Abstract
Current guidelines for vaccination in allogeneic hematopoietic stem cell transplant (HCT) recipients recommend initiation of pneumococcal vaccination series three to six months post-HCT, with most data supporting initiation at six months due to a more robust immune response. This single-center, retrospective, observational chart review aimed to evaluate the impact of initiating the pneumococcal vaccine series at three months post-HCT compared to six months post-HCT. The primary endpoints were defined as a percentage of patients with a serologic response of >1 and >1.3 µg/mL for over 50% of the defined serotypes. Outcomes showed no difference in immunologic response between the two groups.
Introduction
Infections are a significant cause of morbidity and mortality in allogeneic hematopoietic stem cell transplant (HCT) recipients. Pneumonia remains the most common infection, with up to 10% of patients contracting a serious Streptococcus pneumoniae infection within one year. 1 Many factors contribute to the overall risk of pneumonia in this patient population including donor and recipient immunity, graft source, and graft versus host disease. 2 For these patients with prolonged periods of suppressed immunity, vaccine timing, vaccination series adherence, and booster doses are critical for preventing infection. 1
Current guidelines for patients undergoing allogeneic-HCT recommend the pneumococcal conjugate vaccine (PCV13) as a three-dose series followed by a single dose of the pneumococcal polysaccharide vaccine (PPSV23).3–6 Prior to June 2017, the pneumococcal vaccine series was initiated at six months post-HCT at the University of California San Diego Health (UCSDH). In June 2017, the vaccine policy was revised, and the pneumococcal vaccine series was initiated earlier at three months post-HCT.
The primary objective of this study was to evaluate (1) the current practice of initiating the pneumococcal vaccine series at three months post-HCT in comparison to the previous practice of initiating this series at six months post-HCT and (2) the impact on vaccine response and seroprevalence. Secondary objectives include the effect of steroid and immunoglobulin (IVIG) administration on vaccine response, proper administration of the vaccine series, and admissions due to infection.
Methods
This single-center, retrospective, observational cohort study evaluating the impact of initiating the pneumococcal vaccine series at three months post-HCT compared to six months post-HCT was conducted for adult patients at UCSDH between 1 January 2012 and 1 July 2020. Included patients were 18 years or older, recipients of allogeneic-HCT, received at least three doses of PCV13, and had serologic titers obtained at least one month after completion of the three PCV13 doses. Data was collected for two years after the completion of the vaccine series.
Data collected from electronical medical records included the following: age at transplant, gender, race, underlying hematologic disease, date of transplant, prior transplantation(s), conditioning regimen, ABO mismatch, donor human leukocyte (HLA) matching, stem cell source, graft versus host disease prophylaxis, donor and recipient cytomegalovirus serostatus, hematopoietic cell transplant (HCT)-comorbidity index at time of HCT, PCV13 and PPSV23 administration dates, concentrations of IgG specific for PCV13 serotypes following PCV13 series, vaccines co-administered with PCV13 or PPSV23, absolute lymphocyte count prior to each PCV13 and PPSV23 administration, development of graft versus host disease (GVHD) and grade of GVHD through one year post-allogeneic-HCT, IVIG administration within 5 months of PCV13 or PPSV23 administration, presence and dose (mg/kg) of steroids at the time of PCV13 administration, hospitalizations, and length of stay after completion of vaccination, both all-cause and those related to pneumococcal disease, and incidence of infection.
The primary endpoints were defined as a percentage of patients with a serologic response of >1 and >1.3 µg/mL for over 50% of the following serotypes: 1, 3, 4, 8, 9(9N), 12(12F), 14, 19(19F), 23(23F), 26(6B), 51(7F), and 56(18C).7,8 Secondary outcomes included the following: percentage of patients with a serologic response >1 µg/mL for 70% of the defined serotypes, specific serologic responses >1 µg/mL, proper administration of the vaccine series, and incidence of infection post-HCT. Investigated serotypes and threshold limits were determined by a combination of manufacturer recommendations and available published literature.7,8 Serologic responses were measured at least one month after completion of the three PCV13 doses. Bivariate analyses were performed to compare clinical/demographic characteristics between the three- and six-month groups using the chi-square or Fisher's exact test for categorical variables and the Student’s t or Mann–Whitney U tests for continuous variables. A multivariable logistic regression analysis was performed to determine if initiation of pneumococcal vaccine series at three months versus six months was independently associated with outcome.
Results
Demographics
We identified 129 patients that met inclusion criteria. Of the 129 patients, 23 patients received vaccination at three months post-HCT, and 106 patients received vaccination at six months post-HCT. The median (interquartile range) age of the two groups was 41 (33–65) and 55 (42–63), respectively (p = 0.23). The distribution of transplant types (comprised of haploidentical, mismatched unrelated donor (MMUD), matched-related donor (MRD), and matched-unrelated donor (MUD)) significantly differed between the two groups (Table 1). No other statistically significant differences were observed.
Bivariate relationship between clinical/demographic characteristics and treatment group.
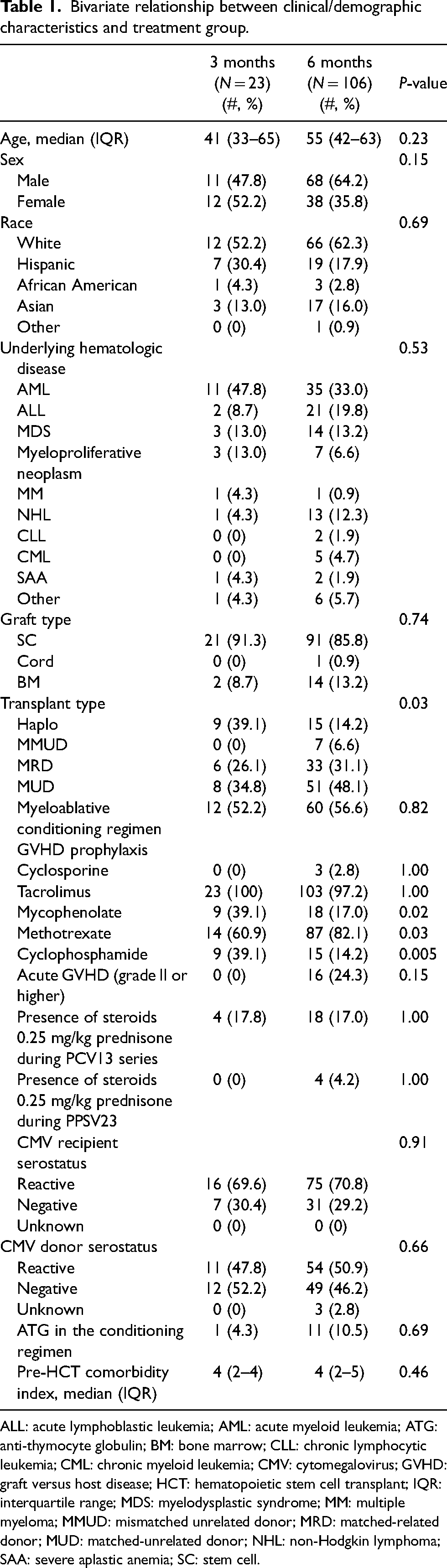
ALL: acute lymphoblastic leukemia; AML: acute myeloid leukemia; ATG: anti-thymocyte globulin; BM: bone marrow; CLL: chronic lymphocytic leukemia; CML: chronic myeloid leukemia; CMV: cytomegalovirus; GVHD: graft versus host disease; HCT: hematopoietic stem cell transplant; IQR: interquartile range; MDS: myelodysplastic syndrome; MM: multiple myeloma; MMUD: mismatched unrelated donor; MRD: matched-related donor; MUD: matched-unrelated donor; NHL: non-Hodgkin lymphoma; SAA: severe aplastic anemia; SC: stem cell.
Primary outcome
There was no significant difference in the percentage of patients with a serologic response of >1 or >1.3 µg/mL for over 50% of the defined serotypes. In the group vaccinated starting at three months post-HCT, 22 patients (95.7%) had measured titers >1 µg/mL for over 50% of the defined serotypes in comparison to 83 patients (78.3%) in those vaccinated at six months post-HCT (p = 0.07). Eighteen (78.3%) patients in the group vaccinated starting three months post-HCT had measured titers >1.3 µg/mL compared to 80 (75.5%) in those vaccinated at six months post-HCT (p = 0.78). After adjustment of variables associated (p < 0.25) with the treatment group, there was no association between treatment group and outcome (odds ratio (OR): 0.15, 95% confidence interval (CI): 0.02–1.24, p = 0.08).
Secondary outcomes
There were no differences in secondary outcomes for the two groups. The percentage of patients with a serologic response of >1 µg/mL for over 70% of the defined serotypes was 14 (16.9%) versus 59 (55.7%) for those vaccinated at three months post-HCT versus those vaccinated six months post-HCT, respectively (p = 0.65). No significant differences were observed between the two groups regarding proper administration of the vaccine series, incidence of infection post-HCT, or use of steroids and IVIG administration between the two groups.
Discussion
United States guidelines recommend initiation of the pneumococcal vaccination series at three to six months post-HCT, with most data supporting initiation at six months due to the possibility of a more robust immune response.1,2,5,6,9 In this study, we aimed to evaluate the current practice of initiating the pneumococcal vaccine series at three months post-HCT in comparison to the previous practice of initiating the pneumococcal vaccine series at six months post-HCT and the impact on pneumococcal serologic response. Our study found no statistically significant differences in serologic response or hospital outcomes between the two groups, suggesting that it is feasible to start the pneumococcal vaccine series at three months post-HCT in this high-risk population. This finding is consistent with current European guidelines. 10
Although our results demonstrate no difference in response or outcomes for patients vaccinated at three months post-HCT versus six months post-HCT, our study does have significant limitations. This was a retrospective chart review with a relatively small sample size. As a result, proportions that appeared to be numerically distinct (95.7% vs. 78.3%) did not differ significantly. Of the patients that met inclusion criteria, there was an imbalance in the number of patients in each group with significantly more patients in the vaccination at six months post-HCT group. This could have contributed to the power issue described above. Due to the non-randomized nature of the study population, confounders including degree of immunosuppression at the time of vaccination may have differed. We attempted to overcome this limitation and control for confounders by performing multivariable logistic regression analyses, which ultimately corroborated the bivariate analyses. Lastly, we used surrogate markers to define vaccine response. Although evaluation of serologic response is aligned with current practice, surrogate biomarkers do not always correlate with clinical response and are not routinely recommended by IDSA guidelines.5,7,10,11 Baseline serotypes and fold increases were not evaluated in this study. If serotypes are used to assist in assessing response, pre-vaccination baseline titers should be obtained for purposes of defining fold increase.5,7 Furthermore, the outcomes of this study resulted in discontinuation of serotype evaluation post-HCT and continued vaccine initiation at three months post-HCT.
To our knowledge, this is the first study to compare the response of the pneumococcal vaccine series in HCT patients at three months versus six months in the United States and therefore provides preliminary data in the post-allogeneic-HCT population. Given the issues listed above, more robust and prospective studies are needed to determine the optimal time to initiate pneumococcal vaccines series after allogeneic-HCT.
Conclusion
Our results demonstrate that initiation of the pneumococcal vaccine series at three months is not associated with differences in serologic response in allogeneic-HCT recipients.
Footnotes
Acknowledgements
All authors contributed to research design, drafting of the paper and its revisions, and have approved the final version. Further delineation is as follows: Halle Cheplowitz contributed to study design, performed the research, analyzed the data, writing—original draft preparation, and writing—review and editing. Nimish Patel contributed to study design, analyzed the data, writing—review and editing. Alexander Kim contributed to study design, writing—review and editing. Cathy Logan contributed to study design, writing—review and editing. Nancy Law contributed to study design, writing—review and editing. Divya Koura contributed to study design, writing—review and editing. Nina Haste contributed to study design. Katherine Medley contributed to study design, writing—review and editing. Julie Trinh contributed to performing the research. Tyler Sanders performed the research. Mahnaz Taremi contributed to writing—review and editing. Ila M. Saunders contributed to designing the research study, analyzing the data, writing—review and editing.
Author contributions
All authors contributed to research design, drafting of the paper and its revisions, and have approved the final version. Further delineation is as follows: HC contributed to study design, performed the research, analyzed the data, writing—original draft preparation, and writing—review and editing. NP contributed to study design, analyzed the data, writing—review and editing. AK contributed to study design, writing—review and editing. CL contributed to study design, writing—review and editing. NL contributed to study design, writing—review and editing. DK contributed to study design, writing—review and editing. Nina Haste contributed to study design. KM contributed to study design, writing—review and editing. JT contributed to performing the research. TS performed the research. MT contributed to writing—review and editing. IMS contributed to designing the research study, analyzing the data, writing—review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
