification.
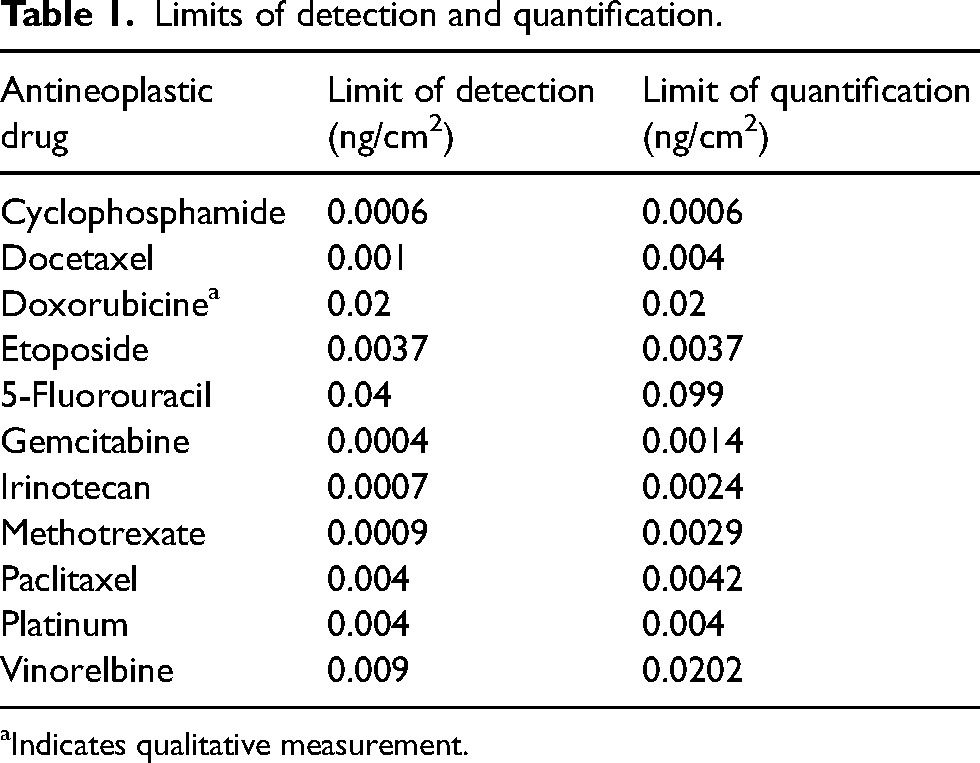
Indicates qualitative measurement.
The laboratory kept the samples refrigerated until analysis. Sample preparation consisted of the addition of 10 mL of the extracting solution (5 mmol/L ammonium acetate in 10% v/v methanol) to each polypropylene tube containing surface wipe samples, which were mechanically stirred for 10 min. Samples were then centrifuged at 2000 r/min for 5 min.
All tests were performed on the same two instruments throughout the study, an ultra-performance liquid chromatography–tandem mass spectrometry (UPLC-MSMS) system for non-platinum-based-antineoplastic drugs and an inductively coupled plasma mass spectrometry (ICP-MS) system for platinum-based-antineoplastic drugs. The [blinded for submission process] staff was not blinded as to the sampling sites.
Quantification and detection of non-platinum-based antineoplastic drugs in the sample extracts were conducted by UPLC–MSMS (Acquity UPLC chromatographic system coupled with a Xevo TQ-S tandem mass spectrometer, Waters, Milford, MA, USA). An aliquot of 1 mL was employed for analysis, diluted, and injected into the UPLC-MSMS system. Chromatographic separation was carried out on an Acquity UPLC HSS (high strength silica) T3 column (2.1 × 100 mm, 1.8 μm; Waters, Milford, MA, USA). Samples were analyzed using two separate chromatographic runs in order to optimize sensitivity. Cyclophosphamide, doxorubicin, 5-fluorouracil, gemcitabine, irinotecan, and methotrexate were analyzed using a gradient elution beginning with 2/98 0.1% formic acid in acetonitrile/0.1% formic acid in water and ending with 60/40 0.1% formic acid in acetonitrile/0.1% formic acid in water over a 3-min period. Docetaxel, etoposide, paclitaxel, and vinorelbine were analyzed using a chromatographic separation based on a gradient elution beginning with 2/98 0.1% formic acid in acetonitrile/0.1% formic acid and 0.085 mM lithium chloride in water and ending with 80/20 0.1% formic acid in acetonitrile/0.1% formic acid and 0.085 mM lithium chloride in water over 2.75 min.
For total soluble platinum analysis, after removing an aliquot of 1 mL for UPLC-MSMS analysis, 1 mL of 40% v/v HCl was added to the remaining sample and mixed again for 10 min using mechanical stirring. Then, the tubes were centrifuged at 3500 r/min for 5 min and placed afterward in an ultrasonic bath for 60 min. The tubes were once again centrifuged for 5 min at 3500 r/min. 100 μL of the extract was withdrawn from each tube and mixed with 10 mL of diluent (1% v/v HCl) and 10 μL internal standard (2 μg/mL iridium solution). Quantification of soluble platinum-based drugs in the sample extracts was conducted by ICP-MS using a NexION 350D simple-quadrupole ICP-MS system (PerkinElmer, Shelton, USA) and an ESI SC-2DX Fast System autosampler. Raw data were processed using Syngistix software (Version 2.3).
Recovery rates from three surfaces (stainless steel, floor, and laminated counter) were calculated with the reference standard Sigma (Ph. Eur. Reference Standard) and described previously. 8
Data analysis
A sample was considered positive for a non-platinum-based drug if the value was above the LOD and if for confirmatory criteria (signal-to-noise ratio > 3, retention time ± 0.02 min, quantifier/qualifier ion ratio within ± 20% of the expected ratio) were observed. For a platinum-based drug, it was considered positive if the value was above the LOQ. Descriptive statistical analyses (percentiles) were carried out with SPSS software (SPSS Statistics for Windows Version 26.0, IBM Corp., Armonk, NY, USA). For calculations, concentrations that fell between the LOD and the LOQ for a given drug were assigned a value corresponding to the LOQ divided by 2, and concentrations that fell below the LOD were assigned a value corresponding to the LOD divided by 2.20,21 Larger centers were defined as centers which prepared at least 5000 antineoplastic drugs preparations per year. A preparation was defined as a finished product. Fisher's exact test was used to compare smaller and larger centers’ characteristics. Kolmogorov–Smirnov test for independent samples was used for sub-analysis of the contamination of some practices (size of centers, existence of a hazardous drug committee, and previous communication of results with workers). A p-value less than 0.05 was considered statistically significant.
Results communication
Each center participating in this monitoring program could access its individual results on a secure-access website. Individual results were compared with the overall results from all participating centers. Sites were alerted that a sampling site may need additional attention with a color code. These contamination thresholds are updated every year with pragmatic and current data. Any result above this year's 75th percentile was highlighted in orange, and any result above this year's 90th percentile was highlighted in red. The website displays both individual and overall historical data. In addition, the research team invited centers to share an infographic summarizing the main results of the program with their staff. Webinars were conducted to share the results with the participating centers and to discuss the interpretation of their local data.
Results
Characteristics of participating centers
The surfaces from 124 centers were sampled between 1 February 2022 and 17 May 2022. Nine centers participated for the first time. The majority of participants came from Quebec (72/124, 58%), followed by Ontario (19/124, 15%), Saskatchewan (19/124, 15%), Manitoba (7/124, 6%), New Brunswick (5/124, 4%), Northwest Territories (1/124, 1%), and Yukon (1/124, 1%). Most facilities were built or had major work done more than 10 years ago (66/123 (54%) for oncology pharmacies and 74/122 (61%) for the oncology outpatient clinics.
Most of the centers prepared less than 5000 antineoplastic drug preparations per year (72/123, 59%). The other centers prepared 5000 or more antineoplastic drug preparations per year (47/123, 38%) or outsourced their antineoplastic drug preparations (4/123, 3%). Over the last year, in decreasing order, centers reported using cyclophosphamide (119/121, 98%), doxorubicin (116/121, 96%), gemcitabine (114/121, 94%), carboplatin (112/119, 94%), cisplatin (111/118, 94%), 5-fluorouracil (113/121, 93%), irinotecan (111/121, 92%), oxaliplatin (106/118, 90%), etoposide (107/120, 89%), paclitaxel (104/120, 87%), vinorelbine (99/120, 83%), methotrexate (97/119, 82%), and docetaxel (98/120, 82%). The most used drug, in grams per year was by far 5-fluorouracil (median [minimum-maximum] 1082.5 [1–12,300]), followed by gemcitabine (215.5 [4–3545] and cyclophosphamide (116.25 [2.76–2205]). Centers used a median of less than 100 g/year per center for the other drugs (Appendix 1).
Surface contamination
1445 compliant samples were analyzed out of a possible 1488 samples. Nine samples were excluded because they did not meet the inclusion criteria (i.e. they did not correspond to the standardized sampling sites). Thirty-three tubes were not sampled by the participating centers for various reasons (e.g. some centers opted to sample only pharmacy areas). One tube was damaged during shipment.
The antineoplastic drugs most frequently measured on surfaces were cyclophosphamide (405/1,445, 28%), gemcitabine (347/1,445, 24%), and platinum (71/756, 9%). Five percent or less of surfaces were contaminated with the other eight drugs (Appendix 1). Half of the centers (66/124, 53%) opted for the optional platinum dosage. The 90th percentile of the concentration measured on the surfaces was 0.0108 ng/cm² for cyclophosphamide and 0.0033 ng/cm2 for gemcitabine. It was lower than the LOD for the other drugs (Appendix 1). The surfaces most frequently contaminated with cyclophosphamide were the armrest of the patient treatment chair, the front grille inside the biological safety cabinet and the floor in front of the biological safety cabinet (Table 2).
Contamination per sampling site.
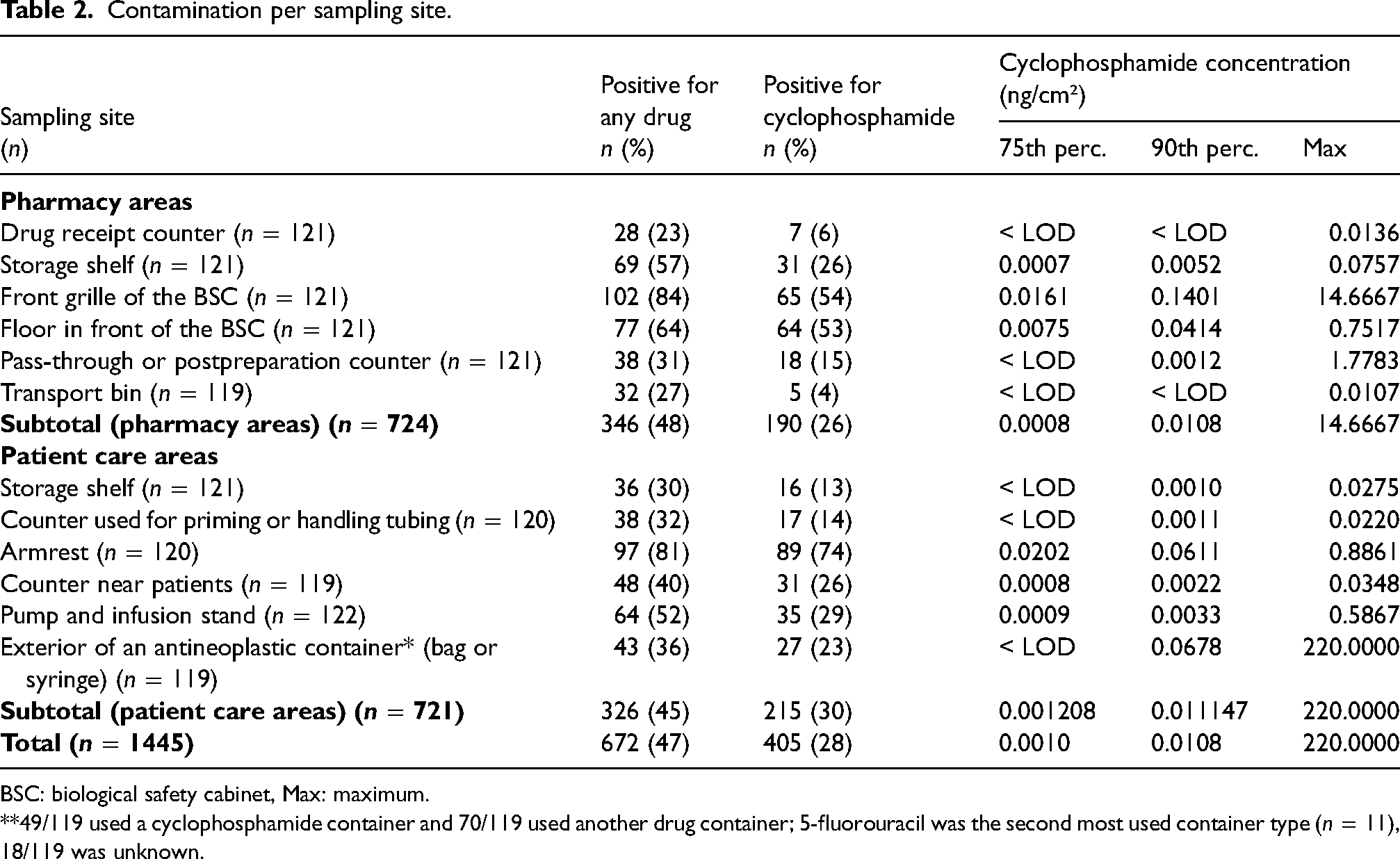
BSC: biological safety cabinet, Max: maximum.
**49/119 used a cyclophosphamide container and 70/119 used another drug container; 5-fluorouracil was the second most used container type (n = 11), 18/119 was unknown.
The larger centers that prepared 5000 or more antineoplastic drug preparations per year had significantly higher concentrations of cyclophosphamide and gemcitabine on their surfaces (p = 0.0001; Figure 1(a) and 1(d)).
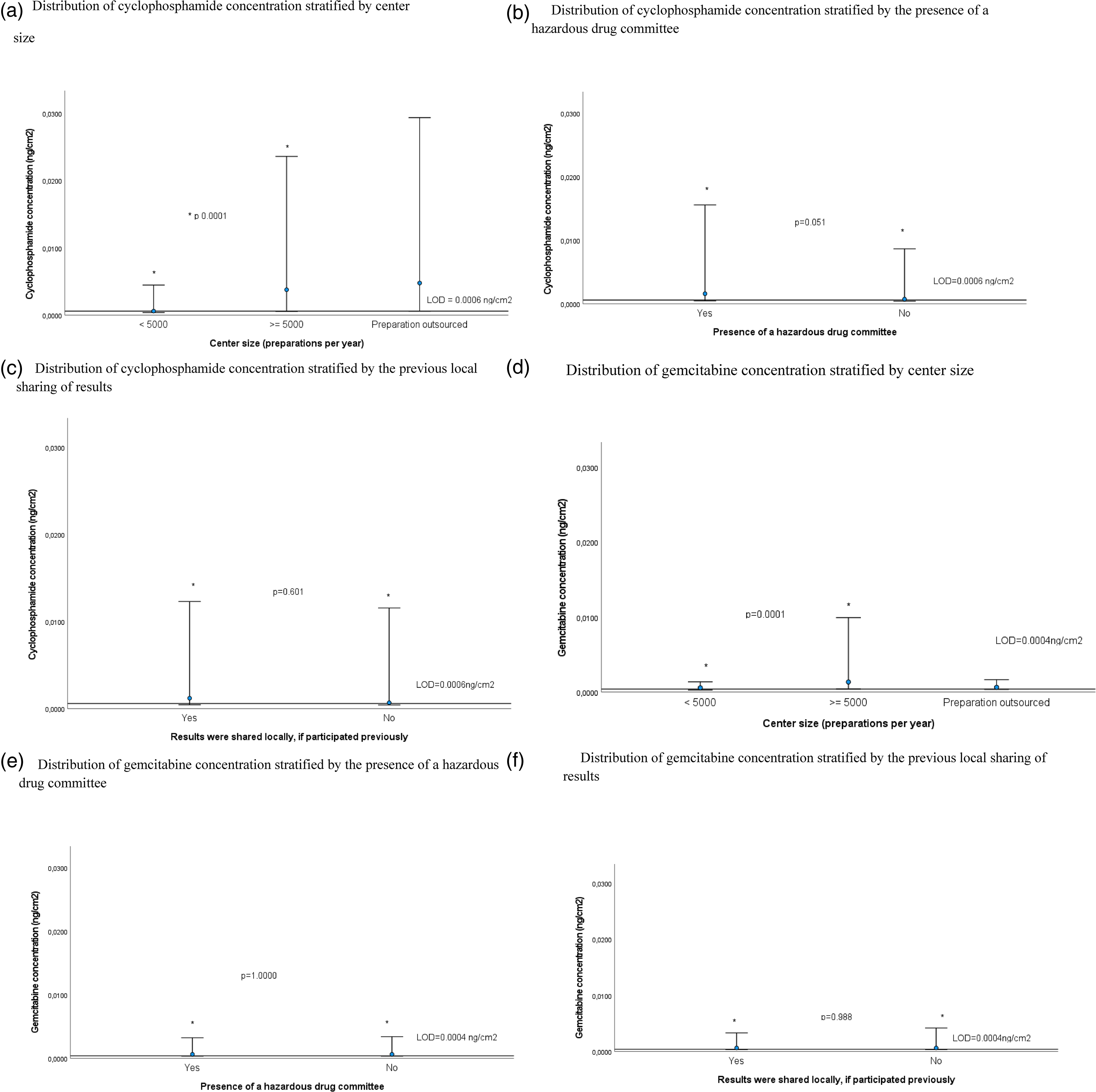
Distribution of cyclophosphamide and gemcitabine concentration stratified by center size, presence of a hazardous drug committee, and results sharing. LOD: limit of detection. Bottom bar = median, circle = 75th percentile, top bar 90th percentile. Horizontal bar y = 0.0006 represents the limit of detection for cyclophosphamide and y = 0.0004 for gemcitabine. * p = Bilateral Kolmogorov–Smirnov test for two independent samples. (a) Distribution of cyclophosphamide concentration stratified by center size. (b) Distribution of cyclophosphamide concentration stratified by the presence of a hazardous drug committee. (c) Distribution of cyclophosphamide concentration stratified by the previous local sharing of results. (d) Distribution of gemcitabine concentration stratified by center size. (e) Distribution of gemcitabine concentration stratified by the presence of a hazardous drug committee. (f) Distribution of gemcitabine concentration stratified by the previous local sharing of results.
Practices
The majority of centers reported the implementation of most general practices sought in this questionnaire (Table 3). Few centers reported having a hazardous drugs committee, but larger centers were significantly more prone to having one (22/71 (31%) vs 23/47 (49%), p = 0.010). Both cyclophosphamide and gemcitabine concentrations were not statistically different in centers with a hazardous drug committee (Figure 1(b) and 1(d)). The distribution of cyclophosphamide concentration stratified by other general practices did not reveal other statistically significant differences (data not shown).
General practices reported by the centers, stratified by center size.
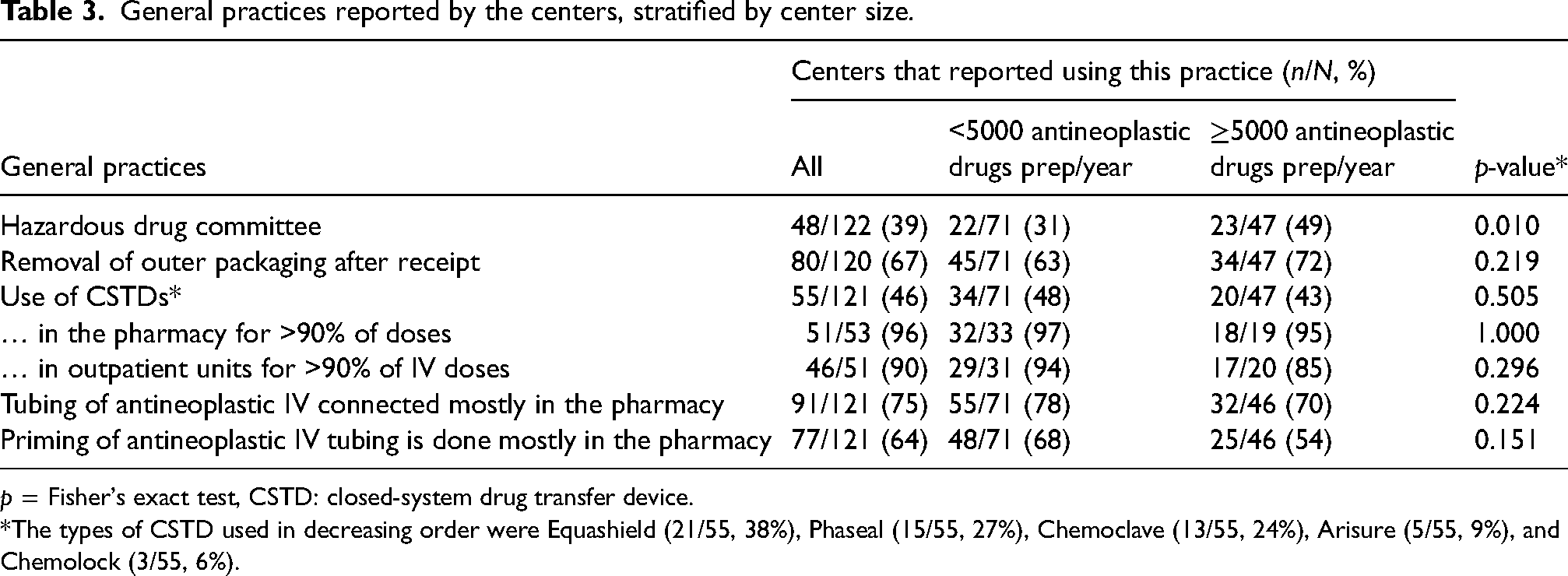
p = Fisher's exact test, CSTD: closed-system drug transfer device.
*The types of CSTD used in decreasing order were Equashield (21/55, 38%), Phaseal (15/55, 27%), Chemoclave (13/55, 24%), Arisure (5/55, 9%), and Chemolock (3/55, 6%).
Centers provided specific training on hazardous drugs for oncology pharmacy and nursing staff, but the proportion was lower for hygiene and sanitation staff (Table 4). However, larger centers provided this training more frequently to hygiene and sanitation workers (hygiene and sanitation in the oncology pharmacy 46/69 (67%) vs 39/46 (85%), p = 0.055 and hygiene and sanitation in the oncology outpatient clinic 25/67 (37%) vs 26/42 (62%), p = 0.004). Most respondents trained their staff upon hiring, but very few reported refreshing this training each year. While the majority of pharmacy staff was evaluated with a theoretical or practical exam, this was infrequent for others.
Training practices reported by the centers.
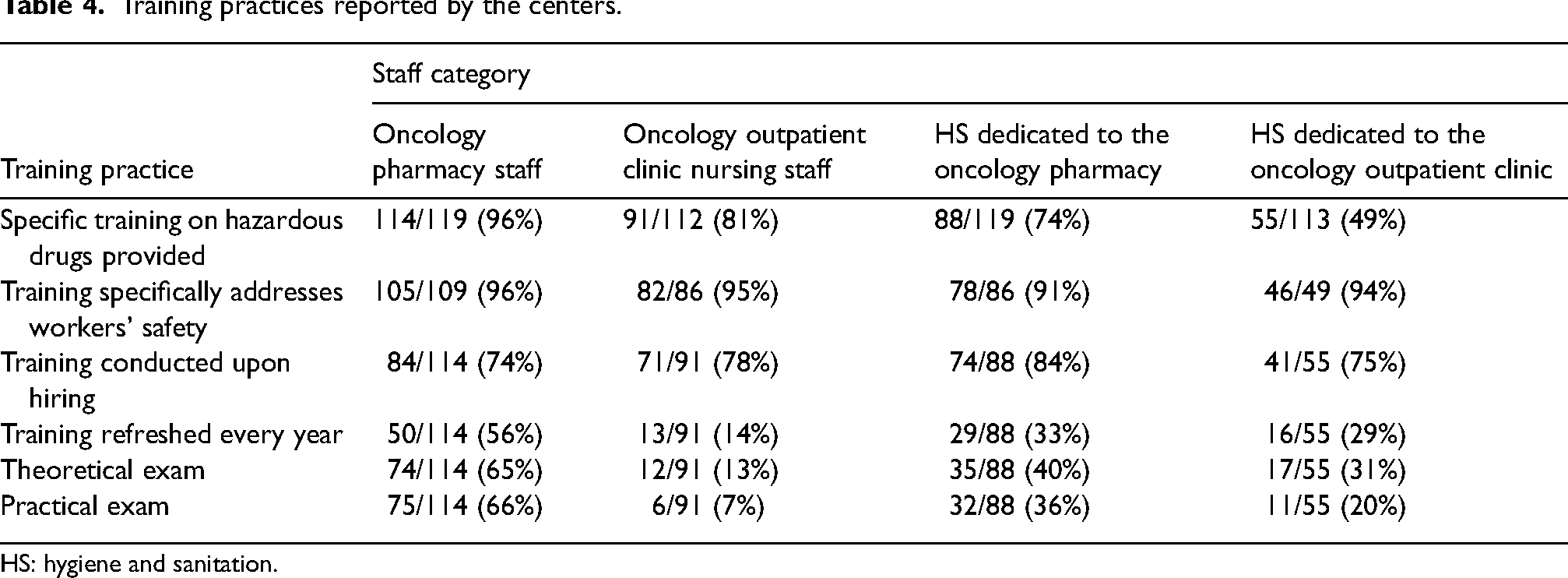
HS: hygiene and sanitation.
Cleaning practices varied in participating centers (Table 5). Significantly more large centers indicated that their cleaning staff was dedicated to the outpatient clinic (22/70 (31%) versus 29/46 (63%), p = 0.002). In addition, a higher proportion of large centers recorded their maintenance activities. Records were kept for the maintenance of the pharmacy cleanroom and in other areas of the oncology pharmacy, but few reported this for outpatient clinics. Similarly, both daily decontamination and monthly deactivation were reported more frequently in the pharmacy clean room, followed by other oncology pharmacy areas and lastly in the outpatient oncology clinic.
Cleaning practices reported by the centers, stratified by center size.
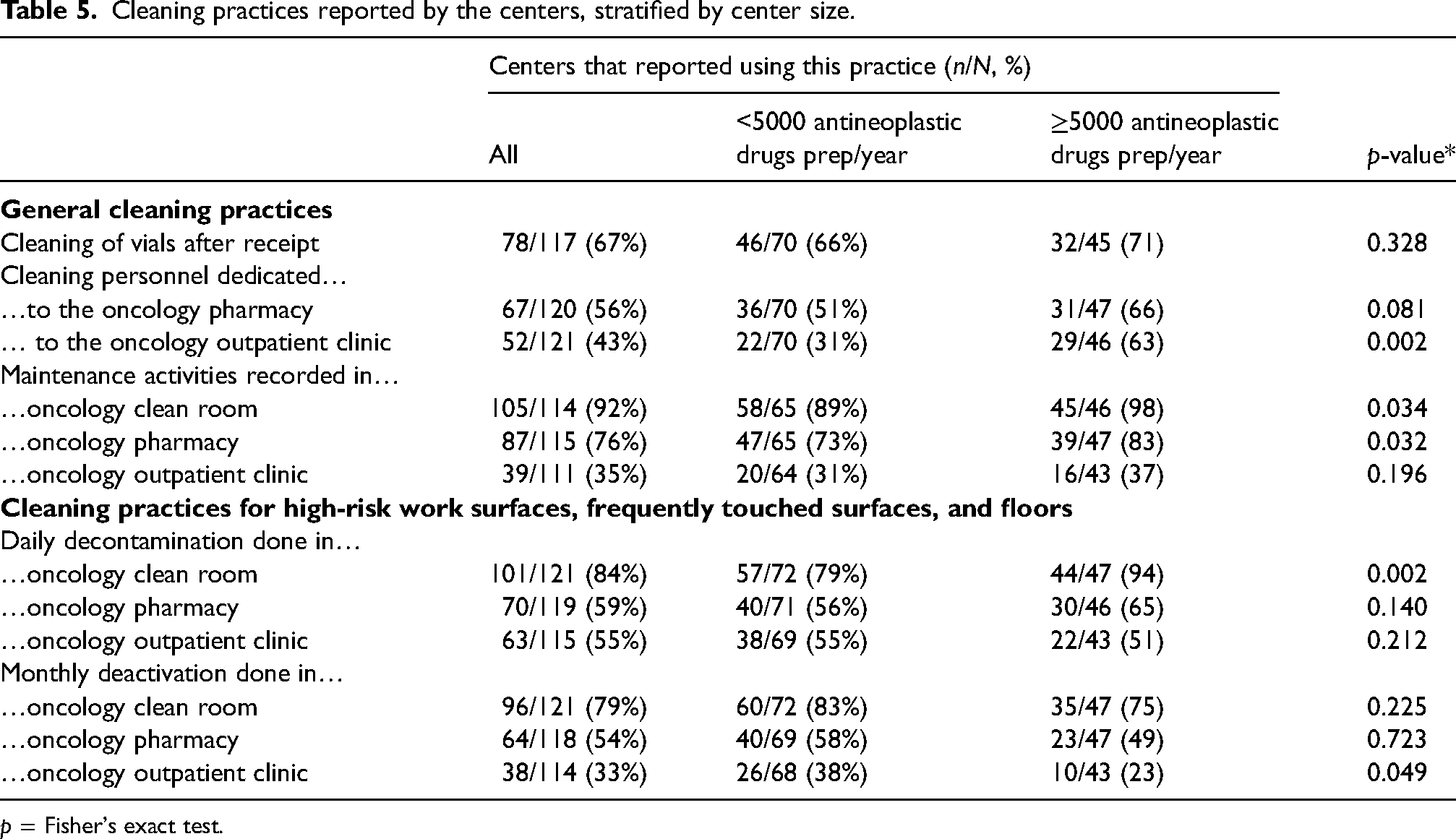
p = Fisher's exact test.
Most centers that had previously participated in this program indicated that they had shared their contamination results locally (95/114, 83%). They were mainly shared with pharmacy (90/95, 95%) and nursing staff (72/95, 76%), but some reported sharing them with hygiene and sanitation staff (45/95, 47%). Receipt of their reports prompted centers to implement diverse corrective actions such as sending reminders (83/124, 67%), performing additional decontamination (71/124, 57%), or implementing a practice change (58/124, 47%). Both cyclophosphamide and gemcitabine concentrations were not statistically different in centers where results were shared locally (Figure 1(c) and 1(e)).
Discussion
Monitoring program
This substantial monitoring program described the 2022 surface contamination in 124 Canadian centers. The availability of historical data obtained with a reproducible method provides valuable benchmarking data for participating centers. The larger centers that reported more than 5000 antineoplastic drug preparations per year were significantly more contaminated with cyclophosphamide and gemcitabine.
Some areas are found to be systematically contaminated by antineoplastic drugs throughout the years, notably the surfaces of biological safety cabinets, floors, and the armrests of the chairs where chemotherapy is administered. Among the 12 surfaces sampled in this program, some surfaces are expected to be contaminated after a workday, such as the places where drugs are compounded. Observing contamination on other surfaces is also anticipated but to a lesser extent. For example, storage areas and infusion pumps should be exempt from contamination if correct handling procedures are followed. Indeed, an intensive decontamination session proved to have a lasting effect on shelves, but biological safety cabinets and floors rapidly became contaminated again after typical daily drug preparation activities were resumed. 22 In addition, workers may be less prone to wearing their PPE in areas where the contamination risk is perceived as lower.
In this monitoring program, cyclophosphamide, gemcitabine, and platinum were the drugs most frequently quantified on surfaces. Contamination with other drugs was seldom measured. Dolezalova et al. described the 2018–2020 contamination with 13 antineoplastic drugs in 24 centers across Czech Republic and Slovak Republic, and they found 5-fluorouracil, cyclophosphamide, and platinum to be the most frequent drugs on surfaces. 23 Sottani et al. reported the 2016–2021 contamination in nine Italian centers with four drugs and found gemcitabine to be the most frequently measured on surfaces, followed by cyclophosphamide and platinum. 11 This group also indicated that while 5-fluorouracil was the less frequent drug on surfaces, its concentration was the highest. This variation in the contamination profile for each drug supports the decision to use contamination thresholds that are drug-specific in our program. Our group also advocates for the regular update on the limits used, considering that many groups have reported a reduction in contamination over the years.
Hazardous drug committee
Less than half of the centers (39%) reported having a hazardous drugs committee, although larger centers reported more frequently having one. This higher proportion of larger centers that implemented a hazardous drug committee probably explains why the surface contamination with cyclophosphamide appeared higher in centers that had one. This may indicate that larger centers may have the ability to allocate more resources for organizational support. Although every center should aim to have such a committee, the proportion is still an improvement over the 2006 Quebec survey where only 23% of centers reported having one. 24 Wrzesniewski and Park concluded that their multidisciplinary oversight committee “positively influenced” their compliance with USP 800 guidelines. 25 Their activities included tracking environmental monitoring reports and action plans. The 2021 ASSTSAS guide reiterated the necessity of having a hazardous drugs committee and the Quebec community of practice on the safe handling of hazardous drugs support this requirement. 7
Other practices
The use of closed-system drug transfer devices was not widespread. Centers were able to achieve low surface contamination with and without these devices. Various sources of contamination coexist in a healthcare center, in many of which a closed system would have no impact, such as the contamination of vials, patient excreta, and spills. A recent Canadian survey identified costs and compatibility as the main issues with these devices. 4
Most participating centers connected and primed IV tubing at the pharmacy; doing so in a controlled environment where pharmacy technicians wear adequate PPE reduces the risks for nursing staff in the healthcare units and outpatient clinics. This monitoring program is not a study designed to assess the efficacy of specific practices; such as study would need to be conducted in a controlled environment to reduce variability and answer specific research questions.
Training
Most oncology pharmacy and nursing staff received specific training on hazardous drugs, but the proportion was lower for hygiene and sanitation staff, especially those working in the oncology outpatient clinics. Surfaces cleaned by hygiene and sanitation staff were shown to be highly contaminated with antineoplastic drugs. 26 A comprehensive training program should involve all potentially exposed workers. Wrzesniewski and Park specified that their oversight committee was responsible for supervising the development of training programs and tracking their completion. 25
Respondents used a wide variety of training methods. Other groups have shown that oncology nurses significantly increased their knowledge and performance scores three months after attending a two-day workshop. 27 Von Grünigen et al. also presented a significant improvement in knowledge scores after respondents from 17 countries participated in their e-learning modules. 28 Zimmer et al. used theoretical and practical means to train nursing students. In addition, the authors simulated handling scenarios and used fluorescence as a marker of good practices. 29
Only 19% of respondents to a French survey indicated that they received hazardous drug content in their initial training, but 47% received continuous education. 14 In this monitoring program, the reverse trend was observed: 74–84% reported initial training and 14–56% reported annual refresher training. Each center is responsible to plan its own training program. The variation in local policies and procedures justifies the creation of content specifically curated for a single center, but people would benefit from more largely available hazardous drug educational content to optimize their resources.
In Quebec, all staff performing sterile hazardous drug compounding must be trained and have a competency assessment. 30 This explains the higher proportion reported for pharmacy staff. In the United States, both the USP 797 and USP 800 also highlighted the need to “demonstrate” competencies after training and every 12 months.5,31 These requirements are time-consuming and are a challenge to plan and to document. 32
Cleaning practices
Two-third of respondents cleaned their antineoplastic drug vials upon receipt, which can reduce this source of contamination. For instance, a center included in the study of Acramel et al. cleaned its vials with a bactericidal wipe. 10 A recent study confirmed that the exterior of antineoplastic drug vials was contaminated. 33
A higher proportion of larger centers reported using the cleaning practices evaluated in our questionnaire. This comes as no surprise as larger centers would be more likely to have dedicated resources. Nonetheless, having a dedicated hygiene and sanitation resource for oncology would facilitate training and make sure that safe practices are used.
Cleaning practices varied tremendously among centers, in terms of frequency, products used, and “goal” (i.e. decontamination, deactivation, disinfection). The cleaning efficacy will also vary according to the drug that needs to be removed from the surface and the type of surface. 34 Recently, Blahova et al. have shown that alcohol disinfectants can even increase the contamination measured after antineoplastic drugs that were encrusted within the floor are rendered accessible. 35
Results communication
Contamination reports should be shared with each center’s hazardous drug committee and with all workers potentially exposed to hazardous drugs, as an opportunity to increase their awareness. 7 The majority of centers that had previously participated in this program indicated that they had shared their contamination results locally. Most had shared their results with their pharmacy staff, but efforts are needed to increase the diffusion to all categories of staff exposed to hazardous drugs. Indeed, Hon et al. measured the highest frequency of contaminated hands with “other” types of job titles, above nurses and pharmacy staff. 36 Very few authors of surface contamination studies provided information about the local diffusion of results. Acramel et al. recently reported that a multidisciplinary team presented contamination results to the staff from two French centers and these were received positively. 10 Indeed, the authors advocate that annual monitoring followed by the presentation of results can help reduce exposure for all workers.37 We believe that the existence of this monitoring program since 2010 has played a significant role in the reduction in surface contamination in Canada.
Limits
Participation in the monitoring program was voluntary and did not include all Canadian hospitals; however historical data from this large-scale study is reproducible. Platinum quantification was optional and represented additional costs for the centers, which explains the lower number of samples for this drug. Surface sampling could not be done by the same research assistant throughout Canada, but staff performing the sampling was trained and standardized sampling sites were used. End of working day sampling made sure that results represented as much as possible the workers’ truthful exposure; however, as with any monitoring, results represent only a snapshot of a single day and may not be representative of every day. Statistical analyses were done with cyclophosphamide and gemcitabine contamination data only, as it was too low with the other drugs.
Conclusion
Regular participation in a surface monitoring program is an excellent opportunity to review practices, identify specific risk areas, inform workers and refresh their training. Local hazardous drug committees can play a key role in ensuring that contamination reports are followed through with corrective measures when warranted and in safeguarding the use of up-to-date procedures. The benchmarking opportunity of this national program is beneficial to centers that can compare their surface contamination with that of similar centers. A low level of contamination will likely persist on surfaces despite the use of safe handling practices. The involvement of all workers is crucial to reduce this contamination as much as possible and to make sure that they use PPE to limit their exposure. More work is needed to increase the awareness of other types of staff such as hygiene and sanitation workers and shared training would be a valuable time-saving resource.
Footnotes
Acknowledgements
The authors would like to thank the Centre de toxicologie du Québec staff for the analysis of samples. We would also like to thank the centers that participate in this monitoring program.
Author Contributions
EP, NC, JFB, and CT contributed significantly to the conception and design of the work. EP, CMC, NC, and CT contributed significantly to the acquisition of data for the work. EP, JFB, and CT contributed significantly to the analysis, and interpretation of data for the work. EP and CT drafted the work. All authors revised it critically for important intellectual content. All authors approved the final version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1.
Detailed results per drug.
